Real-Time Temperature and Location Monitoring Platforms for Cold Chain Market
The Real-Time Temperature And Location Monitoring Platforms For Cold Chain Market Is Segmented By Component (Software Platforms, IoT Trackers, Gateways, Analytics Services), Deployment (Cloud, On-Premise, Hybrid), Connectivity (Cellular, Bluetooth, RFID, Satellite, LPWAN), Asset Scope (Shipments, Storage Sites, Reefers, Containers, Pallets), End Use (Pharmaceuticals, Food, Vaccines, Biologics, Chemicals), And Region. Forecast For 2026 To 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Real-Time Temperature and Location Monitoring Platforms for Cold Chain Market Size, Market Forecast and Outlook By FMI
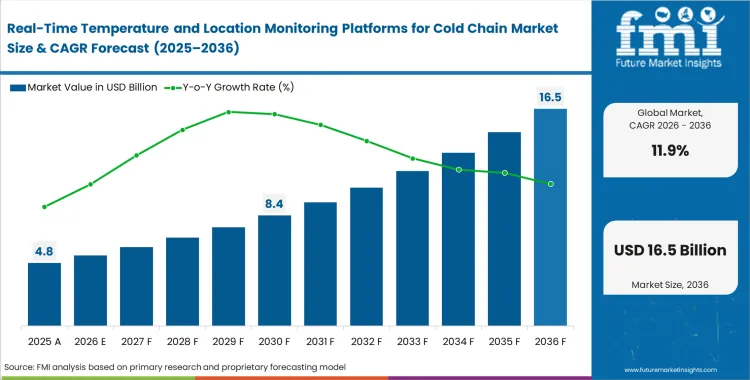
The real-time temperature and location monitoring platforms for cold chain market crossed a valuation of USD 4.3 billion in 2025, with the increasing demand poised to cross USD 4.8 billion in 2026 at a CAGR of 11.9% during forecast parameters. Persistent revenue expansion carries total industry valuation to USD 14.8 billion through 2036 as logistics coordinators transition from retrospective compliance logging to a real-time cold chain tracking platform that allows for predictive load-loss intervention before temperature deviations breach critical thresholds.
Summary of Real-Time Temperature and Location Monitoring Platforms for Cold Chain Market
- The market is forecast to reach USD 14.8 billion by 2036.
- The market is expected to grow at a CAGR of 11.9% from 2026 to 2036.
- The market was estimated at USD 4.3 billion in 2025.
- The forecast period represents an incremental opportunity of USD 10.0 billion.
- The market focuses on digital platforms that combine real-time temperature monitoring, location tracking, alerts, and workflow automation for cold chain logistics.
- Growth is driven by stringent regulatory requirements for pharmaceuticals and perishable goods, requiring continuous digital monitoring instead of manual checks.
- Adoption is increasing as companies shift toward real-time intervention to prevent cargo loss during transit.
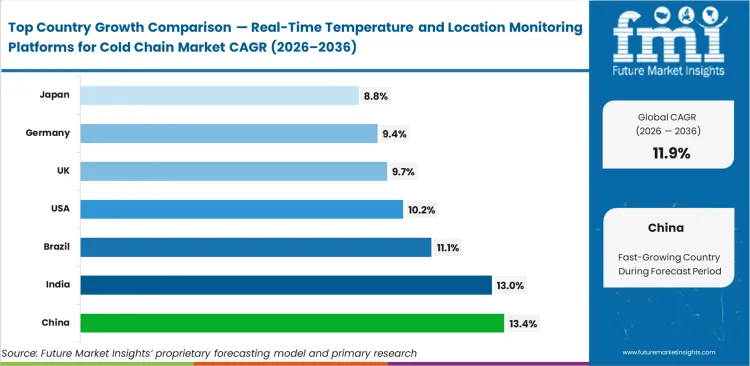
- China, India, and Brazil are among the fastest-growing markets, with China leading at a 13.4% CAGR through 2036.
- Cloud-based software platforms and cellular connectivity dominate due to scalability and reliable long-distance tracking.
- Pharmaceuticals remain the leading end-use segment due to strict compliance and high-value cargo sensitivity.
- Key players in the market include Sensitech, ORBCOMM, Controlant, Tive, ELPRO, and SkyCell.
Quality assurance teams are under growing pressure to reduce product loss when shipment history becomes unclear during carrier handovers. Reviewing cold chain records only after delivery often leaves little room to judge whether a short temperature excursion actually affected product quality. That uncertainty can push companies to discard usable shipments simply because they cannot validate what happened in transit. Demand for cold chain visibility platforms is rising from this need for clearer evidence, faster decisions, and tighter control over temperature-sensitive cargo.
