Cold Chain Integrity and Shock Monitoring Test Devices Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
This report analyzes the cold chain integrity and shock monitoring test devices market across market size, share, shipment demand, revenue forecast, pricing direction, company positioning, competitive landscape, growth drivers, restraints, technology adoption, real-time visibility trends, sensor integration, logistics channel performance, service capacity, regulatory factors, profitability, and strategic opportunities. It also examines segment performance by device type, monitoring parameter, connectivity, deployment mode, packaging level, end use, application stage, and region, while evaluating market attractiveness and future growth prospects across key countries and cold-chain clusters.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Cold Chain Integrity and Shock Monitoring Test Devices Market Size, Market Forecast and Outlook By FMI
Cold chain integrity and shock monitoring test devices market stood at USD 1.0 billion in 2025. Industry valuation is estimated to reach USD 1.1 billion in 2026, and the market is expected to advance at a CAGR of 10.9% through the forecast period. Valuation is likely to climb to USD 3.1 billion by 2036 as shipment qualification in pharmaceuticals, vaccines, diagnostics, and higher-value perishables now relies more on condition evidence that can support release, rejection, and investigation decisions.
Cold Chain Integrity and Shock Monitoring Test Devices Market Summary
- The market is valued at USD 1.1 billion in 2026, rising from USD 1.0 billion in 2025, and is projected to reach USD 3.1 billion by 2036.
- Expansion is expected at 10.9% CAGR through 2036, supported by wider use of shipment-level condition evidence across pharmaceuticals, vaccines, diagnostics, and high-value perishables.
- Shipment monitoring is being aligned with release decisions, deviation review, and route qualification, where device output must support faster acceptance or investigation without adding manual interpretation effort.
- Device selection depends on calibration reliability, deployment simplicity, data clarity, and service availability across shipment lanes rather than feature expansion alone.
- Data loggers are expected to account for 26% share in 2026, reflecting their compatibility with existing shipment-review routines and ease of scaling across mixed route profiles.
- Temperature monitoring is projected to hold 39% share, as it remains the primary threshold for product viability assessment across cold-chain categories.
- Monitoring programs are moving toward lane-based device assignment, where shipment value, route variability, and response requirements determine the level of monitoring applied.
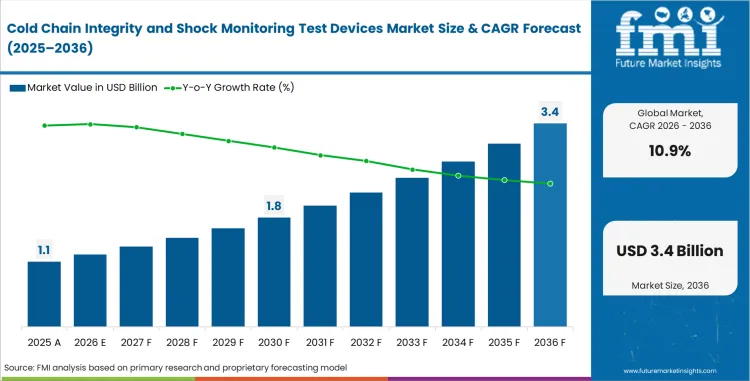
Cold Chain Integrity and Shock Monitoring Test Devices Market Key Takeaways
| Parameter | Details |
|---|---|
| Market value (2026) | USD 1.1 billion |
| Forecast value (2036) | USD 3.1 billion |
| CAGR (2026 to 2036) | 10.9% |
| Estimated market value (2025) | USD 1.0 billion |
| Incremental opportunity | USD 2.0 billion |
| Leading device type | Data loggers |
| Leading monitoring parameter | Temperature monitoring |
| Leading connectivity | Cellular |
| Leading deployment mode | Single-use |
| Leading packaging level | Parcel |
| Leading end use | Pharmaceuticals |
| Leading application stage | In-transit |
| Fastest-growing country | India |
| India CAGR | 12.8% |
| Key supplier brands referenced in market landscape | Sensitech, Controlant, ELPRO, SpotSee |
| Brands referenced in market landscape | Sensitech, Controlant, ELPRO, SpotSee, DeltaTrak, Berlinger, Tive |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Category adoption becomes easier to widen when monitoring devices fit directly into shipping qualification, receiving checks, deviation review, and lane assessment. Pharmaceutical distributors, vaccine handling teams, and specialty food exporters help move that condition forward when device choice is standardized by route type instead of being reconsidered on every shipment. Once that happens, repeat use becomes easier because the device serves a defined operating need rather than acting as a one-off accessory.
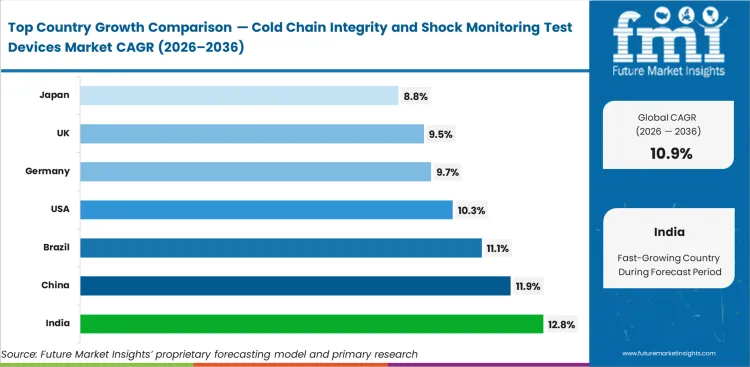
India is expected to post a CAGR of 12.8% in this market through 2036 as cold-chain depth, vaccine handling, and shipment verification tools continue to expand. China follows with a CAGR of 11.9%, supported by manufacturing scale and broader use of monitored export lanes. Brazil is likely to record a CAGR of 11.1% over the forecast period, while the United States is projected to advance at a CAGR of 10.3% as connected monitoring remains closely tied to pharmaceutical release discipline. Germany and the United Kingdom are set to register CAGRs of 9.7% and 9.5% respectively, and Japan is anticipated to move ahead at a CAGR of 8.8% through 2036. Country variation comes from how cold-chain depth, shipment distance, product sensitivity, and review discipline come together in each national setting.
Excursion Evidence Is Becoming A Release Gate
Cold chain buyers are moving beyond simple “in-range” confirmation toward shipment evidence that can stand up in audits, release decisions, and customer disputes. WHO’s 2025 PQS framework separates device types more clearly across indicators, loggers, and advanced monitoring and communication systems, which signals that monitoring choice is now tied to program need rather than treated as interchangeable hardware. The same 2025 WHO testing protocol for data loggers explicitly covers refrigerators, freezers, cold rooms, refrigerated vehicles, and transportable storage, showing how broadly continuous monitoring is now expected across the chain.
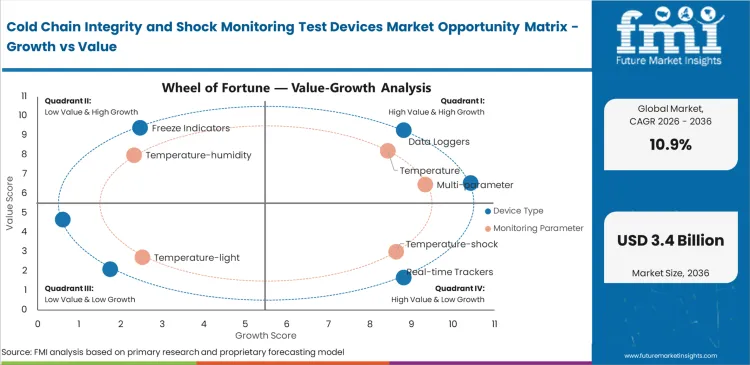
Segmental Analysis
Key Facts About Segments
- Data loggers are expected to hold 26% share in 2026, as they align with shipment qualification routines and allow straightforward post-arrival review without continuous tracking requirements.
