Respiratory Biologics Market
A comprehensive overview of the respiratory biologics market is delivered in this report, featuring market size insights, revenue projections, competitive assessment, demand evaluation, growth propellers, restraining factors, Disease Indication trends, Route of Administration trends, Sales Channel trends, supply chain analysis, and strategic opportunities ahead.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Respiratory Biologics Market Size, Market Forecast and Outlook By FMI
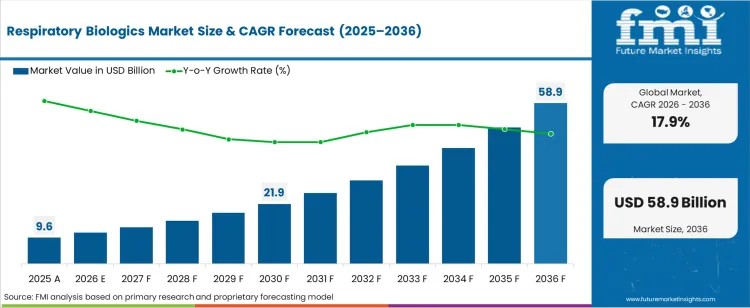
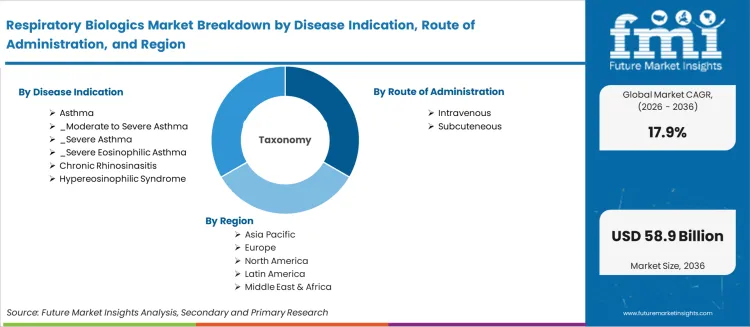
The respiratory biologics market was valued at USD 9.62 billion in 2025, projected to reach USD 11.34 billion in 2026, and is forecast to expand to USD 58.86 billion by 2036 at a 17.9% CAGR. Regulatory approvals for monoclonal antibody therapies targeting IL-5, IL-4/IL-13, and IgE pathways are expanding treatment options for patients with severe eosinophilic asthma, chronic rhinosinusitis with nasal polyps, and hypereosinophilic syndrome. Intravenous administration accounts for 67.0% of the route of administration share as hospital-based infusion centres manage the majority of biologic therapy initiations.
Summary of Respiratory Biologics Market
- Market Snapshot
- The respiratory biologics market is valued at USD 9.62 billion in 2025 and is projected to reach USD 58.86 billion by 2036.
- The industry is expected to grow at a 17.9% CAGR from 2026 to 2036, creating an incremental opportunity of USD 47.52 billion.
- The market is a high-growth specialty pharmaceutical category where clinical indication breadth, reimbursement access, and prescriber network expansion define competitive positioning.
- Demand and Growth Drivers
- Regulatory approvals expanding approved indications for existing respiratory biologics are increasing the prescribable patient population beyond specialist asthma centres into general pulmonology practices.
- Entry of biosimilar alternatives for early-generation respiratory biologics is broadening patient access by reducing per-treatment costs in price-sensitive healthcare systems.
- Growing clinical evidence for biologic therapy in chronic rhinosinusitis with nasal polyps is creating a new indication pathway that expands the addressable market beyond asthma alone.