Integration of IoT-based cold chain monitoring software with business systems is expected to replace manual checks with continuous shipment oversight. Real-time visibility helps teams connect temperature deviations to specific route stages, transfer points, and carrier movements with greater accuracy. Control tower platforms are also gaining importance as businesses look to identify at-risk shipments earlier and respond before product value is lost. Adoption is being shaped by a broader need for stronger traceability across pharmaceutical, biologics, and food logistics networks.
China is projected to record a CAGR of 13.4% in cold chain visibility platform demand during 2026 to 2036, supported by export supply chains that require consistent temperature validation across multiple transit points. Demand in India is likely to expand at a CAGR of 13.0% over the forecast period, driven by wider immunization coverage and stricter handling needs across healthcare distribution. Brazil is anticipated to register an 11.1% CAGR through 2036, reflecting demand tied to agricultural exports that depend on controlled transport conditions. In the U.S., cold chain visibility platform demand is expected to rise at a CAGR of 10.2% during 2026 to 2036, supported by the scale of biologic drug distribution. U.K. demand is estimated to increase at a CAGR of 9.7% over the same period as cross-border shipment complexity keeps environmental tracking in focus. Germany is projected to grow at a CAGR of 9.4% through 2036, with demand linked to specialized chemical and healthcare logistics. Japan is set to expand at a CAGR of 8.8% during the forecast period, supported by healthcare supply chains that require tighter shipment validation.
Segmental Analysis
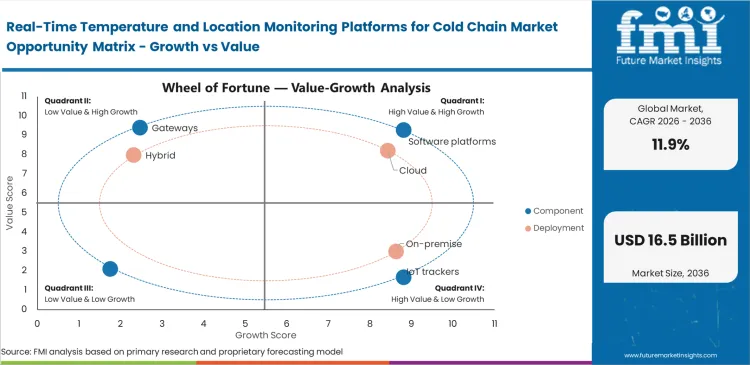
Real-Time Temperature and Location Monitoring Platforms for Cold Chain Market Analysis by Component
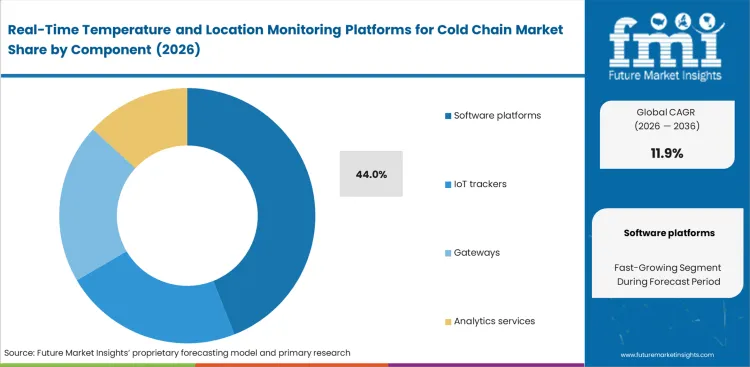
Hardware commoditization is pushing technology providers to build value through specialized exception-management algorithms rather than physical devices. Software-led exception management is estimated to account for 44.0% share in 2026, as demand shifts toward predictive analytics and centralized control. Integrating connected sensors into centralized interfaces allows faster quarantine action when deviations are detected. Standalone software purchases often leave fragmented systems in place when legacy carrier networks limit clean data handoffs. Delayed movement toward unified platforms keep supply chains tied to manual reconciliation and raises administrative burden.
- • Trigger Condition: Compliance audits that expose incomplete transit records are expected to increase demand for centralized data aggregation.
- • Validation Phase: Platform assessment is likely to focus on integration with existing enterprise resource planning systems to support low-latency data flow.
- • Expansion Metric: Wider deployment is projected to follow once quarantine workflows are automated across additional distribution routes.
