Cold Chain Packaging Pooling and Reverse Logistics Services Market
The Cold Chain Packaging Pooling and Reverse Logistics Services Market is segmented by Service Type (Pooling, Reverse logistics, Refurbishment, Conditioning, Tracking), Packaging Format (Parcel shippers, Pallet shippers, ULD containers, Bulk boxes, Totes), Temperature Band (2-8°C, Controlled ambient, Frozen, Ultra-cold), End Use (Biopharma, Vaccines, Clinical trials, Diagnostics, Specialty foods), Contract Model (Rental programs, Pay-per-use, Managed lanes, Enterprise contracts, Hybrid contracts) and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Cold Chain Packaging Pooling and Reverse Logistics Services Market Size, Market Forecast and Outlook By FMI
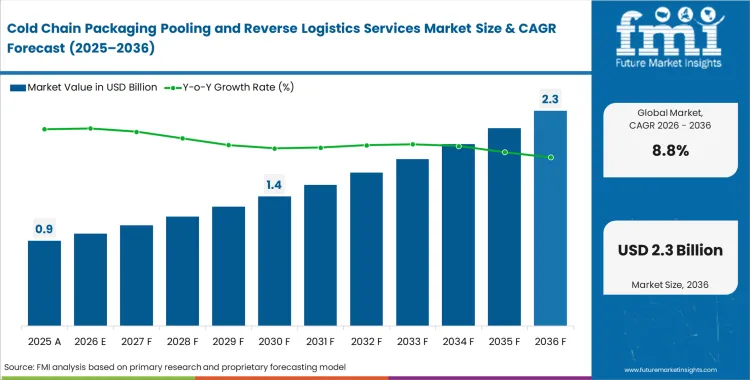
The Cold Chain Packaging Pooling and Reverse Logistics Services Market crossed a valuation of USD 0.8 billion in 2025. Sales are projected to reach USD 0.9 billion in 2026 and USD 2.1 billion by 2036, reflecting an 8.8% CAGR over the forecast period. Growth is being supported by pharmaceutical manufacturers shifting from single-use thermal shippers to leased managed assets that improve chain-of-custody control and reduce packaging waste across regulated distribution networks.
Summary of Cold Chain Packaging Pooling and Reverse Logistics Services Market
- Market Snapshot
- The Cold Chain Packaging Pooling and Reverse Logistics Services Market is valued at USD 0.78 billion in 2025 and is projected to reach USD 2.05 billion by 2036.
- The market is expected to grow at a 9.2% CAGR from 2026 to 2036, creating an incremental opportunity of USD 1.20 billion over the period.
- This is a compliance-heavy logistics service market built around reusable thermal assets, return coordination, conditioning, refurbishment, and controlled redeployment.
- Market structure favors operators that can combine thermal assurance, validated handling, return visibility, and geographically distributed service points.
- Demand and Growth Drivers
- Demand is rising as pharma and healthcare cargo handling requires strict temperature control and coordinated cold-chain partners across cross-border lanes.
- Reusable packaging programs are gaining traction because they lower waste and shift packaging from one-way procurement toward managed circular use.
- Growth is supported by rising cargo throughput, with full-year 2024 global air-cargo demand up 11.3%, which improves utilization of pooled thermal fleets.
- Track-and-trace rules and recall responsiveness are pushing drug distributors toward structured return networks and data-backed asset visibility.
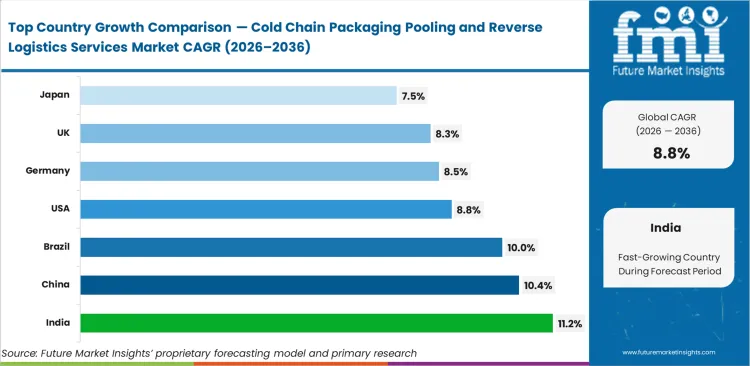
- Among key countries, India leads at 11.2% CAGR, followed by China at 10.4%, Brazil at 10.0%, the United States at 8.8%, Germany at 8.5%, the United Kingdom at 8.3%, and Japan at 7.5%.
- Growth is moderated by high fleet investment, loss-control requirements, lane imbalance, and the need for service-center density to make reusable systems economical.
- Product and Segment View
- The market covers rental and pooling services for parcel shippers, pallet shippers, ULD containers, and bulk thermal boxes used in regulated pharma and specialty cold-chain distribution.
- Core technologies include insulated shells, phase-change materials, telemetry, asset tracking, refurbishment, and return-routing workflows tied to validated temperature bands.
- Pooling leads the Service Type segment with 34.0% share, supported by growing preference for asset-light access to validated reusable fleets.
- Parcel shippers lead the Packaging Format segment with 38.0% share, reflecting dense use in specialty pharma, biologics, and clinical distribution.
