Self-administered Biologics Market
The Self-administered Biologics Market is segmented by Indications (Diabetes, Rheumatoid Arthritis, Anaphylaxis, and Others), Product (Prefilled Syringes, Autoinjectors, Wearable Injectors, Drug Reconstitution Delivery Systems, and Pen Injectors), Mode of Delivery (Subcutaneous, Intramuscular, and Transdermal), End User (Homecare Settings and Hospitals/Clinics), Brand (Branded Biologics and Biosimilars), Technology (Conventional Devices and Smart Devices) and Region. Forecast for 2026 to 2036.
Self-administered Biologics Market Forecast and Outlook By FMI
In 2025, the self-administered biologics market was valued at USD 98.3 billion. Based on Future Market Insights’ analysis, demand for self-administered biologics is estimated to grow to USD 102.7 billion in 2026 and USD 159.5 billion by 2036. FMI projects a CAGR of 4.5% during the forecast period.
Absolute dollar growth of USD 56.8 billion over the decade signals sustained expansion anchored in chronic disease prevalence and care-site shift economics rather than one-time therapy launches. Growth is held back when devices cannot reliably deliver higher-viscosity formulations at home, or when coverage and training pathways limit conversion from infusion suites to patient self-care.
Summary of Self-administered Biologics Market
- Market Definition
- Self-administered biologics are injectable biologic therapies delivered outside clinics using pens, prefilled syringes, autoinjectors, or wearable injectors, where training quality and device performance shape adherence and safety outcomes.
- Demand Drivers in the Market
- Diabetes scale keeps injection-based self-care structurally central, supported by global prevalence and long-duration therapy needs. [2] [3]
- Payers and providers keep pushing site-of-care migration to reduce facility cost load and free capacity for complex care, lifting demand for self-injection formats where protocols allow. [7]
- High-viscosity delivery innovation and usability-led device design raise conversion rates for biologics that historically required supervised administration. [4]
- Key Segments Analyzed in the FMI Report
- Indications: Diabetes remains the largest indication cluster due to routine injection behaviour and high treatment continuity. [2]
- Product: Prefilled syringes and pen-based formats anchor conversion by reducing preparation steps and supporting standardised dosing.
- Geography: China and India lead growth as diagnosis and access expand across large patient pools and urban homecare adoption rises. [2] [3]
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Senior Analyst at Future Market Insights, opines, ‘In the updated version of the Self-administered Biologics Market Report for 2026 to 2036, CXOs will find that device reliability, patient training pathways, and payer acceptance set the real ceiling on self-use conversion, even when biologic pipelines expand.’
- Strategic Implications/Executive Takeaways
- Build payer-facing evidence linking self-administration to reduced facility utilisation, stable adherence, and fewer dose-handling errors, since coverage decisions shape conversion pace. [7]
- Prioritise high-viscosity and higher-volume delivery readiness across device and packaging supply chains, as performance limits can block label expansion into home use. [4] [5]
- Partner with homecare services and nurse educator networks to shorten time-to-competence and protect persistence, since training capacity can cap adoption in fast-growth regions. [9]
- Methodology
- Forecasts triangulate epidemiology, care-site shift assumptions, and device readiness signals, validated through primary interviews and supply-side capacity checks. [1] [2]
- Desk research uses WHO and IDF datasets to ground chronic disease burden and spending pressure that influence payer priorities and access rules. [2] [3]
- Primary interviews test switching protocols, device failure modes, and training time requirements needed for safe home administration across regions.
As Lars Fruergaard Jørgensen, President and CEO of Novo Nordisk, noted regarding injectable capacity scaling tied to chronic disease demand, ‘We are very pleased with the agreement to acquire the three Catalent manufacturing sites which will enable us to serve significantly more people living with diabetes and obesity in the future.’ [1] This matters for the self-administered biologics market because fill-finish capacity, device supply, and pen availability can cap patient access even when prescribing demand exists.
China (7.4% CAGR) and India (6.5% CAGR) drive expansion through faster diagnosis, coverage broadening, and rising self-care acceptance in metro systems. Japan (4.5% CAGR) supports steady demand through ageing demographics and structured reimbursement for chronic therapies. Germany (4.0%) and France (3.8%) grow through protocol-led switching to at-home administration when supported by homecare services. Canada (3.5%) and Italy (3.5%) contribute through gradual conversion of maintenance biologics into home settings, with growth constrained by specialist capacity and payer step-therapy rules.
