Clinical Drug Supply Chain Services Market
Clinical Drug Supply Chain Services Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Clinical Drug Supply Chain Services Market Forecast and Outlook 2026 to 2036
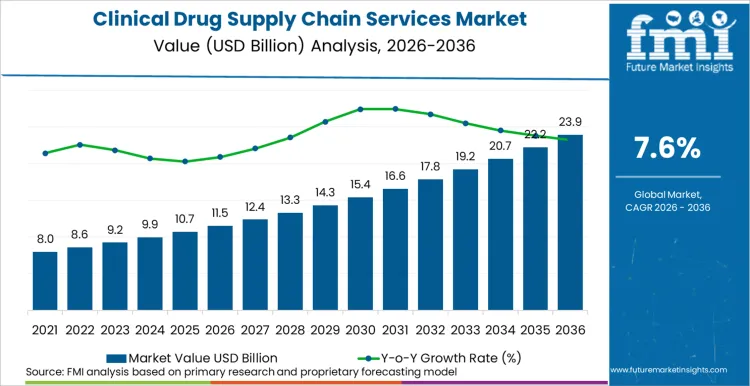
Future Market Insights anticipates the global clinical drug supply chain services market to expand from USD 11.5 billion in 2026 to USD 24.0 billion by 2036, registering a CAGR of 7.6% over the forecast period. The rising number of Phase II and Phase III clinical trials, increasing adoption of biologics and advanced therapies, and the continued globalization of drug development programs, drives this growth. According to FMI’s analysis, late-phase clinical studies are generating significant demand for scalable depot and distribution networks capable of handling large patient populations, complex dosing schedules, and stringent regulatory requirements.
FMI highlights, temperature-controlled logistics is a paradigm shift that is transforming the way sponsor and CROs are thinking about transportation. Clinical studies are being developed with multiple temperature ranges, such as ambient, refrigerated, frozen, ultra-cold, and cryogenic, sometimes in the same study, which is creating a reliance on partners who have validated cold chain capabilities and multimodal execution. At the same time, decentralized and hybrid studies are driving the need for direct-to-patient delivery, particularly in situations where site visits are limited. Purchasing decisions are also being driven by the need to comply with GDP, chain-of-custody, and audit-ready data capture.
These trends are being further reinforced by recent developments among the large service providers. Global logistics companies are continuing to build GMP-aligned depot capacity, enhance temperature-qualified packaging and monitoring expertise, and improve last-mile delivery execution to support the growing complexity of trial designs. In keeping with this trend, the leadership of DHL Supply Chain has consistently emphasized the importance of resilience and end-to-end visibility in supporting the integrity of trial continuity and patient safety as trials become more distributed and global. The clinical supply chain service offering is being repositioned from a transactional transport service to operational infrastructure. “The increasing demand for specialty pharma and healthcare solutions presents significant opportunities for DHL to leverage its scale, expertise, and commitment to operational excellence. With this acquisition, we are expanding our healthcare logistics capabilities, attracting a new segment of healthcare customers, and reinforcing our position as a trusted partner in building resilient and connected healthcare supply chains.," said Mark Kunar, CEO of DHL Supply Chain North America.
The industry is witnessing a shift in service offerings by providers to align with the complexity of clinical development, especially in the areas of biologics, cell and gene therapies, and late-phase studies across multiple countries. The focus of investment is on compliant depot and cold chain infrastructure, improved temperature and chain of custody visibility, and patient-centric delivery models that enable decentralized execution. These capacity-building efforts are helping providers assume earlier and more integrated roles in trial development and continuity management. Differentiation, as predicted by Future Market Insights, will increasingly depend on demonstrated operational mastery, documentation rigor, and reliability at scale through 2036.
Summary of Clinical Drug Supply Chain Services Market
What Is Growth Outlook for Clinical Drug Supply Chain Services Market as per Future Market Insights Projection?