- 2–8°C leads the Temperature Band segment with 41.0% share, as refrigerated biologics and vaccines remain the largest controlled-temperature flow.
- Biopharma leads the End Use segment with 44.0% share, tied to high-value products that require documented thermal protection and controlled handling.
- Rental programs lead the Contract Model segment with 52.0% share, because customers increasingly want access, refurbishment, and return management bundled into one contract.
- Scope includes pooling, collection, inspection, conditioning, refurbishment, and reverse-lane orchestration for reusable cold-chain packs, while excluding one-way disposable shippers, pure freight forwarding, and non-temperature-controlled returnable packaging.
- Geography and Competitive Outlook
- India, China, and Brazil are the fastest-growing markets, while the United States and Germany remain stable high-value demand centers.
- Competition is shaped by rental networks, drop-off coverage, refurbishment capability, digital tracking, and the ability to standardize validated reusable flows across global lanes.
- Key participants include Envirotainer, CSafe, Cold Chain Technologies, Peli BioThermal, SkyCell, Sonoco ThermoSafe, and Sofrigam.
- The market is moderately fragmented, with the leading player benefiting from scale advantages linked more to network reach than to packaging hardware alone.
Supply chain teams are under growing pressure to reduce losses tied to discarded thermal shippers and unstable asset availability. Procurement departments are placing greater emphasis on returnable systems that connect with automated temperature monitoring infrastructure. Gaps in reusable cold chain packaging networks can leave logistics providers exposed to container shortages during peak vaccine and biologics distribution periods. Reverse logistics performance has become a critical operating factor because asset recovery speed directly affects container availability, service reliability, and replacement cost. First-mile injection sites often lack the consolidation infrastructure needed to support smooth return flows, which creates delays in asset collection and turnaround.
Carbon reporting requirements in pharmaceutical logistics are also encouraging wider use of circular shipping models. Third-party logistics providers are responding by expanding localized wash and conditioning capacity to manage higher container volumes more efficiently. Faster cleaning, qualification, and redeployment cycles help improve asset utilization and support more stable network performance.
Demand for cold chain packaging pooling and reverse logistics services in India is anticipated to rise at a CAGR of 11.2% through 2036, supported by expanding clinical trial activity and the need for reliable thermal asset recovery. The market in China is expected to grow at a CAGR of 10.4% during the forecast period as domestic biopharma production scales and distribution systems become more structured. Brazil is set to record a 10.0% CAGR through 2036, with growth supported by tighter regional compliance requirements for temperature-sensitive healthcare logistics. Sales in the United States are projected to increase at a CAGR of 8.8% over the forecast period, driven by strong biologics distribution volumes and established cold chain infrastructure. Germany is likely to post an 8.5% CAGR through 2036 as central European logistics hubs improve reverse-lane efficiency. The market in the United Kingdom is anticipated to expand at a CAGR of 8.3% during the study period, supported by post-Brexit network consolidation and greater focus on asset control. Japan is forecast to register a 7.5% CAGR through 2036 as healthcare demand rises with the country’s aging population.
Segmental Analysis
Cold Chain Packaging Pooling and Reverse Logistics Services Market Analysis by Service Type
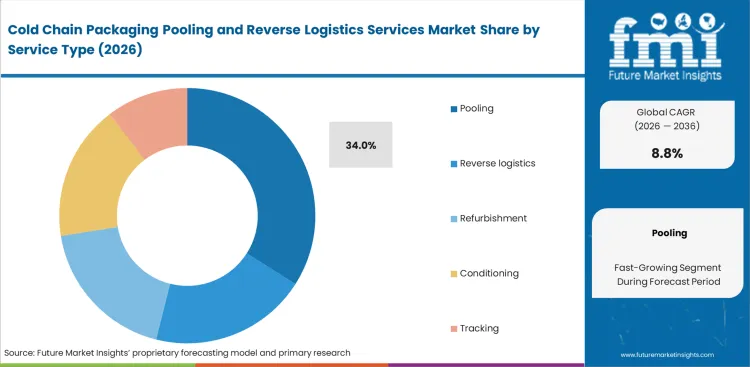
Pharmaceutical manufacturers are moving away from asset upkeep, which keeps demand centered on service models that simplify deployment and retrieval. The pooling segment is estimated to account for 34.0% share in 2026, reflecting this wider move toward outsourced container management. FMI notes that logistics managers using reusable packaging return systems can lower capital pressure while improving operational flexibility. Managed recovery lanes support timely container availability across validated cold chain networks. Asset utilization reports often miss a more important constraint, since conditioning turnaround time has a stronger influence on network performance than total fleet size alone. Large container pools offer limited value when assets remain delayed in poorly coordinated reverse flows. Supply chain leaders that postpone this shift risk tighter availability during seasonal demand surges. Cold chain packaging return management increasingly shapes which providers sustain long-term enterprise relationships.
- Capital reallocation: Eliminating container purchasing requirements allows pharmaceutical companies to redirect funds toward core drug development. Procurement directors secure predictable operating expenses.
- Maintenance offloading: Asset owners handle all thermal validation testing and physical repairs. Quality assurance managers avoid managing complex recalibration schedules for tracking hardware.