Market Definition
Self-administered biologics are injectable biological medicines, mainly monoclonal antibodies, peptide hormones, and related large-molecule therapies, designed to be administered by patients or caregivers outside hospitals using delivery devices such as pens, prefilled syringes, autoinjectors, and wearable injectors. The market measures drug value associated with home or self-injection use across chronic conditions such as diabetes, autoimmune disease, allergy, and select respiratory indications.
Market inclusions
This report covers global and regional market sizing for 2025 with forecasts for 2026 to 2036, including value estimates by indications, product, mode of delivery, end user setting, brand (branded and biosimilars), and device technology (conventional and smart). The scope includes country-level growth assumptions, care-site shift analysis, device enablement factors (training, adherence support), and competitive positioning across pharma and device supply partners.
Market exclusions
The scope excludes orally delivered biologics and small-molecule self-administered drugs that do not require injection devices. It excludes purely clinic-administered infusion biologics unless there is a defined home-administration pathway and device format associated with self-use. It also omits standalone device-only revenue (device sales without a biologic drug value link), downstream digital therapeutics that are not tied to biologic dosing, and non-therapeutic cosmetic injectables.
Self-administered Biologics Market Research methodology
- Primary research: Primary interviews include biologics commercial leaders, homecare pharmacy operators, nurse educators, device engineers, and procurement stakeholders to validate care-site shift rates, device conversion friction, and coverage constraints.
- Desk research: Desk research uses public health burden datasets, payer and regulator publications, and company disclosures on fill-finish capacity and self-injection device development. [1] [2] [3]
- Market-sizing and forecasting: FMI uses hybrid top-down and bottom-up modelling, anchoring addressable patient pools by indication, then applying adoption curves for self-administration by route and device type, adjusted for payer rules and homecare capacity constraints.
- Data validation and update cycle: Outputs are triangulated across epidemiology, device shipment signals, and policy coverage mechanisms. Assumptions are refreshed with major regulatory approvals, reimbursement changes, and manufacturing capacity announcements. [1] [7]
Segmental Analysis
Self-administered Biologics Market Analysis by Indications
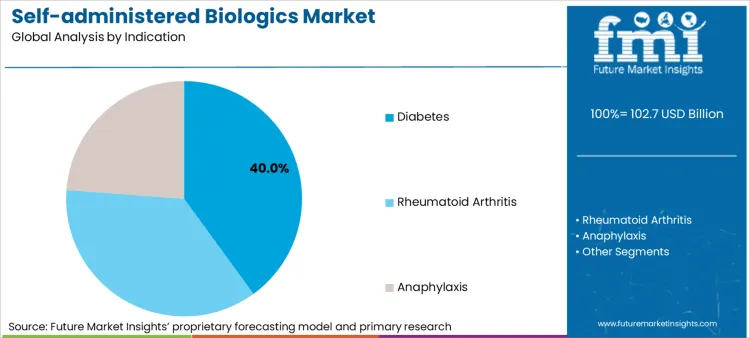
Based on FMI’s self-administered biologics market report, consumption linked to Diabetes is estimated to hold 40.0% share in 2025. Diabetes dominates because routine injection behaviour is already established, dosing is frequent, and device ecosystems are mature, so payer and provider focus shifts toward affordability, adherence, and reliable supply. [2] [3]
- Scale signal: WHO reports diabetes continues to expand as a global chronic burden, keeping self-injection demand structurally high across mature and emerging markets. [2]
- Device adherence: IDF’s 2024 diabetes burden framing supports why device-mediated self-care remains central to treatment continuity and long-term outcomes in large patient bases. [3]
- Supply expansion: Novo Nordisk’s Catalent site acquisition links directly to injectable supply scaling, influencing availability of pen and prefilled formats that support self-administration. [1]
Self-administered Biologics Market Analysis by Product
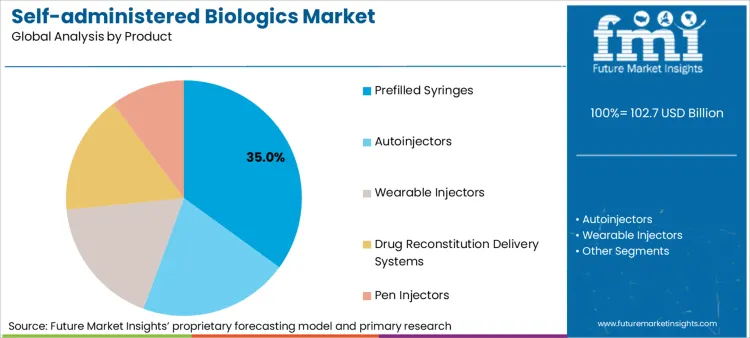
Based on FMI’s self-administered biologics market report, consumption of Prefilled Syringes is estimated to hold 35.0% share in 2025. Prefilled syringes lead because they standardise dosing, reduce preparation errors, and fit both conventional and assisted self-administration pathways, including transitions from hospital clinics to homecare.