- Temperature monitoring is projected to account for 39% share, reflecting its direct link to release decisions and product stability across regulated cold-chain shipments.
- Cellular connectivity is likely to represent 31% share in 2026, where live alert capability supports intervention before shipment completion on higher-value routes.
- Single-use deployment is anticipated to contribute 46% share, supported by reduced recovery effort, simpler logistics, and clearer chain-of-custody handling.
- Parcel-level monitoring is expected to capture 34% share, as shipment-level accountability and receiving decisions depend on device readings tied directly to individual consignments.
- Pharmaceuticals are estimated to account for 36% share, driven by stricter release discipline and the need for defensible shipment condition records.
- In-transit application is projected to hold 58% share, where route variation, handling risk, and delay exposure create the highest need for monitoring visibility.
Cold Chain Integrity and Shock Monitoring Test Devices Market Analysis by Device Type
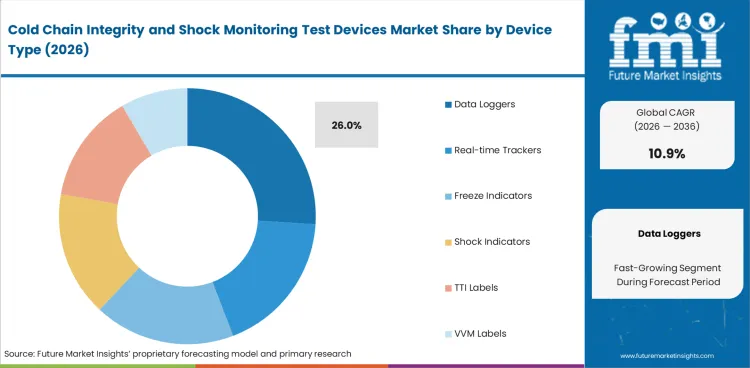
Shipment monitoring decisions often begin with a practical question: how much evidence is enough without making routine deployment harder than the route justifies. Data loggers remain the answer for a large share of that requirement because they fit qualification routines, work across mixed route profiles, and do not require every shipment to be watched in real time. Market estimates place data loggers at 26% share in 2026. Buyers continue to favor this format where post-arrival review is sufficient, especially in lanes where device recovery, subscription cost, and live alert management would add more burden than value. Real-time trackers are rising in premium lanes, but broad category volume still sits with formats that are easier to validate and easier to scale. Freeze indicators and shock indicators remain useful where a single excursion type is the main concern, yet they do not replace the wider role of a logger when teams need a defensible condition record across the full trip. Temperature trackers add value in connected programs, but not every route needs that degree of supervision.
- Qualification fit: Data loggers match existing shipment-review routines with less training burden for quality teams and fewer interpretation disputes after delivery.
- Deployment ease: Single-trip use and simple retrieval options keep adoption broad across lanes where device turnaround speed matters.
- Record clarity: Downloadable shipment evidence supports release, investigation, and lane comparison without requiring continuous monitoring across every shipment.
Cold Chain Integrity and Shock Monitoring Test Devices Market Analysis by Monitoring Parameter
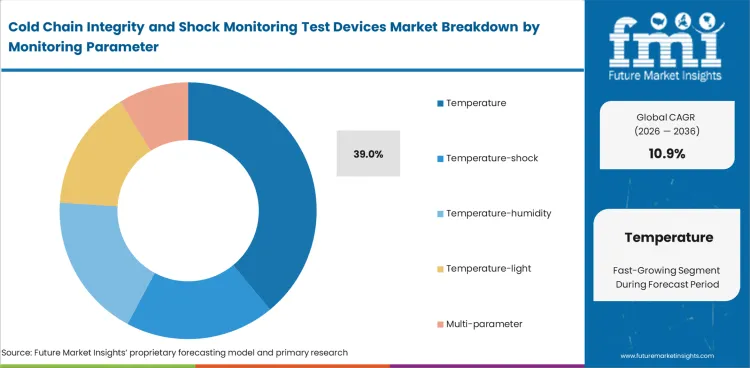
Temperature remains the first condition most teams must verify before any broader handling discussion begins. For that reason, this parameter is expected to account for 39% share in 2026. A temperature reading alone does not explain every handling event, yet it still carries the clearest connection to product viability across pharmaceuticals, vaccines, diagnostics, and chilled perishables. Buyers usually add shock, humidity, or light monitoring only after temperature control is already in place or after a recurring deviation pattern makes secondary evidence necessary. Temperature-shock and temperature-humidity combinations are rising because they reduce blind spots during review, though they also cost more and often require clearer lane justification. Multi-parameter devices are strongest where the product is expensive enough, the route is variable enough, or the release burden is strict enough to reward richer evidence. Temperature-controlled pharma routes are pushing that direction further, though baseline category use still rests on temperature.
- Primary threshold: Temperature remains the first reading quality teams review because it most directly influences release confidence and deviation triage.
- Escalation path: Secondary parameters become more relevant when repeated route issues make temperature-only evidence too narrow for investigation.
- Lane matching: Multi-parameter formats suit premium shipments, but broader category use still favors the parameter that addresses the most common control need.
Cold Chain Integrity and Shock Monitoring Test Devices Market Analysis by Connectivity
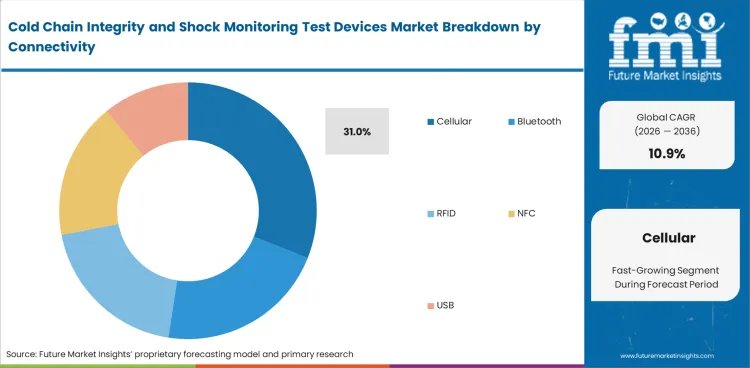
Connected monitoring no longer sits only in pilot programs. Cellular-enabled devices are projected to secure 31% share in 2026 because more high-value routes now benefit from alerts during the trip rather than after the shipment is opened. That advantage is strongest where product value, route complexity, or service risk makes delayed visibility expensive. Bluetooth, RFID, NFC, and USB formats still serve important roles, especially in shorter routes or lower-cost programs where live transmission is not justified. Cellular leads because it reduces the gap between an excursion and the response window, which matters when teams need to intervene before the shipment reaches final review. Battery life, service availability, and subscription cost still guide adoption. Healthcare cold-chain programs are helping normalize connected devices, but buying discipline remains selective.
- Intervention value: Cellular connectivity gives logistics teams a chance to act while the shipment is still in motion instead of discovering the issue at receipt.
- Coverage trade-off: Broader visibility comes with service cost and device-management needs that users weigh against lane value.
