Freeze-Drying Market
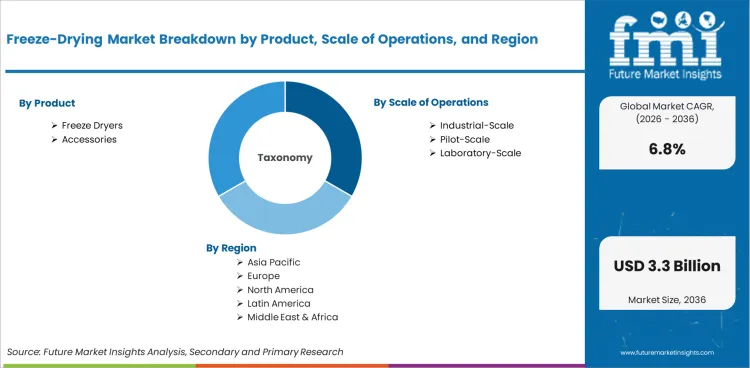
The freeze-drying market is segmented by Product (Freeze Dryers and Accessories); Scale of Operations (Pilot-Scale, Laboratory-Scale, and Industrial-Scale); Application (Aseptic Applications, Non-Aseptic Applications, and Both Aseptic and Non-Aseptic); End User (Pharma, Biotechnology, CRO, CDMO, Academic and Government, Diagnostic, Other Healthcare Markets, Food and Agriculture, General and Environment, Chemicals, and Other); Project Type (Expansion, Greenfield, and Replacement); and Revenue Stream (Equipment Sales, Service Contracts, and Aftermarket Parts). Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Freeze-Drying Market Size, Market Forecast and Outlook By FMI
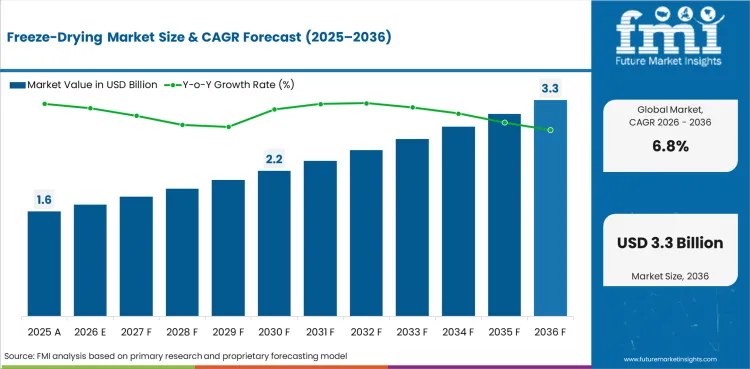
The freeze-drying market is expected to expand from USD 1.6 billion in 2026 to USD 3.1 billion by 2036. The market is anticipated to register a 6.8% CAGR during the forecast period. Freeze dryers are likely to lead with 71.3% share in 2026. Aseptic applications are projected to account for 46.9% of application demand in 2026.
Summary of the Freeze-Drying Market
- Demand and Growth Drivers
- Sterile injectable and biologic programs are expected to raise demand for freeze-drying systems across regulated production sites.
- CDMO and CRO development pipelines are likely to increase orders for pilot systems and flexible lyophilization platforms.
- Stability-focused formulation work is anticipated to lift lyophilization use for products with limited liquid-form shelf life.
- Product and Segment View
- Freeze dryers are expected to lead the product segment as full system installations carry the largest capital outlay.
- Aseptic applications are likely to lead application demand as sterile batch security stays central in injectable production.
- Industrial-scale systems are anticipated to hold strong adoption as commercial plants add larger assets for routine batch output.
- Geography and Competitive Outlook
- India is expected to record the fastest expansion as sterile injectable output and CDMO activity increase system orders.
- China is likely to post strong demand as domestic bioprocessing and aseptic manufacturing programs add new capacity.
- Companies with stronger validation systems and service coverage are anticipated to gain preference across regulated production sites.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Analyst for Healthcare at FMI, opines, “The freeze-drying market is expected to favor suppliers able to deliver repeatable cycles and reliable contamination control across regulated facilities. Equipment makers with strong service reach and validation-ready systems are likely to gain preference as manufacturers work to reduce batch disruption risk.”
- Freeze-Drying Market Value Analysis
- The freeze-drying market is shifting toward larger system investments linked with sterile injectable and biologic manufacturing.
- Demand is likely to rise as lyophilization improves shelf stability for sensitive pharmaceutical and diagnostic products.
- Adoption is expected to gain traction from CDMO programs needing pilot assets and production-ready freeze dryers.
- Spending is anticipated to be backed by capacity expansion and stricter uptime needs across regulated manufacturing sites.
Freeze-Drying Market Definition
The freeze-drying market covers lyophilization systems and related accessories used to remove water from heat sensitive materials under vacuum. It includes equipment used for sterile pharmaceuticals and biologics and diagnostics and food applications and selected chemical programs. Revenue comes from system sales and recurring support linked to installed equipment.
Freeze-Drying Market Inclusions
Scope for the market includes freeze dryers and accessories. Pilot scale and laboratory scale and industrial scale systems are included. Aseptic and non-aseptic and mixed use applications are counted. Demand from pharma and biotechnology and CRO and CDMO and academic and diagnostic and food and chemical users is included.
Freeze-Drying Market Exclusions
Spray drying and vacuum oven drying and hot air-drying equipment are outside the study scope. Contract manufacturing revenue for freeze dried products is not counted. Primary packaging items are excluded unless bundled directly with the equipment offer.