Real-Time Temperature and Location Monitoring Platforms for Cold Chain Market Analysis by Deployment
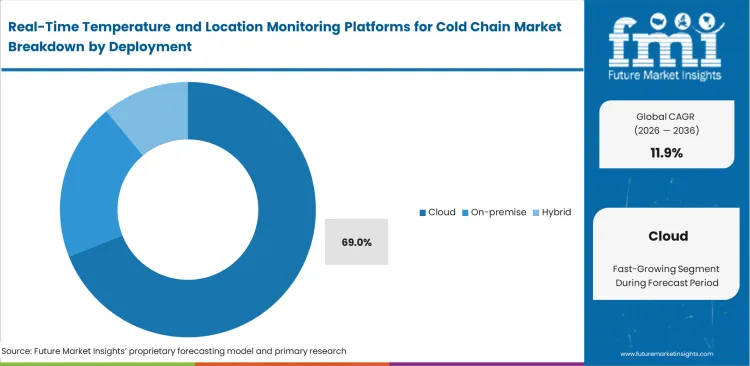
Multi-stakeholder visibility requirements push data hosting outside corporate firewalls for frictionless cross-border accessibility. Utilizing sensor data analytics hosted externally lets contract manufacturers, carriers, and final dispensers monitor identical environmental baselines simultaneously. On-premise installations remain technically viable for closed networks, but shared external liability requires broad system integration. Driven by this necessity for universal access, cloud configurations are likely to hold a share of an expected 69.0% in 2026, solidifying external hosting as the industry standard. Facilities insisting on internal data storage struggle to onboard temporary transport partners during seasonal volume spikes.
- Initial Outlays: Sourcing specialists prioritize cloud subscriptions to shift environmental monitoring costs from capital expenditures to predictable operating expenses.
- Hidden Burdens: Network administrators discover continuous high-frequency data streaming from global pharmaceutical cold chain packaging assets quickly inflates monthly cellular data consumption tiers.
- Lifecycle Comparison: Supply chain heads calculate avoiding single biologic batch losses entirely offsets three years of premium cloud hosting fees.
Real-Time Temperature and Location Monitoring Platforms for Cold Chain Market Analysis by Connectivity
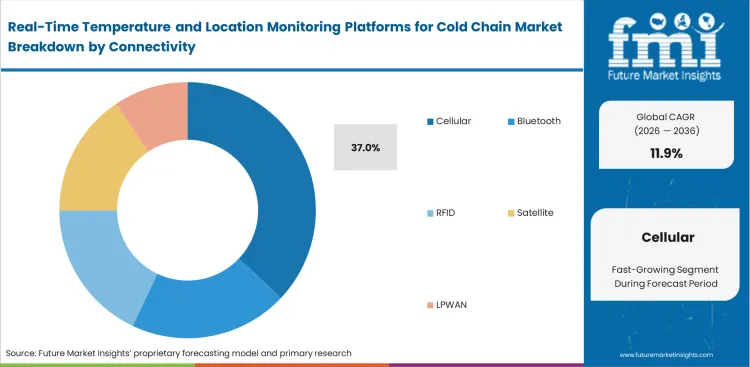
Autonomous reporting capabilities make the cellular category poised to garner 37.0% share in 2026, driven by the operational need to eliminate dependence on fixed warehouse infrastructure. Active tracking nodes transmit location and thermal data directly to central databases without requiring intermediate healthcare cold chain logistics gateways. Relying on short-range alternatives leaves quality assurance units blind during critical multimodal transfer windows. Cellular tracking vulnerability lies strictly in aviation regulations forcing devices into sleep modes when tarmac delays create high thermal risks. Supply chains must balance these localized signal blackouts against the overarching benefit of global network availability.
- Signal Interruption: Aviation compliance mandates force tracking nodes to cease transmission mid-flight, creating blind spots for quality assurance coordinators during transit.
- Residual Vulnerability: Logistics heads acknowledge cellular protocols drain batteries rapidly, risking total data loss on unexpectedly delayed international shipments.
- Recovery Action: Supply chain heads implement predictive algorithms to calculate thermal decay during planned signal blackouts, bridging reporting gaps automatically.
Real-Time Temperature and Location Monitoring Platforms for Cold Chain Market Analysis by Asset Scope
Regulatory scrutiny increasingly targets mobile product units rather than the ambient conditions inside transport vehicles. Tracking individual cold chain logistics packages provides indisputable evidence of viability even if primary cooling units experience temporary compressor failure. With this shift toward item-level telemetry, the shipments segment is estimated to account for a projected 42.0% share in 2026, as enterprises prioritize localized thermal stability over broad trailer metrics. Pallet-level monitoring satisfies general compliance standards but fails to expose exact thermal variations based on physical placement near doors. Organizations relying solely on vehicle-level sensors frequently discard intact products simply because they cannot prove specific container integrity.