- Format coexistence: Offline and near-field options retain relevance where route control is steadier and post-arrival review remains adequate.
Cold Chain Integrity and Shock Monitoring Test Devices Market Analysis by Deployment Mode
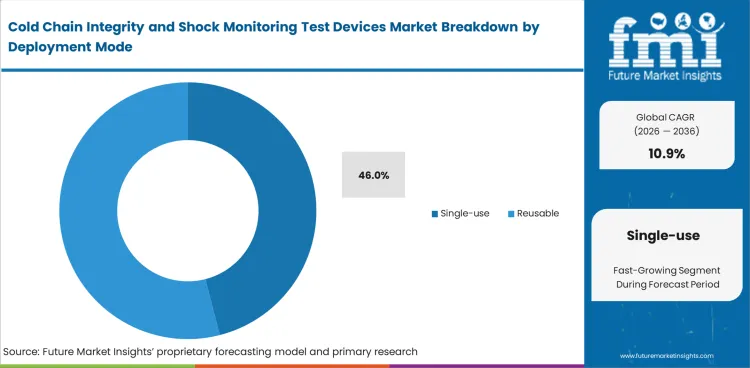
Return handling, device recovery, and turnaround discipline often decide whether a reusable format truly lowers monitoring cost. Single-use devices are likely to represent 46% of the market in 2026 because many shipment programs still prefer a cleaner deployment model with fewer retrieval steps and fewer questions around reuse condition. Preference is strongest in regulated lanes where chain-of-custody clarity matters and where recovery logistics could delay the next shipment cycle. Reusable devices still appeal in closed-loop programs and higher-frequency routes, but they work best when the shipper already has the processes to retrieve, inspect, reset, and redeploy them without service loss. Single-use formats hold broader reach because they match one-trip shipment logic more easily. Reusable biologics programs are widening the conversation, yet one-way monitored movement still fits a large share of day-to-day cold-chain activity.
- Recovery burden: Single-use deployment avoids device return delays that can weaken service consistency across distributed shipping networks.
- Cycle control: Reusable formats make more sense where route repetition is high and device turnaround can be managed without missed dispatches.
- Validation comfort: One-trip use reduces uncertainty around prior handling, reset quality, and readiness for the next monitored shipment.
Cold Chain Integrity and Shock Monitoring Test Devices Market Analysis by Packaging Level
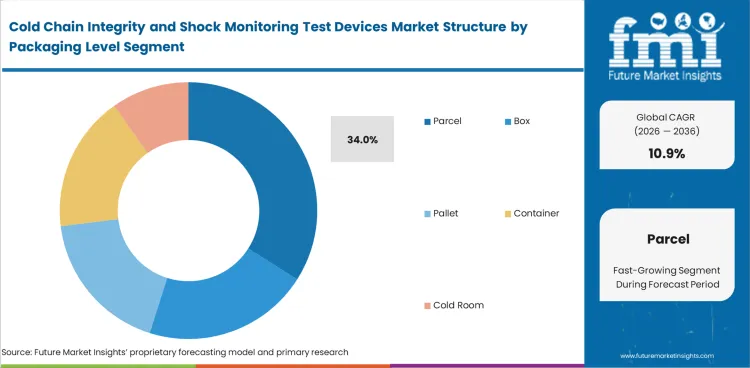
Parcel-level shipping has become an important proving ground for monitored cold-chain movement because more temperature-sensitive consignments now move in smaller, validated units rather than in large consolidated loads alone. Share contribution from parcel-level monitoring is expected to reach 34% in 2026. Parcel use rises when specialty therapies, diagnostics, and urgent replenishment programs require tighter shipment identity and clearer accountability at the unit being received. Box, pallet, container, and cold-room monitoring remain important for broader logistics control, but parcel-level devices are better aligned with the points where release, rejection, or investigation decisions are actually made. Smaller shipment size also raises the value of attaching monitoring to the exact consignment rather than to a larger aggregated load. Vaccine carriers and isothermal boxes support this direction where handling precision matters.
- Unit accountability: Parcel monitoring ties the reading directly to the shipment being accepted, reducing ambiguity during receiving checks.
- Handling precision: Smaller consignments face more handoffs, making device attachment at parcel level more relevant for condition review.
- Review speed: Shipment-level evidence helps receiving teams resolve acceptance decisions without tracing data from a larger load grouping.
Cold Chain Integrity and Shock Monitoring Test Devices Market Analysis by End Use
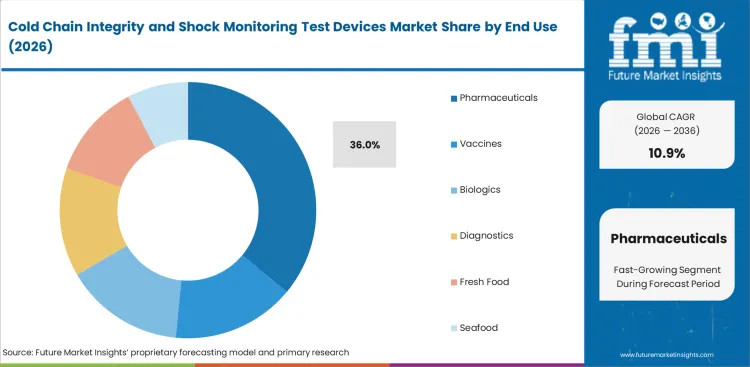
Product sensitivity, release discipline, and documentation burden keep pharmaceuticals at the center of this category. Pharmaceuticals are anticipated to account for 36% share in 2026 across end use because more shipments in this lane require condition evidence that can support quality review rather than a simple delivery confirmation. Lead position comes from the way pharmaceuticals combine higher product value with tighter handling expectations and a lower tolerance for unclear excursion history. Vaccines and biologics follow closely in strategic importance, while diagnostics add another layer of use where time and temperature exposure directly affect usability. Fresh food and seafood use monitoring differently. Their buying logic can be strong in export and premium routes, but it remains more selective when product value cannot absorb premium device deployment. Vaccine temperature control continues to reinforce device awareness, though pharmaceuticals still anchor broad category valuation.
- Release discipline: Pharmaceutical shipments require condition evidence that can stand up to internal review before product is cleared for use.
- Value density: Higher shipment value supports broader use of monitoring devices that would be too expensive for many lower-value perishables.
- Documentation need: Pharmaceutical lanes often need a cleaner record trail, which keeps device adoption deeper than in less regulated cold-chain categories.
Cold Chain Integrity and Shock Monitoring Test Devices Market Analysis by Application Stage
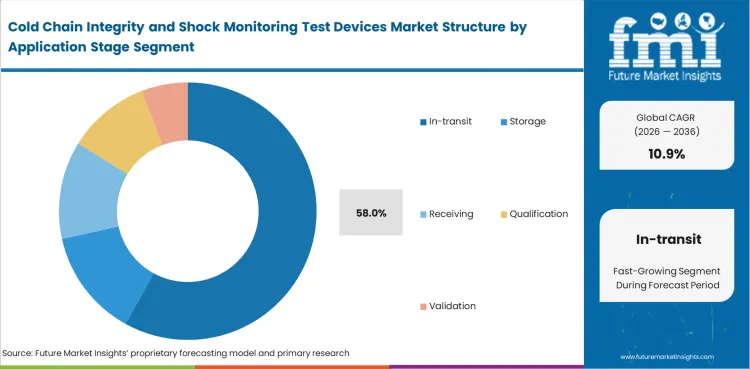
Most device use gathers around the point where product is exposed to route variation, handoff error, delay, or handling damage. In-transit use is set to make up 58% of the market in 2026. That lead reflects where cold-chain risk concentrates most clearly and where device readings carry the greatest practical value for intervention, investigation, or acceptance review. Storage, receiving, qualification, and validation all remain important application stages, especially where facilities need baseline control or route designs must be tested before routine deployment begins. Yet once the shipment is moving, small handling failures can become larger product questions very quickly. Buyers therefore prioritize devices that clarify what happened during movement before they widen monitoring depth elsewhere.