- High-viscosity push: Ypsomed’s collaboration with BD targets self-injection solutions for higher-viscosity biologics, addressing a core technical barrier that limits prefilled and pen expansion into home settings. [4]
- Primary packaging capacity: SCHOTT Pharma’s work on large-volume ready-to-use cartridges supports higher-dose or higher-volume self-administration formats, which is relevant as more biologics target less frequent but larger injections. [5]
- Installed base proof: SHL Medical’s disclosures on autoinjector scale and patient reach signal the real-world volume of device-mediated self-injection, reinforcing why device supply and reliability matter for product mix. [6]
Self-administered Biologics Market Drivers, Restraints, and Opportunities
Future Market Insights analysis finds this market grows because health systems carry large, recurring chronic disease cohorts that need long-duration therapy, and injectables remain standard for many high-value biologics. Diabetes alone sets a structural baseline: global prevalence and complication risk keep injectable maintenance therapy and device ecosystems in active use. [2] [3]
The market’s current shift is shaped by conversion economics. Some biologics still require supervised dosing because of administration complexity, high viscosity, or monitoring needs, limiting self-use penetration. At the same time, device makers and pharma teams are pushing prefilled, pen, and wearable formats that shift labour and site costs away from clinics, which can increase value even when patient growth is steady. Home infusion and homecare service structures also matter because they can act as the bridge between clinic and fully independent self-administration. [7] [9]
- Care-site migration: Health systems and payers support home administration pathways to reduce facility utilisation and staffing pressure, which supports conversion of maintenance biologics into self-use formats when protocols permit. [7]
- High-viscosity delivery: Many modern biologics trend toward higher concentration, which raises injection force and delivery complexity; device advances aimed at high-viscosity delivery expand which therapies can move into self-use. [4] [5]
- Device supply resilience: Fill-finish and device component capacity can become a hard constraint in fast-growing injectable categories, shaping availability and switching pace across indications and countries. [1]
Regional analysis
The market is assessed across North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, and the Middle East & Africa. FMI’s regional view links biologic pipeline conversion into self-use formats with payer rules, homecare infrastructure, and device supply resilience, with the full report providing market attractiveness analysis by country and segment.
