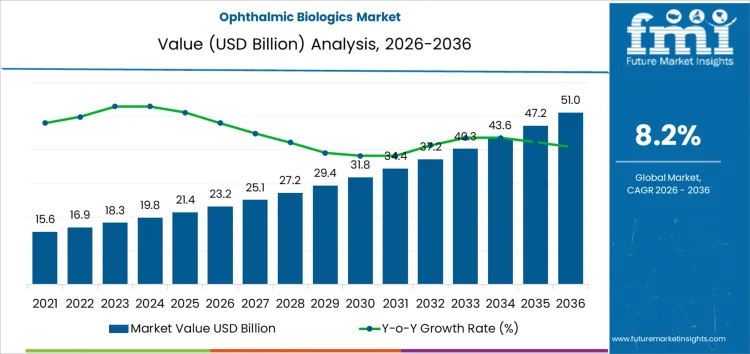
The ophthalmic biologics market is projected to reach USD 23.2 billion in 2026 and advance to USD 51.0 billion by 2036, at a CAGR of 8.2% over the forecast period. Expansion is supported by the growing clinical dependence on biologic agents for managing chronic, vision-threatening retinal conditions where long-term disease control is required. Treatment demand is anchored in disorders such as age-related macular degeneration and diabetic retinopathy, which necessitate repeated intervention rather than episodic care. As diagnostic imaging improves and patient monitoring becomes more precise, biologics are increasingly positioned as sustained therapy rather than rescue treatment, extending average treatment duration per patient.
Market momentum is further influenced by how ophthalmic care delivery is organized and financed. High procedure volumes in outpatient retinal clinics create predictable consumption patterns for intravitreal biologics, while reimbursement frameworks increasingly recognize the cost implications of untreated vision loss. Biosimilar availability is reshaping procurement strategies by enabling broader access without materially altering treatment algorithms, particularly in public healthcare systems. From a supplier standpoint, competitive advantage is linked to dosing durability, supply reliability, and compatibility with clinic workflow constraints. Growth across the forecast horizon therefore reflects structural reliance on biologics as routine components of retinal disease management, rather than incremental uptake driven by short-term therapeutic substitution.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 23.2 billion |
| Market Forecast Value (2036) | USD 51.0 billion |
| Forecast CAGR 2026 to 2036 | 8.2% |
Demand for ophthalmic biologics is rising as treatment approaches for retinal and inflammatory eye diseases emphasize targeted molecular intervention that addresses underlying pathophysiology rather than symptomatic relief. Conditions such as age-related macular degeneration, diabetic macular edema, and uveitis involve pathological signaling pathways that drive vision loss. Ophthalmologists and retinal specialists increasingly specify biologic agents that inhibit vascular endothelial growth factor or modulate immune activity to reduce fluid accumulation, prevent neovascularization, and preserve visual acuity. Clinics and specialty practices invest in injection suites, sterile preparation facilities, and training for intravitreal administration because proper delivery and patient monitoring directly affect clinical outcomes. Procurement teams assess long-term efficacy, safety data, dosing intervals, and cold chain handling requirements to support consistent availability and treatment planning across outpatient care settings.
Growing prevalence of retinal diseases due to aging populations and metabolic health trends reinforces uptake of ophthalmic biologics that align with evidence-based care protocols and visual function preservation goals. Managed care organizations and health systems incorporate these therapies into formulary pathways that recognize impact on quality of life and reduction in severe vision loss. Real-world outcome reporting and longitudinal studies encourage adoption of agents that demonstrate durable response in diverse patient populations. Research investment in next-generation biologics and delivery platforms is expanding options that may extend dosing intervals and reduce treatment burden. These clinical and operational imperatives are contributing to sustained demand growth in the ophthalmic biologics market.
Demand for ophthalmic biologics is driven by rising prevalence of chronic retinal disorders, aging populations, and preference for targeted intravitreal therapies. Clinical management focuses on preserving vision, slowing disease progression, and reducing treatment burden through sustained efficacy. Adoption reflects standardized diagnostic protocols, expanded access to retinal imaging, and guideline-backed biologic use. Therapy selection balances durability of response, injection frequency, and safety monitoring. Segmentation explains how biologic class, ocular indication, and care delivery setting shape utilization intensity, procurement concentration, and treatment workflows across ophthalmic care.
