The reusable biologics cold chain shippers market crossed a valuation of USD 4.9 billion in 2025. Industry is expected to reach approximately USD 5.3 billion in 2026 as biopharma logistics managers orchestrating these closed-loop networks are standardizing their lane qualifications. The market revenue is expected to expand at a CAGR of 6.9% and reach USD 10.4 billion by 2036 as reverse logistics infrastructure matures across secondary clinical trial hubs.
Temperature-controlled packaging engineers face a closing window to validate high-value payload lanes before impending regulatory audits tighten thermal variance allowances. The transition from disposable polyurethane systems to vacuum-insulated reusable configurations compresses the vendor shortlisting process. Clinical logistics directors must deploy qualified shippers integrated with continuous telemetry before the next global sourcing cycle begins. Processors deploying reusable cold chain packaging assets report significant total-cost-of-ownership reductions over multi-year deployment horizons. Supply chain planners operating without these certified return networks forfeit access to the most lucrative biologic distribution contracts.
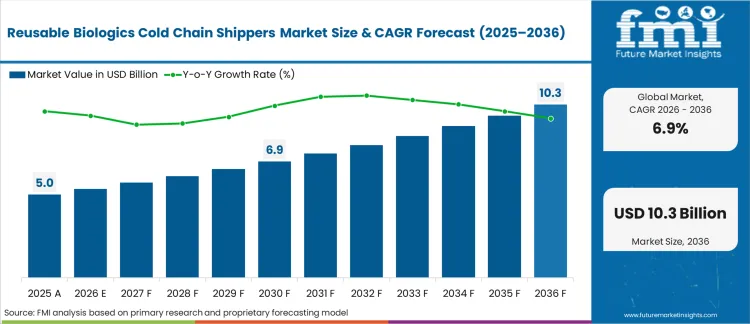
The transition from single-use thermal protection to fleet-managed multi-trip assets represents a fundamental reorganization of pharmaceutical capital allocation. The 6.9% compound expansion rate demonstrates that the initial hurdle of establishing reverse logistics infrastructure has been cleared by major distribution hubs. FMI analysts observe that the financial focus is shifting from asset acquisition costs toward conditioning efficiency and network balancing. This maturation in operational capability dictates the adoption curves mapped across regional regulatory environments.
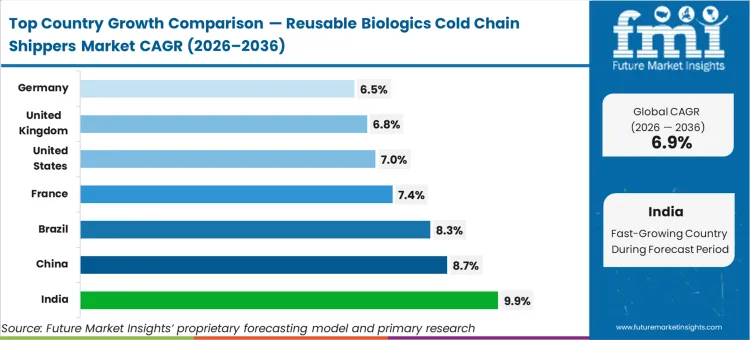
The regional analysis highlights distinct deployment velocities, with Asia Pacific contract manufacturing centers expanding their reusable footprints faster than legacy European hubs. FMI notes that India's 9.9% and China's 8.7% growth rates reflect aggressive export network modernization. Brazil follows closely at 8.3%. Mature markets such as France (7.4%), the United States (7.0%), the United Kingdom (6.8%), and Germany (6.5%) exhibit steady expansion as environmental mandates replace voluntary sustainability initiatives.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 5.3 billion |
| Industry Value (2036) | USD 10.4 billion |
| CAGR (2026-2036) | 6.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The reusable biologics cold chain shippers market represents the engineering, production, and deployment of multi-trip thermal packaging configurations designed to maintain strict temperature tolerances for high-value pharmaceutical payloads. This includes the integration of vacuum-insulated panels, hard-shell plastic composites, and phase-change materials engineered to withstand repeated transit cycles. Solutions ranging from small parcel direct-to-patient boxes to full-scale active pallet shippers are central to the definition.
The market scope includes high-density polyethylene exterior shells, polyurethane rigid foam insulation cores, and proprietary phase-change material conditioning packs. Digital data loggers and integrated IoT tracking sensors physically embedded into the shipper architecture fall within the boundaries. Modular pallet configurations utilizing vacuum-insulated panels for bulk active pharmaceutical ingredient transport are fully incorporated.