Freeze-Drying Market Research Methodology
- Primary Research: FMI analysts reviewed input from sterile manufacturing teams and validation staff and CDMO operations and equipment suppliers.
- Desk Research: The study used public regulatory material and peer reviewed literature and company disclosures and standards references tied to lyophilization and aseptic production.
- Market Sizing and Forecasting: Baseline values were built from the supplied source data and then converted into rounded billion values for 2025 and 2026 and 2036.
- Data Validation and Update Cycle: Additional segment splits for project type and revenue stream were developed from source cues on capacity additions and replacement cycles and lifecycle service intensity.
Why Is the Freeze-Drying Market Growing?
- Sterile injectable and biologic programs are increasing demand for freeze drying capacity in aseptic production.
- CDMO and CRO development activity is raising orders for pilot and laboratory systems.
- Stability needs are lifting use of lyophilization for temperature-sensitive products that do not store well in liquid form.
Demand is witnessing stable expansion as freeze-drying supports product stability across pharmaceuticals and biologics. Aseptic use holds a leading position because contamination control and batch integrity stay central in freeze-dried drug production. CDMOs and CROs influence market growth through pilot and laboratory systems that help development work and method transfer.
Capital budgets can slow equipment orders but pharma manufacturers still invest in systems that improve cycle repeatability and reduce downtime and support qualification documents for audits. Service response and documentation support keep supplier selection tight across regulated plants.
Market Segmentation Analysis
- Freeze dryers are estimated to account for 71.3% of product demand in 2026 since the main system package absorbs most project spending.
- Equipment sales are expected to represent 63.4% of revenue stream demand in 2026 as new system purchases stay larger than yearly service bills.
- Aseptic applications are projected to contribute 46.9% of application demand in 2026 as sterile batch control keeps lyophilization central in injectable production.
- Industrial-scale systems are anticipated to make up 45.2% of operations demand in 2026 because commercial plants keep adding larger assets for repeat batch output.
Freeze-Drying Market Analysis by Product
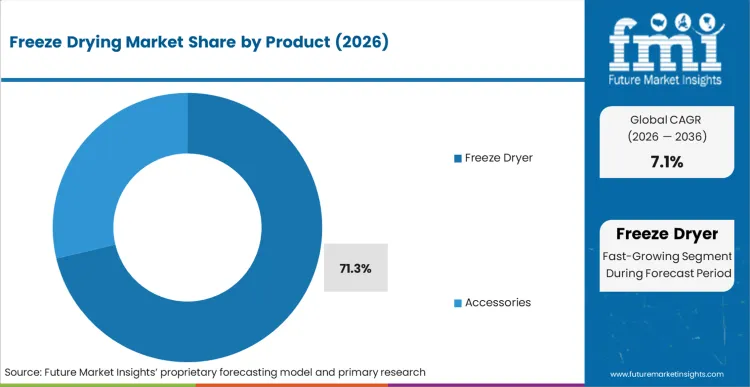
- Freeze dryers are expected to account for 71.3% of product demand in 2026 because full system installations gain the largest share of plant capital budgets.
- Core chambers and condensers and validation-ready shelf controls and software keep this line ahead of accessory purchases in new sterile projects.
- OEMs with strong system integration capability can protect share as plants seek reliable cycle performance and lower commissioning risk.
Insights into the Accessories Product Segment
- Accessories are likely to advance faster than the other product types as installed bases keep adding shelves and control upgrades and sensors and change parts over time.
- Maintenance cycles and performance tuning propel repeat orders across regulated sites that cannot tolerate long downtime windows.
- Accessory demand strengthens further as older systems move into upgrade programs and plants seek longer asset life.
Freeze-Drying Market Analysis by Scale of Operations
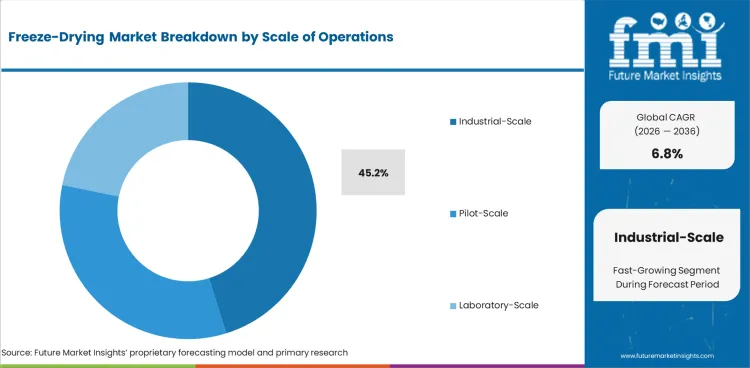
- Industrial-scale systems are projected to contribute 45.2% of operations demand in 2026 as commercial supply programs need larger batch capacity and stable repeat output.
- Large plants favor the systems as each installation can support routine sterile production with stronger throughput per cycle.
- Demand remains strongest in pharma and CDMO projects that add production lines for injectables and higher value therapies.
Insights into the Pilot-Scale Segment
- Pilot-scale systems are likely to rise faster than the other pilot-scale channels since development work and tech transfer activity keep expanding across CDMO and CRO programs.
- Smaller units support formulation trials and process refinement before full commercial installation decisions are locked in.
- Pilot demand remains firm as Asia-based capacity programs keep feeding products from development into commercial supply.