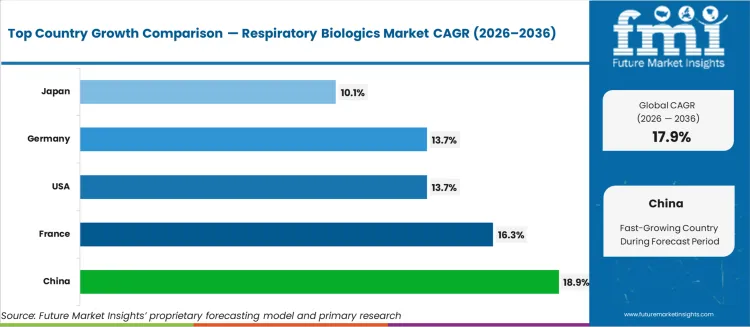
- Among key countries, China at 18.9%, France at 16.3%, USA at 13.7%, Germany at 13.7%, Japan at 10.1%.
- Product and Segment View
- The market includes anti-IgE, anti-IL-5, anti-IL-4/IL-13, and anti-TSLP monoclonal antibody therapies for severe asthma, chronic rhinosinusitis, and hypereosinophilic syndrome.
- Intravenous leads by Route of Administration with 67% share in 2026.
- Hospitals leads by Sales Channel with 44% share in 2026.
- Scope includes market scope includes anti-ige, anti-il-5, anti-il-5r, anti-il-4/il-13, and anti-tslp monoclonal antibody therapies for respiratory indications. Small-molecule respiratory drugs including inhaled corticosteroids and bronchodilators are excluded.
- Geography and Competitive Outlook
- China and France are the fastest-growing markets at 18.9% and 16.3% CAGR respectively, driven by expanding regulatory approvals for biologic therapies and reimbursement coverage expansion.
- USA represents a mature demand base where established biologics prescribing infrastructure and broad insurance coverage sustain respiratory biologic adoption.
- Competition is shaped by clinical pipeline depth, indication breadth, reimbursement access, and prescriber education support, with key players including GSK plc., AstraZeneca, Sanofi, Regeneron Pharmaceuticals, Inc., Genentech USA, Inc., Teva Respiratory, LLC., Novartis Pharmaceuticals Corporation.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare, opines: 'In my analysis, I have observed that respiratory biologics are transitioning from specialist-only prescribing to broader pulmonology practice adoption as clinical evidence accumulates and reimbursement coverage expands. Pharmaceutical companies maintaining launch strategies focused exclusively on academic medical centres face slower market penetration as community pulmonologists gain prescribing confidence. Distribution partners that fail to develop cold-chain logistics capability for biologic therapies will be excluded from the growing specialty pharmacy fulfilment market.'
- Strategic Implications / Executive Takeaways
- Pharmaceutical companies must expand respiratory biologic launch strategies beyond specialist academic centres to include community pulmonology practices where prescribing volume growth is concentrating.
- Payer organizations must develop reimbursement frameworks that accommodate the higher per-patient costs of biologic therapies while capturing the downstream healthcare cost reductions from reduced exacerbation rates.
- Specialty pharmacy networks must build cold-chain logistics capability and patient support programmes to serve the growing demand for biologic therapy fulfilment outside hospital infusion centres.
The incremental opportunity of USD 47.52 billion over the forecast period reflects both expanding approved indications for existing biologics and the entry of biosimilar alternatives that broaden patient access. Hospitals account for 44.0% of sales channel share as biologic therapy initiation and dose titration require clinical supervision, infusion infrastructure, and adverse event monitoring capabilities.
China leads at 18.9% CAGR, driven by NMPA approvals for biologic therapies and expanding reimbursement coverage for severe asthma biologics under the National Reimbursement Drug List. France follows at 16.3%, supported by early access programmes for biologic therapies and comprehensive social security reimbursement for approved respiratory biologics. The United States expands at 13.7%, driven by the established biologics prescribing infrastructure and broad commercial insurance coverage for FDA-approved respiratory monoclonal antibodies. Germany registers 13.7%, anchored by early EMA access and structured reimbursement through the statutory health insurance system for biologic therapies in severe respiratory conditions. Japan registers 10.1% growth as PMDA approvals for respiratory biologics expand and the national health insurance system covers an increasing range of monoclonal antibody therapies for severe asthma.
