Biologics Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
The Biologics market is segmented by Drug Class (Monoclonal Antibodies, Recombinant Hormones/Proteins, Vaccines, Cellular Based Biologics, Gene-Based Biologics, Therapeutic Enzymes, Others), Application (Infectious Diseases, Cancer, Autoimmune Diseases, Rare Diseases, Others), Manufacturing Type (Contract Manufacturing, In-House Manufacturing), Drug Classification (Branded Drugs, Generic Drugs), Mode of Purchase (Prescription Drugs, Over-the-Counter Drugs), Source (Bacterial Cells, Yeast Cells, Plant Cells, Animal Cells, Others), and Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies). Forecast for 2025 to 2035.
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Biologics Market Size, Market Forecast and Outlook By FMI
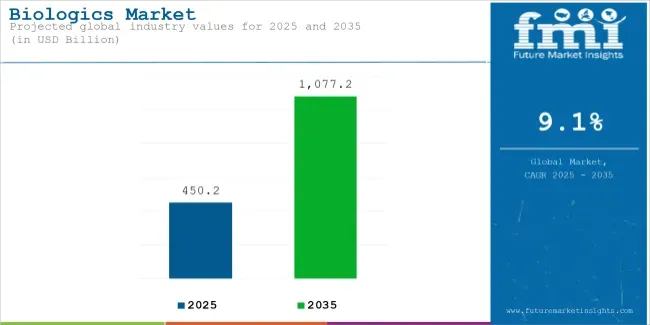
Biologics market value is expected to increase from USD 450.2 billion in 2025 to USD 1,077.2 billion by 2035. Market expansion is anticipated to register a 9.1% CAGR during the forecast period. Monoclonal antibodies are likely to lead drug class demand with 65.1% share in 2025. Oncology is projected to account for 32.4% of application demand in 2025.
Summary of the Biologics Market
- Demand and Growth Drivers
- Rising chronic disease burden is expected to increase demand for targeted therapies across cancer care and autoimmune treatment.
- Expedited approvals are likely to improve patient access to biologics used for serious and rare conditions.
- Better clinical outcomes are anticipated to lift adoption among physicians seeking precise therapies with stronger treatment response.
- Product and Segment View
- Monoclonal antibodies are expected to lead the drug class segment as clinical use broadens across high-value therapies.
- Oncology is likely to stay the largest application area as targeted biologics gain use in cancer treatment plans.
- Hospital pharmacies are anticipated to handle large biologic volumes as complex therapies require controlled storage and administration.
- Geography and Competitive Outlook
- India is expected to record the fastest country-level expansion as access improves across specialty biologic therapies.
- China is likely to see strong adoption as cell therapy investment and domestic innovation increase.
- Larger manufacturers are anticipated to strengthen their position through pipeline depth and biomanufacturing scale.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant for Healthcare at FMI, suggests, “Biologics market expansion is expected to favor companies able to manage clinical innovation and reliable manufacturing together. Firms with strong antibody portfolios and advanced biologic platforms are likely to gain advantage as hospitals seek proven therapies with steady supply.”
- Biologics Market Value Analysis
- Biologics market is shifting into a core pharmaceutical revenue area led by targeted therapies and advanced drug platforms.
- Demand is likely to rise as oncology and autoimmune care use biologics across broader patient groups.
- Adoption is expected to gain pace as approvals improve and healthcare systems prioritize high-efficacy treatment options.
- Spending is anticipated to increase with stronger biologic pipelines and larger investments in GMP-ready production capacity.
The biologics market has maintained strong momentum as biopharmaceutical innovation and targeted therapies reshape the treatment landscape across oncology, immunology, and rare diseases. Rising prevalence of chronic conditions, favorable reimbursement policies, and accelerated regulatory pathways have reinforced widespread adoption.
Biologics, including monoclonal antibodies, recombinant proteins, and cell-based therapies, have demonstrated superior efficacy and specificity, driving demand among clinicians and patients seeking improved outcomes over conventional small-molecule drugs. Manufacturing capabilities have expanded with investments in single-use bioprocessing, modular facilities, and advanced analytics to ensure scalable, GMP-compliant production. Strategic collaborations between biotech innovators and established pharmaceutical companies have supported robust pipelines and global commercialization
Semi Annual Market Update
The global biologics market's compound annual growth rate (CAGR) for the first half of 2024 and 2025 is compared in the table below. This analysis provides important insights into the performance of the industry by highlighting significant shifts and trends in revenue generation.
