Deviation Management and CAPA Platforms for Cold Chain Logistics Market
The deviation management and CAPA platforms for cold chain logistics market is segmented by Platform Type, Workflow, Deployment Scope, End User, Temperature Band, and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Deviation Management and CAPA Platforms for Cold Chain Logistics Market Size, Market Forecast and Outlook By FMI
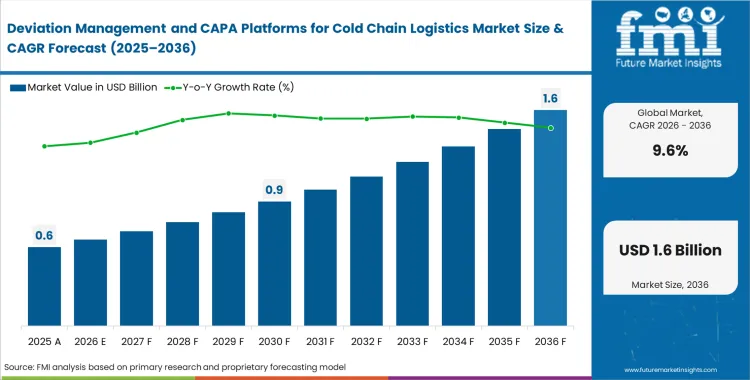
The deviation management and CAPA platforms for cold chain logistics market was valued at USD 0.5 billion in 2025. Industry is expected to cross USD 0.6 billion in 2026 at a CAGR of 9.60% during the forecast period. Pharmaceutical operations are aggressively transitioning from retrospective temperature logging to real-time, automated exception handling tied directly to Good Distribution Practice (GDP) compliance requirements, driving sales value to USD 1.5 billion through 2036.
Summary of Deviation Management and CAPA Platforms for Cold Chain Logistics Market
- The market is estimated at USD 0.6 billion in 2026.
- The market is projected to reach USD 1.5 billion by 2036.
- The market is expected to grow at a CAGR of 9.6% from 2026 to 2036.
- The forecast period represents an incremental opportunity of USD 0.9 billion.
- Cloud-native platforms lead the segment with a 48.0% share.
- Deviation management dominates the workflow segment with a 31.0% share.
- In-transit visibility leads the deployment scope segment with a 39.0% share.
- Pharma manufacturers dominate the end-user segment with a 42.0% share.
- 2-8°C temperature band leads the segment with a 45.0% share.
- India (10.8%), China (10.2%), and Brazil (9.8%) are among the fastest-growing markets.
- Key companies in the market include Controlant, Veeva Systems, ELPRO, Sensitech, SkyCell, TraceLink, and MasterControl.
Deviation Management and CAPA Platforms for Cold Chain Logistics Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 0.6 billion |
| Industry Value (2036) | USD 1.5 billion |
| CAGR (2026-2036) | 9.60% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The industry is currently experiencing a profound operational shift fueled by implementation of stringent regulatory milestones, including the US Drug Supply Chain Security Act (DSCSA) and the EU Falsified Medicines Directive (FMD). These mandates strictly require complete serialization, real-time traceability, and fully documented electronic workflows for batch releases. Consequently, organizations are integrating artificial intelligence (AI) and blockchain technologies into their quality systems to ensure tamper-proof audit trails, predictive risk assessment, and seamless cross-border compliance. Companies are increasingly prioritizing digital traceability to mitigate the financial risks associated with multi-partner logistics networks, elevating these platforms from optional IT upgrades to mission-critical operational imperatives.
Manual quarantine resolution carries heavy financial penalties, forcing quality assurance directors to completely rethink temperature excursions. High-value biologics and specialty medicines concentrate immense capital into single shipments, meaning any delay in determining the root cause directly impacts patient supply and corporate cash flow. Procurement leaders are scrutinizing legacy systems that record breaches without initiating an automated workflow to resolve them, accelerating investment in unified cold chain monitoring tools and dedicated cold chain exception management software. Integrating pharmaceutical logistics CAPA systems across external partners reduces release times from weeks to hours, fundamentally altering working capital requirements for clinical drug supply chain networks.
Integrating third-party dataloggers directly into a manufacturer's enterprise Quality Management System (QMS) changes the operating baseline. A physical temperature spike instantly becomes a locked electronic quality event within the temperature excursion management platform, requiring documented closure before the batch can be legally released. This automation permanently removes the administrative lag of manual transcription, establishing seamless pharmaceutical logistics execution across international borders.
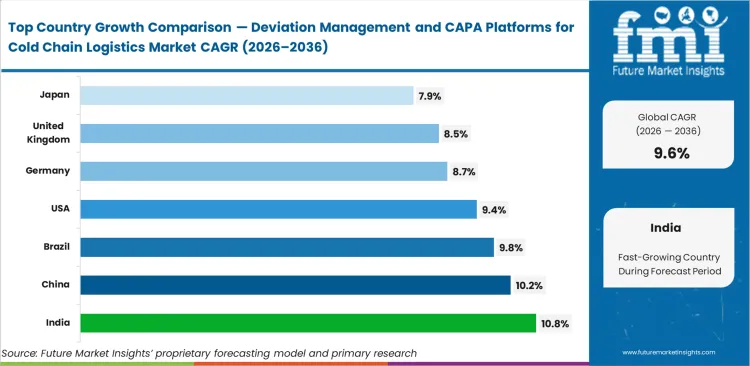
Biologics manufacturing investments require continuous, auditable transit records via localized cold chain compliance software deployments, resulting in India to expand at 10.8% CAGR over the forecast period. China is estimated to register 10.2% CAGR during the forecast period, followed by Brazil (9.8%), the United States (9.4%), Germany (8.7%), the United Kingdom (8.5%), and Japan (7.9%) through 2036 driven by complex automation upgrades and legacy system replacements.