Freeze-Drying Market Analysis by Application
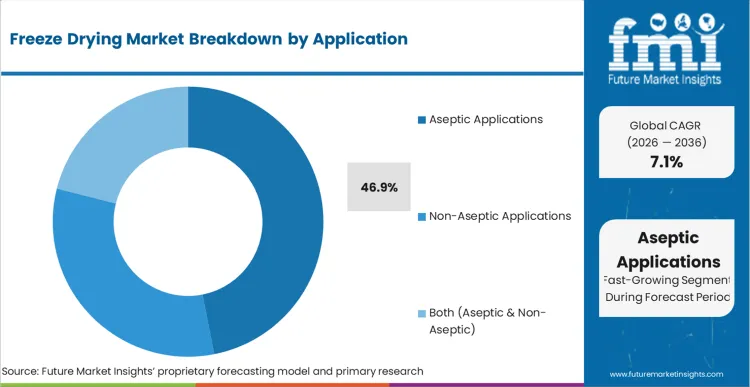
- Aseptic applications are anticipated to represent 46.9% of application demand in 2026 since sterile injectables and biologics need tight contamination control during drying cycles.
- Batch security and traceable cycle settings keep aseptic programs at the center of equipment selection across regulated plants.
- Suppliers that support leak integrity checks and stronger documentation are likely to stay in a better position in this segment.
Insights into the Both Aseptic and Non-Aseptic Segment
- Both aseptic and non-aseptic use is likely to move faster than the other application types as multipurpose sites require flexible assets across development and small batch programs.
- Mixed use platforms help organizations spread equipment time across sterile projects and select non-sterile work without adding separate systems too early.
Freeze-Drying Market Analysis by End User
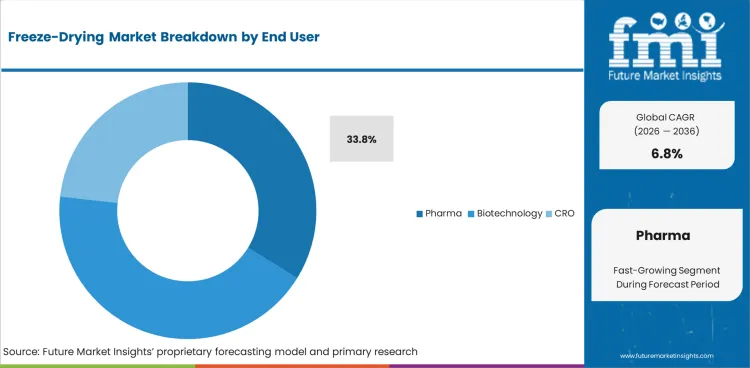
- Pharma is expected to make up 33.8% of end user demand in 2026 since sterile injectable output and biologic manufacturing keep freeze-drying at the center of regulated production.
- Large drug producers continue to order systems that support qualification work and repeatable batch performance across commercial lines.
Insights into the CDMO End User Segment
- CDMOs are likely to record the fastest rise in end user demand as sponsor outsourcing pushes new development and production work into specialist facilities.
- Audit support and fast method transfer are important as contract sites must satisfy many client requirements across the year.
Freeze-Drying Market Analysis by Project Type
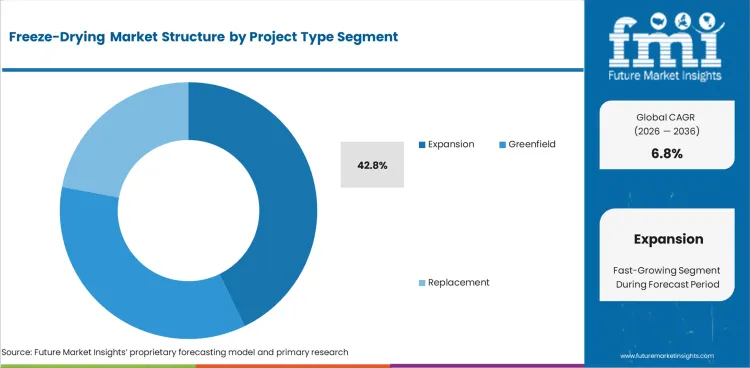
- Expansion projects are expected to account for 42.8% of project type in 2026 as many suppliers add freeze-drying capacity inside existing regulated sites rather than build entirely new plants.
- Brownfield additions reduce qualification burden and shorten timelines compared with full greenfield programs in many regions.
Insights into the Greenfield Project Segment
- Greenfield projects are likely to rise faster than the larger project types as India and China contribute fresh sterile manufacturing space for future output needs.
- New sites often specify automation-ready lyophilizers at an early stage and attach service support to commissioning plans.
Freeze-Drying Market Analysis by Revenue Stream
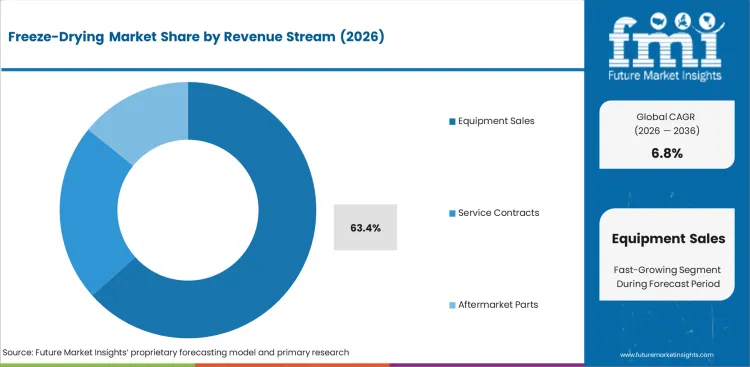
- Equipment sales are likely to secure 63.4% of revenue stream demand in 2026 because the chamber and control package are the main spending event in each new installation.