Respiratory Biologics Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 11.34 billion |
| Industry Value (2036) | USD 58.86 billion |
| CAGR (2026-2036) | 17.9% |
Source: Future Market Insights, 2026
Respiratory Biologics Market Definition
The respiratory biologics market encompasses monoclonal antibody and biologic therapies approved for the treatment of severe respiratory conditions including moderate-to-severe asthma, chronic rhinosinusitis with nasal polyps, and hypereosinophilic syndrome. Products include anti-IgE, anti-IL-5, anti-IL-4/IL-13, and anti-TSLP monoclonal antibodies administered via intravenous and subcutaneous routes through hospital infusion centres, specialty clinics, and pharmacy channels.
Respiratory Biologics Market Inclusions
Market scope includes anti-IgE, anti-IL-5, anti-IL-5R, anti-IL-4/IL-13, and anti-TSLP monoclonal antibody therapies for respiratory indications. Intravenous and subcutaneous formulations distributed through hospital, specialty clinic, retail, and mail order pharmacy channels are covered.
Respiratory Biologics Market Exclusions
Small-molecule respiratory drugs including inhaled corticosteroids and bronchodilators are excluded. The scope omits respiratory devices such as inhalers and nebulizers. Biosimilar products in development without regulatory approval fall outside analytical parameters.
Respiratory Biologics Market Research Methodology
- Primary Research: Analysts engaged with pulmonology department heads, specialty pharmacy managers, and biologic therapy infusion centre directors to map prescribing patterns and reimbursement access dynamics.
- Desk Research: Data collection phases aggregated regulatory approval databases, clinical trial registries, reimbursement policy publications, and biologic therapy prescribing volume statistics.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of procurement and installation tracking data, applying region-specific adoption curves to project future deployment velocity.
- Data Validation and Update Cycle: Projections are tested against publicly reported expenditure guidance from leading industry participants and cross-referenced with regulatory filing data.
Lucrative Opportunities Transforming the Respiratory Biologics Market
Raising awareness and comprehension of the molecular mechanisms underlying respiratory diseases opens up new opportunities for innovative biologics therapies. The rise of precision medicine, which emphasizes tailored therapeutic approaches, fits in well with respiratory biologics' targeted nature and opens up new development opportunities.
The growing elderly population which is more susceptible to respiratory ailments, drives market growth. Government programmes and policies that support healthcare improve accessibility would also push demand. Research & development, partnerships, and collaborations increase market potential and drive the introduction of new treatments.
Factors Restraining the Demand for Respiratory Biologics
Mid-sized companies need help in areas like experience, biologics manufacturing infrastructure, and capital, heightening the challenge. Nevertheless, these mid-sized players possess a distinctive advantage in their capacity for focused efforts, particularly within niche therapeutic areas and cutting-edge technologies.
The high cost of biologics therapies for respiratory diseases is set to restrict access for some patients, especially those without adequate insurance coverage or reimbursement programs. Respiratory biologics are limited by insurance coverage, formulary restrictions, and reimbursement policies, with payers imposing strict eligibility criteria for patients with severe disease or treatment failure.
Analysis of Top Countries Developing, Certifying, Regulating, and Using Respiratory Biologics
The table below shows the estimated growth rates of the five leading countries. China, France, and the United States are set to record high CAGRs of 18.9%, 16.3%, and 13.7%, respectively, through 2036.