Segmental Analysis
Deviation Management and CAPA Platforms for Cold Chain Logistics Market Analysis by Platform Type
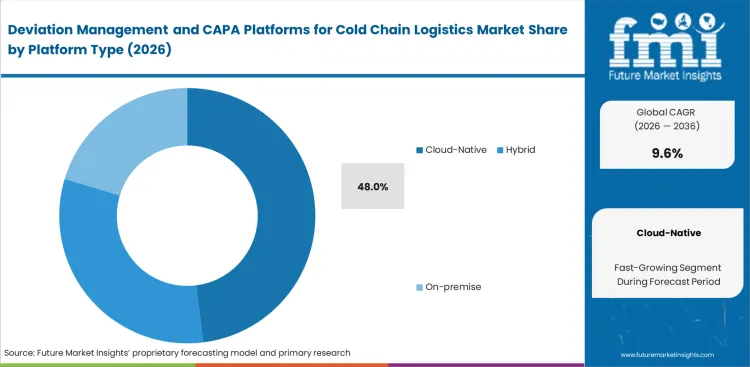
Real-time collaboration across global supply networks exposes the severe operational limits of legacy on-premise quality systems. The cloud-native segment is expected to hold 48.0% revenue share in 2026. This dominant position is supported by four critical factors such as absolute necessity of bridging internal firewalls to allow simultaneous sign-offs by international logistics brokers, and critical requirement to grant immediate and secure read-only access to external regulatory auditors during unannounced inspections. Procurement officers maintaining customized, air-gapped systems for their cold chain packaging operations discover that the administrative friction of manual data sharing costs significantly more than the software itself.
- Workflow initialization: Cloud platforms allow remote warehouse personnel to instantly trigger a deviation record using mobile interfaces the moment a temperature controlled packaging alarm sounds. Quality assurance managers gain immediate, centralized visibility into incident context across multiple geographies, eliminating hours of manual communication, reducing administrative error rates, and accelerating the initial triage of suspected temperature breaches.
- Evidence aggregation: Buyers heavily prioritize platforms capable of seamlessly ingesting high-resolution photographs, detailed logger PDFs, and complex bills of lading directly from third-party logistics partners. Supply chain validation officers require this consolidated, tamper-proof evidence base to confidently determine root causes, assign accurate financial liability, and satisfy stringent regulatory documentation standards required for global commerce.
- Ecosystem expansion: Sustained commercial adoption relies heavily on a vendor's ability to easily onboard new contract manufacturing organizations and distribution partners into secure, shared tenant environments. IT procurement directors face severe operational bottlenecks and escalating overhead costs if adding a new logistics lane requires months of custom integration work, complex security reviews, and redundant system validations.
Deviation Management and CAPA Platforms for Cold Chain Logistics Market Analysis by Workflow
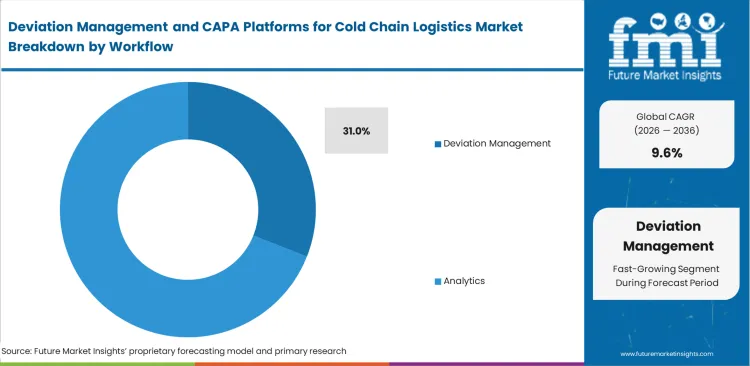
Incident response protocols dictate the initial architecture of compliance environments. Organizations implementing advanced cold chain release decision software without first standardizing their deviation logging procedures routinely fail their regulatory audits due to inconsistent data entry and hidden systemic risks within the cold chain logistics network. The deviation management segment is expected to account for 31.0% revenue share in 2026. This is because identifying and containing a temperature anomaly remains the unavoidable first operational step in any compliance scenario, and sophisticated CAPA modules are functionally useless if initial deviation capture creates an administrative burden for warehouse floor staff. Regulatory affairs directors mandate this workflow because it dictates the absolute speed at which a potentially compromised batch is safely quarantined.
- Initial capture: Floor supervisors at sprawling distribution centers represent the crucial first line of defense, requiring highly intuitive mobile interfaces to log suspected temperature breaches immediately upon discovery. Quality directors strictly mandate simplified data entry screens to ensure every anomaly enters the tracking system without placing excessive administrative burden on fast-moving operational staff.
- Investigation scaling: Mid-level quality engineers attach formal root-cause analysis frameworks to initial deviation reports using dedicated warehouse temperature deviation software. Supply chain validation managers depend entirely on these standardized templates to evaluate whether a pharmaceutical cold chain packaging failure, a prolonged tarmac delay, or a customs hold caused the specific incident.
- Executive oversight: Chief Quality Officers interact with these critical workflows during the final approval stages or when systemic trends demand broader operational interventions. Operations directors who fail to systematically aggregate these individual deviations into meaningful, long-term performance metrics miss vital strategic opportunities to negotiate superior service level agreements with their logistics providers.