- Surge capacity: Shared networks absorb sudden volume spikes efficiently. Logistics directors easily scale operations up during global vaccination campaigns without purchasing excess inventory.
Cold Chain Packaging Pooling and Reverse Logistics Services Market Analysis by Packaging Format
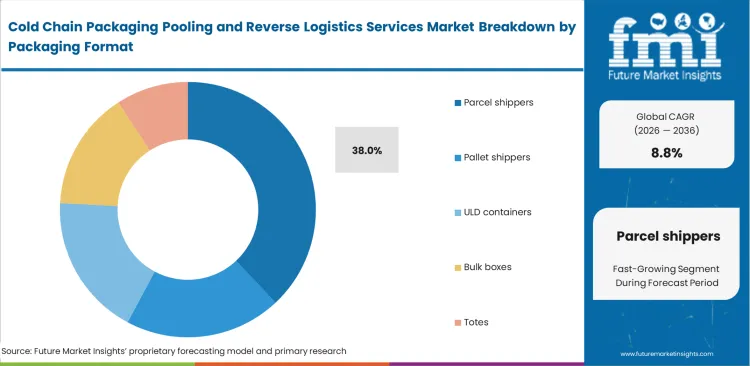
Direct-to-patient biologic delivery protocols require smaller, highly validated thermal containers to protect dose integrity across decentralized routes. Packaging engineers specify these units to support decentralized clinical trials and tighter shipment control at the patient level. The parcel shippers category is expected to hold 38.0% share in 2026, supported by rising use of direct delivery models for sensitive biologics. FMI estimates that integrating cold chain packaging directly into last-mile courier networks depends on robust parcel-sized solutions built for validation and traceability. Clinical managers rely on these formats to monitor individual patient doses and maintain compliance standards. Broad capacity metrics often overlook a key operational strain, since reverse logistics costs for parcel shippers remain structurally higher per unit than pallet systems due to dispersed recovery points. Service providers that fail to optimize parcel return lanes often face pressure on margins and lower asset circulation efficiency. Reusable parcel shipper return programs need close alignment with local courier drop-off networks to keep operations efficient.
- Last-mile flexibility: Small form factors enable direct delivery to patient homes or independent clinics. Logistics managers bypass large central distribution hubs entirely.
- Dose-level tracking: Parcel units accommodate customized conditioning profiles for personalized medicine. Clinical trial managers maintain absolute chain-of-custody data for individual patient samples.
- Recovery friction: Dispersed delivery locations complicate asset collection. Reverse logistics coordinators must partner with local couriers to retrieve empty parcel units efficiently.
Cold Chain Packaging Pooling and Reverse Logistics Services Market Analysis by Temperature Band
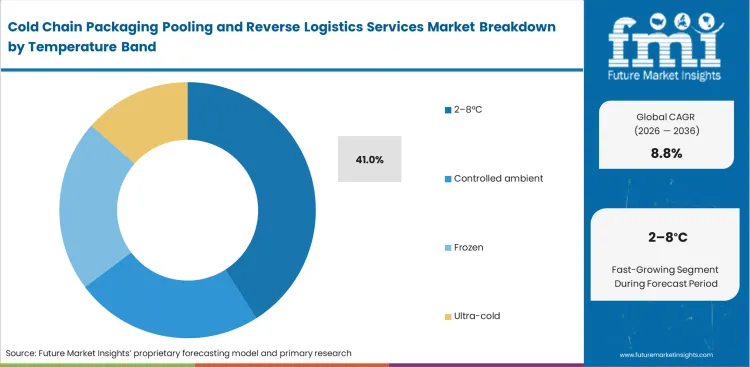
Vaccine distribution networks rely heavily on standard refrigerated conditions for global scale-up. The 2-8°C segment is anticipated to capture 41.0% of the market in 2026. Quality directors mandate strict adherence to this band for most traditional biologics. Based on FMI's assessment, deploying reusable food container pooling frameworks adapted for pharma applications requires precise phase-change material conditioning. Operations managers balance cooling times against dispatch schedules. Procurement dashboards often obscure that ultra-cold infrastructure investments cannibalize capital needed to optimize this dominant 2-8°C segment. Operators neglecting standard refrigerated lanes risk losing high-volume contracts to more specialized thermal shipper rental companies.
- Baseline compliance: Standard biologic formulations require verified 2-8°C maintenance throughout transit. Quality assurance teams rely on pooled assets with proven thermal histories.
- Conditioning complexity: Phase-change materials need specific freezing and thawing cycles before deployment. Operations managers must carefully time this preparation to match daily shipping manifests.
- Infrastructure strain: Handling high volumes of 2-8°C containers demands massive localized refrigeration footprints. Facility managers face continuous space constraints during peak season.