- Risk concentration: Transit is where delay, loading error, route variation, and physical handling issues most often challenge product condition.
- Decision leverage: In-transit readings give receiving and quality teams better context when a shipment arrives with an exception.
- Program sequencing: Many shippers begin with transit monitoring first, then widen use into storage or validation once lane priorities are clearer.
Cold Chain Integrity and Shock Monitoring Test Devices Market Drivers, Restraints, and Opportunities
Condition review is becoming harder to manage with simple pass-fail assumptions when more cold-chain shipments carry higher product value, more route handoffs, and tighter release requirements. Quality teams need device evidence that can explain what happened during transit, not merely confirm that a monitor was attached. That is pushing device choice toward formats that fit lane risk, response needs, and review burden with greater precision. Live monitoring rises where intervention matters. Simpler devices remain preferred where deployment speed and review clarity matter more than continuous visibility. Refrigerated transport and refrigerated trailers increase the importance of route-level control, but device adoption still depends on shipment value and the consequence of uncertainty at receipt.
Internal qualification cycles and inconsistent device deployment rules still slow category expansion. A shipper may recognize the need for richer monitoring, yet adoption stalls when lane definitions are unclear, review teams interpret readings differently, or returns and resets make deployment harder than expected. Reusable programs face that issue more often, though connected device rollouts can face it as well when alert management is not clearly assigned. Cost matters, but operating discipline matters just as much. A monitoring device that creates confusion after delivery can lose favor even if its technical capability is strong. Insulated truck logistics adds route complexity, and that complexity raises the penalty for device-selection mistakes.
Opportunities in the Cold Chain Integrity and Shock Monitoring Test Devices Market
- Lane stratification: Suppliers that help shippers assign the right device to the right route can win share by reducing overspecification on stable lanes and weak visibility on high-risk lanes.
- Multi-sensor adoption: Better uptake is likely where biologics, specialty therapies, and fragile diagnostics need condition evidence that combines temperature with handling context in one reading.
- Service bundling: Device suppliers linked to smart packaging and biologic distribution programs can gain traction when monitoring support is sold as part of a broader shipment-control offering.
Regional Analysis
Key Facts About Country
- At 12.8% CAGR, India is advancing through 2036, supported by expansion in pharmaceutical logistics, vaccine distribution, and increasing use of monitored shipment verification.
- China is progressing at 11.9% CAGR, with adoption linked to manufacturing scale and wider deployment of monitored export and domestic distribution lanes.
- Brazil is expected to grow at 11.1% CAGR, where longer transport routes and variable handling conditions increase reliance on shipment-level condition tracking.
- Supported by established pharmaceutical distribution systems and integration of monitoring into release workflows, the United States is likely to expand steadily, reaching 10.3% CAGR.
- Germany is moving at 9.7% CAGR, where validation discipline and preference for documented shipment control support consistent device use.
- With adoption tied to specialty medicine distribution and the need for clear shipment evidence at receiving, the United Kingdom is projected to register 9.5% CAGR.
- Japan is forecast to expand at 8.8% CAGR, reflecting a mature installed base where replacement cycles and reliability requirements guide procurement decisions.
Based on the regional analysis, the Cold Chain Integrity and Shock Monitoring Test Devices market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 12.8% |
| China | 11.9% |
| Brazil | 11.1% |
| United States | 10.3% |
| Germany | 9.7% |
| United Kingdom | 9.5% |
| Japan | 8.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Cold Chain Integrity and Shock Monitoring Test Devices Market Analysis
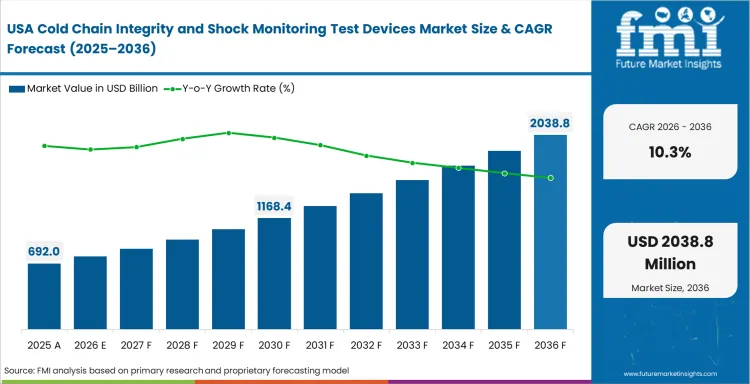
Cold-chain monitoring in North America is being shaped by product value, route accountability, and the need to reduce uncertainty at receipt rather than by hardware novelty alone. Buyers in this region tend to evaluate devices against release confidence, exception handling speed, and fit with established quality routines. That gives an edge to suppliers that can support both device performance and workflow clarity. Cold-chain monitoring requirements run deeper in life science than in many food lanes, which keeps device use concentrated where the cost of a disputed shipment outcome is highest. Route length and handoff density also keep live visibility relevant in premium lanes. Regional industry outlook is on a positive trend, but it remains selective and lane-specific rather than universal across all cold-chain movement.
- United States: Pharmaceutical distribution depth, biologics handling complexity, and a wide installed base of monitored temperature-sensitive routes keep the United States at the center of regional industry outlook. Live visibility is being adopted where product value and route variation justify it, while simpler loggers continue to serve broad shipment programs that need reliable review without constant oversight. Industry outlook in the United States points to a CAGR of 10.3% from 2026 to 2036. Wider use of shipment-level evidence is improving how receiving teams handle deviations, and that is raising the operating value of devices that fit existing release routines.
FMI's report includes Canada and Mexico. Cross-border cold-chain movement and expanding pharmaceutical logistics activity in these countries continue to improve the case for route-specific monitoring rather than one-format deployment across every shipment type.
Latin America Cold Chain Integrity and Shock Monitoring Test Devices Market Analysis
Distance, route variation, and uneven cold-chain execution make device choice in Latin America a more practical shipping decision than a simple compliance purchase. Monitoring depth rises where exporters, pharmaceutical distributors, and vaccine handlers need cleaner shipment evidence across longer transit windows and more variable handoffs. That keeps the region favorable for devices that balance usability with credible condition reporting rather than devices that maximize feature count alone. Vaccine transport and cold-chain packaging needs support device use, but local adoption still depends on whether the monitoring format can be deployed without adding too much retrieval, reset, or interpretation burden.
- Brazil: Brazil is likely to post a CAGR of 11.1% in this sector by 2036. Long domestic distribution routes and a wider need to maintain shipment integrity across varied handling conditions keep Brazil on a stronger upward path than many neighboring markets. Cold-chain programs in pharmaceuticals and immunization logistics increasingly need shipment records that can support acceptance decisions with less ambiguity. Suppliers that match route complexity with practical device deployment are in a better position to gain repeat use, especially where shipment review needs to move quickly after arrival.
FMI's report includes Argentina, Chile, Colombia, and the rest of Latin America. Regional opportunity remains strongest where route conditions make shipment evidence more valuable than a basic temperature check alone.