Deviation Management and CAPA Platforms for Cold Chain Logistics Market Analysis by Deployment Scope
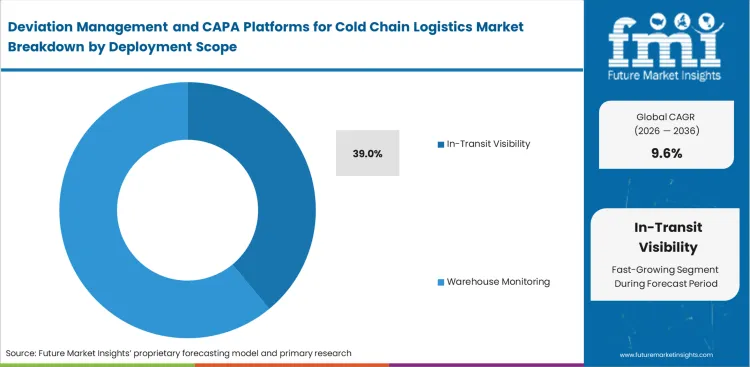
Transit blind spots carry substantially steeper operational costs than static storage monitoring. Shippers limiting their quality management scope to warehouse four walls face immense difficulty disputing liability claims when a reusable biologics shippers shipment arrives degraded at the final destination, making active transit monitoring an absolute commercial necessity. The in-transit visibility segment is expected to secure 39.0% revenue share in 2026. This extensive adoption is supported by factors such as inherent vulnerability of highly sensitive pharmaceutical products moving across multiple transport modes, and necessity for rapid intervention if a logistics transport insulated truck deviates from its planned route. Requirement to establish incontrovertible proof of condition at the exact moment custody transfers between distinct legal entities also supports growth of this segment.
- Handoff blindspots: Relying solely on origin and destination readings dangerously masks temperature cycling occurring during protracted customs delays or exposed tarmac transfers. Supply chain directors systematically avoid this critical failure mode by demanding continuous, integrated data streams that pinpoint precisely where an excursion occurred and exactly which logistics entity possessed the valuable cargo.
- Residual liability: Establishing an accurate root cause requires intelligently correlating real-time sensor data against specific carrier handling procedures and contractual obligations. Quality validation officers struggle immensely to close formal CAPAs if the software cannot map temperature spikes against specific time windows when a pallet sat outside environmentally controlled storage zones.
- Full integration: Capturing the ultimate commercial benefits requires feeding live transit exception data directly into overarching enterprise resource planning systems. Operations managers must ensure that severe in-transit deviations automatically trigger digital inventory quarantine holds, permanently preventing the accidental commercial release of compromised, unsafe pharmaceutical stock.
Deviation Management and CAPA Platforms for Cold Chain Logistics Market Analysis by End User
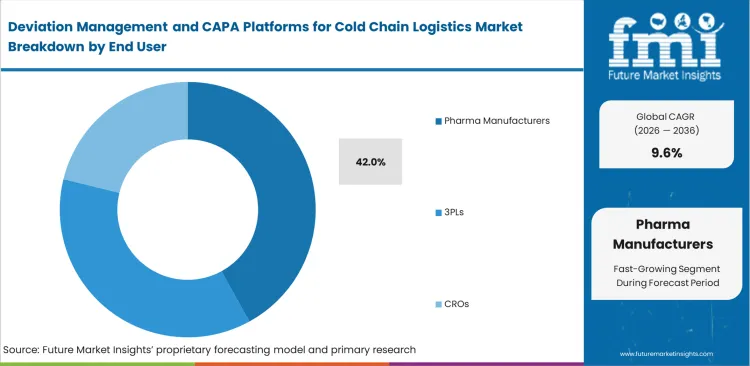
The pharma manufacturers segment is expected to account for 42.0% revenue share in 2026., due to manufacturers retaining the ultimate legal responsibility for patient safety regardless of intermediary touchpoints, and quality directors at these organizations dictate the software architecture that all downstream partners must adopt. These platforms allow manufacturers to forcefully project their quality standards onto third-party providers, and pharmaceutical firms possess the necessary capital to deploy enterprise-grade compliance suites. Global regulators continue shifting product integrity accountability further upstream.
- Platform origination: Software vendors meticulously build core platform architectures around the specific 21 CFR Part 11 compliance requirements demanded by Tier-1 pharmaceutical companies. Product managers at these specialized technology firms heavily prioritize audit-trail robustness and electronic signature capabilities because those specific features directly dictate manufacturer purchasing decisions and enterprise contract renewals.
- Implementation friction: Third-party logistics providers frequently push back against the burden of adopting completely different quality portals for every major pharmaceutical client they actively serve. IT operations directors at global distribution firms must carefully balance rigid customer compliance demands against the crippling operational inefficiencies stemming from managing dozens of distinct, siloed deviation interfaces.
- Network convergence: Future capability roadmaps distinctly point toward interoperable data hubs where multiple global manufacturers and regional carriers can securely exchange standardized quality event records. Supply chain heads eagerly anticipate standardized data formats that could effectively eliminate the need for redundant manual entry across overlapping regulatory reporting systems.