Cold Chain Packaging Pooling and Reverse Logistics Services Market Analysis by End Use
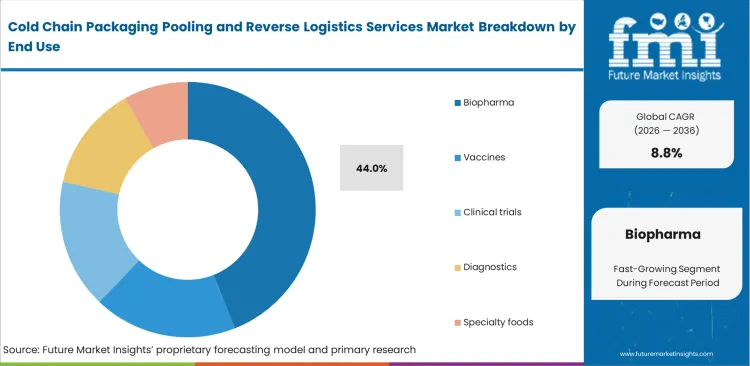
High-value therapeutic development pipelines push demand for zero-failure thermal transport. The biopharma segment is poised to garner 44.0% share in 2026. Clinical operations directors require seamless asset availability for sensitive trial materials. In FMI's view, specialized pharmaceutical packaging frameworks allow drug developers to focus entirely on scientific outcomes. Supply chain managers coordinate complex international hand-offs using pooled shippers. Industry surveys rarely acknowledge that biopharma clients enforce significantly stricter aesthetic cleaning standards for returnable assets than food sectors do. Service providers who fail these visual inspections face immediate contract termination.
- Zero-failure mandate: High-value biologics cannot tolerate any temperature excursions. Clinical directors demand pre-qualified, freshly conditioned shippers for every single dispatch.
- Global coordination: Multi-country trials require standardized packaging formats across borders. Supply chain managers utilize global pooling networks to ensure consistent container availability anywhere.
- Aesthetic rejection: Biopharma clients regularly reject thermally sound shippers due to minor cosmetic damage. Quality control teams must continuously grade and refurbish outer shells.
Cold Chain Packaging Pooling and Reverse Logistics Services Market Analysis by Contract Model
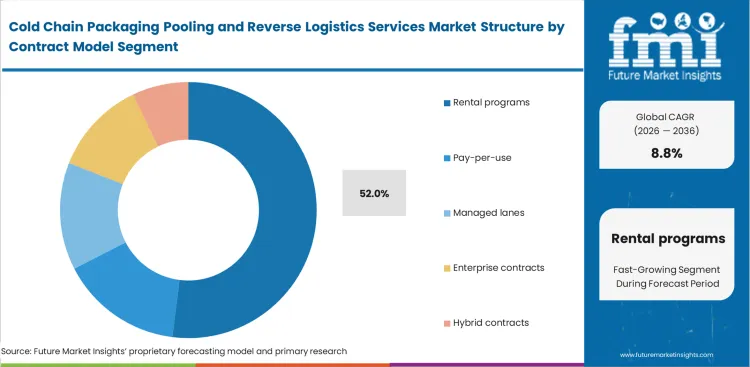
Financial executives favor operating expense models over depreciating capital assets. The rental programs category is set to represent 52.0% of market share in 2026. Procurement officers negotiate these agreements to align shipping costs directly with production volumes. FMI analysts note that flexible reusable packing leasing prevents warehouse congestion during off-peak periods. Operations managers request containers only when production schedules require them. Vendor utilization reports fail to capture that hybrid contracts often mask hidden demurrage fees for delayed returns. Buyers who mismanage asset recovery timelines incur massive penalty charges that erase projected savings. Reusable pharma shipper rental services rely on continuous asset rotation to remain profitable.
- Volume alignment: Rental fees scale proportionally with actual shipping activity. Procurement officers avoid paying for idle thermal assets sitting empty in storage facilities.
- Predictable upgrades: Pooling providers constantly refresh their fleets with newer insulation technologies. Packaging engineers gain access to better performance profiles without requesting new capital budgets.
- Demurrage risks: Delayed asset returns trigger compounding daily fees. Reverse logistics coordinators must actively hunt down empty containers at destination hospitals to protect margins.
Cold Chain Packaging Pooling and Reverse Logistics Services Market Drivers, Restraints, and Opportunities
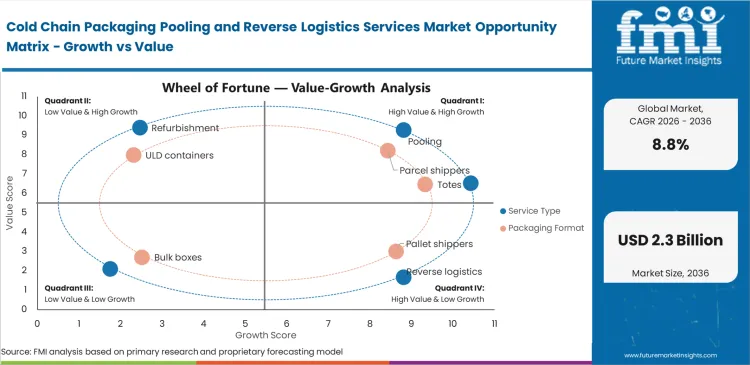
Discarded single-use thermal shippers are becoming a growing cost problem for pharma supply chains, which is accelerating interest in circular asset models. Procurement teams increasingly view disposable packaging as a weak fit for high-frequency temperature-sensitive distribution, especially when raw material prices remain elevated. Companies without access to returnable container contracts can face tighter thermal packaging availability when seasonal demand rises. Reverse logistics services for reusable cold chain packaging help reduce that exposure by keeping validated assets in circulation. Waste-reduction targets are adding another layer of urgency, as distribution teams are under pressure to cut packaging waste without disrupting service reliability.