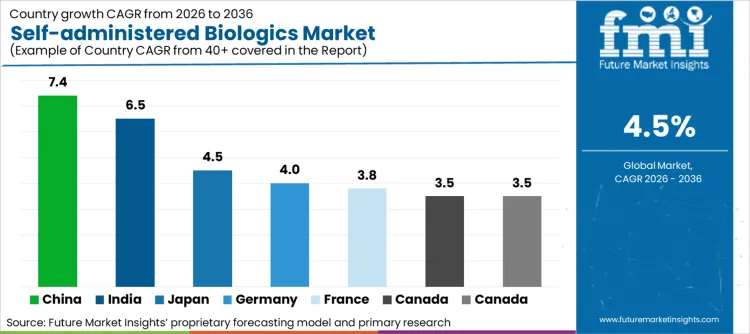
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 7.4% |
| India | 6.5% |
| Japan | 4.5% |
| Germany | 4.0% |
| France | 3.8% |
| Canada | 3.5% |
| Italy | 3.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America market analysis
North America acts as the protocol-and-reimbursement benchmark where payer coverage design and evidence standards shape which biologics shift into self-use, while device supply scale can accelerate adoption. Large pharma and device ecosystems support fast rollouts of new self-injection formats, with demand anchored by diabetes and autoimmune maintenance cohorts. [2] [7]
- United States: Demand for self-administered biologics in the United States is projected to rise at 3.4% CAGR through 2036. Growth is supported by large diabetes prevalence and steady expansion of self-injection formats tied to chronic therapies. WHO’s diabetes burden framing supports sustained therapy need, while the IDF’s 2024 dataset signals high ongoing expenditure pressure that makes at-home dosing economics attractive for payers. [2] [3] The conversion mechanism is shaped by coverage and service capacity: Medicare’s home infusion therapy benefit defines coverage for professional services linked to certain infused drugs and biologicals, supporting site-of-care shifts where a pump or structured home service model applies. [7]
- Canada: Demand for self-administered biologics in Canada is projected to rise at 3.5% CAGR through 2036. Growth is anchored in chronic disease management under provincial health systems, where self-administration can reduce facility load and support continuity of care for stable patients. Diabetes scale remains a baseline demand driver, and global prevalence trends translate into steady treated cohorts. [2] [3] The key constraint is switching friction: patients often need training and ongoing support to move from supervised administration to home dosing, and system capacity for nurse educators can set the pace of conversion. Device reliability in higher-viscosity categories also affects which biologics gain broad self-use penetration, linking Canadian adoption to global device innovation and supply chains. [4] [6]
Western Europe market analysis
Western Europe functions as a service-led conversion market where structured homecare medicine services and strict procurement governance influence the speed of self-administration adoption. The region benefits from strong biologics utilisation in autoimmune care, while conversion depends on homecare capacity, patient training standards, and device safety performance. [9]
- Germany: Demand for self-administered biologics in Germany is projected to rise at 4.0% CAGR through 2036. Growth is driven by steady biologics use in chronic disease pathways and the operational logic of shifting stable patients toward home dosing when protocols allow. Diabetes prevalence trends continue to anchor device-mediated self-care demand, supporting continuous use of pen and syringe formats. [2] [3] A second driver is device capability: as more biologics shift to high-concentration formulations, the ability to deliver higher viscosity reliably at home becomes decisive for broader conversion. Ypsomed’s high-viscosity self-injection focus signals the type of technical pathway that supports higher adoption in markets like Germany where patient training is structured and device usability scrutiny is high. [4]
- France: Demand for self-administered biologics in France is projected to rise at 3.8% CAGR through 2036. France’s growth is shaped by hospital-to-homecare migration where suitable therapies move into community and home settings with supported training. Chronic disease prevalence remains the structural anchor, while payer and provider focus on adherence and reduced facility burden supports self-use conversion. [2] [3] Device innovation provides the practical route: packaging and delivery formats that can handle larger volumes or higher concentrations make it easier to shift dosing away from clinics, which aligns with the direction of primary packaging and device development seen in large-volume ready-to-use cartridges. [5] Patient support infrastructure and homecare medicine standards also matter, since consistent education and safe handling practices increase persistence and reduce avoidable discontinuation. [9]
East Asia market analysis
East Asia is a volume-growth engine where large patient bases, faster diagnosis, and expanding access to chronic therapy drive higher demand, while domestic manufacturing and device supply strategies shape affordability and availability. Conversion toward self-administration tends to accelerate where device formats are standardised and distribution supports refill continuity. [2] [3]