Deviation Management and CAPA Platforms for Cold Chain Logistics Market Analysis by Temperature Band
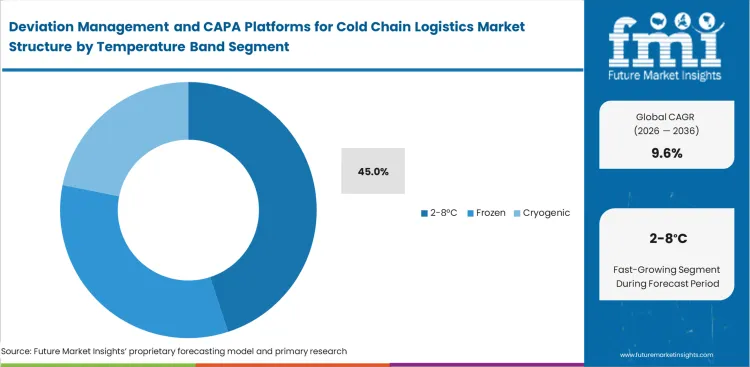
Standard refrigerated logistics consistently generate the highest volume of compliance documentation globally. Software platforms lacking these intelligent filters force validation engineers to waste thousands of highly skilled hours writing superficial CAPA reports for perfectly safe insulated liners shipments. The 2-8°C segment is expected to hold 45.0% revenue share in 2026, because it handles massive global flows of established biologics and routine vaccines, and minor temperature deviations in this narrow band occur frequently requiring extensive documentation to prove stability budget adherence. Specialized algorithms are required to filter out superficial nuisance alarms caused by momentary door openings.
- Baseline processing: Standard vaccine cold chain deviation tracking workflows perform exceptionally well when systematically handling clear, sustained temperature breaches that definitively ruin sensitive products. Quality assurance specialists rely completely on automated workflow templates to quickly log operational failures, digitally quarantine batches, and rapidly initiate financial recovery procedures against responsible freight carriers.
- Edge condition ambiguity: Legacy systems struggle severely when evaluating marginal excursions where ambient temperatures slightly exceed 8°C for durations dangerously close to manufacturer-allowed stability budgets. Regulatory affairs officers actively demand advanced analytical software features calculating cumulative mean kinetic temperature to automatically justify safe release decisions in these complex gray areas.
- Acceptability thresholds: Establishing reliable, automated release protocols requires meticulous technical alignment between core software logic and specific product-validated stability data. Formulation scientists must precisely define exact time-out-of-refrigeration limits to ensure the software only triggers formal CAPA workflows when actual product efficacy and patient safety are genuinely threatened.
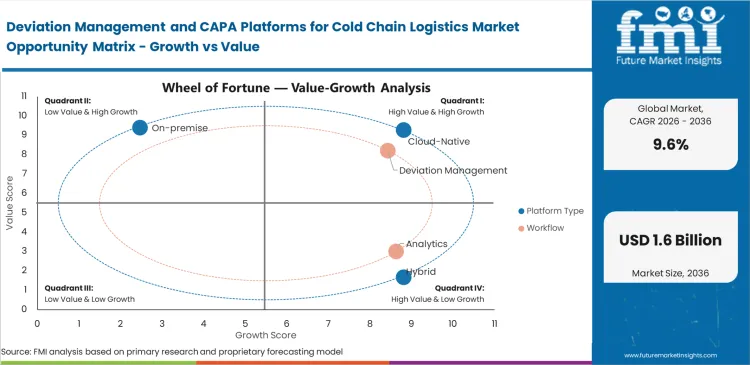
Deviation Management and CAPA Platforms for Cold Chain Logistics Market Drivers, Restraints, and Opportunities
Destroying viable biologics due to undocumented temperature excursions forces supply chain heads to mandate automated quality workflows. When a high-value oncology shipment experiences a temperature spike, the commercial risk extends far beyond physical product loss to the inability to fulfill a critical patient dosage schedule. Manual investigation processes using fragmented emails and spreadsheets introduce days of administrative latency, forcing viable products to sit in expensive quarantine while quality teams hunt for logger data and carrier explanations.
This profound operating friction drives procurement directors to invest in unified cold chain exception management software that instantly aggregates transit data, assigns liability, and precisely calculates stability budgets. By automating the root-cause analysis process, organizations drastically reduce time to disposition, converting week-long compliance headaches into rapid, digitally signed electronic release decisions preserving cash flow. Integrating these tools with paperboard cartons tracking systems further tightens closed-loop control.
Cloud software validation across heterogeneous IT environments introduces operational friction slowing enterprise-wide rollouts. Pharmaceutical quality systems must adhere to strict Computer Software Assurance (CSA) and 21 CFR Part 11 requirements, meaning every significant platform update requires extensive re-validation testing to prove the system functions exactly as intended. IT operations directors struggle to maintain compliance when SaaS vendors push mandatory feature updates altering underlying deviation logic or critical data fields.
This persistent validation burden forces many organizations to stick with sub-optimal legacy platforms or manual workarounds rather than risk falling out of regulatory compliance during complex software migrations. Integrating continuous validation tools with varied cold chain antifreeze glycols monitoring hardware remains difficult, leaving large segments of the industry reliant on tedious manual testing protocols.
Opportunities in the Deviation Management and CAPA Platforms for Cold Chain Logistics Market
- AI-assisted root cause analysis and predictive maintenance: Deploying advanced machine learning models that analyze massive sets of historical deviation patterns to suggest probable causes for new temperature excursions. Quality assurance managers significantly reduce investigation time, resolving routine Canada cold chain packaging nonconformances faster and focusing expert attention strictly on complex, systemic failures via advanced cold chain root cause analysis software. Furthermore, AI capabilities allow for predictive maintenance of refrigeration units, identifying potential equipment failures before they compromise the cargo.
- Blockchain integration for tamper-proof ESG and regulatory compliance: Utilizing decentralized, immutable blockchain frameworks provides highly secure and transparent platforms for deviation record management. This completely eliminates the risk of data manipulation during critical transit handoffs, facilitating immediate trust during complex international audits, optimizing regulatory reporting speed, and enhancing supply chain sustainability transparency for global stakeholders.
- API-driven carrier integration and automated MKT calculation: Providing pre-built digital connectors that automatically pull real-time tracking data from major global freight forwarders directly into manufacturer QMS. IT procurement directors gain immediate deployment speed, completely avoiding massive custom integration costs typically stalling global supply chain visibility projects. Embedding Mean Kinetic Temperature (MKT) algorithms into these workflows instantly determines if minor excursions actually degrade products, freeing up massive operational capacity.