Future Market Insights projects the clinical drug supply chain services market to expand at a CAGR of 7.6% from 2026 to 2036, increasing from USD 11.5 Billion in 2026 to USD 24.0 Billion by 2036.
FMI Research Approach: Historical market size, clinical trial volume trends, service-type utilization patterns, temperature-controlled logistics demand, and late-stage trial intensity were analyzed to model demand scenarios through 2036.
How Do FMI Analysts Perceive Clinical Drug Supply Chain Services Market to Evolve?
As per Future Market Insights, the clinical drug supply chain services are evolving from transactional logistics functions toward integrated, end-to-end orchestration models. Demand is increasingly driven by decentralized trials, adaptive protocols, and higher biologic trial complexity, where visibility, traceability, and temperature integrity are critical across the clinical lifecycle.
FMI Research Approach: Evaluation focused on shifts in trial design, biologics pipeline expansion, decentralization trends, and growing reliance on bundled supply, packaging, and distribution services.
Which Country Holds Largest Share in Global Clinical Drug Supply Chain Services Market?
The United States holds the largest share in the global clinical drug supply chain services market, supported by high clinical trial density, strong sponsor and CRO presence, and advanced regulatory infrastructure. Concentration of Phase III trials and biologics development further reinforces sustained demand for specialized supply chain services.
FMI Research Approach: Country-level assessment included trial activity concentration, regulatory maturity, sponsor presence, logistics infrastructure depth, and temperature-controlled distribution capacity.
How Large Will Clinical Drug Supply Chain Services Market Be by 2036?
The global clinical drug supply chain services market is projected to reach USD 24.0 Billion by 2036, reflecting sustained demand from late-stage clinical development and expanding global trial footprints.
FMI Research Approach: Market sizing was derived by modeling service demand across clinical phases, temperature bands, and shipment modes with weighted utilization factors.
How are Clinical Development and Regulatory Requirements Fueling Market Growth?
According to Future Market Insights, tightening regulatory expectations for traceability, temperature compliance, and material accountability are accelerating demand for specialized clinical supply chain services. Growth is further supported by increasing biologics trials, decentralized delivery models, and sponsor emphasis on risk mitigation and trial continuity.
FMI Research Approach: Emerging trends including decentralized trials, biologics-led pipelines, and evolving Good Distribution Practice requirements were incorporated into forecasting models.
Clinical Drug Supply Chain Services Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 11.5 Billion |
| Industry Value (2036) | USD 24.0 Billion |
| CAGR (2026-2036) | 7.6% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Why is the Demand for Clinical Drug Supply Chain Services Growing?
The demand for clinical drug supply chain services is increasing as clinical development is becoming more global, biologics-driven, and complex. The increase in Phase II and Phase III studies, the reliance on temperature-sensitive investigational products, and the adoption of decentralized and hybrid trials are increasing the need for compliant end-to-end execution. The service providers are being engaged beyond distribution, and the responsibility is extending to comparator sourcing, packaging, labeling, returns, and direct-to-patient coordination. As the sponsors and CROs are increasing their focus on continuity, compliance, and risk management, these services are increasingly being viewed as core operational infrastructure for trials.
How is the Clinical Drug Supply Chain Services Market Segmented?
The clinical drug supply chain services market is segmented by service type, clinical phase, temperature band, customer type, and shipment mode. By service type, the market includes depot and warehousing, cold-chain logistics, comparator sourcing, packaging and labeling, distribution and transportation, direct-to-patient delivery, and returns and destruction services. Based on clinical phase, demand spans Phase I, Phase II, Phase III, and Phase IV or real-world evidence studies. By temperature band, services are categorized into ambient/controlled room temperature (CRT), 2-8°C, frozen (-20°C), ultra-cold (-70°C), and cryogenic (LN2). By customer type, the market serves pharma and biopharma sponsors, CROs, CDMOs/CMOs, and academic or NGO organizations. Shipment modes include air, road, sea, and courier express.
How Do Phase III Trials Secure Market Leadership?