- Hardware Production: Sensor manufacturers miniaturize components continuously, enabling logistics coordinators to place discrete trackers inside smaller shipment volumes.
- Capacity Constraints: Supply chain officers struggle to source single-use tracking nodes during peak pharmaceutical distribution seasons, disrupting compliance protocols.
- Trajectory Alignment: Hardware recycling programs emerge to recover and refurbish shipment-level sensors, reducing per-trip operational costs for distributors.
Real-Time Temperature and Location Monitoring Platforms for Cold Chain Market Analysis by End Use
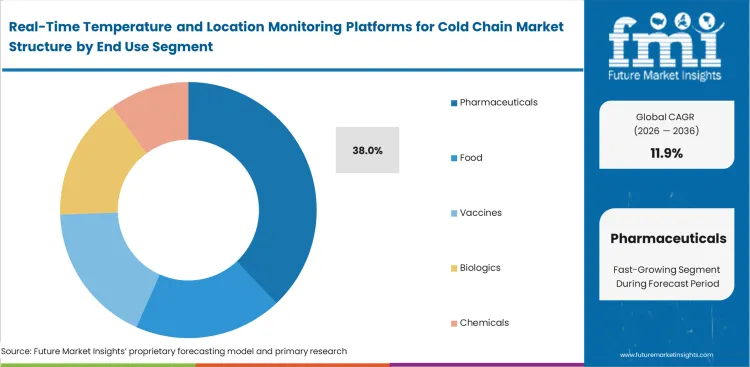
Zero-tolerance therapeutic efficacy thresholds compel immediate digitization of thermal records across highly regulated supply networks. Utilizing advanced temperature controlled packaging solutions combined with real-time telemetry prevents the administration of inactive drug compounds. Dispensaries failing to verify complete transit histories face severe regulatory penalties and immediate license suspensions. Driven by personalized cell therapies requiring ultra-low temperature maintenance with zero allowable deviations, pharmaceuticals are set to represent an estimated 38.0% of market share in 2026, setting the baseline for precision. General food distributors accept ambient monitoring, but medical formulation compliance demands granular fraction-of-a-degree sensitivity.
- Baseline Expectations: General food distributors accept ambient monitoring, but quality assurance heads in the pharmaceutical industry cold chain monitoring vertical require granular fraction-of-a-degree sensitivity.
- Edge Conditions: Ultra-low temperature transit utilizing liquid nitrogen challenges sensor resilience, forcing logistics officers to source specialized cryogenic probes.
- Qualification Metrics: Supply chain coordinators validate hardware exclusively against strict pharmacopeial standards to ensure legal compliance during cross-border transfers.
Real-Time Temperature and Location Monitoring Platforms for Cold Chain Market Drivers, Restraints, and Opportunities
Insurance providers now demand absolute proof of proper temperature control before paying spoilage claims. Strict requirement forces shipping companies to digitize their thermal records completely. Manufacturers relying on outdated manual transit logs face immediate premium increases or total coverage denial for high-value biological shipments. Third-party logistics firms currently rewrite their standard service agreements to require continuous data streaming. Such contractual change creates an urgent need for live tracking just to remain competitive in the transport sector.
Getting reusable tracking devices back from their final destination creates a major operational hurdle. Companies calculate their shipping budgets expecting to use a single sensor dozens of times. Local clinics or pharmacies are encouraged to mail these physical trackers back after receiving a delivery proves surprisingly difficult in daily practice. Constant loss of hardware forces manufacturers to continuously buy replacements, erasing the expected cost savings. Single-use cellular trackers solve the return problem entirely, thereby, leading to their high individual purchase price currently restricts their use to highly expensive shipments like specialized pharmaceuticals rather than everyday perishable goods.
- Automated Claims Execution: Direct links between temperature data and digital insurance contracts allow providers to authorize spoilage payouts the exact moment a thermal breach is verified.
- Predictive Thermal Forecasting: Analyzing past route data helps carriers anticipate exact temperature shifts inside a temperature monitoring device long before a transport vehicle leaves the loading dock.
- Carbon Emission Tracking: Tying cooling engine performance to live environmental data allows organizations to measure the precise carbon footprint of every refrigerated delivery.