Regional Analysis
Based on regional analysis, the deviation management and CAPA platforms for cold chain logistics market is segmented into North America, Latin America, Europe, Asia Pacific, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 10.8% |
| China | 10.2% |
| Brazil | 9.8% |
| United States | 9.4% |
| Germany | 8.7% |
| United Kingdom | 8.5% |
| Japan | 7.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
South Asia & Pacific Deviation Management and CAPA Platforms for Cold Chain Logistics Market Analysis
Export-oriented biologics manufacturing forces domestic operations in South Asia to aggressively adopt European-grade quality management architectures. Pharmaceutical producers in this rapidly evolving region recognize that competing for lucrative international supply contracts demands incontrovertible, auditable proof of cold-chain integrity from the factory floor to the destination port. Local contract manufacturing organizations are systematically abandoning manual spreadsheet tracking because international pharmaceutical clients refuse to accept batches without fully digitized, Part 11-compliant deviation records. This foundational transition radically alters the operational baseline, forcefully pushing local logistics providers to upgrade their tracking capabilities or face permanent exclusion from high-value global distribution networks.
- India: India is projected to expand at a 10.8% CAGR during the forecast period, driven by the rapid expansion of export-oriented biologics manufacturing requiring stringent digital documentation, the enforcement of stricter domestic Good Supply Practice guidelines that push wholesale distributors to implement formal excursion protocols, and rising strategic investments in localized infrastructure evolving toward predictive, lane-based risk assessment driving immense local demand for dedicated India cold chain compliance software.
Asia Pacific Deviation Management and CAPA Platforms for Cold Chain Logistics Market Analysis
Revised Good Supply Practice (GSP) regulations in Asia Pacific fiercely compel massive distribution networks to entirely overhaul their legacy tracking systems. Regulatory authorities now require rigorous, unalterable documentation of temperature breaches across highly fragmented regional logistics hubs, effectively eliminating the commercial viability of basic temperature dataloggers lacking integrated CAPA workflows. Leading domestic pharmaceutical firms proactively deploy sophisticated, cloud-based quality portals to assert absolute control over sprawling networks of third-party carriers. By strictly mandating that all logistics partners log excursions into centralized, manufacturer-controlled systems, these companies drastically reduce their exposure to severe regulatory penalties and catastrophic product recalls.
- China: China is estimated to register a 10.2% CAGR during the forecast period, owing to aggressive enforcement actions against regional distributors failing to properly document temperature abuse, targeted investments by procurement officers consolidating logistics networks around highly digitized partners, and the urgent need for domestic firms to prove superior compliance to secure contracts with global pharmaceutical brands entering the ecosystem in China.
- Japan: Presence of highly established quality frameworks demanding complex system integration rather than basic digitization, and necessity to automatically correlate vast transit data streams with uniquely stringent domestic batch release requirements are key factors driving steady expansion. Japan is expected to exhibit a 7.9% CAGR during the forecast period which is driven by strategic deployment of advanced workflows to command premium service pricing from multinational clients is also supporting market growth.
Latin America Deviation Management and CAPA Platforms for Cold Chain Logistics Market Analysis
The expanding distribution of highly sensitive specialty therapeutics demands robust, fail-safe temperature control architectures operating in challenging climatic and infrastructural environments. Multinational pharmaceutical brands importing high-value biologics into the region require absolute, real-time visibility into final-mile deliveries, driving substantial capital investments in digital quality platforms that bridge the gap between global regulatory standards and local execution realities. Local logistics providers increasingly realize that adopting validated, enterprise-grade deviation management software remains the only viable path to retain lucrative, long-term contracts with major international shippers.
- Brazil: Brazil market is anticipated to record a 9.8% CAGR during the forecast period. Growth is driven by regulatory oversight across specialized distribution networks, compelling carriers to put in place rigorous Corrective Action Preventive Action (CAPA) protocols. Operations managers rely heavily on automated reporting mechanisms to substantiate product handling methods during stringent audits performed by manufacturers. Operational necessity present to substantially lower the occurrence of devastating product losses is frequently stemming from infrastructural delays lacking appropriate tracking.
North America Deviation Management and CAPA Platforms for Cold Chain Logistics Market Analysis
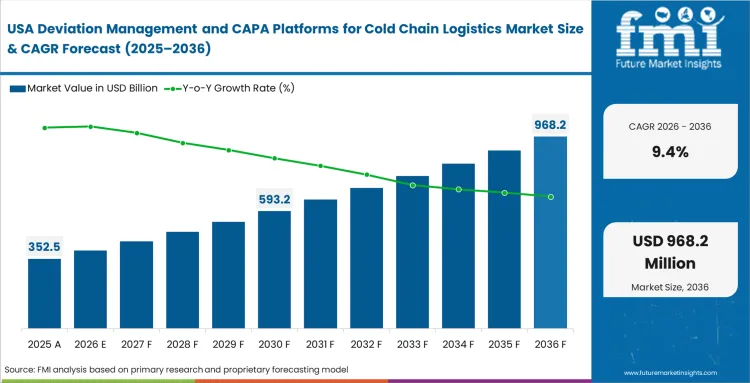
Deep system integration firmly dictates advanced software adoption across the mature North American landscape. Stringent compliance with the Drug Supply Chain Security Act (DSCSA) establishes uncompromising baselines of item-level traceability, pushing quality directors to demand software capable of instantly linking specific serialized pharmaceutical units to their corresponding temperature deviation investigations. The operational focus has decisively shifted from simply logging isolated incidents to automating complex, multi-party root cause analysis workflows required to legally and rapidly release delayed shipments into the commercial supply chain.