The shift is not easy to execute because return recovery at the first mile is still highly fragmented. Coordinators managing reusable flows often need to collect empty containers from large numbers of pharmacies and clinics that operate on a small scale. Many of these sites have limited room to store used assets, which reduces pickup flexibility and slows turnaround. Recovery costs are harder to control in these settings, since collecting one parcel shipper from a remote clinic is far less efficient than retrieving pallet loads from a centralized hub. Tracking visibility also weakens when local courier systems are not tightly connected with broader international asset pools.
Opportunities in the Cold Chain Packaging Pooling and Reverse Logistics Services Market
- Automated cleaning hubs: Deploying robotic sanitization lines accelerates container turnaround times. Operations managers increase overall network capacity without adding new thermal assets.
- Cross-industry lane sharing: Combining pharmaceutical reverse lanes with circular cold chain systems improves truck utilization. Logistics directors dramatically lower per-unit retrieval costs.
- Predictive repositioning AI: Implementing machine learning for asset demand forecasting prevents regional shortages. Supply chain heads position empty shippers exactly where needed before volume spikes occur.
Regional Analysis
Based on regional analysis, cold chain packaging pooling and reverse logistics services market is segmented into North America, Latin America, Europe, Asia Pacific, Middle East and Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 11.2% |
| China | 10.4% |
| Brazil | 10.0% |
| United States | 8.8% |
| Germany | 8.5% |
| United Kingdom | 8.3% |
| Japan | 7.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Cold Chain Packaging Pooling and Reverse Logistics Services Market Analysis
Dense clinical trial network expansion drives immediate demand for reliable thermal asset recovery. Logistics managers across this region face fragmented transportation infrastructure that complicates reverse flow coordination. FMI observes that third-party logistics providers are investing heavily in localized wash stations near major manufacturing hubs to solve this exact bottleneck. Pharmaceutical export requirements push domestic manufacturers toward globally validated pooling networks.
- India: Clinical trial network expansion requires highly reliable thermal asset recovery from remote testing sites. Supply chain managers utilize third-party pooling services to avoid managing these complex reverse lanes internally. Demand for Cold Chain Packaging Pooling and Reverse Logistics Services in India is anticipated to rise at a CAGR of 11.2% through 2036. Regional providers that successfully solve rural container retrieval unlock massive competitive advantages.
- China: Localized biopharma scale-up pushes domestic manufacturers to adopt international shipping standards quickly. Packaging engineers specify pre-qualified returnable shippers to satisfy stringent export requirements. China is set to record a CAGR of 10.4% during the assessment period. Integrating automated tracking hardware into every pooled asset becomes standard operational practice here.
- Japan: Aging population healthcare demands drive continuous high-volume biological distributions. Quality assurance teams require zero-failure thermal protection for sensitive age-related therapeutics. The Japan sector is expected to register a CAGR of 7.5% through 2036. High population density allows reverse logistics coordinators to optimize retrieval routes efficiently.
Europe Cold Chain Packaging Pooling and Reverse Logistics Services Market Analysis
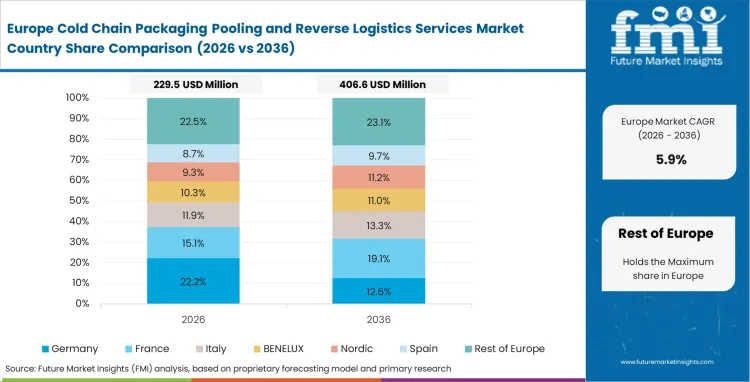
Stringent sustainability mandates compel pharmaceutical manufacturers to eliminate single-use thermal packaging waste completely. Supply chain directors navigate complex cross-border reverse logistics networks to maintain high asset utilization rates. As per FMI's projection, temperature controlled pharma packaging providers who establish dense conditioning nodes across central transit corridors capture dominant share. Cross-border asset recovery networks are essential to supporting pan-European clinical trials.
- Germany: Central European logistics hubs optimize reverse lanes by consolidating returns from multiple neighboring countries. Operations managers rely on vast regional wash centers to maintain high fleet readiness. Sales of Cold Chain Packaging Pooling and Reverse Logistics Services in Germany are expected to increase at a CAGR of 8.5% during the forecast period. Dense infrastructure networks allow asset owners to achieve industry-leading container turnaround speeds.
- United Kingdom: Post-Brexit supply chain consolidation requires localized asset pools to prevent border delays. Procurement officers favor domestic pooling providers to ensure uninterrupted container availability. The United Kingdom is projected to witness 8.3% CAGR through 2036. Providers dominating this domestic circuit insulate themselves from broader continental supply shocks.