Single-use expanded polystyrene coolers and disposable gel packs are explicitly omitted from the valuation. Standalone software platforms managing logistics networks without associated physical hardware are excluded. Refrigerated ocean containers and active temperature-controlled cargo aircraft compartments fall outside the defined packaging parameters.
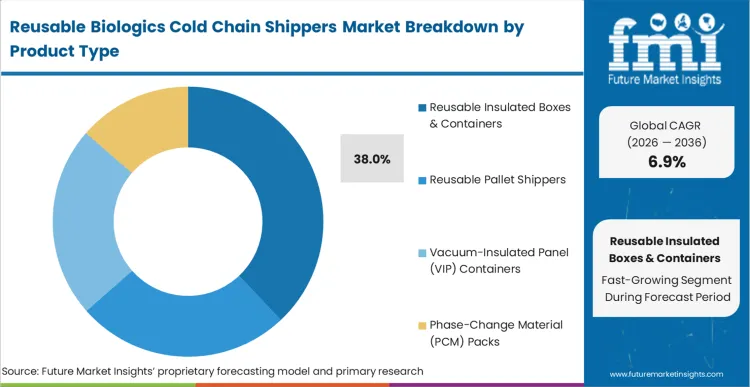
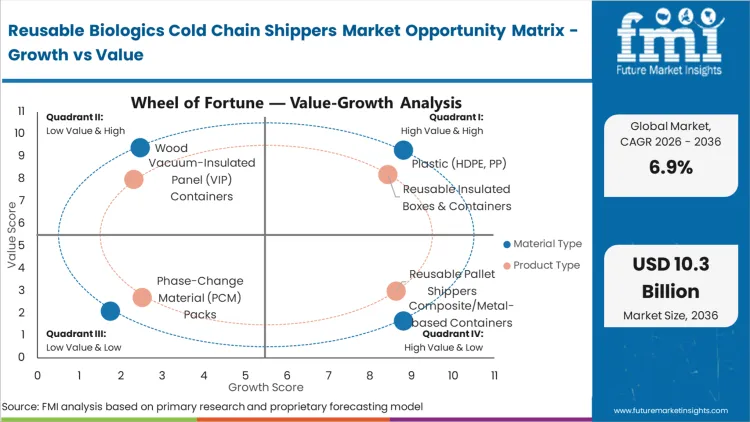
Every clinical supply chain director bidding on advanced therapy distribution contracts now faces strict adherence criteria for deep-frozen payload stability. Reusable Insulated Boxes & Containers capture a commanding 38.0% share in 2026, reflecting the massive scale of direct-to-patient clinical trial deliveries required by modern research protocols. Manufacturers deploying passive temperature controlled packaging solutions directly into localized distribution hubs establish the foundation for agile response networks. As per FMI's projection, clinical supply chain directors utilizing these modular assets significantly reduce volumetric shipping penalties on long-haul air freight lanes. Logistics providers unable to supply certified multi-trip configurations face exclusion from the next wave of decentralized clinical trial RFPs.
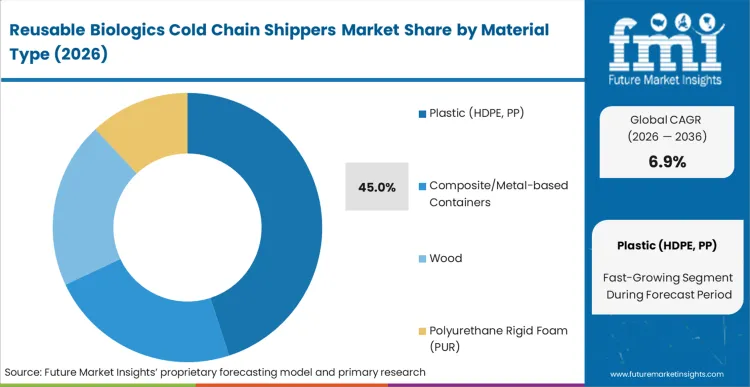
Traditional single-use polyurethane configurations lacked the structural integrity necessary for continuous reverse logistics recovery. High-density plastic composites engineered for impact resistance broke this barrier, establishing a new operational standard. Plastic (HDPE, PP) dominates with a 45.0% segment share in 2026, driven by the material's superior strength-to-weight ratio and sanitization characteristics. According to FMI's estimates, packaging qualification engineers specifying these resilient shells drastically extend the amortization lifecycle of their thermal assets. Material suppliers focusing on closed-loop recyclability secure long-term contracts with major contract development organizations.