The first half (H1) is the period from January to June, and the second half (H2) is July to December. In the first half (H1) of the decade from 2024 to 2034, the business is predicted to surge at a CAGR of 10.3%, followed by a slightly lower growth rate of 9.8% in the second half (H2) of the same decade.
| Particular | Value CAGR |
|---|---|
| H1 (2024 to 2034) | 10.3% |
| H2 (2024 to 2034) | 9.8% |
| H1 (2025 to 2035) | 9.1% |
| H2 (2025 to 2035) | 8.8% |
Moving into the subsequent period, from H1 2025 to H2 2035, the CAGR is projected to decrease slightly to 9.1% in the first half and remain relatively moderate at 8.8% in the second half. In the first half (H1) the industry witnessed a decrease of 120 BPS while in the second half (H2), the industry witnessed a decrease of 100 BPS.
Analyzing Biologics Market by Top Investment Segments
Monoclonal Antibodies Lead the Market with Proven Efficacy Across Multiple High-Value Therapeutic Areas
Drug Class Analysis: Monoclonal Antibodies
Monoclonal Antibodies sales contributes a revenue share of 65.1% due their prominence as the backbone of modern biologics portfolios. Utilization has been driven by their proven ability to selectively target disease-specific antigens, enabling higher response rates and improved safety profiles relative to conventional treatments. Regulatory approvals have endorsed monoclonal antibodies for oncology, autoimmune disorders, and infectious diseases, reinforcing clinician confidence and adoption.
Advances in antibody engineering, including humanization and bispecific formats, have expanded therapeutic applications and enhanced clinical outcomes. Biomanufacturers have invested heavily in production capacity and process optimization to meet rising demand and ensure supply continuity. These factors have established monoclonal antibodies as the dominant drug class, with sustained growth expected as innovation and market penetration increase globally.
Oncology Dominates the Market with High Adoption of Targeted Biologics and Immunotherapies
Oncology segments holds a revenue share of 32.4% which has been attributed to the segment’s critical role in driving biologics adoption. Utilization has been driven by the increasing incidence of cancer worldwide and the urgent need for therapies that improve survival while minimizing toxicity.
Biologics have been integrated into first-line and adjuvant treatment protocols, supported by strong clinical evidence demonstrating superior outcomes over chemotherapy alone. Advances in precision medicine, biomarker-guided therapy selection, and immuno-oncology approaches have further expanded the role of biologics in cancer care.
Regulatory bodies have prioritized expedited approvals and expanded indications for innovative oncology biologics. Hence, oncology as the leading application segment, with continued growth expected as research pipelines advance and patient access improves.
Hospital Pharmacies Anchor the Market with Centralized Dispensing and Support for Complex Biologic Therapies
Hospital pharmacies have accounted for 48.2% of market revenue, driven by their role as primary distribution channels for biologics requiring specialized handling, storage, and administration. The segment is driven by the complexity of biologic therapies, which often require infusion services, cold chain logistics, and pharmacist oversight to ensure patient safety and adherence.
Hospitals have prioritized investments in biologics management programs, including electronic prescribing, tracking systems, and clinician training. Reimbursement frameworks in many regions have incentivized hospital-based delivery of high-cost biologics, supporting consistent volume growth.
Key Industry Highlights
Rising prevalence of chronic diseases is driving the market growth
Chronic diseases, such as cancer, diabetes, and autoimmune disorders, require long-term management and are often related to complex biological pathways that traditional therapies cannot effectively manage. Biologics are targeted to act upon these specific molecules or pathways, thus providing a more precise and effective approach toward the treatment of these conditions.
For instance, according to the World Health Organization (WHO), Cancer is a leading cause of death worldwide, accounting for nearly 10 million deaths in 2020, or nearly one in six deaths
Unlike traditional therapies, biologics are designed to interface with the body's normal processes to provide treatments complementary to the underlying causes of chronic diseases and are not just symptomatic. This targeted approach enhances therapeutic outcomes while reducing the likelihood of adverse effects and makes biologics a preferred choice among healthcare providers and patients.
Besides, increasing chronic diseases burdens have put a greater emphasis on treatment options through innovation. The solution has come to be biologics and provided a way to solve unmet medical needs.
Increasing awareness among physicians and patients regarding its benefits has increased demand for, and hence driving, the biologics market. This trend is indicative of the critical role biologics are playing to change how chronic diseases will be managed and in the future of healthcare.
Advent of next-generation biologics is driving the industry growth
The scope and potential of the biologics market is widened with the advent of next-generation biologics. Indeed, newer treatments like antibody-drug conjugates, bispecific antibodies, and fusion proteins are already shifting the paradigms of treatment with much higher efficacy, precision, and diversity than their predecessors.