Regional Analysis
Regional demand heavily depends on local regulatory strictness and the density of pharmaceutical manufacturing. Strict export mandates drive rapid adoption in Asian supply chains, while mature Western networks focus on compliance integration and navigating cross-border customs friction.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 13.4% |
| India | 13.0% |
| Brazil | 11.1% |
| United States | 10.2% |
| United Kingdom | 9.7% |
| Germany | 9.4% |
| Japan | 8.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia-Pacific Real-Time Temperature and Location Monitoring Platforms for Cold Chain Market Analysis
Export mandates regarding active pharmaceutical ingredients dictate immediate upgrades to continuous nodal visibility. Facilities handling chemical intermediates recognize international buyers reject batches lacking verifiable environmental transit histories. Integrating with maritime tracking networks helps manufacturers maintain line-of-sight during lengthy oceanic voyages. Sourcing reefer container telematics allows origin facilities to monitor internal ambient conditions while assets sit in congested port terminals.
- China: Export pharmaceutical mandates shift enforcement from end-point verification to continuous nodal visibility. Demand for the cold chain monitoring market China is anticipated to rise at a CAGR of 13.4% from 2026 to 2036, as organizations rely on telemetry to bypass extended customs quarantine procedures at destination ports. Sourcing specialized hardware secures lucrative supply contracts with strict European pharmaceutical buyers.
- India: Massive infrastructure upgrades to national healthcare distribution networks expose systemic vulnerabilities in manual temperature logging. Pharmaceutical companies implement continuous tracking to reduce vaccine spoilage during extended rural transit, pushing sales of a cold chain visibility platform in India to increase at an expected CAGR of 13.0% during the forecast period. Demonstrating verifiable chain-of-custody opens participation in heavily regulated international clinical trials.
- Japan: Demand for cold chain monitoring in Japan is projected to expand at a CAGR of 8.8% during 2026 to 2036, supported by healthcare distribution needs tied to an aging population and rising movement of temperature-sensitive biologics. Advanced sensors are increasingly used to reduce spoilage risk across dense urban last-mile delivery networks. Wider platform adoption is expected to improve shipment validation and lower product rejection across hospital supply chains.
FMI assesses that regional tracking architectures are increasingly standardizing around selected cellular bandwidths to support more consistent cross-border data continuity. Coverage in the report also extends to South Korea and ASEAN countries.
North America & Latin America Real-Time Temperature and Location Monitoring Platforms for Cold Chain Market Analysis
Biologics cold chain visibility establishes baseline expectations for fraction-of-a-degree thermal precision across the Americas. Quality assurance officers discard legacy passive indicators in favor of active nodes transmitting live intervention alerts. Supply chains prioritize software platforms capable of aggregating data from multiple competing hardware vendors. Securing specialized reefer container for pharmaceutical transport ensures compliance with strict federal sanitary transportation regulations.
- Brazil: Specialized agricultural export requirements demand verifiable transit histories to clear strict international phytosanitary inspections. Brazil is set to record a CAGR of 11.1% in refrigerated logistics monitoring platform demand during the assessment period because logistics providers rely on continuous telemetry to prove perishable cargo maintained optimal conditions during lengthy maritime journeys. Securing these capabilities differentiates premium logistics providers in highly commoditized regional transport sectors.
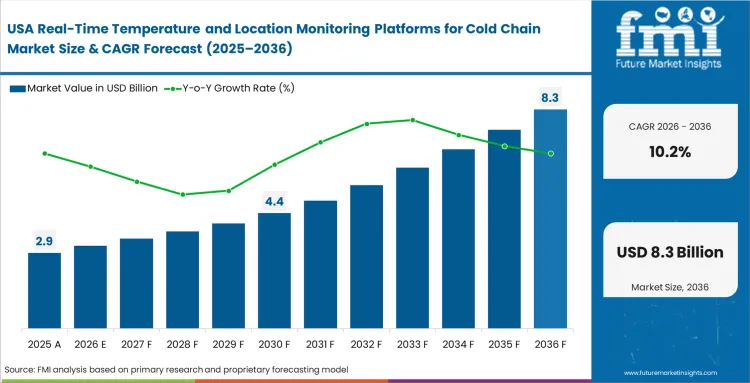
- United States: Federal supply chain security acts mandate comprehensive traceability for all commercial pharmaceutical distributions. Manufacturers deploy advanced analytics dashboards to automate compliance reporting and isolate liability instantly, poising US adoption to advance at a CAGR of 10.2% by 2036. Integrating life sciences logistics visibility software fundamentally shifts organizational posture from reactive damage control to proactive yield protection.
FMI's report includes Canada and Mexico. Cross-border customs friction necessitates tighter environmental controls to preserve product viability during unpredictable border inspection delays.