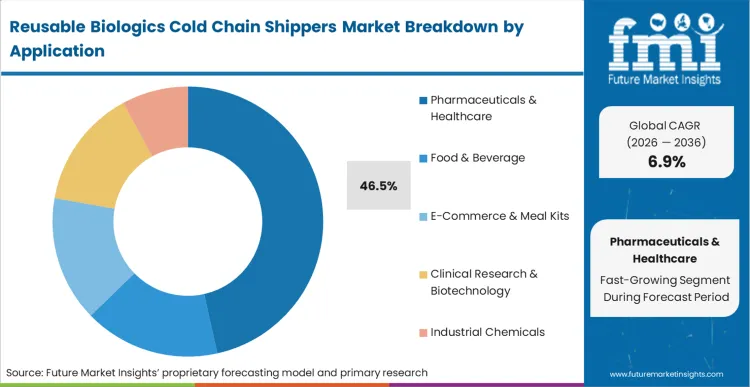
Strict cybersecurity and data visibility mandates across multi-tier defense supply chains force the deployment of converged network architectures. Pharmaceuticals & Healthcare represent the primary application sector, capturing 46.5% of the market share in 2026. Biopharma logistics managers operating continuous production facilities reject packaging designs introducing variable temperature excursions into the supply chain. FMI analysts opine that thermal integrity requirements for mRNA vaccines amplify the demand for high-performance pharmaceutical cold chain packaging. Suppliers failing to demonstrate seamless integration with established digital data loggers lose priority status in critical infrastructure upgrade cycles.
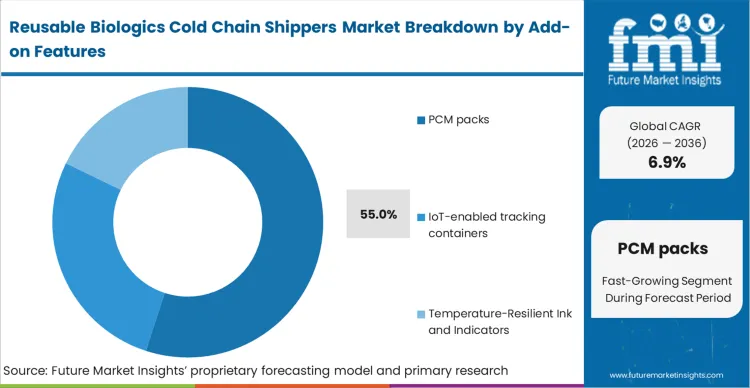
Prior reliance on localized, uncoordinated hardware clocks caused significant synchronization drift across expansive production facilities. The integration of advanced thermal energy storage resolves this fundamental disconnect. PCM packs account for a commanding 55.0% share in 2026 by establishing a highly accurate thermal buffer capable of absorbing extreme external temperature variations. As per FMI's projection, digital lane monitoring combined with temperature controlled pharma packaging assets coordinate complex multi-axis motion sequences with unprecedented reliability. Facilities operating without this foundational timing architecture face systemic production errors as control domains drift out of phase.
The convergence of clinical trial complexity and stringent environmental mandates forces biopharma logistics managers to extract actionable carbon data directly from their distribution networks. This architectural requirement renders fragmented, disposable packaging systems obsolete. Asset owners upgrading international lanes face a strict binary choice between absorbing carbon tax penalties or overhauling their base physical infrastructure. According to FMI's estimates, reverse logistics execution capabilities enable healthcare cold chain logistics operators to lower per-trip costs over a five-year horizon.
The intricate reverse logistics required to optimize asset recovery create steep learning curves for traditional clinical supply planners. Recovering insulated shippers from remote clinical sites demands specialized network coordination that most pharmaceutical producers lack internally. Managing this complex repositioning flow while ensuring precise pre-conditioning at regional depots requires dedicated third-party service centers. Supply chain planners mitigate this constraint by transitioning from outright purchase models to lease-based network access agreements.