- Plants commit the largest outlay during system purchase and qualification while service income builds more gradually across the asset life cycle.
Insights into the Service Contracts Segment
- Service contracts are likely to expand faster than the other revenue streams because uptime targets and spare parts planning are becoming stricter across sterile plants.
- Sites want faster field response and preventive maintenance plans that reduce deviation risk during critical batches.
Freeze-Drying Market Drivers, Restraints, Opportunities
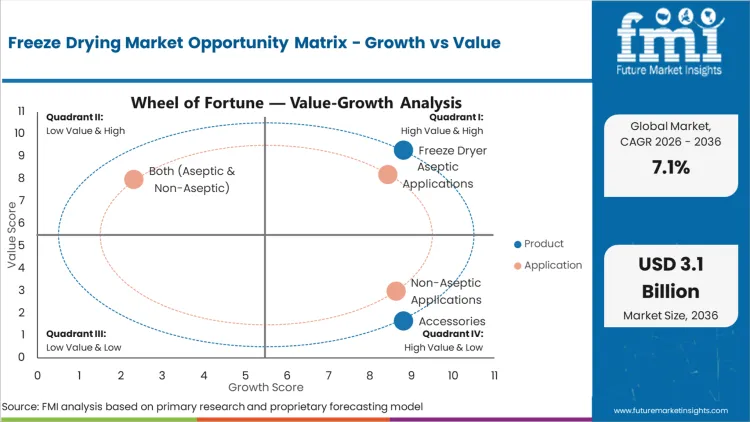
- Sterile injectable and biologic pipelines sustain a consistent system demand growth across regulated plants.
- Long validation cycles and high capex slow full-scale additions at some sites.
- Lifecycle service contracts and upgrade packages can lift recurring income as installed bases expand.
Freeze-drying demand advances with sterile capacity additions and the extensive use of lyophilized formats in biologics and injectables. Plants value stable cycles because one failed batch can delay supply and add high recovery cost.
High capital budgets and long qualification timelines slow some projects. Service quality and documentation quality matter in supplier selection because regulated sites need support that extends beyond equipment delivery.
Regional Market Analysis
.webp)
| Country | CAGR |
|---|---|
| United States | 6.5% |
| China | 8.4% |
| India | 8.5% |
| Japan | 7.2% |
| United Kingdom | 6.6% |
| Germany | 5.4% |
| Brazil | 5.9% |
Source. Future Market Insights analysis based on the provided study inputs for 2026.
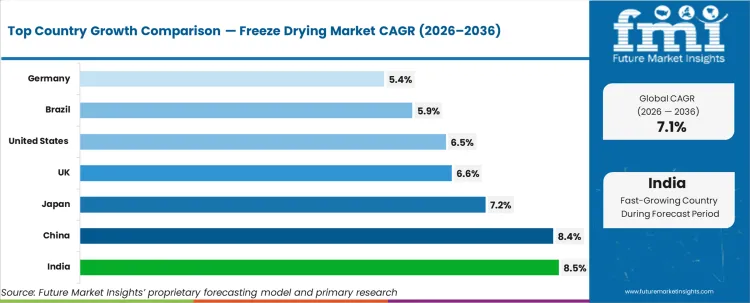
Freeze‑Drying Market CAGR Analysis by Country
- India is anticipated to rise at an 8.5% CAGR and China (8.4% CAGR) lead global growth, advancing faster than the global average due to rapid expansion of sterile manufacturing capacity and rising Contract Development and Manufacturing Organization (CDMO) activity. These markets are seeing strong demand for new freeze‑drying systems to support pharmaceutical and biologics production.
- The United States, projected to grow at a 6.5% CAGR, reflects steady demand driven by its large installed base of freeze‑drying equipment.
- Japan is predicted to grow at a CAGR of 7.2% through 2036 and the United Kingdom (6.6%) remain in the mid‑growth band, as system demand is shaped by highly regulated pharmaceutical operations.
- Brazil is expected to rise at a CAGR of 5.9% and Germany at 5.4% CAGR through 2036 reflecting mature market conditions. In these countries, a well‑established installed base results in a higher proportion of replacement and modernization demand compared to new capacity additions.
The freeze‑drying market continues to expand at a steady pace while developed markets progress through equipment replacement and incremental capacity upgrades. The study covers more than 30 countries, and the main markets are listed below.
Demand Outlook for Freeze-Drying Market in the United States
The United States sector is expected to grow at a 6.5% CAGR through 2036 as established aseptic manufacturing and high-value biologics production propel freeze-drying demand growth across regulated pharmaceutical operations. Sterile capacity expansion influences orders for systems that deliver repeatable cycles and stable batch performance. Pharma manufacturers and CDMOs keep service response and spare parts support high on the selection list. The country shows a measured growth pace because it starts from a mature production base.
- Established aseptic manufacturing keeps equipment demand steady.
- Biologics production supports repeat need for validated cycles.
- Service support and spare parts availability influence supplier choice.
Sales Analysis of Freeze-Drying Market in China
Industry sales in China are projected to record an 8.4% CAGR through 2036 as rapid sterile manufacturing build-out and higher aseptic lyophilization use for injectables lift demand across production and development settings. Industrial-scale equipment orders influence sector growth as pharma plants expand local and export-facing output. Laboratory and pilot demand expansion tracks growth in formulation work.
- Sterile manufacturing build-out lifts national equipment demand.
- Aseptic injectable production supports industrial-scale orders.
- Formulation work increases pilot and laboratory system use.