- United States: United States is likely to grow at a rate of 9.4% during the forecast period. This dominance is driven by immense volume of high-value biologic shipments generating millions of data points that overwhelm manual review processes, widespread implementation of AI-assisted deviation routing to automatically triage nuisance alarms, and pioneering efforts of local software vendors establishing seamless API connections between active monitoring hardware and enterprise QMS architectures for reusable cold chain packaging fleets.
Europe Deviation Management and CAPA Platforms for Cold Chain Logistics Market Analysis
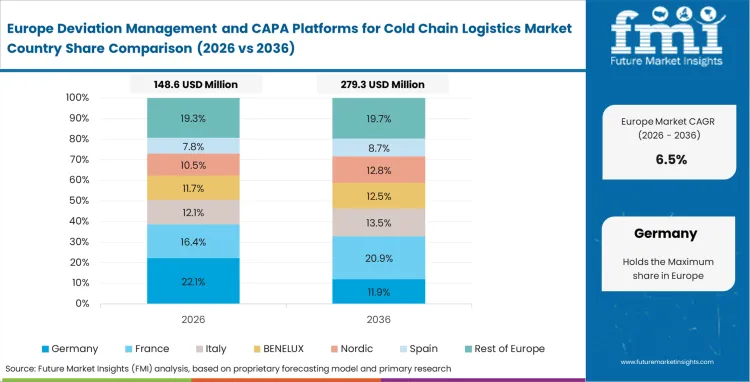
Rigorous EU GDP guidelines strictly dictate the fundamental architectures of every sophisticated cold-chain software deployment across the region. European regulators expect manufacturers and distributors to maintain tightly interconnected webs of electronic quality agreements, automated risk assessments, and fully documented CAPA closures for every single temperature excursion regardless of severity. Organizations are actively and aggressively replacing siloed, departmental tools with unified, cloud-native platforms to survive increasingly complex, multi-country regulatory inspections demanding verifiable, GDP compliant cold chain software.
- Germany: The consistent growth of Germany is driven by uncompromising data integrity demands bridging pharmaceutical manufacturing and precision logistics, and intense operational pressure on quality validation officers to harmonize excursion data across specialized cross-border carriers. Germany is expected to exhibit an 8.7% CAGR during the forecast period. The strategic push by leading enterprises to overcome the reluctance of competing logistics firms to openly share failure data.
- United Kingdom: United Kingdom is projected to garner 8.5% CAGR during the forecast period, driven by complex post-Brexit regulatory realignments forcing distributors to maintain meticulous digital records for uninterrupted pharmaceutical flows. Deployment of targeted deviation modules designed to rapidly isolate and resolve customs checkpoint delays, and broader industry shift toward collaborative platforms allowing regulatory bodies direct, read-only access are other factors driving market growth in the country.
FMI's report includes extensive coverage of additional countries not featured in the primary bullets, spanning specialized distribution hubs in the Middle East and emerging pharmaceutical manufacturing centers in Eastern Europe. Multinational buyers insist that all regional nodes operate on the exact same auditable software standard, functionally erasing local regulatory leniency.
Competitive Aligners for Market Players
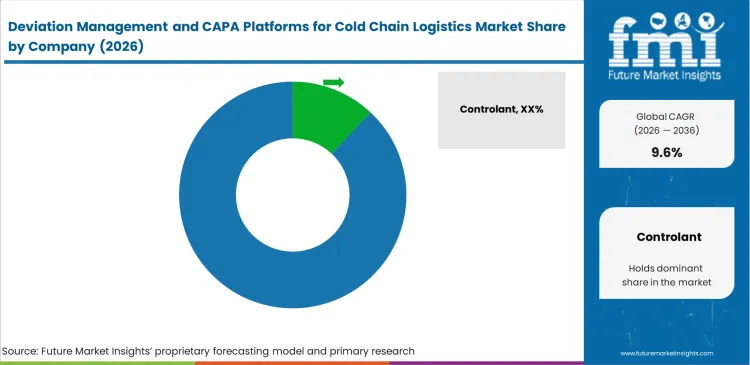
Pure-play cold chain visibility vendors and established enterprise Quality Management System (QMS) developers approach the market from entirely opposite strategic directions. Companies like Veeva Systems and MasterControl approach from the top down, successfully leveraging their deep entrenchment in the manufacturer's core compliance and regulatory approval workflows. Specialized tracking entities such as Controlant and Sensitech approach from the bottom up, utilizing their direct control over physical temperature sensors and real-time data streams to offer highly contextualized deviation alerts. Procurement directors evaluate these competing architectures based on a critical operational capability: the software's ability to seamlessly map a physical transit failure directly to the specific regulatory batch release requirement without requiring manual data transcription.
Enterprise QMS developers rely heavily on massive, pre-validated libraries of 21 CFR Part 11 compliant workflows and sophisticated electronic signature protocols. Challengers attempting to displace these entrenched systems must build highly robust, bidirectional integration layers allowing their circular packaging systems visibility data to automatically trigger and intelligently populate the incumbent's CAPA forms. Software vendors attempting to build closed ecosystems forcing customers to use proprietary hardware face immense, organized resistance from logistics directors managing heterogeneous transport networks. Winning players secure market share by offering open APIs that effortlessly ingest excursion data from any logger, instantly translating raw temperature spikes into standardized, actionable quality events within existing pharmaceutical packaging industry analysis compliance frameworks.