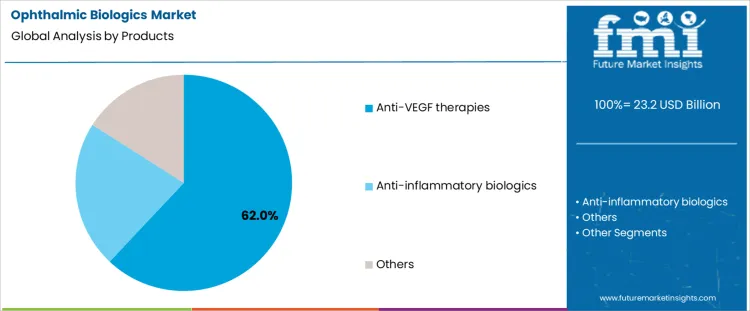
Anti-VEGF therapies hold 62.0%, representing the largest share among ophthalmic biologic product types. These agents directly inhibit pathological angiogenesis responsible for vision loss in major retinal diseases. Strong clinical evidence supports consistent visual acuity improvement and disease stabilization. Established dosing regimens enable predictable treatment planning and follow-up scheduling. Anti-inflammatory biologics serve specific inflammatory eye conditions with narrower patient pools. Other biologics address less common ocular pathways. Product segmentation reflects dominance of therapies targeting vascular endothelial growth where disease prevalence, treatment effectiveness, and standardized protocols drive sustained utilization.
Key Points
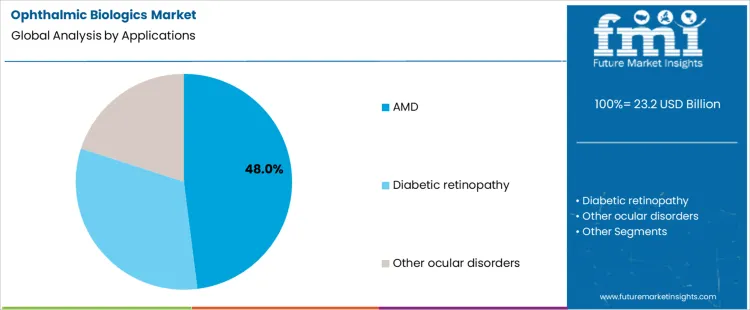
Age-related macular degeneration accounts for 48.0%, representing the largest share among ophthalmic biologic applications. High prevalence in older populations creates sustained demand for long-term treatment. Neovascular disease progression requires repeated biologic intervention to preserve central vision. Screening programs and early diagnosis increase eligible patient volumes. Diabetic retinopathy follows with biologic use driven by rising diabetes prevalence and retinal complications. Other ocular disorders involve smaller, heterogeneous patient groups. Application segmentation reflects concentration where disease burden, chronic progression, and proven biologic responsiveness converge most strongly.
Key Points
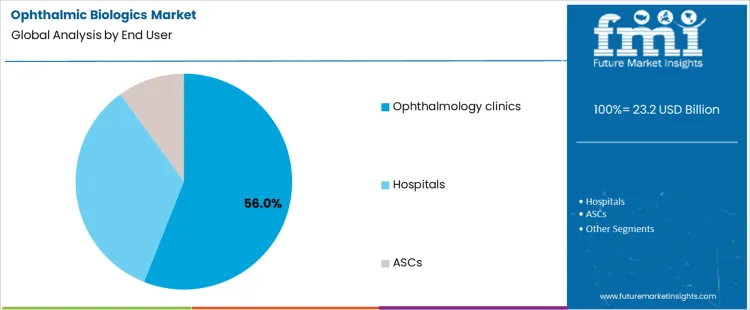
Ophthalmology clinics account for 56.0%, representing the largest share among end-user settings. Most biologic injections are administered in specialized outpatient retinal practices. Clinics provide integrated diagnostics, injection suites, and follow-up monitoring. High patient throughput supports efficient scheduling of repeat treatments. Hospitals manage complex cases and inpatient coordination with lower routine volume. Ambulatory surgical centers perform procedures for selected patients requiring controlled environments. End-user segmentation reflects concentration where specialized expertise, outpatient infrastructure, and procedural efficiency support routine biologic administration.
Key Points
Demand for ophthalmic biologics reflects need to manage progressive retinal and inflammatory eye diseases that threaten vision preservation. Adoption concentrates in ophthalmology clinics, specialty eye hospitals, and outpatient injection centers. Global scope aligns with aging populations and increased diagnosis of chronic ocular disorders. Usage centers on biologic agents administered primarily through intravitreal injection to control pathological angiogenesis, inflammation, or immune-mediated tissue damage.
How do disease burden characteristics and treatment precision requirements shape adoption?