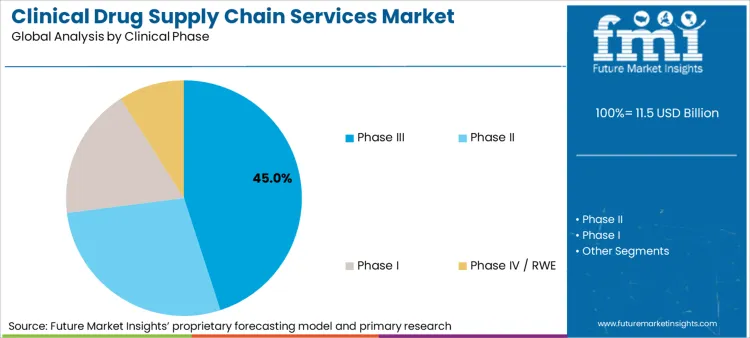
As per Future Market Insights, Phase III trials are currently driving the demand for clinical drug supply chain services due to the size and complexity of Phase III trials, as well as the greater regulatory risk. With an estimated 45.0% share of total service demand, Phase III trials involve high volume distribution, extended depot reach, and carefully coordinated resupply to maintain the momentum of enrollment and study protocol at geographically separated sites. The level of compliance is also high, with documented temperature validation, chain-of-custody documentation, and near real-time inventory visibility being necessary. Because of the disproportionate financial and timeline risk at this point, sponsors are turning to expert help to ensure continuity, minimize deviations due to logistics, and remain audit-ready, thus making Phase III the prime demand anchor.
How Does Ambient and Controlled Room Temperature Handling Lead Demand?
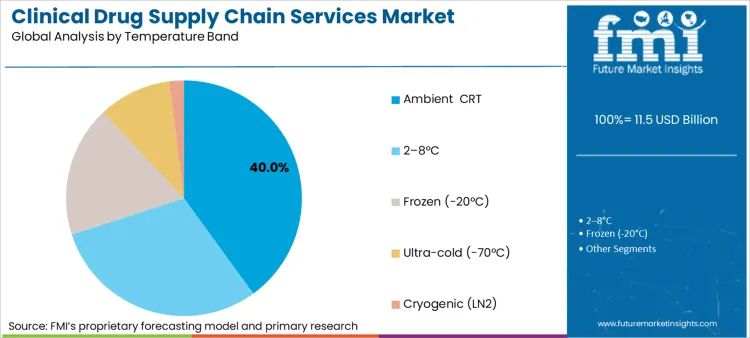
Future Market Insights highlights that the ambient and controlled room temperature (CRT) handling continues to be the dominant temperature segment, accounting for 40.0% of the total service demand. This is because a majority of the small molecule investigational and oral solid dosage forms has retained stability at ambient temperatures, thus making refrigerated, frozen, or cryogenic storage less necessary. Ambient and CRT routes also facilitate faster site start-up and flexible routing, which is especially important in multi-regional Phase II and Phase III trials where time and resupply flexibility are of utmost importance. These temperature segments make it possible for sponsors to mitigate risks associated with costs while remaining in sync with stability requirements, thus maintaining ambient/CRT as the most used temperature segment.
How Are Regulatory Frameworks Influencing Clinical Supply Chain Design?
The demands related to the integrity of trials, patient safety, and traceability are increasingly influencing the design of the clinical drug supply chain. The end-to-end control of investigational products is being demanded, including the management of excursions, accountability reconciliation, and returns and destruction. These demands are further emphasizing the need for validated logistics processes and quality systems designed for inspection readiness. In this scenario, service providers are enhancing their capabilities related to documentation discipline, audit readiness, and real-time monitoring and reporting to minimize data gaps and deviation risks. With increasing levels of scrutiny in multinational trials, companies with well-developed compliance infrastructure are being given priority by sponsors to avoid operational disruptions and regulatory risks.
Why Is Trial Globalization Reshaping Service Demand Patterns?