An example of this is antibody-drug conjugates or ADCs, which combine the specificity of monoclonal antibodies with the potent cytotoxicity of small molecule drugs, thus allowing for the targeted killing of cancer cells without destroying healthy tissues. So far, these treatments have proven highly effective in oncology, hence the investments in ADC development.
Bispecific antibodies, which can bind two different antigens simultaneously, open new mechanisms of action, including engaging immune cells to directly attack tumors. They are gaining ground in oncology and immunology as a way of addressing complex disease pathways.
Emerging options in conditions as diverse as hemophilia or immune-mediated disease benefit from newer possibilities in therapy derived from so-called fusion proteins where all the functionality from multiple biologics could potentially be addressed simultaneously.
As scalable and potentially economic, future success will only expand further driven by the strength gained from development work in variously improved bioprocessing techniques alongside engineering advancement to support more promising next-generation products.
Increased government incentives for rare disease therapies creates further growth opportunity
Government incentives for rare diseases' therapies are one good opportunity in the biologics market. Such policies as the USA orphan drug act, like others it has with other countries, were enacted to stimulate the development of treatments for rare and underserved conditions.
These incentives provided are tax credits in form of cost of clinical trials, grants for research as well as the waiver of application fees, and extended market exclusivity at approval.
The biologics sector, particularly addresses rare diseases because many diseases have genetic or molecular origins; monoclonal antibodies, gene therapies, and recombinant proteins have provided hope in the cure of conditions as diverse as Duchenne muscular dystrophy, cystic fibrosis, and rare cancers.
Globally, all markets outside of the United States such as the European Medicines Agency (EMA) and Japan's Pharmaceuticals and Medical Devices Agency (PMDA) had agreed on similar frameworks.
For biopharmaceutical companies, these incentives reduce financial risk and provide lucrative opportunities in niche markets, thereby fostering innovation and encouraging investment in biologics tailored for rare and orphan diseases. Increased rare diseases, with better diagnostic ability, are putting a greater demand on targeted biologic therapies.
High cost of biologics may restrict market growth
High cost of biologics is the major restricting factor in the global biologics market, which often prevents their accessibility and adoption-mainly in low-income and some middle-income countries. In contrast to the traditional small-molecule drugs, biologics require more complex biotechnological processes, skilled manufacturing facilities, and strict controls over their quality.
Consequently, it costs much to produce biologics and makes them many times costlier than the traditional therapy. Research and development (R&D) of biologics also comes at great expense, including considerable direct clinical trials and regulatory approval which add to the expenditure.
Biologics also require cold chain logistics, where the product remains stable during distribution, adding to their costs. These high costs make biologics unaffordable for patients and healthcare systems, especially in resource-poor settings where insurance coverage is either minimal or nonexistent. Even in high-income countries, the cost of biologics is a burden on healthcare budgets, which can result in restrictive reimbursement policies.
However, biosimilars are quite cheaper and hence not sufficient in filling the gap of access most parts of the world. Therefore, manufacturing innovations, government subsidies, or differential pricing strategies will be the keys to widening access to biologics and realizing their potential market.
Market Concentration
Tier 1 companies are the industry leaders with 45.2% of the global industry. These companies stand out for having a large product portfolio and a high production capacity.
These industry leaders also stand out for having a wide geographic reach, a strong customer base, and substantial experience in manufacturing and having enough financial resources, which enables them to enhance their research and development efforts and expand into new industries.
The companies within tier 1 have a good reputation and high brand value. These companies frequently get involved in strategies such as acquisition and product launches. Prominent companies within tier 1 include Amgen, Inc., Eli Lilly and Company, GlaxoSmithKline PLC and AbbVie Inc.
Tier 2 companies are relatively smaller as compared with tier 1 players. The tier 2 companies hold a market share of 29.1% worldwide. These firms may not have cutting-edge technology or a broad global reach, but they do ensure regulatory compliance and have good technology.
The players are more competitive when it comes to pricing and target niche markets. Key Companies under this category include F. Hoffmann-La Roche Ltd, Novartis AG, Pfizer Inc. and AstraZeneca.
Compared to Tiers 1 and 2, Tier 3 companies offer biologics, but with smaller revenue spouts and less influence. These companies mostly operate in one or two countries and have limited customer base. The companies such as Bayer AG, and others falls under tier 3 category. They specialize in specific products and cater to niche markets, adding diversity to the industry.
Country-wise Insights
The market analysis for biologics in various nations is covered in the section below. An analysis of important nations in North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and Middle East & Africa of the world has been mentioned below.