Europe Real-Time Temperature and Location Monitoring Platforms for Cold Chain Market Analysis
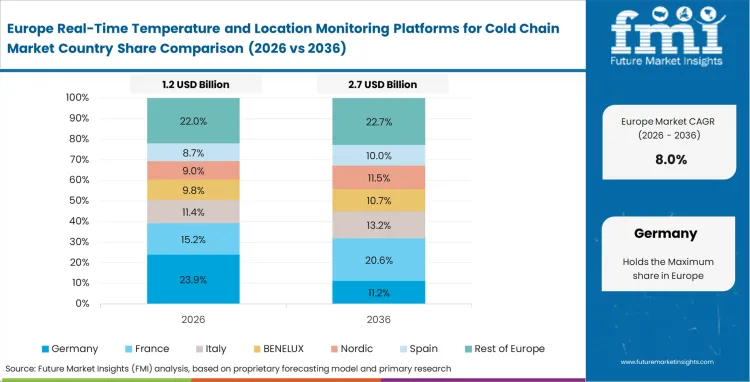
Heavy concentration of specialized chemical manufacturing necessitates rigorous environmental controls during intra-continental transport. Logistics providers face immense pressure to maintain strict good distribution practices across multimodal handoffs. Supply chains evaluate platforms based on the ability to execute automated quality release protocols immediately upon shipment arrival. Utilizing advanced telemetry eliminates traditional administrative delays typically required to review manual temperature logs.
- United Kingdom: Demand for pharma cold chain visibility software in the United Kingdom is projected to expand at a CAGR of 9.7% during 2026 to 2036, supported by cross-border customs complexity that is increasing the need for tighter environmental monitoring. Real-time sensors are increasingly used to track biologic shipments held at border inspection points and to maintain clearer in-transit condition records. Adoption is expected to remain linked to spoilage control, shipment validation, and stronger visibility across temperature-sensitive pharmaceutical flows.
- Germany: High-value automotive and chemical precursor exports require precise thermal conditioning to prevent material degradation. The Germany’s industry sector is expected to register a CAGR of 9.4% through 2036 as logistics providers utilize live reefer temperature alerts to intercept failing refrigerated trailer assets. This standardization synchronizes quality control across disparate contract manufacturing networks.
FMI analysis suggests that retrospective data records are becoming less acceptable, which is pushing cold chain operations toward continuous active monitoring. Coverage in the report also includes France, Italy, and Spain.
Competitive Aligners for Market Players
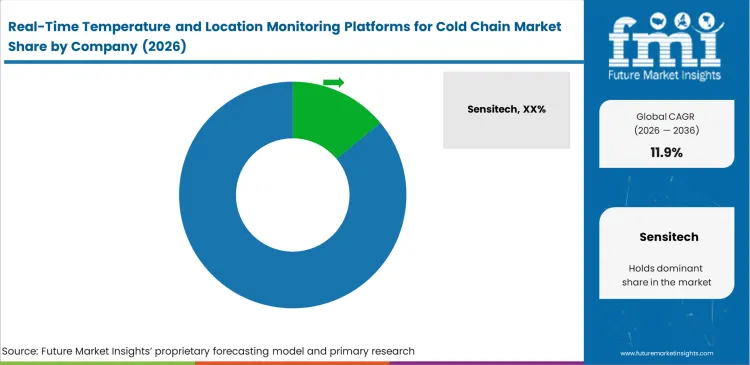
Cold chain monitoring hardware is becoming harder to differentiate, so competition is shifting toward software quality and how well platforms connect across logistics networks. Buyers are placing more weight on whether a system can pull data from multiple transport partners without extra manual work. Even capable tracking devices can lose relevance when links with inventory, planning, and shipment systems remain weak. Value in this market is moving toward cleaner interfaces, stronger connectivity, and consistent data visibility from one shipment point to the next.
Established suppliers still benefit from compliance readiness built over years of working within air and sea freight conditions. Experience in transport safety, device handling, and route acceptance gives those providers an edge that newer entrants cannot build quickly. Pharmaceutical cold chain logistics depends on more than device accuracy alone. Shipment approval, dependable execution, and validated handling standards all shape buyer confidence in tightly controlled transport settings.
Large end users are also leaning toward life science logistics software that can work with more than one sensor brand. That approach helps preserve shipment history and monitoring continuity when hardware vendors change. As a result, hardware competition is narrowing around battery life, durability, and performance in the field. Software competition is becoming more important, especially where providers can offer better route visibility, faster exception handling, and stronger control across connected sensor networks.