The globalization of clinical trials is also affecting the demand for depot, distribution, and transport services. Clinical trials are being conducted in North America, Europe, Asia, and other emerging countries to accelerate enrollment and better represent diverse populations, which is also creating a demand for multi-regional supply strategies. The increased geographic reach is also creating demands for centralized planning and localized execution, such as regional depot network optimization and country-specific labeling to meet trial protocols and regulations. Air transport is also continuing to be the preferred transport mode for time-sensitive routes, and road and courier networks are being utilized for managed last-mile delivery.
How Are Digital Platforms and Advanced Monitoring Tools Driving Innovation?
Digital solutions are being leveraged to enhance the control of the clinical supply chain through improved forecasting, inventory, and temperature management at remote sites. Risk management is being transformed from reactive to proactive through analytics-driven planning, which is also minimizing buffer overbuild, expiration-driven waste, and resupply timing. Meanwhile, advances in temperature sensor technology and data integration are enhancing the reliability of cold chain and ultra-cold chain lanes by allowing for quicker detection of excursions and improved handoff points. With increasingly decentralized trial designs and operational complexities, digital enablement is being considered a differentiator that can drive both execution and compliance in clinical distribution.
How Will Clinical Drug Supply Chain Services Market Expansion Unfold Across Key Global Regions?
Future Market Insights identifies the United States (CAGR ~7.2%), India (~9.1%), China (~8.4%), Germany (~6.3%), the United Kingdom (~6.8%), and Japan (~5.9%) as the six core demand drivers for clinical drug supply chain services globally. The United States is driving demand anchored by complex trial profiles and high protocol complexity, enabled by integrated depot, packaging, and direct-to-site execution requirements. Growing trial volumes and the expansion of outsourcing, in addition to a rapidly increasing need for compliant cold chain distribution across multi-city investigator networks, are fueling China and India. Germany is maintaining regulated European execution enabled by documentation discipline, managed comparator procurement, and quality-focused depot execution. Stable Phase II-III volumes and ongoing needs for specialized labeling and cross-border distribution are fueling the United Kingdom. High compliance needs, controlled temperature handling, and precision-focused last-mile execution are driving Japan. According to FMI, the combined challenges of regulatory complexity, trial complexity, and temperature-sensitive logistics are maintaining a robust demand base through 2036.
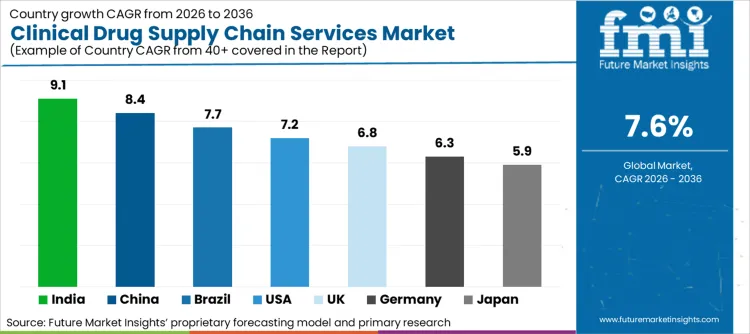
| Country | CAGR (2026-2036) |
|---|---|
| India | 9.1% |
| China | 8.4% |
| United States | 7.2% |
| United Kingdom | 6.8% |
| Germany | 6.3% |
| Japan | 5.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Is Integrated Trial Logistics Redefining Clinical Supply Chain Demand in the United States?
Future Market Insights analysts note that the United States clinical drug supply chain services market is expected to grow at a 7.2% CAGR, driven primarily by higher trial complexity, decentralized execution models, and stringent chain-of-custody expectations across investigational sites. Demand is being reinforced for integrated service bundles combining depot and warehousing, packaging and labeling, direct-to-patient and direct-to-site distribution, and returns and destruction management. FMI highlights that protocol amendments and adaptive trial designs are increasing resupply frequency and temperature-controlled shipment planning rather than simplifying operations. The requirement for standardized documentation and rapid turnaround timelines is further sustaining demand for specialized clinical logistics capabilities.