Ocular diseases such as age-related macular degeneration, diabetic macular edema, and uveitis require sustained control to prevent irreversible vision loss. Demand increases where conventional topical or systemic therapies fail to achieve localized efficacy. Biologics deliver targeted action at the retinal or intraocular level, limiting systemic exposure. Treatment regimens are structured around loading doses and maintenance intervals tailored to disease activity and imaging findings. Real-world adoption depends on visual acuity outcomes, injection frequency, and durability of response. Clinic workflows adapt to high injection volumes supported by standardized protocols. Patient monitoring relies on imaging-guided decision-making. Adoption reflects clinical emphasis on preserving functional vision through localized, high-efficacy biologic intervention.
How does administration burden, cost exposure, and long-term adherence influence market scalability?
Intravitreal delivery requires sterile settings, trained specialists, and repeat clinic visits, increasing operational burden. Demand sensitivity rises where injection frequency affects patient adherence and clinic capacity. Safety concerns include endophthalmitis risk and cumulative ocular stress. High therapy cost drives reimbursement scrutiny and dosing optimization strategies. Biosimilar entry affects pricing dynamics yet requires clinician confidence in equivalence. Cold chain handling and shelf-life constraints affect distribution logistics. Long-term treatment persistence varies due to disease progression and patient tolerance. Scalability remains constrained by procedural intensity, payer controls, and reliance on sustained specialist-led care delivery.
Demand for ophthalmic biologics is expanding globally as retinal disease prevalence rises and treatment protocols increasingly favor biologic interventions over conventional therapies. Anti-VEGF biologics and newer agents dominate management of age-related macular degeneration, diabetic retinopathy, and retinal vein occlusion. Longer treatment duration, repeat dosing schedules, and improved diagnostic screening sustain volume growth. Expansion of ophthalmology services and reimbursement inclusion influence access dynamics. Growth rates in India at 9.9%, China at 9.7%, Brazil at 9.4%, USA at 8.8%, and the UK at 7.7% reflect strong expansion driven by patient identification, therapy persistence, and healthcare system capacity building rather than short-term product switching.
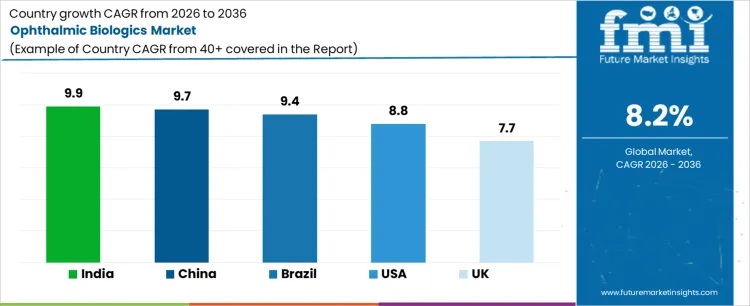
| Country | CAGR (%) |
|---|---|
| India | 9.9% |
| China | 9.7% |
| Brazil | 9.4% |
| USA | 8.8% |
| UK | 7.7% |
Ophthalmic biologic demand in India is growing at a CAGR of 9.9%, shaped by rising retinal disease diagnosis and expanding access to specialized eye care. Large private hospital chains and dedicated eye institutes perform increasing volumes of intravitreal injections. Improved availability of optical coherence tomography supports earlier detection and sustained monitoring. Insurance participation for retinal therapies improves affordability for chronic treatment cycles. Biosimilar entry reduces therapy discontinuation caused by cost pressures. High diabetes prevalence sustains long-term need for retinal biologic management. Growth reflects service expansion, affordability improvements, and treatment continuity rather than rapid adoption of premium next-generation agents.
Demand for ophthalmic biologics in China is expanding at a CAGR of 9.7%, supported by large hospital networks and standardized retinal treatment pathways. Public hospitals increasingly adopt protocol-based intravitreal injection schedules for retinal disorders. Reimbursement inclusion for selected biologics improves initiation and persistence rates. Domestic biologic manufacturing strengthens supply reliability and pricing control. Expansion of diabetic retinopathy screening programs increases eligible patient identification. Growth reflects coordinated scaling of diagnosis, reimbursement, and service delivery rather than concentration of biologic use in high-end private clinics.
Ophthalmic biologic demand in Brazil is growing at a CAGR of 9.4%, driven by public healthcare engagement and increasing retinal disease burden. National treatment protocols prioritize biologics for advanced retinal conditions where vision preservation is critical. Judicial access mechanisms frequently mandate biologic coverage, expanding patient eligibility. Public hospitals adopt biosimilars to manage budget impact while maintaining treatment access. Growth of regional ophthalmology centers improves availability beyond metropolitan areas. Expansion is supported by access enforcement, chronic disease prevalence, and public system scale rather than rapid private-sector driven uptake.