Key Players in Deviation Management and CAPA Platforms for Cold Chain Logistics Market
- Controlant
- Veeva Systems
- ELPRO
- Sensitech
- SkyCell
- TraceLink
- MasterControl
Scope of the Report
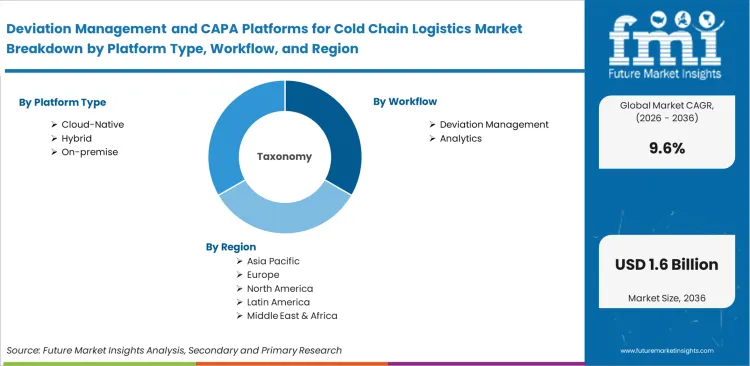
| Metric | Value |
|---|---|
| Quantitative Units | USD 0.6 billion to USD 1.5 billion, at a CAGR of 9.60% |
| Market Definition | Software architectures configured to identify, investigate, document, and resolve temperature excursions or handling nonconformances during the storage and transit of sensitive goods, serving as definitive compliance records for regulatory bodies enforcing GDP standards. |
| Segmentation | Platform Type, Workflow, Deployment Scope, End User, Temperature Band, Region |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | United States, Canada, Germany, United Kingdom, France, Italy, Spain, Russia, Poland, China, Japan, India, South Korea, ASEAN, Australia, New Zealand, Brazil, Mexico, Argentina, GCC, South Africa, Israel |
| Key Companies Profiled | Controlant, Veeva Systems, ELPRO, Sensitech, SkyCell, TraceLink, MasterControl |
| Forecast Period | 2026 to 2036 |
| Approach | Baseline anchored by active software licensing revenue mapped against the volume of biologics requiring active monitoring, cross-validated against enterprise QMS deployment rates and Tier-1 logistics provider capital expenditure. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Deviation Management and CAPA Platforms for Cold Chain Logistics Market Analysis by Segments
Platform Type
- Cloud-native
- Hybrid
- On-premise
Workflow
- Deviation management
- CAPA management
- Release evaluation
- Audit trails
- Analytics
Deployment Scope
- In-transit visibility
- Warehouse monitoring
- End-to-end orchestration
End User
- Pharma manufacturers
- 3PLs
- Wholesalers
- Hospitals
- CROs
Temperature Band
- 2-8°C
- Controlled room
- Frozen
- Cryogenic
Region
- North America
- United States
- Canada
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Asia Pacific
- China
- Japan
- South Korea
- Taiwan
- Singapore
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East & Africa
- GCC Countries
- South Africa
- Israel
- Rest of Middle East & Africa
Bibliography
- USA Food and Drug Administration. (2024, October 9). Waivers and exemptions beyond the stabilization period.
- Ferraz, K., Cato, M., Fox, E., Rawlins, M., & Misko, J. (2024, September). Temperature excursions in cold chain management-Assessing the accuracy of refrigerator temperature probes. Hospital Pharmacy, 60(1), 66-69.
- USA Food and Drug Administration. (2025, April). Q1 stability testing of drug substances and drug products.
- Nozari, H., Rahmaty, M., Foukolaei, P. Z., Movahed, H., & Bayanati, M. (2025, January). Optimizing cold chain logistics with artificial intelligence of things (AIoT): A model for reducing operational and transportation costs. Future Transportation, 5(1), 1.
- Ran, W., & Li, Z. (2025, September). Literature review: Current trends and future prospects of digital vaccine supply chain support technology. Human Vaccines & Immunotherapeutics, 21(1), 2553454.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- How Quality Assurance Directors evaluate cloud-native architectures for immediate cross-border deviation visibility.
- Why manual temperature excursion investigations create severe quarantine bottlenecks for high-value biologics shipments.
- The integration challenges logistics providers face when connecting active dataloggers to enterprise QMS APIs.
- Which specific Good Distribution Practice mandates force wholesale distributors to upgrade legacy tracking systems.
- How automated mean kinetic temperature algorithms reduce false-positive CAPA reports for refrigerated specialty medicines.
- Why North American pharmaceutical manufacturers dictate compliance software standards adopted by global logistics partners.
- The operational limitations of relying purely on predictive lane analytics without robust incident response workflows.
- How continuous validation protocols impact the deployment speed of vital Part 11-compliant software updates.
Frequently Asked Questions
What is deviation management in cold chain logistics?
Deviation management refers to the structured digital process of identifying, investigating, documenting, and resolving temperature excursions or handling nonconformances during the transit of sensitive goods, ensuring compliance with Good Distribution Practice standards.
Why do pharma cold chains need CAPA platforms?
Pharmaceutical supply networks rely on these systems because high-value biologics require immediate, auditable root-cause analysis when temperature breaches occur. Manual workflows introduce days of administrative latency, leaving viable products stranded in quarantine facilities.
How do temperature excursions trigger CAPA?
When active dataloggers register a temperature reading outside validated stability budgets, the integrated software automatically opens an electronic quality event, locking the batch from release until a formal Corrective and Preventive Action is documented, investigated, and digitally signed by liable parties.
What software is used for cold chain excursion investigations?
Quality assurance teams utilize dedicated cold chain deviation management software and enterprise Quality Management Systems (QMS) featuring specialized modules that aggregate in-transit sensor data, carrier handling records, and product stability budgets to determine root cause.
How does GDP affect cold chain software selection?