.webp)
| Countries | Value CAGR (2026 to 2036) |
|---|---|
| United States | 13.7% |
| China | 18.9% |
| Japan | 10.1% |
| Germany | 13.7% |
| France | 16.3% |
Rising Patient and Healthcare Provider Awareness to Propel the United States
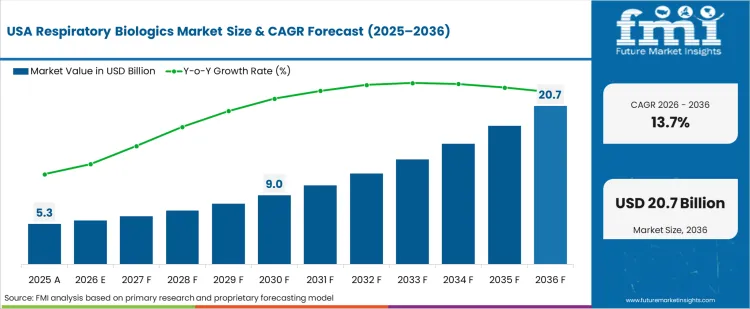
The United States is expected to exhibit a CAGR of 13.7% throughout the forecast period. Patient and healthcare provider awareness about the benefits of respiratory biologics therapy is growing. It is taking place through education initiatives, advocacy groups, and disease awareness campaigns, which is set to significantly boost market growth and treatment adherence.
The United States is home to several cutting-edge biotechnology and pharmaceutical enterprises. These companies make significant investments in research and development, which results in identifying and releasing innovative respiratory biologics.
Growing Aging Population to Uplift China
China is expected to grow at a CAGR of 18.9% by 2036. China is becoming a key force in the respiratory biologics market because of its rapidly developing healthcare infrastructure and rising awareness of respiratory disorders.
The demand for cutting-edge treatments is increasing due to the rising prevalence of diseases, including COPD and asthma. The market is also growing because of the government's efforts to improve healthcare access and concentrate on developing biotechnology.
China presents a sizable market for respiratory biologics due to its aging population and growing healthcare awareness. Both local and foreign companies are drawn to the area to take advantage of this potential.
Germany's Strong Healthcare System to Spur Market Growth
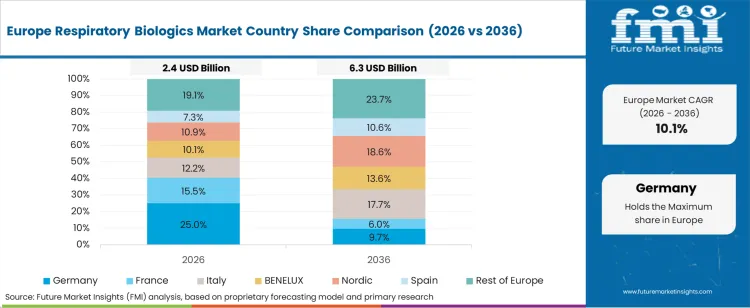
Germany is expected to surge at a CAGR of 13.7% during the forecast period. Germany's strong healthcare system, huge research resources, and focus on innovative treatments are driving the country's respiratory biologics industry.
With Germany's dedication to innovation and first-rate healthcare, the nation is well-positioned to advance respiratory biologics. Germany creates an atmosphere favorable to developing and using innovative biological medicines for respiratory disorders, propelling growth in this rapidly expanding healthcare industry.
Emergence of Affordable Treatments in Japan to Augment Sales
Japan is expected to surge at a CAGR of 10.1% by 2036. Japan faces a growing burden of respiratory conditions like asthma, COPD, and allergic rhinitis, necessitating the need for effective treatment options, including respiratory biologics.
Respiratory biologics in Japan offer targeted treatment options for severe or difficult-to-treat respiratory conditions, providing better symptom control and reducing exacerbations compared to traditional medications. Japan's respiratory biologics market is growing due to efforts to improve patient access and affordability, including patient assistance programs and negotiated pricing agreements.
Presence of Local and Foreign Firms in France to Propel Sales by 2036
France is projected to rise at 16.3% CAGR during the forecast period. The biotechnology sector in France is fostering innovation in respiratory biologics through research and development, resulting in novel molecules, improved delivery devices, and advanced diagnostic tools for personalized patient treatment.