Ophthalmic biologic demand in the United States is expanding at a CAGR of 8.8%, reflecting high screening penetration and long-term therapy persistence. Early diagnosis through routine retinal examinations enables prompt biologic initiation. Broad insurance coverage supports sustained repeat dosing across approved indications. High specialist density allows individualized regimen optimization and switching. Aging population demographics increase prevalence of macular degeneration and diabetic eye disease. Growth is driven by treatment duration, patient retention, and service capacity rather than rapid introduction of entirely new biologic classes.
Demand for ophthalmic biologics in United Kingdom is growing at a CAGR of 7.7%, supported by structured NHS retinal care pathways and centralized commissioning. National screening programs increase early identification of eligible patients requiring biologic intervention. Managed access agreements regulate utilization within defined cost-effectiveness thresholds. Biosimilar adoption expands treatment capacity without proportional escalation in public spending. Centralized ophthalmology clinics enable consistent dosing, monitoring, and adherence across regions. Growth remains steady, reflecting governance-led access expansion and capacity planning rather than unrestricted biologic utilization or rapid protocol escalation.
Demand for ophthalmic biologics is anchored in rising prevalence of retinal disorders, aging populations, and chronic management needs for vision-threatening diseases. Retina specialists evaluate visual acuity outcomes, injection frequency, durability between doses, safety signals, and real-world persistence. Hospital and clinic buyers assess cold-chain handling, indication-specific approvals, patient support programs, and reimbursement alignment. Procurement behavior reflects preference for therapies reducing treatment burden while maintaining consistent efficacy.
Purchasing decisions emphasize long-term disease control, predictable supply, and integration into established retinal care workflows. Trend in the ophthalmic biologics market shows continued shift toward longer-acting biologics and expanded use beyond late-stage disease.
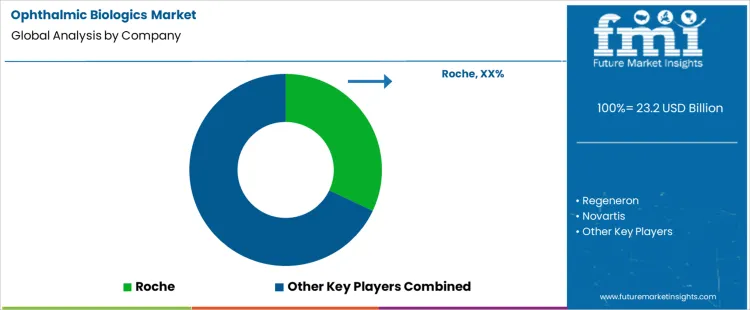
Roche leads competitive positioning through biologics with strong clinical adoption and broad utilization across retinal indications. Regeneron competes through high-efficacy agents widely embedded in standard retinal treatment protocols. Novartis supports demand with ophthalmology-focused biologics positioned for differentiated dosing schedules and sustained outcomes. Bayer maintains relevance through biologic therapies supported by global commercialization partnerships and strong presence in retinal care. Santen participates by advancing biologic innovation aligned with ophthalmology specialization and regional market strengths. Competitive differentiation centers on dosing durability, visual outcome consistency, payer access, and depth of engagement with retinal specialists.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Products | Anti-VEGF therapies; Anti-inflammatory biologics; Others |
| Applications | AMD; Diabetic retinopathy; Other ocular disorders |
| End User | Ophthalmology clinics; Hospitals; ASCs |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, Brazil, USA, UK, and 40+ countries |
| Key Companies Profiled | Roche; Regeneron; Novartis; Bayer; Santen; Others |
| Additional Attributes | Dollar sales by product type, application, end user, and sales channel; treatment frequency and durability profiles influencing anti-VEGF utilization; administration setting preferences across clinics, hospitals, and ASCs; cold-chain handling and intravitreal injection logistics; reimbursement structures and procedure bundling affecting uptake; competitive dynamics shaped by biosimilar entry, pipeline innovation, and long-acting formulation development. |
How big is the ophthalmic biologics market in 2026?
The global ophthalmic biologics market is estimated to be valued at USD 23.2 billion in 2026.
What will be the size of ophthalmic biologics market in 2036?
The market size for the ophthalmic biologics market is projected to reach USD 51.0 billion by 2036.
How much will be the ophthalmic biologics market growth between 2026 and 2036?
The ophthalmic biologics market is expected to grow at a 8.2% CAGR between 2026 and 2036.
What are the key product types in the ophthalmic biologics market?
The key product types in ophthalmic biologics market are anti-vegf therapies, anti-inflammatory biologics and others.
Which applications segment to contribute significant share in the ophthalmic biologics market in 2026?
In terms of applications, amd segment to command 48.0% share in the ophthalmic biologics market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.