Good Distribution Practice regulations mandate auditable records of product handling, forcing procurement directors to evaluate platforms strictly on their 21 CFR Part 11 compliance, electronic signature robustness, and ability to securely onboard third-party logistics partners.
Can cold chain monitoring tools replace CAPA systems?
No, monitoring tools only capture and alert users to the temperature data point. Resolving the resulting operational bottleneck requires an entirely separate, formal CAPA workflow architecture to assign liability, document the investigation, and secure legal batch release.
What data should a cold chain CAPA platform capture?
Robust systems ingest precise geolocation coordinates, continuous temperature readings, carrier bills of lading, photographic evidence from warehouse staff, and calculated mean kinetic temperature values to support defensible release or destroy decisions.
Which countries are expected to grow fastest for cold chain quality software?
India leads the global trajectory at a 10.8% CAGR, followed closely by China at 10.2%, as rapidly expanding export-oriented biologics manufacturing hubs face pressure to digitize their compliance infrastructure to meet European and North American import standards.
Compare cold chain monitoring software with CAPA platforms.
Monitoring software provides real-time visibility and alerts regarding physical sensor status. CAPA platforms handle the downstream regulatory consequence, managing the complex, multi-party root cause analysis and corrective action workflow required to legally clear the compromised batch.
Which vendors are active in cold chain deviation management software?
The competitive landscape features specialized cold chain CAPA software vendors like Controlant, ELPRO, Sensitech, and SkyCell offering highly contextualized transit platforms, alongside enterprise QMS developers such as Veeva Systems, TraceLink, and MasterControl providing regulatory batch release integration.
What makes a system the best cold chain deviation management platform?
Winning architectures provide open APIs capable of ingesting excursion data from any heterogeneous logger hardware while offering intuitive, low-friction portals allowing external logistics partners to upload investigation evidence without severe operational fatigue.
How does pharma cold chain quality software pricing impact adoption?
Procurement directors actively move away from basic per-user subscription models, evaluating Total Cost of Ownership based on the platform's ability to seamlessly onboard external logistics partners without incurring custom integration fees.
Why is validated excursion management software for biologics critical?
High-value, advanced therapies possess exceptionally narrow stability windows, demanding software that instantly calculates cumulative exposure limits and executes automated release logic to prevent destroying viable products over superficial, nuisance temperature alarms.
How does a cold chain CAPA platform vs QMS evaluation unfold?
Buyers weigh the immediate, lane-level contextual visibility offered by dedicated cold chain platforms against the deep, pre-validated regulatory reporting frameworks inherently built into incumbent enterprise Quality Management Systems.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Platform Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Platform Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Platform Type , 2026 to 2036
- Cloud-Native
- Hybrid
- On-premise
- Cloud-Native
- Y to o to Y Growth Trend Analysis By Platform Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Platform Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Workflow
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Workflow, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Workflow, 2026 to 2036
- Deviation Management
- Analytics
- Deviation Management
- Y to o to Y Growth Trend Analysis By Workflow, 2021 to 2025
- Absolute $ Opportunity Analysis By Workflow, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment Scope
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment Scope, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment Scope, 2026 to 2036
- In-Transit Visibility
- Warehouse Monitoring
- In-Transit Visibility
- Y to o to Y Growth Trend Analysis By Deployment Scope, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment Scope, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Pharma Manufacturers
- 3PLs
- CROs
- Pharma Manufacturers
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Temperature Band
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Temperature Band, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Temperature Band, 2026 to 2036
- 2-8°C
- Frozen
- Cryogenic
- 2-8°C
- Y to o to Y Growth Trend Analysis By Temperature Band, 2021 to 2025
- Absolute $ Opportunity Analysis By Temperature Band, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- By Country
- Market Attractiveness Analysis
- By Country
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Platform Type
- By Workflow
- By Deployment Scope
- By End User
- By Temperature Band
- Competition Analysis
- Competition Deep Dive
- Controlant
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Veeva Systems
- ELPRO
- Sensitech
- SkyCell
- TraceLink
- MasterControl
- Controlant
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Deployment Scope, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Deployment Scope, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Deployment Scope, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Deployment Scope, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Deployment Scope, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Deployment Scope, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Deployment Scope, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Platform Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Workflow, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Deployment Scope, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Platform Type
- Figure 6: Global Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Workflow
- Figure 9: Global Market Value Share and BPS Analysis by Deployment Scope, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Deployment Scope, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Deployment Scope
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Temperature Band
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Platform Type
- Figure 32: North America Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Workflow
- Figure 35: North America Market Value Share and BPS Analysis by Deployment Scope, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Deployment Scope, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Deployment Scope
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Temperature Band
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Platform Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Workflow
- Figure 51: Latin America Market Value Share and BPS Analysis by Deployment Scope, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Deployment Scope, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Deployment Scope
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Temperature Band
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Platform Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Workflow
- Figure 67: Western Europe Market Value Share and BPS Analysis by Deployment Scope, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Deployment Scope, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Deployment Scope
- Figure 70: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End User
- Figure 73: Western Europe Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Temperature Band
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Platform Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Workflow
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Deployment Scope, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Deployment Scope, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Deployment Scope
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Temperature Band
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Platform Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Workflow
- Figure 99: East Asia Market Value Share and BPS Analysis by Deployment Scope, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Deployment Scope, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Deployment Scope
- Figure 102: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End User
- Figure 105: East Asia Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Temperature Band
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Platform Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Workflow
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Deployment Scope, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment Scope, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Deployment Scope
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Temperature Band
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Platform Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Platform Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Platform Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Workflow, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Workflow, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Workflow
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Deployment Scope, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment Scope, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Deployment Scope
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Temperature Band
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