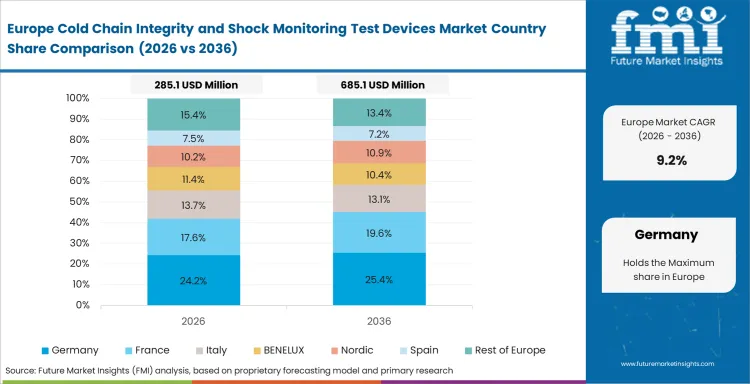
Europe and Asia Pacific Cold Chain Integrity and Shock Monitoring Test Devices Market Analysis
Mature quality systems in Europe and expanding cold-chain depth across Asia Pacific create two different adoption paths inside one broader regional discussion. Europe favors device consistency, validation comfort, and clearer fit with established release processes. Asia Pacific is adding device use through manufacturing scale, healthcare logistics expansion, and wider use of monitored transport in developing cold-chain networks. That mix supports both stable replacement demand and first-time deployment. Healthcare cold-chain, pharma logistics, and life-science movement all reinforce the need for route-matched monitoring. Device suppliers that can serve both mature validation-heavy programs and faster-rising distribution networks have a wider opening across this combined region.
- Germany: A CAGR of 9.7% is expected for this market in Germany through 2036. Validation discipline, pharmaceutical manufacturing presence, and a steady preference for documented shipment control keep Germany relevant even though its expansion rate is below faster-rising Asian countries. Quality teams in this market tend to favor monitoring devices that can be integrated cleanly into receiving review and deviation handling rather than tools that add reading complexity. Reliable deployment and documentation matter as much as device sophistication, which gives consistency real commercial weight in supplier selection.
- United Kingdom: Specialty medicine distribution, biologics handling requirements, and a strong need for defensible shipment evidence continue to support the United Kingdom market. Buyers in this country often evaluate monitoring formats through the lens of service reliability and review clarity, especially where product availability and patient timing can be affected by a disputed shipment outcome. Demand analysis in the United Kingdom is expected to move ahead at a CAGR of 9.5% over the forecast period. Programs that reduce ambiguity after receipt are likely to gain ground faster than tools that add more data without simplifying action.
- India: India is projected to record a CAGR of 12.8% in this market through 2036. Cold-chain expansion in pharmaceuticals, vaccines, and temperature-sensitive healthcare distribution keeps India on the strongest upward path among the countries assessed. Shipment monitoring is becoming more relevant because wider healthcare distribution and broader product sensitivity make undocumented handling harder to absorb. Lane-based device selection is still evolving, which leaves room for suppliers that can support both simple deployment and clearer shipment review.
- China: Manufacturing scale, export exposure, and increasing use of monitored transport in life science distribution keep China close behind India in forecast pace. Buyers in China are balancing larger cold-chain volumes with rising expectations around handling visibility, which favors devices that can support both domestic and export-facing shipment programs. Industry outlook in China points to a CAGR of 11.9% through 2036. Broader uptake of connected and multi-parameter devices is likely to appear first in routes where handling complexity and shipment value justify richer evidence.
- Japan: Adoption in Japan is anticipated to move ahead at a CAGR of 8.8% during the assessment period. Replacement demand carries more weight in Japan than in faster-rising markets, yet that does not reduce the country’s importance in regional valuation. Mature cold-chain handling practices and a preference for reliability keep device use tied to consistency, product quality, and operating fit. Buyers that prioritize deployment discipline over feature expansion are likely to keep simpler, dependable formats relevant for longer.
FMI's report includes France, Italy, Spain, Benelux, Nordic regions, South Korea, ASEAN, Australia, New Zealand, and the rest of Asia Pacific and Europe. Mature Western European programs continue to favor validation comfort and service consistency, while several Asia Pacific countries are building device use through expanding healthcare logistics, wider manufacturing activity, and a greater need for shipment-level condition evidence.
Competitive Aligners for Market Players
Competition in this market is moderate rather than tightly concentrated because users still have room to choose among specialist monitoring suppliers, but not every supplier can satisfy regulated shipment needs with equal consistency. Sensitech, Controlant, ELPRO, SpotSee, DeltaTrak, Berlinger, and Tive compete on reliability of readings, ease of deployment, quality-review fit, and the ability to support different shipment lanes without creating extra confusion after delivery. Users do not choose on device capability alone. Service reliability, calibration credibility, reading clarity, and support during exception handling often matter just as much as the device hardware itself.
Incumbents hold an advantage where they can pair known device performance with dependable support across pharmaceuticals and other temperature-sensitive lanes. That advantage is difficult to replicate quickly because replacement decisions are rarely based on price alone once a device has already been accepted into established monitoring routines. Challengers can still gain share when they reduce setup burden, improve live visibility for premium lanes, or support clearer route-level monitoring decisions across connected tracking and shipment control. Users will continue to reward suppliers that make condition evidence easier to interpret and easier to act on.
Large shippers usually resist vendor lock-in by assigning different device types to different route categories instead of giving every lane to one supplier. That behavior keeps room open for specialists in freeze indication, shock detection, or connected tracking even when broader monitoring programs sit with larger vendors. Category direction through 2036 is likely to favor suppliers that can hold consistency in established programs while adapting device choice to lane-specific need. Workflow fit matters more than feature escalation alone.
Key Players in Cold Chain Integrity and Shock Monitoring Test Devices Market
- Sensitech
- Controlant
- ELPRO
- SpotSee
- DeltaTrak
- Berlinger
- Tive
Scope of the Report

| Metric | Value |
|---|---|
| Quantitative Units | USD 1.1 billion to USD 3.1 billion, at a CAGR of 10.9% |
| Market Definition | Cold chain integrity and shock monitoring test devices include hardware used to record, indicate, or transmit shipment condition during storage and transport for temperature-sensitive and impact-sensitive goods. Scope centers on the device layer that supports condition evidence, release review, and deviation handling across monitored cold-chain movement. |
| Device Type Segmentation | Data Loggers, Real-time Trackers, Freeze Indicators, Shock Indicators, TTI Labels, VVM Labels |
| Monitoring Parameter Segmentation | Temperature, Temperature-shock, Temperature-humidity, Temperature-light, Multi-parameter |
| Connectivity Segmentation | Cellular, Bluetooth, RFID, NFC, USB |
| Deployment Mode Segmentation | Single-use, Reusable |
| Packaging Level Segmentation | Parcel, Box, Pallet, Container, Cold Room |
| End Use Segmentation | Pharmaceuticals, Vaccines, Biologics, Diagnostics, Fresh Food, Seafood |
| Application Stage Segmentation | In-transit, Storage, Receiving, Qualification, Validation |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | India, China, Brazil, United States, Germany, United Kingdom, Japan, and 40 plus countries |
| Key Companies Profiled | Sensitech, Controlant, ELPRO, SpotSee, DeltaTrak, Berlinger, Tive |
| Forecast Period | 2026 to 2036 |
| Approach | FMI combined primary interviews with shipment-monitoring category review, supplier triangulation, and end-use lane assessment. Baseline sizing was anchored to monitored shipment use, installed monitoring points, and vendor exposure across regulated and higher-value cold-chain programs. Forecasts were validated against replacement cycle logic, device deployment patterns, and end-use adoption behavior. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Cold Chain Integrity and Shock Monitoring Test Devices Market Analysis by Segments
Device Type
- Data Loggers
- Real-time Trackers
- Freeze Indicators
- Shock Indicators
- TTI Labels
- VVM Labels
Monitoring Parameter
- Temperature
- Temperature-shock
- Temperature-humidity
- Temperature-light
- Multi-parameter
Connectivity
- Cellular
- Bluetooth
- RFID
- NFC
- USB
Deployment Mode
- Single-use
- Reusable
Packaging Level
- Parcel
- Box
- Pallet
- Container
- Cold Room
End Use
- Pharmaceuticals
- Vaccines
- Biologics
- Diagnostics
- Fresh Food
- Seafood
Application Stage
- In-transit
- Storage
- Receiving
- Qualification
- Validation
Region
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Colombia
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Benelux
- Nordic Regions
- East Asia
- China
- Japan
- South Korea
- South Asia
- India
- ASEAN
- Oceania
- Australia
- New Zealand
- Middle East & Africa
- GCC Countries
- South Africa
- Rest of Middle East & Africa
Bibliography
- World Health Organization. (2024). Temperature monitoring devices. WHO Performance, Quality and Safety (PQS) system.