Based on the regional analysis, the reusable biologics cold chain shippers market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 9.9% |
| China | 8.7% |
| Brazil | 8.3% |
| France | 7.4% |
| United States | 7.0% |
| United Kingdom | 6.8% |
| Germany | 6.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Greenfield pharmaceutical infrastructure development across the East Asia region accelerates the bypass of legacy single-use packaging constraints. Capital projects directors constructing new biologics manufacturing facilities specify unified reusable shipping networks in their initial logistics blueprints. FMI analysts opine that regional payload consolidation entirely eliminates the costly and time-consuming disposable material procurement cycles that plague older European centers. By building native conditioning capabilities into the foundation, regional asset owners establish highly flexible distribution environments capable of rapid volume scaling. The specific therapeutic export requirements of individual nations dictate the precise thermal implementation parameters for these shipping technologies.
FMI's report includes extensive coverage of the Asia Pacific reusable packaging landscape. It incorporates detailed analysis of Japan, South Korea, Australia, and the broader ASEAN region. A primary trend shaping these nations is the rapid localization of active pharmaceutical ingredient formulation, forcing contract manufacturers to deploy unified reverse logistics architectures to satisfy stringent global brand traceability requirements.
Industrial modernization mandates across North America target the systematic eradication of single-use plastics from advanced clinical trial supply chains. Biopharma logistics managers leading critical infrastructure overhauls face strict directives to unify disparate regional distribution domains under a single leased-asset architecture. Implementing a standardized, reusable backbone enables clinical research organizations to deploy advanced predictive telemetry algorithms without compromising real-time payload stability. This strategic shift requires significant capital allocation toward robust composite hardware capable of isolating mixed-criticality biologic streams. National pharmaceutical frameworks and aerospace cargo qualification standards govern the exact specifications required for these critical network deployments.
FMI's report includes comprehensive evaluation of the North American cold chain sector. It features specific analysis of the Canadian and Mexican distribution markets. A defining dynamic in these countries involves the integration of cross-border clinical trial supply chains, which requires standardized conditioning protocols to coordinate just-in-time delivery sequences and maintain synchronized therapeutic efficacy across multiple trial sites.
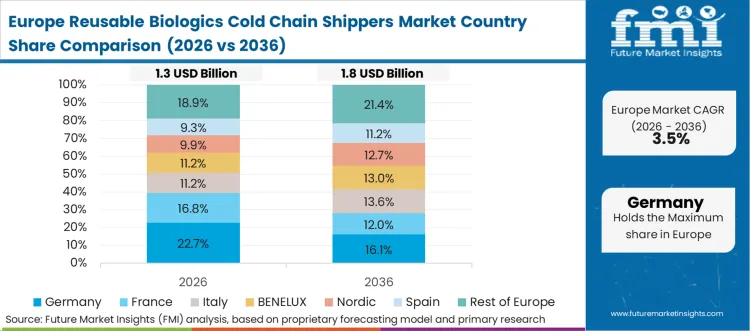
European environmental policy actively penalizes the continued operation of waste-generating and technologically isolated pharmaceutical supply chains. Packaging qualification engineers redesigning legacy distribution lines must integrate continuous carbon footprint data alongside critical payload temperature signals. The transition requires a complete overhaul of existing procurement topologies, shifting from linear consumption models to circular, event-driven asset architectures. Upgrading the core physical infrastructure provides the necessary thermal duration and physical durability to support advanced sustainable healthcare practices. Regional extended producer responsibility standards heavily influence the technical requirements for these network transformations.
FMI's report includes thorough investigation of the European pharmaceutical networking framework. The analysis encompasses Italy, Spain, the Nordics, and the Benelux region. A prevailing structural condition across these nations is the mandatory compliance with strict corporate sustainability directives, forcing asset owners to specify leased networks that can reliably transport fail-safe therapeutics while minimizing secondary packaging waste.
Stringent import qualification standards across Latin America target the systematic eradication of temperature excursions in biologic supply chains. Biopharma logistics managers leading critical distribution overhauls face strict directives to unify disparate national customs clearance domains under a single extended-duration packaging architecture. Implementing a standardized, highly insulated backbone enables regional distributors to deploy advanced passive systems capable of surviving prolonged tarmac delays without active refrigeration. This strategic shift requires significant capital allocation toward robust vacuum-insulated hardware capable of isolating mixed-criticality traffic streams. Regional health surveillance frameworks govern the exact thermal performance specifications required for these critical network deployments.