Key Players in Real-Time Temperature and Location Monitoring Platforms for Cold Chain Market
- Sensitech
- ORBCOMM
- Controlant
- Tive
- ELPRO
- SkyCell
Scope of the Report
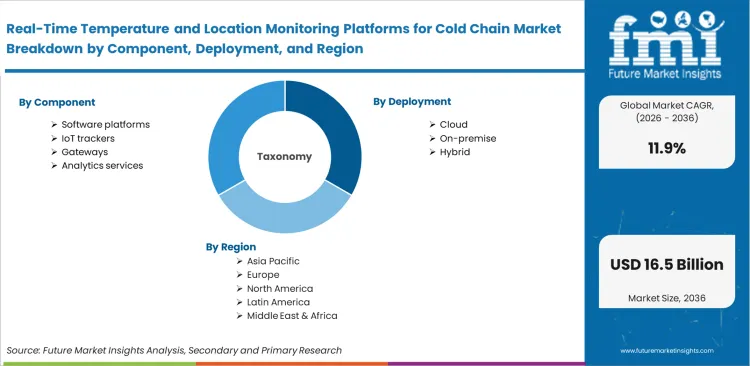
| Metric | Value |
|---|---|
| Quantitative Units | USD 4.8 billion to USD 14.8 billion, at a CAGR of 11.9% |
| Market Definition | Integrated hardware, connectivity layers, and software architectures deployed to continuously capture, transmit, and analyze environmental data during temperature-sensitive transit. Operations depend on these systems to maintain unbroken chain-of-custody records, trigger automated excursion alerts, and model product viability from origin facilities to final dispensation points. |
| Segmentation | Component, Deployment, Connectivity, Asset scope, End use, and Region |
| Regions Covered | Asia-Pacific, North America & Latin America, Europe, Middle East & Africa |
| Countries Covered | China, India, Brazil, United States, United Kingdom, Germany, Japan |
| Key Companies Profiled | Sensitech, ORBCOMM, Controlant, Tive, ELPRO, SkyCell |
| Forecast Period | 2026 to 2036 |
| Approach | Active telematics subscription volumes and specialized reefer container production rates anchor baseline measurements. Independent carrier API integration logs cross-validate enterprise software deployment forecasts. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Segments
Component
- Software platforms
- IoT trackers
- Gateways
- Analytics services
Deployment
- Cloud
- On-premise
- Hybrid
Connectivity
- Cellular
- Bluetooth
- RFID
- Satellite
- LPWAN
Asset Scope
- Shipments
- Storage sites
- Reefers
- Containers
- Pallets
End Use
- Pharmaceuticals
- Food
- Vaccines
- Biologics
- Chemicals
Regions:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- Centers for Disease Control and Prevention. (2024). Vaccine storage and handling toolkit.
- European Medicines Agency. (2024). GDP inspection procedure.
- World Health Organization. (2026). Cold chain equipment and dry store temperature mapping tool.
- Ayowole, D. J., Adebajo, G. O., Lasisi, T. O., & Bakai, J. G. (2025). Effects of climate change on vaccine storage and cold chain logistics: a qualitative study in Ogun State, Nigeria. BMJ Global Health, 10(7), e018990.
- Chan, Y. J., Anwar, N., & Reuel, N. F. (2024). Cold and ultra-cold chain integrity monitoring via embedded resonant sensor indicators. Sensors and Actuators A: Physical, 372, 115346.
- Kamyod, C., Arwatchananukul, S., Aunsri, N., Saengrayap, R., Tontiwattanakul, K., Prahsarn, C., Trongsatitkul, T., Lerslerwong, L., Mahajan, P., Kim, C.-G., Wu, D., & Chaiwong, S. (2025). IoT-Based System for Real-Time Monitoring and AI-Driven Energy Consumption Prediction in Fresh Fruit and Vegetable Transportation. Sensors, 25(24), 7475.
- Meng, X., Xie, R., Liao, J., Shen, X., & Yang, S. (2024). A cost-effective over-temperature alarm system for cold chain delivery. Journal of Food Engineering, 368, 111914.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Software platforms driving margin capture through specialized exception-management algorithms.
- Cloud cold chain dashboard architectures enabling frictionless cross-border accessibility for multi-stakeholder supply chains.
- Cellular connectivity specifications executing autonomous reporting without fixed local gateways.
- Shipment-level telemetry providing localized thermal stability evidence for biologics cold chain visibility.
- Strict pharmacopeial standards dictating sensor validation requirements for clinical trial shipment monitoring.
- China export mandates forcing transitions from end-point verification to continuous nodal visibility.
- Insurance underwriters demanding real-time temperature and location monitoring platforms for cold chain to process spoilage claims.
- Hardware-agnostic cold chain control tower software preventing single-vendor lock-in across pharmaceutical networks.
Frequently Asked Questions
What triggers initial purchase decisions for software dashboards?