- World Health Organization. (2025). Data logger and machine-to-machine interface for Equipment Monitoring Systems: PQS independent type-testing protocol, E006/DL01-VP.3. WHO Performance, Quality and Safety (PQS) system.
- United States Pharmacopeia. (2024). General Chapter <1079.4> Temperature Mapping for the Qualification of Storage Areas. USP.
- Controlant hf. (2025). Results of Controlant’s Annual General Meeting - 5 May 2025. Controlant.
- Duman, E., Yıldız, B., & Aytekin, O. (2025). Enhancing Traceability and Reliability in Cold Chain Logistics Through Hyperledger Fabric and IoT.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Market intelligence to support decision making across data loggers, real-time trackers, freeze indicators, shock indicators, TTI labels, and VVM labels
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, supported by monitored-shipment logic, supplier triangulation, and end-use lane evaluation
- Opportunity mapping across device type, monitoring parameter, connectivity, deployment mode, packaging level, end use, and application stage with emphasis on lane-based device selection
- Segment and regional revenue forecasts covering pharmaceuticals, temperature monitoring, cellular connectivity, single-use deployment, parcel-level use, and in-transit application across regulated and high-value cold-chain programs
- Competition assessment including service reliability, reading clarity, calibration credibility, route fit, and support during exception handling
- Device-category tracking including data loggers, real-time trackers, freeze indicators, shock indicators, multi-parameter monitoring, and shipment-level connected visibility
- Market access analysis covering monitored pharmaceutical distribution, vaccine handling, biologics transport, diagnostics movement, and validated cold-chain receiving review
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive planning, cold-chain planning, and operating benchmarking use
Frequently Asked Questions
How big is this market in 2026?
Cold Chain Integrity and Shock Monitoring Test Devices Market is expected to reach USD 1.1 billion in 2026.
How much could it be worth by 2036?
Market valuation is projected to reach USD 3.1 billion by 2036, supported by wider use across monitored shipment lanes.
What CAGR is expected from 2026 to 2036?
A CAGR of 10.9% is projected from 2026 to 2036, indicating steady category expansion through the forecast period.
What are cold chain integrity and shock monitoring test devices?
These devices check whether shipments stayed within acceptable temperature and handling limits during storage, transit, and receiving review.
Which device type leads the market?
Data loggers lead the device type segment and are expected to account for 26% share in 2026.
Which monitoring parameter leads the market?
Temperature monitoring leads this segment and is projected to represent 39% share in 2026 across the market.
Which connectivity type leads the market?
Cellular connectivity leads the connectivity segment, with 31% share expected in 2026 due to stronger real-time visibility needs.
Cellular connectivity leads the connectivity segment, with 31% share expected in 2026 due to stronger real-time visibility needs.
More temperature-sensitive shipments need clearer condition records, making monitoring devices more relevant across regulated and high-value lanes.
What is the main limit on wider use?
Adoption slows when route rules, alert handling, and review steps are not clearly defined within shipping programs.
Adoption slows when route rules, alert handling, and review steps are not clearly defined within shipping programs.
India is moving ahead the fastest, with a projected CAGR of 12.8% through 2036 in this market.
Why does route type matter so much?
Route type matters because shipment value, handling variation, and response needs differ widely across cold-chain delivery programs.
Why are multi-sensor devices getting more attention?
Multi-sensor devices attract more attention because temperature alone cannot explain every shipment event or handling problem.
What do users compare when choosing suppliers?
Users compare reading reliability, deployment ease, support quality, and fit with shipment review routines.
Why do single-use devices still stay ahead of reusable ones?
Single-use devices stay ahead because they avoid return handling, reset work, and uncertainty around previous device use.
Why is parcel-level monitoring important?
Parcel-level monitoring matters because it links the reading directly to the shipment being accepted or rejected.
Why do pharmaceuticals lead end use?
Pharmaceuticals lead end use because shipments need tighter handling control, clearer records, and better release review support.
Why is in-transit application the biggest one?
In-transit application leads because most delay, route variation, and handling risk appears while goods are moving.
How is the United States positioned in this market?
United States industry outlook remains strong due to established pharmaceutical logistics, biologics handling, and monitored shipment programs.
Why is Brazil on a stronger upward path?
Brazil is on a stronger upward path because longer routes increase the value of shipment condition evidence.
Why does Germany still matter with a lower CAGR than India or China?
Germany still matters because mature pharmaceutical handling and documentation discipline keep device use commercially important.
What supports the United Kingdom market?
Specialty medicine distribution and biologics handling support the United Kingdom market, where clear shipment evidence remains important.
What supports China’s outlook?
China’s outlook is supported by manufacturing scale and wider use of monitored cold-chain transport across domestic and export lanes.
Why does Japan rise more moderately?
Japan rises more moderately because replacement demand carries more weight in an already mature monitored-shipment environment.
What is included in this market?
This market includes data loggers, real-time trackers, indicators, and related monitoring hardware used in cold-chain storage.
What is the difference between a logger and an indicator?
A logger records condition data for later review, while an indicator shows whether a specific limit was crossed.
How was this market assessed?