The government is a key regulatory body responsible for overseeing respiratory biologics therapies' approval and post-market surveillance, significantly influencing their development timelines and commercialization strategies in France. France's respiratory biologics market faces fierce competition from pharmaceutical companies, influenced by factors like pricing strategies, market share, and product differentiation.
Respiratory Biologics Industry Analysis by Top Investment Segments
The section below shows the intravenous segment dominating by route of administration. It is set to hold a market share of 67.0% in 2026. Based on sales channels, the hospital segment is anticipated to generate a dominant share through 2024. It is likely to generate a share of 44.0% in 2026.
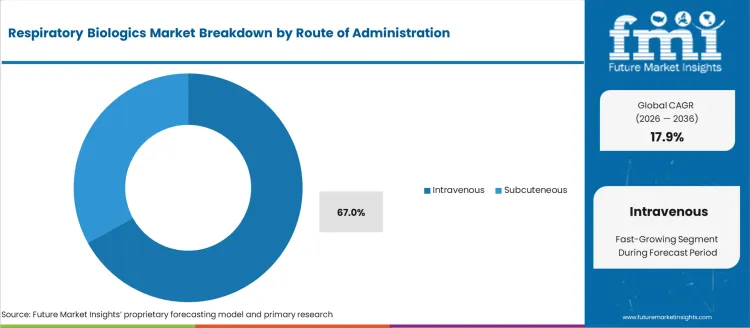
| Segment | Market Share (2026) |
|---|---|
| Intravenous (Route of Administration) | 67.0% |
| Hospital (Sales Channel) | 44.0% |
Demand for Respiratory Biologics to Remain High in Asthma Disease Indication
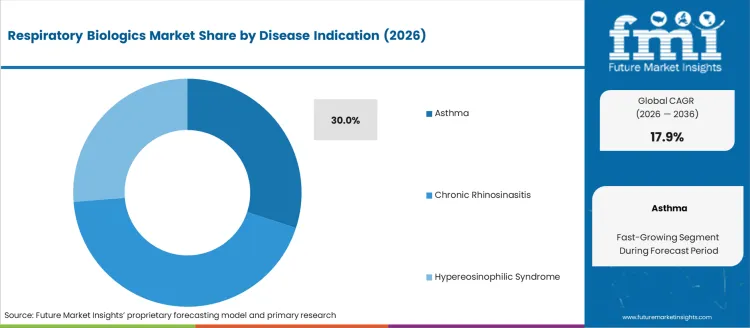
Respiratory biologics are in high demand because of the growing prevalence of asthma and its profound impact on patients' lives. These cutting-edge therapies effectively relieve asthma by addressing its underlying causes.
Respiratory biologics provide accurate solutions in response to the growing emphasis on individualized therapy, fueling the market's expansion. Owing to their special capacity to target particular pathways, biologics play a pivotal role in respiratory treatment, particularly given the intricate nature of managing asthma.
Hospital Segment to Lead by Sales Channel in the Market Through 2036
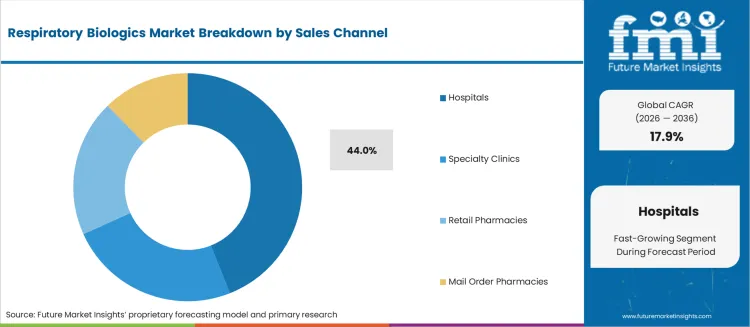
With their robust infrastructure and resources, hospitals are adept at delivering comprehensive respiratory care services. This includes the diagnosis, treatment, and management of respiratory conditions.