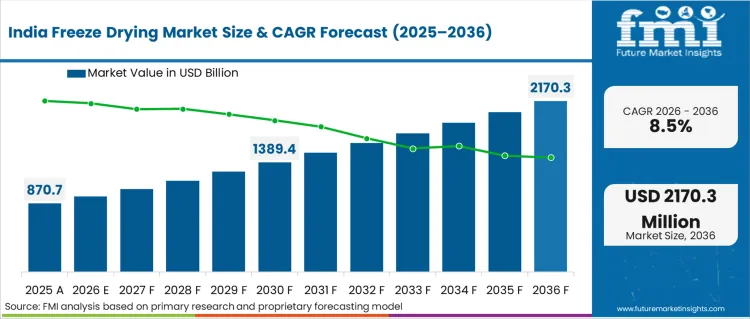
Opportunity Analysis of Freeze-Drying Market in India
Adoption for the sector in India is expected to expand at an 8.5% CAGR through 2036 because sterile injectable output is rising. CDMO activity is increasing orders for pilot systems and production lyophilizers. Audit-linked project work keeps qualification support important in supplier selection. Uptime reliability and service response remain central for manufacturers that cannot absorb extended downtime.
- Sterile injectable output is increasing equipment demand.
- CDMO activity raises orders for pilot and production units.
- Qualification support influences supplier selection in regulated work.
In-Depth Analysis of Freeze-Drying Market in Germany
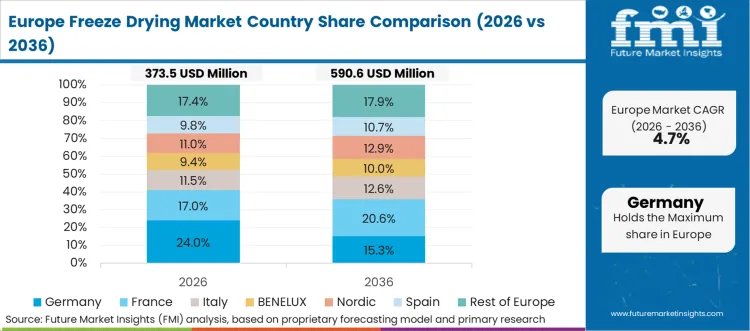
Germany’s market is likely to post a 5.4% CAGR through 2036 as a strong pharmaceutical manufacturing base and steady investment in process consistency keep demand stable across regulated facilities. Industrial-scale systems draw a large share of spending in aseptic applications. Laboratory and pilot units support development programs and tech transfer work. Germany reflects demand growth led by replacement and compliance from a mature installed base.
- Pharmaceutical manufacturing depth keeps system demand firm.
- Process consistency needs support industrial-scale spending.
- Development work sustains pilot and laboratory demand.
In-Depth Analysis of Freeze-Drying Market in Japan
Industry expansion for Japan is forecast to grow at a 7.2% CAGR through 2036 as stable pharmaceutical production and high-quality governance influence growth for repeatable cycles and strong documentation traceability. Laboratory-scale activity influences further expansion in formulation and process optimization work. Smaller systems remain important for development timelines in regulated settings. Japan shows healthy growth without the sharper pace recorded in India and China.
- Stable pharmaceutical production supports recurring system demand.
- Quality governance keeps traceability and repeatability important.
- Formulation work supports laboratory-scale equipment use.
Freeze-Drying Market Analysis in the United Kingdom
Demand for the sector in the United Kingdom is expected to register a 6.6% CAGR through 2036 as CDMO activity and sterile injectable capacity investment support orders for systems with validated cycles and repeatable performance. Project-based expansions keep installation timelines important in buying decisions. Commissioning support and audit documentation also influence equipment selection.
- CDMO activity supports recurring demand for freeze drying systems.
- Sterile injectable investment lifts validated equipment orders.
- Installation and commissioning support influence purchase decisions.
Freeze-Drying Market Analysis in Brazil
The industry in Brazil is projected to record a 5.9% CAGR through 2036 as steady investment in pharmaceutical manufacturing and rising need for stable shelf-life oriented formats support selected sterile and non-sterile applications. Local service engineer availability also affects supplier preference and ownership cost. Brazil posts moderate growth because deployment is tied closely to import flow and service access.
- Pharmaceutical investment supports steady equipment demand.
- Shelf-life oriented formats raise freeze drying use in selected applications.
- Import lead times and local service access influence supplier choice.
Competitive Landscape and Strategic Positioning
- Competition is fragmented. Buyer selection narrows around suppliers that support validated cycles and fast service response.
- Large equipment groups compete through installed base support or qualification documentation and accessory compatibility across regulated plants.
- Specialist firms compete through laboratory systems or automation controls and region-specific distributor support.
- Entry is harder in regulated projects that require GMP-ready records and uptime assurance and method transfer support.
Competition in freeze-drying is influenced by validated cycle performance and documentation quality after installation. Pharma manufacturers and CDMOs do not select on hardware alone because uptime support and spare-parts access affect batch continuity. Suppliers with service reach across industrial and development systems hold an edge in regulated projects. Larger portfolios influence account retention because one supplier can cover dryers and accessories with lower compatibility risk.
Established suppliers benefit from application knowledge and installed-base service coverage. Cleanability design and automation readiness improve their position in aseptic work because IQ and OQ support can lengthen vendor review. Academic and government laboratories keep price pressure tighter through tenders and approved vendor lists. Pricing power is stronger in projects that need qualification support at specific temperature and pressure targets.