Assessment combined primary interviews, supplier triangulation, monitored-shipment logic, and end-use lane evaluation for forecast validation.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Device Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Device Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Device Type , 2026 to 2036
- Data Loggers
- Real-time Trackers
- Freeze Indicators
- Shock Indicators
- TTI Labels
- VVM Labels
- Data Loggers
- Y-o-Y Growth Trend Analysis By Device Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Device Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Monitoring Parameter
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Monitoring Parameter, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Monitoring Parameter, 2026 to 2036
- Temperature
- Temperature-shock
- Temperature-humidity
- Temperature-light
- Multi-parameter
- Temperature
- Y-o-Y Growth Trend Analysis By Monitoring Parameter, 2021 to 2025
- Absolute $ Opportunity Analysis By Monitoring Parameter, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Connectivity
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Connectivity, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Connectivity, 2026 to 2036
- Cellular
- Bluetooth
- RFID
- NFC
- USB
- Cellular
- Y-o-Y Growth Trend Analysis By Connectivity, 2021 to 2025
- Absolute $ Opportunity Analysis By Connectivity, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment Mode
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment Mode, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment Mode, 2026 to 2036
- Single-use
- Reusable
- Single-use
- Y-o-Y Growth Trend Analysis By Deployment Mode, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment Mode, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Packaging Level
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Packaging Level, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Packaging Level, 2026 to 2036
- Parcel
- Box
- Pallet
- Container
- Cold Room
- Parcel
- Y-o-Y Growth Trend Analysis By Packaging Level, 2021 to 2025
- Absolute $ Opportunity Analysis By Packaging Level, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharmaceuticals
- Vaccines
- Biologics
- Diagnostics
- Fresh Food
- Seafood
- Pharmaceuticals
- Y-o-Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application Stage
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application Stage, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application Stage, 2026 to 2036
- In-transit
- Storage
- Receiving
- Qualification
- Validation
- In-transit
- Y-o-Y Growth Trend Analysis By Application Stage, 2021 to 2025
- Absolute $ Opportunity Analysis By Application Stage, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- By Country
- Market Attractiveness Analysis
- By Country
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Device Type
- By Monitoring Parameter
- By Connectivity
- By Deployment Mode
- By Packaging Level
- By End Use
- By Application Stage
- Competition Analysis
- Competition Deep Dive
- Sensitech
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Controlant
- ELPRO
- SpotSee
- DeltaTrak
- Berlinger
- Tive
- Sensitech
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Monitoring Parameter, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Packaging Level, 2021 to 2036
- Table 7: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 8: Global Market Value (USD Million) Forecast by Application Stage, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Monitoring Parameter, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 13: North America Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 14: North America Market Value (USD Million) Forecast by Packaging Level, 2021 to 2036
- Table 15: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 16: North America Market Value (USD Million) Forecast by Application Stage, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 19: Latin America Market Value (USD Million) Forecast by Monitoring Parameter, 2021 to 2036
- Table 20: Latin America Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 21: Latin America Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 22: Latin America Market Value (USD Million) Forecast by Packaging Level, 2021 to 2036
- Table 23: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 24: Latin America Market Value (USD Million) Forecast by Application Stage, 2021 to 2036
- Table 25: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Western Europe Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 27: Western Europe Market Value (USD Million) Forecast by Monitoring Parameter, 2021 to 2036
- Table 28: Western Europe Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 29: Western Europe Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 30: Western Europe Market Value (USD Million) Forecast by Packaging Level, 2021 to 2036
- Table 31: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 32: Western Europe Market Value (USD Million) Forecast by Application Stage, 2021 to 2036
- Table 33: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 34: Eastern Europe Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 35: Eastern Europe Market Value (USD Million) Forecast by Monitoring Parameter, 2021 to 2036
- Table 36: Eastern Europe Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 37: Eastern Europe Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 38: Eastern Europe Market Value (USD Million) Forecast by Packaging Level, 2021 to 2036
- Table 39: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 40: Eastern Europe Market Value (USD Million) Forecast by Application Stage, 2021 to 2036
- Table 41: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 42: East Asia Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 43: East Asia Market Value (USD Million) Forecast by Monitoring Parameter, 2021 to 2036
- Table 44: East Asia Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 45: East Asia Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 46: East Asia Market Value (USD Million) Forecast by Packaging Level, 2021 to 2036
- Table 47: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 48: East Asia Market Value (USD Million) Forecast by Application Stage, 2021 to 2036
- Table 49: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 50: South Asia and Pacific Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 51: South Asia and Pacific Market Value (USD Million) Forecast by Monitoring Parameter, 2021 to 2036
- Table 52: South Asia and Pacific Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 53: South Asia and Pacific Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 54: South Asia and Pacific Market Value (USD Million) Forecast by Packaging Level, 2021 to 2036
- Table 55: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 56: South Asia and Pacific Market Value (USD Million) Forecast by Application Stage, 2021 to 2036
- Table 57: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 58: Middle East & Africa Market Value (USD Million) Forecast by Device Type , 2021 to 2036
- Table 59: Middle East & Africa Market Value (USD Million) Forecast by Monitoring Parameter, 2021 to 2036
- Table 60: Middle East & Africa Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 61: Middle East & Africa Market Value (USD Million) Forecast by Deployment Mode, 2021 to 2036
- Table 62: Middle East & Africa Market Value (USD Million) Forecast by Packaging Level, 2021 to 2036
- Table 63: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 64: Middle East & Africa Market Value (USD Million) Forecast by Application Stage, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Device Type
- Figure 6: Global Market Value Share and BPS Analysis by Monitoring Parameter, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Monitoring Parameter, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Monitoring Parameter
- Figure 9: Global Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Connectivity
- Figure 12: Global Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Deployment Mode
- Figure 15: Global Market Value Share and BPS Analysis by Packaging Level, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Packaging Level, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Packaging Level
- Figure 18: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by End Use
- Figure 21: Global Market Value Share and BPS Analysis by Application Stage, 2026 and 2036
- Figure 22: Global Market Y-o-Y Growth Comparison by Application Stage, 2026-2036
- Figure 23: Global Market Attractiveness Analysis by Application Stage
- Figure 24: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 25: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 26: Global Market Attractiveness Analysis by Region
- Figure 27: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 29: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 30: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 31: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 32: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 33: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 34: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 35: North America Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Device Type
- Figure 38: North America Market Value Share and BPS Analysis by Monitoring Parameter, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Monitoring Parameter, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Monitoring Parameter
- Figure 41: North America Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Connectivity
- Figure 44: North America Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 45: North America Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 46: North America Market Attractiveness Analysis by Deployment Mode
- Figure 47: North America Market Value Share and BPS Analysis by Packaging Level, 2026 and 2036
- Figure 48: North America Market Y-o-Y Growth Comparison by Packaging Level, 2026-2036
- Figure 49: North America Market Attractiveness Analysis by Packaging Level
- Figure 50: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 51: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 52: North America Market Attractiveness Analysis by End Use
- Figure 53: North America Market Value Share and BPS Analysis by Application Stage, 2026 and 2036
- Figure 54: North America Market Y-o-Y Growth Comparison by Application Stage, 2026-2036
- Figure 55: North America Market Attractiveness Analysis by Application Stage