It is projected that the United States will maintain its leading position in North America through 2035, holding a value share of 88.9%. By 2035, China is expected to experience a CAGR of 9.0% in the Asia-Pacific region.
| Countries | Value CAGR (2025 to 2035) |
|---|---|
| United States | 3.7% |
| Germany | 3.8% |
| France | 4.7% |
| UK | 4.1% |
| China | 9.0% |
| India | 9.5% |
| South Korea | 8.7% |
Growing use of Artificial Intelligence in Biologic Development is driving growth on Germany
Germany’s biologics market is poised to exhibit a CAGR of 3.8% between 2025 and 2035. The Germany holds highest market share in European market.
AI in biologic development is a significant growth factor in the German biologics market. Artificial intelligence technologies are changing the ways of identification, development, and optimization of biologics while accelerating R&D.
In Germany, where much focus lies on innovative and state-of-the-art technologies in the pharmaceutical industry, AI is increasingly being used to identify novel therapeutic targets, predict protein structures, and streamline drug design.
AI-powered tools grant the opportunity for more effective analysis of big biological data, which may lead to biomarker identification, optimization of clinical trial designs, and personalized treatments. This would decrease the overall time and expenses spent on new biologic development and enhance the capability of the market to meet the growing demand by the use of innovative therapies.
Coupled with AI, Germany has a strong biotechnology ecosystem that positions it as an international leader in the development of biologics. AI accelerates the discovery of new biologic therapies, enhances treatment efficiency and effectiveness, thereby driving growth within Germany's biologics market.
Favorable regulatory approvals is a growth factor in the United States
United States is anticipated to show a CAGR of 3.7% between 2025 and 2035.
Favorable regulatory approvals are one of the major growth drivers for the biologics market in the United States. The USA FDA offers expedited approval pathways, which include the breakthrough therapy designation, accelerated approval, and priority review programs, to expedite promising biologic therapies.
These programs are implemented with an aim of speeding up the development and approval of drugs in respect of significant potential in the treatment of serious conditions, especially where there are limited or no existing treatments.
For biologics, it means sooner speed to the market for therapies targeted to diseases like cancer, autoimmune disorders, and rare genetic conditions. The flexible regulatory framework that the FDA offers encourages innovation and investment because companies can bring life-saving treatments to patients more rapidly.
Moreover, biotech companies engage closely with the FDA to assure that scientific achievements in biologics, gene and cell therapy, are adapted efficiently into health care. All these factors foster a favorable environment for the regulations, which expedites penetration into the markets and fuels further growth in the USA market.
Increased investments for research and development for biologics in China drives the market
China is anticipated to show a CAGR of 9.0% between 2025 and 2035.
The increasing adoption of cell and gene therapies acts as a major growth factor in the biologics market of China. Innovations in the use of breakthrough gene-editing technologies like CRISPR, together with new developments around CAR-T-cell therapies, present an ever-changing treatment paradigm-very significantly within oncology and for conditions considered genetic in nature.
These revolutionary treatments have begun to speedily take shape and gain swift acceptance with advances in China's healthcare structure and government-friendly policies.
All this has created a very good environment for both domestic and international companies to develop and bring innovative biologics to the market. Besides, China's huge patient population and the high incidence of diseases such as cancer and genetic disorders create a huge demand for personalized treatments.
As cell and gene therapies continue to evolve, the increasing usage of biologics in clinical practice within China propels the market forward. The ability to offer curative options for previously untreatable diseases will further accelerate this growth, making China a vital part of the development of biologics worldwide.
Competition Outlook
The competitive landscape has been shaped by companies advancing monoclonal antibodies, biosimilars, and novel biologic modalities to address unmet clinical needs. Leading manufacturers have prioritized strategic collaborations to accelerate pipeline development and secure global commercialization rights.
Investments in advanced biomanufacturing technologies and supply chain expansion have strengthened production capacity. Educational initiatives and patient access programs have been launched to support adoption and adherence. These activities are expected to drive continued innovation and sustain competitive differentiation as biologics uptake increases worldwide.
Key Development:
- In 2025, Abeona Therapeutics announced FDA approval for ZEVASKYN™ (pz-cel), making it the first and only autologous cell-based gene therapy for treating wounds in recessive dystrophic epidermolysis bullosa (RDEB) patients. This single-application treatment offers a significant breakthrough for the serious genetic skin disease, which currently has no cure.
- In 2025, Biocon Biologics Ltd (BBL) announced that the USA FDA has approved Jobevne™ (bevacizumab-nwgd), a biosimilar to Avastin®. This recombinant humanized monoclonal antibody is a VEGF inhibitor that works by restricting blood supply to tumors, and it's approved for intravenous use in treating several different types of cancer. Companies:
Key Players of Biologics Industry
- Amgen, Inc.