Smaller specialists expand in narrower spaces tied to laboratory systems or controls integration. Yamato Scientific is well placed in research and process development systems while Azbil supports cycle consistency through controls and automation in Asia Pacific. Regional partner models help companies like GEA and IMA support installation and maintenance in import-led markets. Entry stays difficult because service depth must match equipment sales or downtime risk rises after commissioning.
Key Companies in the Freeze-Drying Market
Key companies in the freeze-drying industry include global system suppliers and specialist laboratory manufacturers along with regional service-focused providers.
- Global Leaders: GEA Group AG and IMA SpA and SP Industries Inc. and Azbil Corporation maintain stronger industrial installations and deeper regulated site support.
- Laboratory and Development Specialists: Yamato Scientific Co. Ltd. and Martin Christ Gefriertrocknungsanlagen GmbH and BUCHI Labortechnik AG keep a strong role in laboratory and pilot-scale systems.
- Regional and other Participants: Cuddon Freeze Dry and Lyophilization Systems Ltd. support selective projects through focused equipment and service capabilities.
Key Developments for the Freeze-Drying Market
- In October 2024, GEA launched the RAY Plus batch freeze dryer line for food processing with attention on efficiency and hygiene.
- In February 2025, CryoDry partnered with Spinco to strengthen distribution support for freeze-drying systems in India.
Competitive Benchmarking for the Freeze-Drying Market
| Company | Product Breadth | Sterile Workflow Fit | Service Reach | Footprint |
|---|---|---|---|---|
| GEA Group AG | High | Strong | Strong | Europe and Global |
| IMA SpA | High | Strong | Moderate | Europe and Global |
| SP Industries Inc. | Moderate | Strong | Strong | North America |
| Azbil Corporation | Moderate | Strong | Moderate | Japan and Asia |
| Yamato Scientific Co. Ltd. | Moderate | Moderate | Moderate | Asia |
| Martin Christ Gefriertrocknungsanlagen GmbH | Moderate | Moderate | Moderate | Europe |
| BUCHI Labortechnik AG | Moderate | Moderate | Moderate | Europe and Global |
Source. Future Market Insights analysis based on the provided study inputs for 2026.
Key Players in the Freeze-Drying Market
Major Companies
- GEA Group AG
- IMA S.p.A.
- SP Industries, Inc.
- Azbil Corporation
- Martin Christ Gefriertrocknungsanlagen GmbH
- BÜCHI Labortechnik AG
Regional Participants
- Yamato Scientific Co., Ltd.
- Cuddon Freeze Dry
- Lyophilization Systems Ltd.
Report Scope and Coverage for Freeze-Drying Market
| Item | Value |
|---|---|
| Market | Freeze-Drying Market |
| Estimated Value In 2025 | USD 1.5 billion |
| Expected Value In 2026 | USD 1.6 billion |
| Forecast Value In 2036 | USD 3.1 billion |
| CAGR 2026 To 2036 | 6.8% |
| Quantitative Units | Revenue in USD billion |
| Parent Segments Covered | Product and Scale of Operations and Application and End User and Project Type and Revenue Stream |
| Regions Covered | North America and Latin America and Europe and East Asia and South Asia and Oceania and Middle East and Africa |
| Countries Covered | United States and China and India and Japan and United Kingdom and Germany and Brazil |
| Key Companies Profiled | GEA Group AG and IMA SpA and SP Industries Inc. and Azbil Corporation and Yamato Scientific Co. Ltd. and Martin Christ Gefriertrocknungsanlagen GmbH and BUCHI Labortechnik AG |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up model validated through primary interviews and installed base review |
Source. Future Market Insights analysis based on the provided study inputs for 2026.
Freeze-Drying Market Analysis By Segments
Product
- Freeze Dryer
- Accessories
Scale of Operations
- Pilot-Scale
- Laboratory-Scale
- Industrial-Scale
Application
- Aseptic Applications
- Non-Aseptic Applications
- Both Aseptic And Non-Aseptic
End User
- Pharma
- Biotechnology
- CRO
- CDMO
- Academic And Government
- Diagnostic
- Other Healthcare Markets
- Food And Agriculture
- General And Environment
- Chemicals
- Other
Project Type
- Expansion
- Greenfield
- Replacement
Revenue Stream
- Equipment Sales
- Service Contracts
- Aftermarket Parts
Research Sources and Bibliography
- Samedan Pharmaceuticals Publisher. (2025). Fast precise efficient the freeze-drying revolution.
- GEA Group. (2025). LYOVAC pharma freeze dryer.
- USA Food and Drug Administration. Guide to inspections of lyophilization of parenterals.
- CryoDry. (2025). CryoDry welcomes Spinco as our official distributor in India.
- EuropaWire. (2024). GEA unveils RAY Plus freeze dryers for energy efficiency and hygiene in food processing.
- USA Food and Drug Administration. (2014, August 26). Lyophilization of parenterals.
- International Pharmaceutical Industry. (2025). Fast, precise, efficient: The freeze-drying revolution.
- Pharmaceutical Technology. (2025, May 8). Parenteral formulation: Deciding when to go frozen or freeze-dried.
- Pharmaceutical Technology. (2024, July 6). The rising iceberg of lyophilized innovations.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
This Report Answers
- What size is the freeze-drying market in 2026 and how large can it become by 2036?
- How fast is freeze-drying demand expected to expand between 2026 and 2036?
- Which product and scale of operations and application and end user and project type and revenue stream segments lead 2026 demand?
- Why are freeze dryers and equipment sales holding the largest 2026 shares?
- How are pilot systems and service contracts and CDMO demand changing the mix across the industry?
- Which countries are expected to post the fastest growth through 2036?