North America Cold Chain Packaging Pooling and Reverse Logistics Services Market Analysis
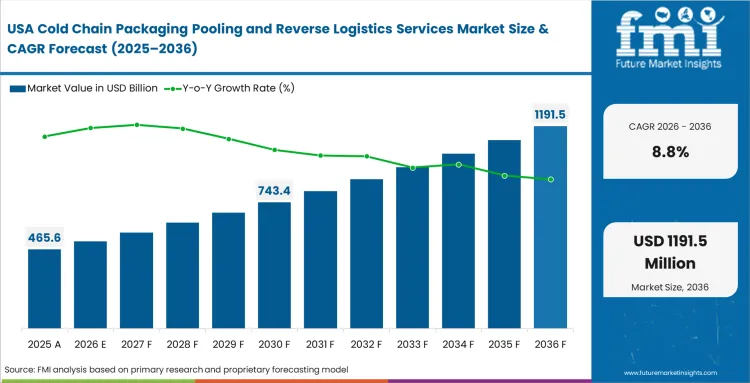
High-value biologics distribution networks require fleets of ultra-reliable thermal shippers. Financial executives push operations teams toward rental models to align packaging costs directly with unpredictable therapeutic demand cycles. FMI's analysis indicates that independent clinics often fail to return assets promptly, forcing providers to enforce strict demurrage penalty structures. Integrating digital tracking capabilities into regional asset pools provides logistics coordinators with real-time location data.
- United States: Dispersed direct-to-patient biological deliveries strain traditional reverse logistics pathways. Reverse logistics coordinators must negotiate retrieval agreements with major courier networks to recover parcel-sized shippers. The United States market is poised to expand at a CAGR of 8.8% through 2036. Providers offering seamless courier integrations dominate this decentralized distribution landscape.
Latin America Cold Chain Packaging Pooling and Reverse Logistics Services Market Analysis
Tightened regional compliance mandates force local distributors to abandon unvalidated passive coolers. Packaging engineers partner with international pooling networks to immediately access highly validated thermal fleets. Establishing localized drop-off points reduces retrieval friction for remote medical facilities.
- Brazil: Tightened regional compliance mandates force local distributors to upgrade thermal protection standards rapidly. Supply chain managers lease pre-conditioned shippers to bypass extensive internal validation testing. The adoption of Cold Chain Packaging Pooling and Reverse Logistics Services in Brazil is expected to move ahead at a CAGR of 10.0% through 2036. Upgrading basic distribution lanes to international thermal standards opens lucrative export opportunities for local drug manufacturers.
Competitive Aligners for Market Players
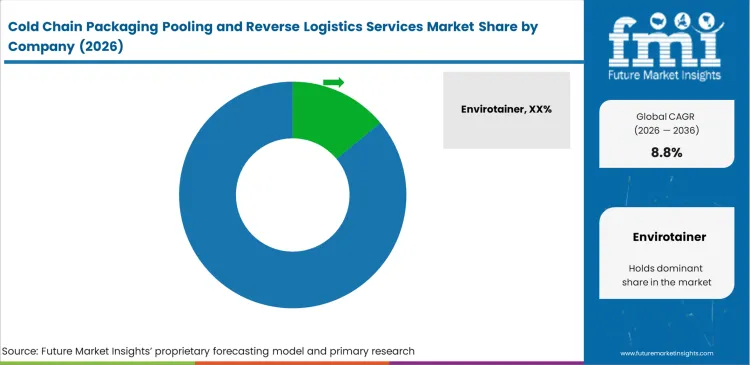
Envirotainer and CSafe dictate international air-freight thermal container standards through massive global fleet scale. Airline cargo managers heavily favor these established pooling networks because their containers feature ubiquitous regulatory pre-approval. Competitors attempting to enter this specific ULD segment face near-insurmountable capital barriers to build comparable global conditioning infrastructure. High-volume pharmaceutical manufacturers rely on these incumbent networks to guarantee capacity during peak distribution periods.
Mid-tier innovators like SkyCell and Sofrigam focus entirely on advanced phase-change material efficiency and digital tracking integration. Packaging engineers specify these healthcare cold chain logistics enabled shippers to guarantee absolute chain-of-custody visibility for high-value biologics. These specialized providers utilize lighter tare weights to offer pharmaceutical clients significant reductions in volumetric freight charges. Differentiation relies on software integration and specific thermal endurance profiles rather than raw container counts.
Procurement directors actively split their contracts across multiple pooling providers to prevent single-vendor pricing monopolies. Quality assurance teams routinely audit competing wash stations to maintain leverage during contract renewals. Smaller niche players survive by dominating localized domestic reverse lanes where major international operators lack sufficient density to operate profitably. Regional providers build specialized drop-off networks to service independent clinical sites effectively.