- Eli Lilly and Company
- GlaxoSmithKline PLC
- AbbVie Inc.
- Novartis AG
- Pfizer Inc
- Sanofi S.A.
- Novavax Inc.
- Moderna TX, Inc.
- Biocon Limited.
Key Segments of Biologics Industry
By Drug Class:
In terms of drug class, the industry is divided into monoclonal antibodies, recombinant hormones/proteins, vaccines, cellular based biologics, gene-based biologics, therapeutic enzymes and others.
By Application:
In terms of application, the industry is segregated into infectious diseases, cancer, autoimmune diseases, rare diseases, and others
By Manufacturing Type:
In terms of manufacturing type, the industry is divided into contract manufacturing and in-house manufacturing.
By Drug Classification:
In drug classification, the industry is segregated into branded drugs and generic drugs
By Mode of Purchase:
In terms of mode of purchase, the industry is divided into prescription drugs and over-the-counter (OTC) drugs.
By Source:
In terms of source, the industry is segregated into bacterial cells, yeast cells, plant cells, animal cells and others source
By Distribution Channel:
In terms of distribution channel, the industry is divided into hospital pharmacies, retail pharmacies and online pharmacies.
By Region:
Key countries of North America, Latin America, East Asia, South Asia & Pacific, Western Europe, Eastern Europe and Middle East and Africa (MEA) have been covered in the report.
Frequently Asked Questions
What is the future of global biologics industry?
The global biologics industry is projected to witness CAGR of 9.1% between 2025 and 2035.
What was the worth of the global biologics industry in 2024?
The global biologics industry stood at USD 417.1 billion in 2024.
What will the worth of global biologics industry by 2035 end?
The global biologics industry is anticipated to reach USD 1,077.2 billion by 2035 end.
What is the expected CAGR for China during forecast period?
China is expected to show a CAGR of 9.0% in the assessment period.
Who are the key manufacturer of global biologics industry?
The key players operating in the global biologics industry Amgen, Inc., Eli Lilly and Company, GlaxoSmithKline PLC, AbbVie Inc., F. Hoffmann- La Roche AG, Novartis AG, Pfizer Inc, AstraZeneca, Bayer AG, Merck & Co. Inc., Others.
Table of Content
- Executive Summary
- Industry Introduction, including Taxonomy and Market Definition
- Market Trends and Success Factors, including Macro-economic Factors, Market Dynamics, and Recent Industry Developments
- Global Market Demand Analysis 2020 to 2024 and Forecast 2025 to 2035, including Historical Analysis and Future Projections
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035
- By Drug Class
- By Application
- By Manufacturing Type
- By Drug Classification
- By Mode of Purchase
- By Source
- By Distribution Channel
- By Region
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Drug Class
- Monoclonal Antibodies
- Recombinant Hormones/Proteins
- Vaccines
- Cellular Based Biologics
- Gene-Based Biologics
- Therapeutic Enzymes
- Others
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application
- Infectious Diseases
- Cancer
- Autoimmune Diseases
- Rare Diseases
- Others
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Manufacturing Type
- Contract Manufacturing
- In-house Manufacturing
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Drug Classification
- Branded Drugs
- Generic Drugs
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Mode of Purchase
- Prescription Drugs
- Over-The-Counter (OTC) Drugs
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, by Source
- Bacterial Cells
- Yeast Cells
- Plant Cells
- Drug Classification Cells
- Others Source
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Distribution Channel
- Hospital Pharmacies
- Retail Pharmacies
- Online Pharmacies
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, by Region
- North America
- Latin America
- East Asia
- South Asia and Pacific
- Western Europe
- Eastern Europe
- Middle East and Africa
- North America Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Latin America Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- East Asia Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- South Asia & Pacific Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Western Europe Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Eastern Europe Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Middle East and Africa Sales Analysis 2020 to 2024 and Forecast 2025 to 2035, by Key Segments and Countries
- Sales Forecast 2025 to 2035 by drug class, by application, by manufacturing type, by drug classification, by mode of purchase, by source, and by distribution channel for 30 Countries
- Competition Outlook, including Market Structure Analysis, Company Share Analysis by Key Players, and Competition Dashboard
- Company Profile
- Amgen, Inc.
- Eli Lilly and Company
- GlaxoSmithKline PLC
- AbbVie Inc.
- Novartis AG
- Pfizer Inc
- Sanofi S.A.
- Novavax Inc.
- Moderna TX, Inc.
- Biocon Limited.
- Others