- Which companies stay most prominent across industrial and pilot and laboratory freeze-drying systems?
Frequently Asked Questions
How large is the freeze-drying market in 2026?
The freeze-drying market is expected to reach USD 1.6 billion in 2026 after rounding the supplied value into single decimal billion terms.
How big can the freeze-drying market become by 2036?
The freeze-drying market is projected to reach USD 3.1 billion by 2036 based on the supplied outlook converted into single decimal billion terms.
What growth rate is expected for the freeze-drying market through the forecast period?
The freeze-drying market is projected to expand at a 6.8% CAGR from 2026 to 2036 after rounding the stated values into billion terms.
Which product leads the freeze-drying market by 2026?
Freeze dryers lead the freeze-drying market with an expected 71.3% share in 2026 because full system purchases stay larger than accessory spending.
Which scale of operations holds the highest share in the freeze-drying market by 2026?
Industrial-scale systems lead the freeze-drying market with a projected 45.2% share in 2026 as commercial plants keep adding larger batch assets.
Which application remains largest in the freeze-drying market by 2026?
Aseptic applications stay largest in the freeze-drying market with an anticipated 46.9% share in 2026 due to sterile batch control needs.
Which country grows fastest in the freeze-drying market study?
India records the fastest freeze-drying market growth at a projected 8.5% CAGR through 2036 as sterile injectable output and CDMO demand rise.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2026 to 2036
- Freeze Dryers
- Accessories
- Freeze Dryers
- Y to o to Y Growth Trend Analysis By Product , 2021 to 2025
- Absolute $ Opportunity Analysis By Product , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Scale of Operations
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Scale of Operations, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Scale of Operations, 2026 to 2036
- Industrial-Scale
- Pilot-Scale
- Laboratory-Scale
- Industrial-Scale
- Y to o to Y Growth Trend Analysis By Scale of Operations, 2021 to 2025
- Absolute $ Opportunity Analysis By Scale of Operations, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Aseptic Applications
- Non-Aseptic Applications
- Both Aseptic And Non-Aseptic
- Aseptic Applications
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Pharma
- Biotechnology
- CRO
- Pharma
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Project Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Project Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Project Type, 2026 to 2036
- Expansion
- Greenfield
- Replacement
- Expansion
- Y to o to Y Growth Trend Analysis By Project Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Project Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Revenue Stream
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Revenue Stream, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Revenue Stream, 2026 to 2036
- Equipment Sales
- Service Contracts
- Aftermarket Parts
- Equipment Sales
- Y to o to Y Growth Trend Analysis By Revenue Stream, 2021 to 2025
- Absolute $ Opportunity Analysis By Revenue Stream, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- By Country
- Market Attractiveness Analysis
- By Country
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Scale of Operations
- By Application
- By End User
- By Project Type
- By Revenue Stream
- Competition Analysis
- Competition Deep Dive
- GEA Group AG
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- IMA S.p.A.
- SP Industries, Inc.
- Azbil Corporation
- Martin Christ Gefriertrocknungsanlagen GmbH
- BÜCHI Labortechnik AG
- GEA Group AG
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Scale of Operations, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Project Type, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by Revenue Stream, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Scale of Operations, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Project Type, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Revenue Stream, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Scale of Operations, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Project Type, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Revenue Stream, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Scale of Operations, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Project Type, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Revenue Stream, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 31: Eastern Europe Market Value (USD Million) Forecast by Scale of Operations, 2021 to 2036
- Table 32: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Project Type, 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Revenue Stream, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: East Asia Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 38: East Asia Market Value (USD Million) Forecast by Scale of Operations, 2021 to 2036
- Table 39: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 40: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Project Type, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Revenue Stream, 2021 to 2036
- Table 43: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: South Asia and Pacific Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 45: South Asia and Pacific Market Value (USD Million) Forecast by Scale of Operations, 2021 to 2036
- Table 46: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 47: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: South Asia and Pacific Market Value (USD Million) Forecast by Project Type, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Revenue Stream, 2021 to 2036
- Table 50: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 51: Middle East & Africa Market Value (USD Million) Forecast by Product , 2021 to 2036
- Table 52: Middle East & Africa Market Value (USD Million) Forecast by Scale of Operations, 2021 to 2036
- Table 53: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 54: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 55: Middle East & Africa Market Value (USD Million) Forecast by Project Type, 2021 to 2036
- Table 56: Middle East & Africa Market Value (USD Million) Forecast by Revenue Stream, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product
- Figure 6: Global Market Value Share and BPS Analysis by Scale of Operations, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Scale of Operations, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Scale of Operations
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Project Type, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Project Type, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Project Type
- Figure 18: Global Market Value Share and BPS Analysis by Revenue Stream, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Revenue Stream, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Revenue Stream
- Figure 21: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 23: Global Market Attractiveness Analysis by Region
- Figure 24: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 29: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 30: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 31: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 32: North America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Product
- Figure 35: North America Market Value Share and BPS Analysis by Scale of Operations, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Scale of Operations, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Scale of Operations
- Figure 38: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Application
- Figure 41: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by End User
- Figure 44: North America Market Value Share and BPS Analysis by Project Type, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Project Type, 2026-2036
- Figure 46: North America Market Attractiveness Analysis by Project Type