FMI's report includes extensive coverage of the Latin American cold chain logistics landscape. It incorporates detailed analysis of Argentina, Chile, Colombia, and the broader Andean region. A primary trend shaping these nations is the rapid localization of clinical trial execution, forcing global pharmaceutical sponsors to deploy unified reverse logistics architectures to satisfy stringent international thermal traceability requirements.
As per FMI's projection, thermal validation data sharing between biopharmaceuticals packaging providers and drug manufacturers is redefining supplier qualification criteria. Instead of accepting static laboratory test results, clinical supply chain directors now demand continuous, real-world lane telemetry data in all new tenders. This architectural pivot forces incumbent hardware providers to integrate IoT silicon into every new reusable product line to maintain data continuity. Packaging operations directors utilizing these dynamic analytics platforms streamline their deployment topologies and significantly reduce overall payload loss rates. Asset manufacturers unable to supply certified interoperable data streams face a disqualification window they cannot close after shortlisting begins.
Asset leasing providers deploying high-density global conditioning networks establish a formidable barrier to entry for regional competitors. Maintaining pre-conditioned phase-change material inventories adjacent to major clinical trial hubs requires substantial capital investment and precise predictive demand modeling. Biopharma logistics managers specifying these agile networks streamline their reverse logistics topologies and significantly reduce overall capital expenditure. Hardware engineers executing factory floor expansions now reject proprietary conditioning processes that fragment operational efficiency. Logistics firms failing to demonstrate seamless integration with established global depot networks lose priority status in critical clinical trial upgrade cycles.
The integration of sustainable material science into the core physical hardware fundamentally alters the lifecycle cost calculations for major pharmaceutical shippers. Chemical engineers formulating biodegradable or infinitely recyclable structural components address the stringent end-of-life disposal requirements demanded by European regulatory bodies. Clinical logistics planners utilizing these advanced materials coordinate complex global distribution sequences with unprecedented environmental compliance reliability. Regulatory frameworks enforcing absolute carbon accountability force asset owners to abandon highly toxic insulating materials and commit significant capital to sustainable backbones.

| Metric | Value |
|---|---|
| Quantitative Units | USD 5.32 billion to USD 10.45 billion, at a CAGR of 6.9% |
| Market Definition | The reusable biologics cold chain shippers market represents the engineering, production, and deployment of multi-trip thermal packaging configurations designed to maintain strict temperature tolerances for high-value pharmaceutical payloads. |
| Product Type Segmentation | Reusable Insulated Boxes & Containers, Reusable Pallet Shippers, Vacuum-Insulated Panel (VIP) Containers, Phase-Change Material (PCM) Packs |
| Material Type Segmentation | Plastic (HDPE, PP), Composite/Metal-based Containers, Wood, Polyurethane Rigid Foam (PUR) |
| Application Segmentation | Pharmaceuticals & Healthcare, Food & Beverage, E-Commerce & Meal Kits, Clinical Research & Biotechnology, Industrial Chemicals |
| Add-on Features Segmentation | PCM packs, IoT-enabled tracking containers, Temperature-Resilient Ink and Indicators |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | India, China, Brazil, France, United States, United Kingdom, Germany, and 40 plus countries |
| Key Companies Profiled | Sonoco ThermoSafe, Pelican BioThermal, Cold Chain Technologies, va-Q-tec AG, Softbox Systems, Envirotainer, Cryopak |
| Forecast Period | 2026 to 2036 |
| Approach | The baseline value derives from a bottom-up aggregation of reusable shipper fleet deployments, applying region-specific modernization curves to project future adoption velocity. |
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary research documentation.
How large is the demand for reusable biologics cold chain shippers in the global market in 2026?
Demand for reusable biologics cold chain shippers in the global market is estimated to be valued at USD 5.32 billion in 2026.
What will be the market size of reusable biologics cold chain shippers in the global market by 2036?
Market size for reusable biologics cold chain shippers is projected to reach USD 10.45 billion by 2036.
What is the expected demand growth for reusable biologics cold chain shippers in the global market between 2026 and 2036?
Demand for reusable biologics cold chain shippers is expected to grow at a CAGR of 6.9% between 2026 and 2036.
Which Product Type is poised to lead global sales by 2026?
Reusable Insulated Boxes & Containers accounts for 38.0% in 2026 as clinical supply chain directors execute full-scale direct-to-patient logistics programs.