Why Is India Emerging as the Fastest-Growing Clinical Drug Supply Chain Services Market?
Future Market Insights identifies India as the fastest-growing market, with an estimated 9.1% CAGR, driven by expanding clinical trial activity, rising sponsor and CRO outsourcing intensity, and broader deployment of cold-chain handling capabilities across investigator networks. Clinical supply execution is being increasingly structured around compliant 2-8°C distribution, controlled ambient handling, and time-sensitive delivery for biologics-linked trials. FMI highlights that operational scale is being reinforced by the increasing use of centralized depots and coordinated last-mile distribution to reduce site-level burden and improve trial continuity. Increased focus on standardized labeling, comparator availability management, and returns processing is expected to sustain service demand growth through the forecast period.
How Is China’s Trial Scale and Cold-Chain Expansion Accelerating Service Adoption?
According to Future Market Insights, China’s clinical drug supply chain services market is projected to expand at an 8.4% CAGR, underpinned by increasing trial volumes, broader geographic dispersion of sites, and accelerating demand for compliant temperature-controlled distribution. Multi-city trial execution is increasing reliance on depot networks, validated packaging solutions, and monitored shipments across ambient, 2-8°C, and frozen lanes where required. FMI analysts observe that regulatory expectations around documentation and product integrity are intensifying the use of qualified service partners for comparator sourcing, labeling control, and controlled destruction workflows. As trial complexity rises, service demand is being driven more by execution precision and risk control than by shipment volume alone.
Does Germany’s Regulatory Rigor Sustain High-Value Clinical Supply Chain Demand?
As per FMI, Germany’s clinical drug supply chain services market to grow at a 6.3% CAGR, supported by regulatory-driven execution requirements rather than volume-led expansion. Clinical logistics in Germany is being shaped by stringent documentation standards, validated storage and handling expectations, and strong emphasis on product integrity across controlled distribution workflows. The demand is being reinforced for comparator sourcing discipline, high-accuracy labeling operations, and reliable depot and warehousing capabilities aligned to multi-country European trial needs. Quality-led temperature monitoring and compliant returns and destruction services are sustaining consistent demand for premium clinical supply chain services.
How Is the United Kingdom’s Multi-Country Trial Role Influencing Service Demand?
FMI states, the United Kingdom clinical drug supply chain services market is expected to expand at a 6.8% CAGR, shaped by sustained Phase II-III activity and its continued role in multi-country trial execution requiring labeling precision and coordinated distribution planning. Service demand is being supported by the need for validated packaging formats, rapid release cycles, and controlled shipment execution across ambient and 2-8°C lanes. FMI analysts emphasize that centralized planning and standardized documentation are being prioritized to reduce site-level operational strain and minimize deviation risk. As trial designs continue to evolve, demand is expected to remain structurally supported by workflow integration and compliance consistency.
Is Japan’s Compliance Intensity Creating Stable, High-Precision Service Demand?
Future Market Insights projects Japan’s clinical drug supply chain services market to grow at a 5.9% CAGR, driven by high compliance expectations, careful handling requirements, and a strong preference for execution reliability across clinical distribution workflows. Demand is being reinforced for validated storage, controlled-temperature shipment execution, and precise labeling and documentation practices aligned with strict quality expectations. FMI analysts note that operational planning is being shaped by punctual delivery requirements and risk-averse handling norms, sustaining steady demand for specialized clinical logistics services. Growth is expected to remain stable through continued emphasis on integrity-first trial execution rather than rapid shipment expansion.
What Strategic Moves Are Defining Competitive Landscape?
According to Future Market Insights, the competition is increasingly being shaped by specialization in clinical phases, temperature-controlled execution depth, and end-to-end trial integrity, rather than the scale of transportation per se. Infrastructure development is being reinforced in the areas of depot and warehousing, cold chain logistics, and comparator sourcing to support multi-country studies, especially in late-stage programs where risks of trial discontinuity are greatest. Differentiation is also being reinforced through builds of capabilities tailored to temperature bands, including ambient/CRT, 2-8°C, frozen (-20°C), ultra-cold (-70°C), and cryogenic (LN2) temperature ranges to support handling of biologics and CGTs.