- China: Demand for self-administered biologics in China is projected to rise at 7.4% CAGR through 2036. Growth is propelled by scale: large chronic disease cohorts, rising diagnosis, and expanding access to maintenance therapies create volume headroom for self-use biologics. WHO’s diabetes framing and the IDF’s 2024 burden dataset support the demand base and the incentive to move stable dosing out of hospital settings to reduce system strain. [2] [3] Conversion into self-administration is supported when devices reduce dosing complexity and when supply chains can provide consistent availability. Packaging and device innovations aimed at high-volume or high-viscosity delivery become relevant as more biologics move toward higher concentration and less frequent dosing schedules. [4] [5]
- Japan: Demand for self-administered biologics in Japan is projected to rise at 4.5% CAGR through 2036. Japan’s demand is driven by ageing demographics and high chronic disease management intensity, which supports steady biologics utilisation and a consistent market for self-injection devices. Diabetes remains a baseline cohort, and prevalence trends maintain long-duration therapy demand. [2] [3] Japan’s adoption also reflects device quality expectations: delivery formats need high reliability and user safety for older patients and caregivers, which links market performance to the maturity of autoinjector and pen ecosystems. [6] The ability to administer higher-viscosity biologics in home settings influences the breadth of therapies that can convert into self-use pathways, tying future conversion to the technical direction of high-viscosity self-injection solutions. [4]
South Asia & Pacific market analysis
South Asia & Pacific acts as an access expansion zone where growing chronic disease burden and rising healthcare capacity increase treated populations. Self-administration adoption depends on affordability, patient education infrastructure, and the ability to maintain cold-chain and refill continuity for biologics. [2] [3]
- India: Demand for self-administered biologics in India is projected to rise at 6.5% CAGR through 2036. Growth is driven by expanding chronic disease burden and a rising treated patient base in metro and tier-1 systems, where self-care pathways reduce travel time and facility dependence. WHO’s diabetes assessment and the IDF’s 2024 data reinforce the size of the demand pool and the economic pressure to manage long-duration therapy efficiently. [2] [3] India’s conversion pace is linked to practical enablers: devices need to be simple to use with clear instructions, and reliable supply is essential because interruptions can disrupt adherence for chronic therapies. Device and packaging innovations that support higher-dose delivery while maintaining usability can widen the set of biologics suitable for self-use, while shortages of training capacity and variability in access across states can slow adoption outside top centres. [4] [5]
Competitive aligners for market players
The self-administered biologics market is concentrated around large pharma portfolios that control high-value chronic therapy franchises, supported by a specialised device and primary packaging ecosystem. Competitive advantage is shaped by three variables: (1) portfolio breadth across high-prevalence chronic indications, (2) device and packaging readiness for higher-viscosity and higher-volume delivery, and (3) the ability to secure supply at scale.
Companies with structural advantages tend to control both demand creation and delivery feasibility. They invest in fill-finish capacity, pen and syringe supply, and device partnerships to prevent availability bottlenecks. The Catalent site acquisition linked to Novo Nordisk’s injectable supply strategy is an example of vertical capacity reinforcement that protects therapy continuity when demand rises. [1]
Buyer behaviour varies by setting. Payers and hospital systems prefer formats that reduce total episode cost and shift labour away from facilities while maintaining safety. This creates leverage for buyers to demand real-world adherence evidence, training programmes, and risk mitigation plans for device failure or misuse. Where coverage supports home-based administration services, care-site migration can accelerate, but service capacity constraints can still cap conversion rates. [7] [9]
Device ecosystem leverage has increased because many biologics now face formulation and delivery complexity. Device makers that can handle higher viscosity and deliver consistent dose performance become strategic partners rather than commodity suppliers, shaping switching success for biologics attempting to move from clinic pathways to home use. [4] [5]
Recent developments:
- In February 2024, Novo Nordisk announced an agreement to acquire three Catalent fill-finish sites, aiming to expand capacity for injectable therapies used in chronic diseases. [1]
- In October 2024, Ypsomed announced a strategic collaboration with BD to advance self-injection systems designed for high-viscosity biologics, targeting a central technical barrier to home administration. [4]
- In 2024, IDF published updated global diabetes facts indicating 589 million adults live with diabetes and health expenditure reached at least USD 1 trillion, reinforcing payer pressure to support efficient long-duration treatment pathways. [3]
Key players in Self-administered Biologics Market
- Novo Nordisk A/S
- Eli Lilly and Company
- Johnson & Johnson
- Pfizer Inc.
- F. Hoffmann-La Roche Ltd.
- AbbVie Inc.