Compliance audits revealing incomplete records force quality assurance specialists to seek centralized data aggregation.
How do aviation regulations impact cellular nodes?
Strict aviation mandates force tracking nodes to cease transmission mid-flight, creating temporary blind spots requiring predictive modeling.
What is a real-time temperature and location monitoring platform for cold chain?
It is an integrated hardware and software solution that provides continuous environmental telemetry during transit.
How is a live monitoring platform different from a standard data logger?
Connected platforms transmit data actively to trigger real-time shipment temperature monitoring alerts, while loggers require manual extraction.
Which industries use real-time cold chain visibility tools the most?
The pharma, vaccine, and perishable food sectors utilize these platforms to maintain product efficacy and safety.
Why do pharma companies prefer real-time cold chain monitoring over offline logging?
Live tracking allows for immediate intervention during excursions, preventing catastrophic batch losses for sensitive biologics.
What standards apply to cold chain monitoring software?
Compliance with GDP, FSMA, and WHO PQS guidelines ensures software meets rigorous data integrity and thermal accuracy requirements.
Which countries are growing fastest in this market?
China and India lead global growth as they modernize logistics networks and satisfy international pharmaceutical export mandates.
Who are the main vendors in real-time cold chain visibility?
Market leaders include Sensitech, ORBCOMM, Controlant, and Tive each providing specialized IoT and software solutions.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Component
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Component , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Component , 2026 to 2036
- Software platforms
- IoT trackers
- Gateways
- Analytics services
- Software platforms
- Y to o to Y Growth Trend Analysis By Component , 2021 to 2025
- Absolute $ Opportunity Analysis By Component , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment, 2026 to 2036
- Cloud
- On-premise
- Hybrid
- Cloud
- Y to o to Y Growth Trend Analysis By Deployment, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Connectivity
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Connectivity, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Connectivity, 2026 to 2036
- Cellular
- Bluetooth
- RFID
- Satellite
- LPWAN
- Cellular
- Y to o to Y Growth Trend Analysis By Connectivity, 2021 to 2025
- Absolute $ Opportunity Analysis By Connectivity, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Asset Scope
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Asset Scope, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Asset Scope, 2026 to 2036
- Shipments
- Storage sites
- Reefers
- Containers
- Pallets
- Shipments
- Y to o to Y Growth Trend Analysis By Asset Scope, 2021 to 2025
- Absolute $ Opportunity Analysis By Asset Scope, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharmaceuticals
- Food
- Vaccines
- Biologics
- Chemicals
- Pharmaceuticals
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Component
- By Deployment
- By Connectivity
- By Asset Scope
- By End Use
- Competition Analysis
- Competition Deep Dive
- Sensitech
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- ORBCOMM
- Controlant
- Tive
- ELPRO
- SkyCell
- Sensitech
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Asset Scope, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Asset Scope, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Asset Scope, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Asset Scope, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Asset Scope, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Asset Scope, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Asset Scope, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Component , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Connectivity, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Asset Scope, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Component
- Figure 6: Global Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Deployment
- Figure 9: Global Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Connectivity
- Figure 12: Global Market Value Share and BPS Analysis by Asset Scope, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Asset Scope, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Asset Scope
- Figure 15: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by End Use
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Component
- Figure 32: North America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Deployment
- Figure 35: North America Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Connectivity
- Figure 38: North America Market Value Share and BPS Analysis by Asset Scope, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Asset Scope, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Asset Scope
- Figure 41: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by End Use
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Component
- Figure 48: Latin America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Deployment
- Figure 51: Latin America Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Connectivity
- Figure 54: Latin America Market Value Share and BPS Analysis by Asset Scope, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Asset Scope, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Asset Scope
- Figure 57: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by End Use
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Component
- Figure 64: Western Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Deployment
- Figure 67: Western Europe Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Connectivity
- Figure 70: Western Europe Market Value Share and BPS Analysis by Asset Scope, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Asset Scope, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Asset Scope
- Figure 73: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by End Use
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Component
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Deployment
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Connectivity
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Asset Scope, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Asset Scope, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Asset Scope
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Component
- Figure 96: East Asia Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Deployment
- Figure 99: East Asia Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Connectivity
- Figure 102: East Asia Market Value Share and BPS Analysis by Asset Scope, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Asset Scope, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Asset Scope
- Figure 105: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by End Use
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Component
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Deployment
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Connectivity
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Asset Scope, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Asset Scope, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Asset Scope
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Component , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Component , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Component
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Deployment
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Connectivity, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Connectivity, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Connectivity
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Asset Scope, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Asset Scope, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Asset Scope
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