Companies such as DHL Supply Chain, UPS Healthcare, and FedEx Logistics are being positioned as integrated clinical partners by integrating storage, packaging and labeling, distribution and transportation, and direct-to-patient coordination into unified operating models. According to FMI, the trend of customization by type of customer, including Pharma and Biopharma sponsors, CROs, CDMO/CMOs, and academic or NGO studies, is being supported by investments in digital visibility, returns and destruction management, and multimodal optimization over air, road, sea, and courier/express modes, and is reinforcing compliance-driven, execution-oriented ecosystems through 2036.
Recent Developments:
- On 9 Sept 2025, DHL Supply Chain strengthened its Life Sciences and Healthcare (LSHC) offerings by acquiring SDS Rx, enhancing final-mile delivery and healthcare transportation services for long-term care and specialty pharmacies, radiopharmacies, and health system networks. The acquisition was set to further enhance DHL Health Logistics by optimizing integrated and time-critical delivery execution for multiple steps of the LSHC supply chain.
- On 26 August 2025, EVERSANA and Waltz Health merged to create a new healthcare platform focused on enhancing pharmaceutical commercialization and patient access. The merger was set to combine EVERSANA’s global commercialization offerings with Waltz Health’s software-driven drug price marketplace and direct-to-payer platform to optimize drug access and value realization in the USA healthcare system.
Key Players in Clinical Drug Supply Chain Services Market
- DHL Supply Chain
- UPS Healthcare
- FedEx Logistics
- World Courier (Cencora)
- Marken (UPS)
- Kuehne+Nagel
- DB Schenker
- Catalent
- Almac Clinical Services
- Thermo Fisher Scientific
Market Definition
The clinical drug supply chain services market is described as the global market that is engaged in the planning, warehousing, handling, distribution, and management of investigational medicinal products for different clinical trial programs. The services are a critical component of the process that ensures the timely, compliant, and temperature-qualified distribution of clinical supplies across the drug development process, from early-phase clinical trials to late-stage trials and Phase IV or real-world evidence studies. The market is expressed in USD billion and projected from 2026 to 2036.
The market includes depot and warehousing, cold chain logistics, comparator sourcing, packaging and labeling, distribution and transportation, direct to patient delivery, and returns and destruction services. The demand is for Phase I, Phase II, Phase III, and Phase IV/RWE, with execution requirements including ambient/CRT, 2-8°C, frozen (-20°C), ultra-cold (-70°C), and cryogenic (LN2) temperature ranges. The services are provided to Pharma and Biopharma sponsors, CROs, CDMO/CMOs, and Academic or NGO institutions, with the execution of shipments by air, road, sea, and courier/express services based on clinical and regulatory needs.