- Bristol-Myers Squibb Company
- GlaxoSmithKline plc
- Novartis AG
- Sanofi SA
- Takeda Pharmaceutical Company Limited
- AstraZeneca PLC
Scope of the report
| Metric | Value |
|---|---|
| Quantitative Units | USD 102.7 billion (2026) to USD 159.5 billion (2036), at a CAGR of 4.5% |
| Market Definition | The self-administered biologics market covers biologic drug therapies delivered outside clinical infusion settings using patient-operated injection formats such as prefilled syringes, autoinjectors, pen injectors, wearable injectors, and reconstitution systems, used across chronic indications where at-home dosing, training, and device reliability influence adherence and outcomes. |
| Indications Segmentation | Diabetes, Rheumatoid Arthritis, Anaphylaxis, Others |
| Product Segmentation | Prefilled Syringes, Autoinjectors, Wearable Injectors, Drug Reconstitution Delivery Systems, Pen Injectors |
| Mode of Delivery Segmentation | Subcutaneous, Intramuscular, Transdermal |
| End User Segmentation | Homecare Settings, Hospitals/Clinics |
| Brand Segmentation | Branded Biologics, Biosimilars |
| Technology Segmentation | Conventional Devices, Smart Devices |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | United States, Canada, Mexico, Brazil, Argentina, Germany, France, United Kingdom, Italy, Spain, China, India, Japan, South Korea, Australia and 40 plus countries |
| Key Companies Profiled | Novo Nordisk, Eli Lilly and Company, Johnson & Johnson, Pfizer Inc., F. Hoffmann-La Roche Ltd., AbbVie Inc., Bristol-Myers Squibb Company, GlaxoSmithKline plc, Novartis AG, Sanofi, Takeda Pharmaceutical Company Limited, AstraZeneca PLC, Bayer AG |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with biologics manufacturers, device suppliers, and homecare operators, supported by WHO and IDF burden datasets and policy benchmarking |
Self-administered Biologics Market Analysis by Segments
By Indications:
- Diabetes
- Rheumatoid Arthritis
- Anaphylaxis
- Others
By Product:
- Prefilled Syringes
- Autoinjectors
- Wearable Injectors
- Drug Reconstitution Delivery Systems
- Pen Injectors
By Mode of Delivery:
- Subcutaneous
- Intramuscular
- Transdermal
By End User:
- Homecare Settings
- Hospitals/Clinics
By Brand:
- Branded Biologics
- Biosimilars
By Technology:
- Conventional Devices
- Smart Devices
By Region:
- North America (United States, Canada)
- Latin America
- Western Europe (Germany, France, Italy)
- Eastern Europe
- East Asia (China, Japan)
- South Asia & Pacific (India)
- Middle East & Africa
Bibliography
- [1] Novo Nordisk. (2024, February). Novo Nordisk to acquire three fill-finish sites from Novo Holdings A/S in connection with the Catalent, Inc. transaction.
- [2] World Health Organization. (2024, November). Diabetes (fact sheet).
- [3] International Diabetes Federation. (2024). IDF diabetes atlas: Global diabetes data & statistics (2024).
- [4] Ypsomed. (2024, October). Ypsomed and BD collaborate to advance self-injection systems for high viscosity biologics (press release).
- [5] SCHOTT Pharma. (2024). Large format ready-to-use cartridges/primary packaging for higher-volume injections (press materials).
- [6] SHL Medical. (2025, November). SHL Medical hits milestone: 1.5 billion autoinjectors shipped.
- [7] Centers for Medicare & Medicaid Services. (2025, March). Home infusion therapy/Home IVIG services.
- [8] USA Food and Drug Administration. (2025). Drug label with subcutaneous administration instructions (PDF label).
- [9] East of England Collaborative Procurement Hub. (2024). Homecare medicines framework (homecare services and governance).
This Report Addresses
- Market sizing for 2025 with forecast values for 2026 and 2036 under a consistent CAGR framework
- Segment forecasts across indications, product formats, routes of delivery, end user settings, brand types, and device technology
- Country-level CAGR outlook and regional market attractiveness assessment
- Care-site shift analysis linking clinic-to-home conversion with payer rules and service capacity
- Competitive review of pharma portfolios and device ecosystem partnerships that govern self-administration scaling
- Risk tracking for device performance limits in high-viscosity delivery and supply continuity constraints
- Regulatory and reimbursement context shaping home administration pathways and covered services
- Delivery as analyst narrative with tables for metrics, country outlook, segment positioning, and competitive developments
Frequently Asked Questions
How large is the self-administered biologics market in 2025?
The market was valued at USD 98.3 billion in 2025.