- Figure 56: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 57: Latin America Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Device Type
- Figure 60: Latin America Market Value Share and BPS Analysis by Monitoring Parameter, 2026 and 2036
- Figure 61: Latin America Market Y-o-Y Growth Comparison by Monitoring Parameter, 2026-2036
- Figure 62: Latin America Market Attractiveness Analysis by Monitoring Parameter
- Figure 63: Latin America Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 64: Latin America Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 65: Latin America Market Attractiveness Analysis by Connectivity
- Figure 66: Latin America Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 67: Latin America Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 68: Latin America Market Attractiveness Analysis by Deployment Mode
- Figure 69: Latin America Market Value Share and BPS Analysis by Packaging Level, 2026 and 2036
- Figure 70: Latin America Market Y-o-Y Growth Comparison by Packaging Level, 2026-2036
- Figure 71: Latin America Market Attractiveness Analysis by Packaging Level
- Figure 72: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 73: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 74: Latin America Market Attractiveness Analysis by End Use
- Figure 75: Latin America Market Value Share and BPS Analysis by Application Stage, 2026 and 2036
- Figure 76: Latin America Market Y-o-Y Growth Comparison by Application Stage, 2026-2036
- Figure 77: Latin America Market Attractiveness Analysis by Application Stage
- Figure 78: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 79: Western Europe Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 80: Western Europe Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 81: Western Europe Market Attractiveness Analysis by Device Type
- Figure 82: Western Europe Market Value Share and BPS Analysis by Monitoring Parameter, 2026 and 2036
- Figure 83: Western Europe Market Y-o-Y Growth Comparison by Monitoring Parameter, 2026-2036
- Figure 84: Western Europe Market Attractiveness Analysis by Monitoring Parameter
- Figure 85: Western Europe Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 86: Western Europe Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 87: Western Europe Market Attractiveness Analysis by Connectivity
- Figure 88: Western Europe Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 89: Western Europe Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 90: Western Europe Market Attractiveness Analysis by Deployment Mode
- Figure 91: Western Europe Market Value Share and BPS Analysis by Packaging Level, 2026 and 2036
- Figure 92: Western Europe Market Y-o-Y Growth Comparison by Packaging Level, 2026-2036
- Figure 93: Western Europe Market Attractiveness Analysis by Packaging Level
- Figure 94: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 95: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 96: Western Europe Market Attractiveness Analysis by End Use
- Figure 97: Western Europe Market Value Share and BPS Analysis by Application Stage, 2026 and 2036
- Figure 98: Western Europe Market Y-o-Y Growth Comparison by Application Stage, 2026-2036
- Figure 99: Western Europe Market Attractiveness Analysis by Application Stage
- Figure 100: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 101: Eastern Europe Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 102: Eastern Europe Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 103: Eastern Europe Market Attractiveness Analysis by Device Type
- Figure 104: Eastern Europe Market Value Share and BPS Analysis by Monitoring Parameter, 2026 and 2036
- Figure 105: Eastern Europe Market Y-o-Y Growth Comparison by Monitoring Parameter, 2026-2036
- Figure 106: Eastern Europe Market Attractiveness Analysis by Monitoring Parameter
- Figure 107: Eastern Europe Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 108: Eastern Europe Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 109: Eastern Europe Market Attractiveness Analysis by Connectivity
- Figure 110: Eastern Europe Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 111: Eastern Europe Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 112: Eastern Europe Market Attractiveness Analysis by Deployment Mode
- Figure 113: Eastern Europe Market Value Share and BPS Analysis by Packaging Level, 2026 and 2036
- Figure 114: Eastern Europe Market Y-o-Y Growth Comparison by Packaging Level, 2026-2036
- Figure 115: Eastern Europe Market Attractiveness Analysis by Packaging Level
- Figure 116: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 117: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 118: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 119: Eastern Europe Market Value Share and BPS Analysis by Application Stage, 2026 and 2036
- Figure 120: Eastern Europe Market Y-o-Y Growth Comparison by Application Stage, 2026-2036
- Figure 121: Eastern Europe Market Attractiveness Analysis by Application Stage
- Figure 122: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 123: East Asia Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 124: East Asia Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 125: East Asia Market Attractiveness Analysis by Device Type
- Figure 126: East Asia Market Value Share and BPS Analysis by Monitoring Parameter, 2026 and 2036
- Figure 127: East Asia Market Y-o-Y Growth Comparison by Monitoring Parameter, 2026-2036
- Figure 128: East Asia Market Attractiveness Analysis by Monitoring Parameter
- Figure 129: East Asia Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 130: East Asia Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 131: East Asia Market Attractiveness Analysis by Connectivity
- Figure 132: East Asia Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 133: East Asia Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 134: East Asia Market Attractiveness Analysis by Deployment Mode
- Figure 135: East Asia Market Value Share and BPS Analysis by Packaging Level, 2026 and 2036
- Figure 136: East Asia Market Y-o-Y Growth Comparison by Packaging Level, 2026-2036
- Figure 137: East Asia Market Attractiveness Analysis by Packaging Level
- Figure 138: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 139: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 140: East Asia Market Attractiveness Analysis by End Use
- Figure 141: East Asia Market Value Share and BPS Analysis by Application Stage, 2026 and 2036
- Figure 142: East Asia Market Y-o-Y Growth Comparison by Application Stage, 2026-2036
- Figure 143: East Asia Market Attractiveness Analysis by Application Stage
- Figure 144: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 145: South Asia and Pacific Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 146: South Asia and Pacific Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 147: South Asia and Pacific Market Attractiveness Analysis by Device Type
- Figure 148: South Asia and Pacific Market Value Share and BPS Analysis by Monitoring Parameter, 2026 and 2036
- Figure 149: South Asia and Pacific Market Y-o-Y Growth Comparison by Monitoring Parameter, 2026-2036
- Figure 150: South Asia and Pacific Market Attractiveness Analysis by Monitoring Parameter
- Figure 151: South Asia and Pacific Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 152: South Asia and Pacific Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 153: South Asia and Pacific Market Attractiveness Analysis by Connectivity
- Figure 154: South Asia and Pacific Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 155: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 156: South Asia and Pacific Market Attractiveness Analysis by Deployment Mode
- Figure 157: South Asia and Pacific Market Value Share and BPS Analysis by Packaging Level, 2026 and 2036
- Figure 158: South Asia and Pacific Market Y-o-Y Growth Comparison by Packaging Level, 2026-2036
- Figure 159: South Asia and Pacific Market Attractiveness Analysis by Packaging Level
- Figure 160: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 161: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 162: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 163: South Asia and Pacific Market Value Share and BPS Analysis by Application Stage, 2026 and 2036
- Figure 164: South Asia and Pacific Market Y-o-Y Growth Comparison by Application Stage, 2026-2036
- Figure 165: South Asia and Pacific Market Attractiveness Analysis by Application Stage
- Figure 166: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 167: Middle East & Africa Market Value Share and BPS Analysis by Device Type , 2026 and 2036
- Figure 168: Middle East & Africa Market Y-o-Y Growth Comparison by Device Type , 2026-2036
- Figure 169: Middle East & Africa Market Attractiveness Analysis by Device Type
- Figure 170: Middle East & Africa Market Value Share and BPS Analysis by Monitoring Parameter, 2026 and 2036
- Figure 171: Middle East & Africa Market Y-o-Y Growth Comparison by Monitoring Parameter, 2026-2036
- Figure 172: Middle East & Africa Market Attractiveness Analysis by Monitoring Parameter
- Figure 173: Middle East & Africa Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 174: Middle East & Africa Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 175: Middle East & Africa Market Attractiveness Analysis by Connectivity
- Figure 176: Middle East & Africa Market Value Share and BPS Analysis by Deployment Mode, 2026 and 2036
- Figure 177: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment Mode, 2026-2036
- Figure 178: Middle East & Africa Market Attractiveness Analysis by Deployment Mode
- Figure 179: Middle East & Africa Market Value Share and BPS Analysis by Packaging Level, 2026 and 2036
- Figure 180: Middle East & Africa Market Y-o-Y Growth Comparison by Packaging Level, 2026-2036
- Figure 181: Middle East & Africa Market Attractiveness Analysis by Packaging Level
- Figure 182: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 183: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 184: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 185: Middle East & Africa Market Value Share and BPS Analysis by Application Stage, 2026 and 2036
- Figure 186: Middle East & Africa Market Y-o-Y Growth Comparison by Application Stage, 2026-2036
- Figure 187: Middle East & Africa Market Attractiveness Analysis by Application Stage
- Figure 188: Global Market - Tier Structure Analysis
- Figure 189: Global Market - Company Share Analysis