The market does not include commercial pharmaceutical distribution, post-approval bulk drug logistics, non-clinical research sample transport, and general healthcare supply chain services that are not intended for clinical trial materials. Laboratory testing services, clinical data management systems, and manufacturing that are not directly related to the storage, handling, or distribution of clinical supplies are also excluded.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 11.5 Billion |
| Service Type | Depot & warehousing, Cold-chain logistics, Comparator sourcing, Packaging & labeling, Distribution & transportation, Direct-to-patient delivery, Returns & destruction |
| Clinical Phase | Phase I, Phase II, Phase III, Phase IV / RWE |
| Temperature Band | Ambient / CRT, 2-8°C, Frozen (-20°C), Ultra-cold (-70°C), Cryogenic (LN2) |
| Customer Type | Pharma / Biopharma sponsor, CRO, CDMO / CMO, Academic / NGO |
| Shipment Mode | Air, Road, Sea, Courier / express |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | USA, Germany, UK, China, Japan, India, Brazil, and 40+ countries |
| Key Companies Profiled | DHL Supply Chain, UPS Healthcare, FedEx Logistics, World Courier (Cencora), Marken (UPS), Kuehne+Nagel, DB Schenker |
| Additional Attributes | Revenue analysis by service type and clinical phase; temperature-band compliance assessment; customer-type service customization; shipment-mode optimization and regulatory execution capability |
Clinical Drug Supply Chain Services Market by Segments
Service Type
- Depot & Warehousing
- Cold-chain logistics
- Comparator sourcing
- Packaging & labeling
- Distribution & transportation
- Direct-to-patient delivery
- Returns & destruction
Clinical Phase
- Phase III
- Phase II
- Phase I
- Phase IV / RWE
Temperature Band
- Ambient / CRT
- 2-8°C
- Frozen (-20°C)
- Ultra-cold (-70°C)
- Cryogenic (LN2)
Customer Type
- Pharma / Biopharma sponsor
- CRO
- CDMO / CMO
- Academic / NGO
Shipment Mode
- Air
- Road
- Sea
- Courier / express
By Region:
- North America
- Europe
- East Asia
- South Asia
- Latin America
- Middle East & Africa
Bibliography
- USA Food and Drug Administration. (2024). Supply Chain Security Requirements for Clinical Investigational Drugs and Biologics. FDA.
- European Medicines Agency. (2024). Guidelines on Good Distribution Practice for Investigational Medicinal Products. EMA.
- World Health Organization. (2024). Technical Guidance on Cold Chain and Temperature-Controlled Distribution for Clinical Trials. WHO.
- International Council for Harmonisation. (2023). ICH Q9 and Q10: Quality Risk Management and Pharmaceutical Quality Systems in Clinical Supply Chains. ICH.
- United States Pharmacopeia. (2024). USP <1079> and <1083>: Good Storage and Distribution Practices for Drug Products. USP.
- Organisation for Economic Co-operation and Development. (2023). Pharmaceutical Logistics, Traceability, and Cross-Border Clinical Supply Compliance. OECD.
Frequently Asked Questions
What is the current global market size for clinical drug supply chain services?
The global clinical drug supply chain services market is valued at USD 11.5 billion in 2026, reflecting the growing operational complexity of multi-country clinical trials and expanding biologics pipelines.
What is the projected CAGR for the market over the next 10 years?
The market is projected to grow at a 7.6% CAGR from 2026 to 2036, supported by sustained growth in late-stage clinical trials and increasing outsourcing of supply chain functions.
Which regions are experiencing the fastest expansion?
India and China are experiencing the fastest expansion, with growth rates of 9.1% and 8.4%, respectively, driven by rising clinical trial activity, cost-efficient logistics infrastructure, and expanding CRO/CDMO ecosystems.
What are the primary market drivers?
Rising Phase III trial volumes, increasing temperature-sensitive biologic shipments, comparator sourcing complexity, and regulatory emphasis on GDP-compliant distribution are collectively driving demand for specialized clinical supply chain services.
Who are the leading service providers in the industry?