Key Players in Cold Chain Packaging Pooling and Reverse Logistics Services Market
- Envirotainer
- CSafe
- Cold Chain Technologies
- Peli BioThermal
- SkyCell
- Sonoco ThermoSafe
- Sofrigam
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 0.9 billion to USD 2.1 billion, at a CAGR of 8.8% |
| Market Definition | Commercial arrangements where temperature-controlled shipping assets are rented, recovered, cleaned, and redeployed to eliminate capital expenditure for pharmaceutical manufacturers. |
| Segmentation | Service Type, Packaging Format, Temperature Band, End Use, Contract Model and Region |
| Regions Covered | North America, Latin America, Europe, Asia Pacific, Middle East and Africa |
| Countries Covered | United States, Brazil, Germany, United Kingdom, India, China, Japan |
| Key Companies Profiled | Envirotainer, CSafe, Cold Chain Technologies, Peli BioThermal, SkyCell, Sonoco ThermoSafe, Sofrigam |
| Forecast Period | 2026 to 2036 |
| Approach | Active returnable thermal asset fleet sizes across global trade lanes |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Cold Chain Packaging Pooling and Reverse Logistics Services Market Analysis by Segments
Service Type:
- Pooling
- Reverse logistics
- Refurbishment
- Conditioning
- Tracking
Packaging Format:
- Parcel shippers
- Pallet shippers
- ULD containers
- Bulk boxes
- Totes
Temperature Band:
- 2-8°C
- Controlled ambient
- Frozen
- Ultra-cold
End Use:
- Biopharma
- Vaccines
- Clinical trials
- Diagnostics
- Specialty foods
Contract Model:
- Rental programs
- Pay-per-use
- Managed lanes
- Enterprise contracts
- Hybrid contracts
Region:
- North America
- United States
- Canada
- Latin America
- Brazil
- Mexico
- Rest of Latin America
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Rest of Europe
- Asia Pacific
- China
- India
- Japan
- South Korea
- Australia
- Rest of Asia Pacific
- Middle East and Africa
- GCC Countries
- South Africa
- Rest of Middle East and Africa
Bibliography
- Food and Drug Administration. (2025). CBER-CDER Data Standards Program annual assessment 2024. U.S. Department of Health and Human Services.
- International Air Transport Association. (2025). Global air cargo demand achieves record growth in 2024.
- Jackman, S., Mc Guinness, P., Brennan, L., Pereira, R., Tyrrell, A., Barry, A. M., Brennan, C., & Naughton, B. D. (2025). Global innovations in sustainable pharmaceutical packaging in the last 25 years: A scoping review. Sustainability, 17(23), 10716.
- Pfoser, S., Kienberger, T., & collaborators. (2025). Environmental assessment of reusable packaging for pharmaceutical distribution. Procedia Computer Science, 268, Article 127650.
- World Health Organization. (2024). WHO expert committee on specifications for pharmaceutical preparations: Fifty-seventh report.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Capital expenditure elimination models for pharmaceutical cold chain distribution networks.
- Reverse logistics bottleneck resolutions for direct-to-patient parcel shipper recovery.
- Aesthetic rejection criteria impacts on high-value biopharma container utilization rates.
- Localized wash station deployment strategies across rural clinical trial networks.
- Demurrage fee structures and hidden costs within hybrid rental agreements.
- Standard refrigerated 2-8°C phase-change material conditioning timelines.
- Digital tracking hardware integration into pooled thermal asset fleets.
- Post-Brexit supply chain consolidation effects on regional packaging networks.
Frequently Asked Questions
What is the projected size in 2036?
Sales are poised to cross USD 2.1 billion by 2036. This expansion reflects pharmaceutical manufacturers aggressively shifting from disposable thermal shippers to managed multi-trip assets.
What is the baseline valuation in 2026?
Demand reaches USD 0.9 billion in 2026. Supply chain directors facing margin degradation recognize that capital-intensive container ownership models destroy profitability.
What is the expected CAGR?
Growth maintains an 8.8% pace through 2036. Strict carbon reporting mandates force packaging engineers to eliminate disposable packaging waste entirely.
Why does pooling lead the service segment?
Pooling is estimated to account for 34.0% share in 2026 because it eliminates capital expenditure requirements. Procurement directors secure predictable operating expenses while offloading complex thermal validation testing to asset owners.
Why do parcel shippers dominate packaging formats?
Parcel shippers are expected to hold 38.0% share in 2026 due to direct-to-patient biologic delivery protocols. Clinical managers require these smaller validated containers to track individual patient doses seamlessly.
Why is the 2-8°C band critical?
This standard refrigerated range is anticipated to capture 41.0% of the market in 2026. Quality directors mandate strict adherence to this band for traditional biologics, necessitating highly precise phase-change material conditioning.
Why do biopharma users hold major share?
Biopharma is set to represent 44.0% of market share in 2026 because high-value therapeutics demand zero-failure thermal transport. Clinical operations directors require seamless asset availability for highly sensitive trial materials.
Why are rental programs preferred?
Rental contracts are likely to account for 52.0% share in 2026. Financial executives favor operating expense models that align shipping costs directly with unpredictable production volumes.
How does India compare to China?
Demand in India is anticipated to rise at a CAGR of 11.2% driven by dispersed clinical trial network expansion requiring reliable rural retrieval. China is set to record a CAGR of 10.4% on localized biopharma scale-up demanding international export standards.
What drives United States growth?
The United States market is poised to expand at a CAGR of 8.8%. Dispersed direct-to-patient biological deliveries strain traditional reverse logistics pathways, forcing reliance on specialized courier integrations.
What speeds adoption in Germany?