What is the projected value of the self-administered biologics market in 2026?
FMI estimates the market will reach USD 102.7 billion in 2026.
What will be the size of the self-administered biologics market by 2036?
The market is forecast to reach USD 159.5 billion by 2036.
What CAGR is expected for the self-administered biologics market from 2026 to 2036?
FMI projects a 4.5% CAGR from 2026 to 2036.
What is the absolute dollar opportunity between 2026 and 2036?
The market adds USD 56.8 billion in value over the period (USD 102.7 billion to USD 159.5 billion).
Which indication segment leads the market in the base year?
Diabetes leads, accounting for 40.0% share in 2025.
Which product format holds the leading share in 2025?
Prefilled syringes hold the leading product share at 35.0% in 2025.
Which mode of delivery is structurally most aligned with self-administration?
Subcutaneous delivery is the main route for many self-administered biologics due to ease of use and device compatibility.
Which end user setting drives most self-administered biologics demand?
Homecare settings drive demand where training and support allow safe dosing outside clinics.
How important are biosimilars to market growth through 2036?
Biosimilars support value growth by expanding access and encouraging payer-driven switching into self-administration formats.
Which country posts the fastest CAGR in the outlook table?
China is the fastest-growing market in the table at 7.4% CAGR through 2036.
Which country is the second-fastest in the outlook table?
India follows at 6.5% CAGR through 2036.
How fast is Japan expected to grow through 2036?
Japan is projected to grow at 4.5% CAGR through 2036.
How fast is Germany expected to grow through 2036?
Germany is projected to grow at 4.0% CAGR through 2036.
How fast is France expected to grow through 2036?
France is projected to grow at 3.8% CAGR through 2036.
How fast is Canada expected to grow through 2036?
Canada is projected to grow at 3.5% CAGR through 2036.
How fast is Italy expected to grow through 2036?
Italy is projected to grow at 3.5% CAGR through 2036.
What is the single biggest adoption constraint for self-administered biologics?
Device limits in delivering high-viscosity formulations and constrained training capacity can slow conversion to home dosing. [4]
What is the primary strategic lever for manufacturers to accelerate self-administration uptake?
Securing device performance plus supply continuity, alongside payer-ready evidence and homecare support pathways, increases conversion rates. [1] [7]
Which numbers should buyers use as the master forecast benchmarks in this report?
Use USD 98.3 billion (2025), USD 102.7 billion (2026), USD 159.5 billion (2036), and 4.5% CAGR (2026 to 2036) as the master forecast set.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Indication
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Indication , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Indication , 2026 to 2036
- Diabetes
- Rheumatoid Arthritis
- Anaphylaxis
- Diabetes
- Y to o to Y Growth Trend Analysis By Indication , 2021 to 2025
- Absolute $ Opportunity Analysis By Indication , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product, 2026 to 2036
- Prefilled Syringes
- Autoinjectors
- Wearable Injectors
- Drug Reconstitution Delivery Systems
- Pen Injectors
- Prefilled Syringes
- Y to o to Y Growth Trend Analysis By Product, 2021 to 2025
- Absolute $ Opportunity Analysis By Product, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Indication
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Product
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Indication
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Product
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Indication
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Product
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Indication
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Product
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Indication
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Product
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Indication
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Product
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Indication
- By Product
- By Country
- Market Attractiveness Analysis
- By Country
- By Indication
- By Product
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Indication
- By Product
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Indication
- By Product
- Competition Analysis
- Competition Deep Dive
- Novo Nordisk A/S
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Eli Lilly and Company
- Johnson & Johnson
- Pfizer Inc.
- F. Hoffmann-La Roche Ltd.
- AbbVie Inc.
- Bristol-Myers Squibb Company
- GlaxoSmithKline plc
- Novartis AG
- Sanofi SA
- Novo Nordisk A/S
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Product, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Indication , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Product, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Indication
- Figure 6: Global Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Product
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Indication
- Figure 23: North America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Product
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Indication
- Figure 30: Latin America Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Product
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Indication
- Figure 37: Western Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Product
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Indication
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Product
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Indication
- Figure 51: East Asia Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Product
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Indication
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Product
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Indication , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Indication , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Indication
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Product, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Product, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Product
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