DHL Supply Chain, UPS Healthcare, and FedEx Logistics are among the leading providers, differentiated by global depot networks, validated cold-chain capabilities, and integrated clinical trial logistics platforms.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Service Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Service Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Service Type , 2026 to 2036
- Depot & warehousing
- Cold-chain logistics
- Comparator sourcing
- Packaging & labeling
- Distribution & transportation
- Direct-to-patient delivery
- Returns & destruction
- Depot & warehousing
- Y to o to Y Growth Trend Analysis By Service Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Service Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Clinical Phase
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Clinical Phase, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Clinical Phase, 2026 to 2036
- Phase III
- Phase II
- Phase I
- Phase IV / RWE
- Phase III
- Y to o to Y Growth Trend Analysis By Clinical Phase, 2021 to 2025
- Absolute $ Opportunity Analysis By Clinical Phase, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Temperature Band
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Temperature Band, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Temperature Band, 2026 to 2036
- Ambient CRT
- 2–8°C
- Frozen (-20°C)
- Ultra-cold (-70°C)
- Cryogenic (LN2)
- Ambient CRT
- Y to o to Y Growth Trend Analysis By Temperature Band, 2021 to 2025
- Absolute $ Opportunity Analysis By Temperature Band, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Customer Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Customer Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Customer Type, 2026 to 2036
- Pharma Biopharma sponsor
- CRO
- CDMO / CMO
- Academic / NGO
- Pharma Biopharma sponsor
- Y to o to Y Growth Trend Analysis By Customer Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Customer Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Shipment Mode
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Shipment Mode, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Shipment Mode, 2026 to 2036
- Air
- Road
- Sea
- Courier / express
- Air
- Y to o to Y Growth Trend Analysis By Shipment Mode, 2021 to 2025
- Absolute $ Opportunity Analysis By Shipment Mode, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Service Type
- By Clinical Phase
- By Temperature Band
- By Customer Type
- By Shipment Mode
- Competition Analysis
- Competition Deep Dive
- DHL Supply Chain
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- UPS Healthcare
- FedEx Logistics
- World Courier (Cencora)
- Marken (UPS)
- Kuehne+Nagel
- DB Schenker
- Catalent
- Almac Clinical Services
- Thermo Fisher Scientific
- DHL Supply Chain
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Clinical Phase, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Shipment Mode, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Clinical Phase, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Shipment Mode, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Clinical Phase, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Shipment Mode, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Clinical Phase, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Shipment Mode, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Clinical Phase, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Shipment Mode, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Clinical Phase, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Shipment Mode, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Clinical Phase, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Shipment Mode, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Clinical Phase, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Temperature Band, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Customer Type, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Shipment Mode, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Service Type
- Figure 6: Global Market Value Share and BPS Analysis by Clinical Phase, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Clinical Phase, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Clinical Phase
- Figure 9: Global Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Temperature Band
- Figure 12: Global Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Customer Type
- Figure 15: Global Market Value Share and BPS Analysis by Shipment Mode, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Shipment Mode, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Shipment Mode
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Service Type
- Figure 32: North America Market Value Share and BPS Analysis by Clinical Phase, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Clinical Phase, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Clinical Phase
- Figure 35: North America Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Temperature Band
- Figure 38: North America Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Customer Type
- Figure 41: North America Market Value Share and BPS Analysis by Shipment Mode, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Shipment Mode, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Shipment Mode
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Service Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Clinical Phase, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Clinical Phase, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Clinical Phase
- Figure 51: Latin America Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Temperature Band
- Figure 54: Latin America Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Customer Type
- Figure 57: Latin America Market Value Share and BPS Analysis by Shipment Mode, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Shipment Mode, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Shipment Mode
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Service Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Clinical Phase, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Clinical Phase, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Clinical Phase
- Figure 67: Western Europe Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Temperature Band
- Figure 70: Western Europe Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Customer Type
- Figure 73: Western Europe Market Value Share and BPS Analysis by Shipment Mode, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Shipment Mode, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Shipment Mode
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Service Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Clinical Phase, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Clinical Phase, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Clinical Phase
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Temperature Band
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Customer Type
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Shipment Mode, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Shipment Mode, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Shipment Mode
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Service Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Clinical Phase, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Clinical Phase, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Clinical Phase
- Figure 99: East Asia Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Temperature Band
- Figure 102: East Asia Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Customer Type
- Figure 105: East Asia Market Value Share and BPS Analysis by Shipment Mode, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Shipment Mode, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Shipment Mode
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Service Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Clinical Phase, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Clinical Phase, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Clinical Phase
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Temperature Band
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Customer Type
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Shipment Mode, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Shipment Mode, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Shipment Mode
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Service Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Clinical Phase, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Clinical Phase, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Clinical Phase
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Temperature Band, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Temperature Band, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Temperature Band
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Customer Type, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Customer Type, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Customer Type
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Shipment Mode, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Shipment Mode, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Shipment Mode
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