Sales in Germany are expected to increase at a CAGR of 8.5%. Central European logistics hubs optimize reverse lanes by consolidating returns from multiple neighboring countries into massive regional wash centers.
What slows adoption?
Fragmented first-mile return infrastructure complicates asset retrieval. Reverse logistics coordinators struggle to recover empty containers from thousands of independent clinics efficiently.
How do buyers resist lock-in?
Procurement directors actively split their contracts across multiple pooling providers. Quality assurance teams audit competing wash stations to maintain leverage during negotiations.
What defines incumbent advantage?
Competitors like Envirotainer possess massive global fleet scale. Airline cargo managers favor these networks because their containers feature ubiquitous regulatory pre-approval.
What role do digital trackers play?
Packaging engineers specify tracking-enabled shippers to guarantee absolute chain-of-custody visibility. This allows clinical trial managers to monitor individual high-value doses continuously.
How do demurrage fees impact buyers?
Delayed asset returns trigger compounding daily penalties. Reverse logistics coordinators must actively hunt down empty containers at destination hospitals to protect projected savings.
Why is aesthetic cleaning important?
Biopharma clients regularly reject thermally sound shippers due to minor cosmetic damage. Quality control teams must continuously grade and refurbish outer shells.
How do wash stations improve efficiency?
Conditioning turnaround times dictate true network efficiency. Automated robotic sanitization lines drastically increase overall network capacity without adding new thermal assets.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Service Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Service Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Service Type , 2026 to 2036
- Pooling
- Reverse logistics
- Refurbishment
- Conditioning
- Tracking
- Pooling
- Y to o to Y Growth Trend Analysis By Service Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Service Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Packaging Format
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Packaging Format, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Packaging Format, 2026 to 2036
- Parcel shippers
- Pallet shippers
- ULD containers
- Bulk boxes
- Totes
- Parcel shippers
- Y to o to Y Growth Trend Analysis By Packaging Format, 2021 to 2025
- Absolute $ Opportunity Analysis By Packaging Format, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Temperature Band
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Temperature Band, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Temperature Band, 2026 to 2036
- 2–8°C
- Controlled ambient
- Frozen
- Ultra-cold
- 2–8°C
- Y to o to Y Growth Trend Analysis By Temperature Band, 2021 to 2025
- Absolute $ Opportunity Analysis By Temperature Band, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Biopharma
- Vaccines
- Clinical trials
- Diagnostics
- Specialty foods
- Biopharma
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Contract Model
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Contract Model, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Contract Model, 2026 to 2036
- Rental programs
- Pay-per-use
- Managed lanes
- Enterprise contracts
- Hybrid contracts
- Rental programs
- Y to o to Y Growth Trend Analysis By Contract Model, 2021 to 2025
- Absolute $ Opportunity Analysis By Contract Model, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Service Type
- By Packaging Format
- By Temperature Band
- By End Use
- By Contract Model
- Competition Analysis
- Competition Deep Dive
- Envirotainer
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- CSafe
- Cold Chain Technologies
- Peli BioThermal
- SkyCell
- Sonoco ThermoSafe
- Sofrigam
- Envirotainer
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Packaging Format, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Contract Model, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Packaging Format, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Contract Model, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Packaging Format, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Contract Model, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Packaging Format, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Contract Model, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Packaging Format, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Contract Model, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Packaging Format, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Contract Model, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Packaging Format, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Contract Model, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Packaging Format, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Contract Model, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Service Type
- Figure 6: Global Market Value Share and BPS Analysis by Packaging Format, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Packaging Format, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Packaging Format
- Figure 9: Global Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Temperature Band
- Figure 12: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End Use
- Figure 15: Global Market Value Share and BPS Analysis by Contract Model, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Contract Model, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Contract Model
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Service Type
- Figure 32: North America Market Value Share and BPS Analysis by Packaging Format, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Packaging Format, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Packaging Format
- Figure 35: North America Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Temperature Band
- Figure 38: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End Use
- Figure 41: North America Market Value Share and BPS Analysis by Contract Model, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Contract Model, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Contract Model
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Service Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Packaging Format, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Packaging Format, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Packaging Format
- Figure 51: Latin America Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Temperature Band
- Figure 54: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End Use
- Figure 57: Latin America Market Value Share and BPS Analysis by Contract Model, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Contract Model, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Contract Model
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Service Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Packaging Format, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Packaging Format, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Packaging Format
- Figure 67: Western Europe Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Temperature Band
- Figure 70: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End Use
- Figure 73: Western Europe Market Value Share and BPS Analysis by Contract Model, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Contract Model, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Contract Model
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Service Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Packaging Format, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Packaging Format, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Packaging Format
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Temperature Band
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Contract Model, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Contract Model, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Contract Model
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Service Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Packaging Format, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Packaging Format, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Packaging Format
- Figure 99: East Asia Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Temperature Band
- Figure 102: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End Use
- Figure 105: East Asia Market Value Share and BPS Analysis by Contract Model, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Contract Model, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Contract Model
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Service Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Packaging Format, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Packaging Format, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Packaging Format
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Temperature Band
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Contract Model, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Contract Model, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Contract Model
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Service Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Packaging Format, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Packaging Format, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Packaging Format
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Temperature Band
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Contract Model, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Contract Model, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Contract Model
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