How significant is the role of Pharmaceuticals & Healthcare in driving reusable biologics cold chain shippers adoption in 2026?
Pharmaceuticals & Healthcare represents 46.5% of segment share as biologic manufacturers standardize on unified reusable backbones to eliminate thermal excursion risks.
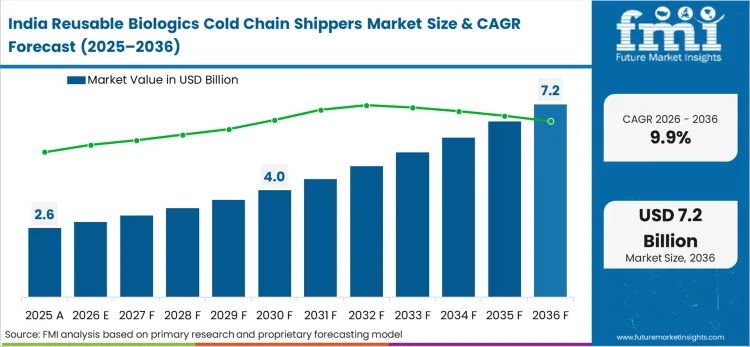
What is driving demand in India?
India's contract development and manufacturing sector relies heavily on closed-loop asset recovery programs to maintain gross margins on temperature-sensitive therapies exported to North American distribution hubs.
What compliance standards or regulations are referenced for France?
The French Ministry of Health enforces strict eco-modulation tariffs on all single-use pharmaceutical transit packaging.
What is the India growth outlook in this report?
India is projected to grow at a CAGR of 9.9% during 2026 to 2036.
Why is North America described as a priority region in this report?
The North American advanced therapy medicinal product sector actively drives network consolidation to secure clinical trial payloads against complex multi-tier supply chain delays.
What type of demand dominates in North America?
Demand heavily focuses on replacing vulnerable disposable coolers with standardized multi-trip architectures that comply with strict federal thermal visibility mandates.
What is China's growth outlook in this report?
China is projected to expand at a CAGR of 8.7% during 2026 to 2036.
Does the report cover Brazil in its regional analysis?
Yes, Brazil is included within Latin America under the regional scope of analysis.
What are the sources referred to for analyzing Brazil?
Federal health regulatory mandates and ANVISA cold chain validation standards form the analytical basis.
What is the main demand theme linked to Brazil in its region coverage?
Strict continuous temperature visibility mandates across multi-tier pharmaceutical importation ecosystems force the deployment of converged reusable architectures.
Does the report cover France in its regional analysis?
Yes, France is included within Europe under the regional coverage framework.
What is the main France related demand theme in its region coverage?
Stringent validation criteria for zero-waste hospital logistics operations compel the adoption of multi-trip shipping assets.
Which product formats or configurations are strategically important for Asia Pacific supply chains?
Embedded vacuum-insulated panels integrated directly into modular pallet shippers are critical for streamlining localized active pharmaceutical ingredient export assembly.
What is reusable biologics cold chain shippers and what is it mainly used for?
Reusable biologics cold chain shippers provide extended thermal protection and zero physical degradation over multiple transit cycles. They are primarily used to synchronize high-value cell therapies and critical biologic distributions in pharmaceutical environments.
What does reusable biologics cold chain shippers mean in this report?
The scope encompasses specialized hardware, phase-change materials, and embedded telemetry built on global compliance standards to guarantee thermal stability for clinical traffic.
What is included in the scope of this reusable biologics cold chain shippers report?
The market covers high-density plastic shells, vacuum-insulated panels, phase-change packs, and integrated tracking modules featuring native reusable capabilities.
What is excluded from the scope of this report?
Standard single-use expanded polystyrene coolers, pure software platforms lacking physical hardware, and active refrigerated cargo containers are explicitly excluded.
What does market forecast mean on this page?
The market forecast represents a model-based projection built on defined clinical trial and logistics adoption assumptions for strategic planning purposes.
How does FMI build and validate the reusable biologics cold chain shippers forecast?
The model applies a bottom-up methodology starting with global installed base metrics and cross-validates projections against publicly reported biological product approval rates.
What does zero reliance on speculative third-party market research mean here?
Primary interviews, verified regulatory filings, and official thermal compliance registries are used exclusively instead of unverified syndicated estimates.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.