Specialized respiratory care units, dedicated pulmonary function testing labs, and teams of pulmonologists, respiratory therapists, and other healthcare professionals operate within these facilities. Their collective expertise is pivotal in effectively managing respiratory diseases, positioning hospitals as key contributors to the respiratory biologics market.
Key Respiratory Biologics Market Players and Concentration
Respiratory biologics companies are investing heavily in respiratory biologics research & development to advance scientific understanding, expand treatment options, and gain market competitiveness.
Leading Suppliers of Respiratory Biologics
- GSK plc.
- AstraZeneca
- Sanofi and Regeneron Pharmaceuticals, Inc.
- Genentech USA, Inc.
- Teva Respiratory, LLC.
- Novartis Pharmaceuticals Corporation
- GlaxoSmithKline
- Novartis
- Roche
- Boehringer Ingelheim
- Regeneron Pharmaceuticals
- Merck & Co.
- Johnson & Johnson
- Amgen
- Biogen
- Vertex Pharmaceuticals
- Genentech (a member of the Roche Group)
- AbbVie
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 11.34 billion to USD 58.86 billion, at a CAGR of 17.9% |
| Market Definition | The respiratory biologics market encompasses monoclonal antibody and biologic therapies approved for the treatment of severe respiratory conditions including moderate-to-severe asthma, chronic rhinosi... |
| Segmentation | Route of Administration: Intravenous, Subcutaneous; Sales Channel: Hospitals, Specialty Clinics, Retail Pharmacies, Mail Order Pharmacies |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | China, France, USA, Germany, Japan, and 40 plus countries |
| Key Companies Profiled | GSK plc., AstraZeneca, Sanofi, Regeneron Pharmaceuticals, Inc., Genentech USA, Inc., Teva Respiratory, LLC., Novartis Pharmaceuticals Corporation, Roche, Boehringer Ingelheim, Merck & Co., Johnson & Johnson, Amgen, Biogen, Vertex Pharmaceuticals, AbbVie |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with procurement tracking data and projecting adoption rates using region-specific deployment curves. |
Top Segments Studied in the Respiratory Biologics Market Report
By Disease Indication:
- Asthma
- Moderate to Severe Asthma
- Severe Asthma
- Severe Eosinophilic Asthma
- Chronic Rhinosinasitis
- Hypereosinophilic Syndrome
By Route of Administration:
- Intravenous
- Subcuteneous
By Sales Channel:
- Hospitals
- Specialty Clinics
- Retail Pharmacies
- Mail Order Pharmacies
By Region:
- North America
- Latin America
- East Asia
- South Asia & Pacific
- Western Europe
- Eastern Europe
- Middle East & Africa
Bibliography
- U.S. Food and Drug Administration. (2024). Biological products for respiratory indications approval database. FDA.
- European Medicines Agency. (2024). CHMP assessment reports for respiratory biologic therapies. EMA.
- National Medical Products Administration, China. (2024). Biologic therapy approval and reimbursement guidance for respiratory conditions. NMPA.
- National Institute for Health and Care Excellence. (2024). Technology appraisal guidance for respiratory biologics. NICE.
- World Health Organization. (2024). Global asthma report: Biologic therapy access and utilization. WHO.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for Respiratory Biologics Market in the global market in 2026?
Demand for respiratory biologics in the global market is estimated to be valued at USD 11.34 billion in 2026.
What will be the market size of Respiratory Biologics Market in the global market by 2036?
Market size for respiratory biologics is projected to reach USD 58.86 billion by 2036.
What is the expected demand growth for Respiratory Biologics Market between 2026 and 2036?
Demand for respiratory biologics is expected to grow at a CAGR of 17.9% between 2026 and 2036.
Which Route of Administration is poised to lead global sales by 2026?
Intravenous accounts for 67% share in 2026 as hospital-based infusion centres manage the majority of biologic therapy initiations requiring clinical supervision, dose titration, and adverse event monitoring.