- Figure 47: North America Market Value Share and BPS Analysis by Revenue Stream, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Revenue Stream, 2026-2036
- Figure 49: North America Market Attractiveness Analysis by Revenue Stream
- Figure 50: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 51: Latin America Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Product
- Figure 54: Latin America Market Value Share and BPS Analysis by Scale of Operations, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Scale of Operations, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Scale of Operations
- Figure 57: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Application
- Figure 60: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 62: Latin America Market Attractiveness Analysis by End User
- Figure 63: Latin America Market Value Share and BPS Analysis by Project Type, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Project Type, 2026-2036
- Figure 65: Latin America Market Attractiveness Analysis by Project Type
- Figure 66: Latin America Market Value Share and BPS Analysis by Revenue Stream, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Revenue Stream, 2026-2036
- Figure 68: Latin America Market Attractiveness Analysis by Revenue Stream
- Figure 69: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 70: Western Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Product
- Figure 73: Western Europe Market Value Share and BPS Analysis by Scale of Operations, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Scale of Operations, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Scale of Operations
- Figure 76: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 77: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 78: Western Europe Market Attractiveness Analysis by Application
- Figure 79: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 81: Western Europe Market Attractiveness Analysis by End User
- Figure 82: Western Europe Market Value Share and BPS Analysis by Project Type, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Project Type, 2026-2036
- Figure 84: Western Europe Market Attractiveness Analysis by Project Type
- Figure 85: Western Europe Market Value Share and BPS Analysis by Revenue Stream, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Revenue Stream, 2026-2036
- Figure 87: Western Europe Market Attractiveness Analysis by Revenue Stream
- Figure 88: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Product
- Figure 92: Eastern Europe Market Value Share and BPS Analysis by Scale of Operations, 2026 and 2036
- Figure 93: Eastern Europe Market Y-o-Y Growth Comparison by Scale of Operations, 2026-2036
- Figure 94: Eastern Europe Market Attractiveness Analysis by Scale of Operations
- Figure 95: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 96: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 97: Eastern Europe Market Attractiveness Analysis by Application
- Figure 98: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 99: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 100: Eastern Europe Market Attractiveness Analysis by End User
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Project Type, 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Project Type, 2026-2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Project Type
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Revenue Stream, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Revenue Stream, 2026-2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Revenue Stream
- Figure 107: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 108: East Asia Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 109: East Asia Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 110: East Asia Market Attractiveness Analysis by Product
- Figure 111: East Asia Market Value Share and BPS Analysis by Scale of Operations, 2026 and 2036
- Figure 112: East Asia Market Y-o-Y Growth Comparison by Scale of Operations, 2026-2036
- Figure 113: East Asia Market Attractiveness Analysis by Scale of Operations
- Figure 114: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 115: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 116: East Asia Market Attractiveness Analysis by Application
- Figure 117: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 118: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 119: East Asia Market Attractiveness Analysis by End User
- Figure 120: East Asia Market Value Share and BPS Analysis by Project Type, 2026 and 2036
- Figure 121: East Asia Market Y-o-Y Growth Comparison by Project Type, 2026-2036
- Figure 122: East Asia Market Attractiveness Analysis by Project Type
- Figure 123: East Asia Market Value Share and BPS Analysis by Revenue Stream, 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Revenue Stream, 2026-2036
- Figure 125: East Asia Market Attractiveness Analysis by Revenue Stream
- Figure 126: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 127: South Asia and Pacific Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 128: South Asia and Pacific Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 129: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 130: South Asia and Pacific Market Value Share and BPS Analysis by Scale of Operations, 2026 and 2036
- Figure 131: South Asia and Pacific Market Y-o-Y Growth Comparison by Scale of Operations, 2026-2036
- Figure 132: South Asia and Pacific Market Attractiveness Analysis by Scale of Operations
- Figure 133: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 134: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 135: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 136: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 137: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 138: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 139: South Asia and Pacific Market Value Share and BPS Analysis by Project Type, 2026 and 2036
- Figure 140: South Asia and Pacific Market Y-o-Y Growth Comparison by Project Type, 2026-2036
- Figure 141: South Asia and Pacific Market Attractiveness Analysis by Project Type
- Figure 142: South Asia and Pacific Market Value Share and BPS Analysis by Revenue Stream, 2026 and 2036
- Figure 143: South Asia and Pacific Market Y-o-Y Growth Comparison by Revenue Stream, 2026-2036
- Figure 144: South Asia and Pacific Market Attractiveness Analysis by Revenue Stream
- Figure 145: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 146: Middle East & Africa Market Value Share and BPS Analysis by Product , 2026 and 2036
- Figure 147: Middle East & Africa Market Y-o-Y Growth Comparison by Product , 2026-2036
- Figure 148: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 149: Middle East & Africa Market Value Share and BPS Analysis by Scale of Operations, 2026 and 2036
- Figure 150: Middle East & Africa Market Y-o-Y Growth Comparison by Scale of Operations, 2026-2036
- Figure 151: Middle East & Africa Market Attractiveness Analysis by Scale of Operations
- Figure 152: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 153: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 154: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 155: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 156: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 157: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 158: Middle East & Africa Market Value Share and BPS Analysis by Project Type, 2026 and 2036
- Figure 159: Middle East & Africa Market Y-o-Y Growth Comparison by Project Type, 2026-2036
- Figure 160: Middle East & Africa Market Attractiveness Analysis by Project Type
- Figure 161: Middle East & Africa Market Value Share and BPS Analysis by Revenue Stream, 2026 and 2036
- Figure 162: Middle East & Africa Market Y-o-Y Growth Comparison by Revenue Stream, 2026-2036
- Figure 163: Middle East & Africa Market Attractiveness Analysis by Revenue Stream
- Figure 164: Global Market - Tier Structure Analysis
- Figure 165: Global Market - Company Share Analysis