How is the role of Hospitals in driving Respiratory Biologics Market adoption in 2026?
Hospitals represents 44% of Sales Channel share in 2026 as hospitals account for the highest concentration of specialist prescribers and infusion infrastructure for respiratory biologic administration.
What is driving demand in China?
China is projected to grow at a CAGR of 18.9% during 2026 to 2036, driven by NMPA approvals for biologic therapies and expanding reimbursement coverage for severe asthma biologics under the National Reimbursement Drug List.
What is Respiratory Biologics and what is it mainly used for?
Respiratory biologics are monoclonal antibody therapies targeting specific immune pathways in severe respiratory conditions. They are used to treat moderate-to-severe asthma, chronic rhinosinusitis, and hypereosinophilic syndrome in patients who do not respond adequately to conventional therapies.
How does FMI build and validate the Respiratory Biologics Market forecast?
Forecasting models apply a bottom-up methodology starting with biologic therapy prescription volume records and specialty pharmacy fulfilment tracking data and cross-validate projections against publicly reported expenditure data from leading industry participants.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Disease Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Disease Indication , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Disease Indication , 2026 to 2036
- Asthma
- Moderate to Severe Asthma
- Severe Asthma
- Severe Eosinophilic Asthma
- Chronic Rhinosinasitis
- Asthma
- Y to o to Y Growth Trend Analysis By Disease Indication , 2021 to 2025
- Absolute $ Opportunity Analysis By Disease Indication , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Route of Administration
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Route of Administration, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Route of Administration, 2026 to 2036
- Intravenous
- Subcuteneous
- Intravenous
- Y to o to Y Growth Trend Analysis By Route of Administration, 2021 to 2025
- Absolute $ Opportunity Analysis By Route of Administration, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Sales Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Sales Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Sales Channel, 2026 to 2036
- Hospitals
- Specialty Clinics
- Retail Pharmacies
- Mail Order Pharmacies
- Hospitals
- Y to o to Y Growth Trend Analysis By Sales Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Sales Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Disease Indication
- By Route of Administration
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Disease Indication
- By Route of Administration
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Disease Indication
- By Route of Administration
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Disease Indication
- By Route of Administration
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Disease Indication
- By Route of Administration
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Disease Indication
- By Route of Administration
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Disease Indication
- By Route of Administration
- By Sales Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Disease Indication
- By Route of Administration
- By Sales Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Disease Indication
- By Route of Administration
- By Sales Channel
- Competition Analysis
- Competition Deep Dive
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Disease Indication , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Disease Indication , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Disease Indication , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Disease Indication , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Disease Indication , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Disease Indication , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Disease Indication , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Disease Indication , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Route of Administration, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Sales Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Disease Indication , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Disease Indication , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Disease Indication
- Figure 6: Global Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Route of Administration
- Figure 9: Global Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Sales Channel
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Disease Indication , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Disease Indication , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Disease Indication
- Figure 26: North America Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Route of Administration
- Figure 29: North America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Sales Channel
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Disease Indication , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Disease Indication , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Disease Indication
- Figure 36: Latin America Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Route of Administration
- Figure 39: Latin America Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Sales Channel
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Disease Indication , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Disease Indication , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Disease Indication
- Figure 46: Western Europe Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Route of Administration
- Figure 49: Western Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Sales Channel
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Disease Indication , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Disease Indication , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Disease Indication
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Route of Administration
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Sales Channel
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Disease Indication , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Disease Indication , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Disease Indication
- Figure 66: East Asia Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Route of Administration
- Figure 69: East Asia Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Sales Channel
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Disease Indication , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Disease Indication , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Disease Indication
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Route of Administration
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Sales Channel
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Disease Indication , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Disease Indication , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Disease Indication
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Route of Administration, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Route of Administration, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Route of Administration
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Sales Channel, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Sales Channel, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Sales Channel
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

