Allergy Clinical Trial Services Market
Allergy Clinical Trial Services Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Allergy Clinical Trial Services Market Forecast and Outlook 2026 to 2036
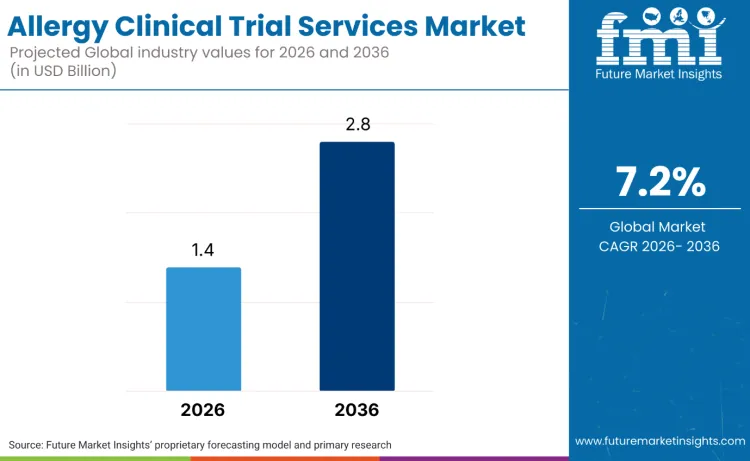
The global allergy clinical trial services market is forecasted to reach USD 1.4 billion in 2026 and expand to USD 2.8 billion by 2036, advancing at a CAGR of 7.2%. As per FMI analysis, market expansion shows growing pharmaceutical investment in allergy drug development, driven by rising allergy prevalence and unmet medical needs across allergic rhinitis, asthma, atopic dermatitis, and food allergy indications.
Companies are investing in specialized service capabilities to support complex allergy trial protocols. AI is also significantly enhancing allergy clinical trial services by improving efficiency, accuracy, and personalized treatment approaches. Thus, companies are harnessing the benefits of AI by improving services in the life sciences industry. For instance, in June 2025, IQVIA, a prominent clinical research services provider, rolled out AI agents at GTC Paris.
“IQVIA AI Assistant provides the precision, speed and trust required by our life sciences and healthcare customers. Through this purpose-built AI technology, we are fulfilling our mission of accelerating innovation to create a healthier world,” -Bernd Haas, senior vice president, IQVIA Digital Products and Solutions.
Market growth is supported by increasing complexity of allergy trial designs, requiring specialized expertise in patient phenotyping, biomarker analysis, and exposure challenge methodologies. Service providers are developing integrated platforms that combine traditional clinical operations with advanced allergy-specific capabilities including environmental exposure units, food challenge protocols, and immunological endpoint assessment.
The industry focus is shifting toward precision medicine approaches in allergy clinical development. Advanced clinical research organizations are integrating biomarker-guided patient selection, real-world evidence collection, and digital endpoint capture to improve trial efficiency and regulatory success rates. Research-focused service providers are gaining preference for their ability to support novel therapeutic modalities including biologics, cell therapies, and allergen immunotherapies.
Summary of the Allergy Clinical Trial Services Market
What Is Growth Outlook on the Allergy Clinical Trial Services Market as per Future Market Insights Projection?
Future Market Insights projects the allergy clinical trial services market to grow at a CAGR of 7.2% from 2026 to 2036, expanding from USD 1.4 billion in 2026 to USD 2.8 billion by 2036.
FMI Research Approach:FMI proprietary forecasting model using bottom-up revenue estimation
How Do FMI Analysts Perceive the Allergy Clinical Trial Services Market to Evolve?
FMI analysts perceive the market shifting from traditional clinical trial execution toward specialized, biomarker-driven platforms positioned as comprehensive allergy research solutions.
FMI Research Approach: Transition toward precision medicine approaches in allergy clinical development
Which Country Holds the Largest Share in the Global Allergy Clinical Trial Services Market?
The United States holds the largest share of the global allergy clinical trial services market by value.
FMI Research Approach: FMI country-level revenue modeling across pharmaceutical and biotech sponsors
How Large Will the Allergy Clinical Trial Services Market Be by 2036?
The global allergy clinical trial services market is estimated to reach USD 2.8 billion by 2036.
FMI Research Approach: Long-term revenue projections by service type and therapeutic indication
What Is the Definition of the Allergy Clinical Trial Services Market?
The allergy clinical trial services market comprises specialized research platforms designed to support clinical development of allergy therapeutics, including protocol development, patient recruitment, clinical operations, and regulatory support across pharmaceutical and biotech sponsors.
FMI Research Approach: FMI market taxonomy and definition framework, Inclusion of study design, site management, clinical monitoring, and data management services
What Are Globally Unique Trends Shaping the Allergy Clinical Trial Services Market?
Globally unique trends include the rise of precision allergy medicine, environmental exposure chamber adoption, and the integration of real-world evidence collection in allergy clinical development.
FMI Research Approach: Increased use of biomarker-guided patient stratification and personalized treatment approaches, Growth in controlled allergen challenge studies and environmental exposure protocols
Allergy Clinical Trial Services Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 1.4 billion |
| Market Forecast Value (2036) | USD 2.8 billion |
| Forecast CAGR (2026-2036) | 7.2% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research.
How Do Regulatory Standards and Protocol Requirements Shape Market Demand?
Evolving regulatory guidance for allergy diagnostics and allergy clinical trials is driving demand toward specialized service providers with expertise in complex trial designs and allergen exposure methodologies. Regulatory authorities in North America and Europe are establishing specific requirements for allergen immunotherapy trials, environmental exposure studies, and biomarker validation, creating opportunities for service providers with demonstrated experience in these specialized areas.
This environment favors organizations with established relationships with regulatory agencies, validated exposure chamber facilities, and expertise in allergy-specific endpoints. Companies are prioritizing service partners with track records in successful regulatory submissions and familiarity with evolving guidance documents for allergy therapeutic development. They are also focusing on securing FDA approvals to cater to huge patient profiles. In April 2025, Dupixent secured the USA FDA approval for the treatment of adults and adolescents aged 12 years and older diagnosed with chronic spontaneous urticaria (CSU) who remain symptomatic despite histamine-1 (H1) antihistamine treatment.
How is the Allergy Clinical Trial Services Market Segmented?
The allergy clinical trial services market has been segmented based on service type, therapeutic indication, end user, and region. In terms of service type, the market is divided into study design and protocol development, site management and patient recruitment, clinical operations and monitoring, bioanalytical and laboratory services, data management and biostatistics, and regulatory affairs and medical writing. By therapeutic indication, the market is divided into allergic rhinitis, allergic asthma, atopic dermatitis, food allergies, and drug hypersensitivity. By end user, the market is segmented into pharmaceutical companies, biotechnology firms, academic research institutions, and government organizations. Regionally, the market is divided into North America, Europe, Asia Pacific, Latin America, and Middle East and Africa.
Why Do Clinical Operations and Monitoring Services Dominate the Allergy Clinical Trial Services Market?
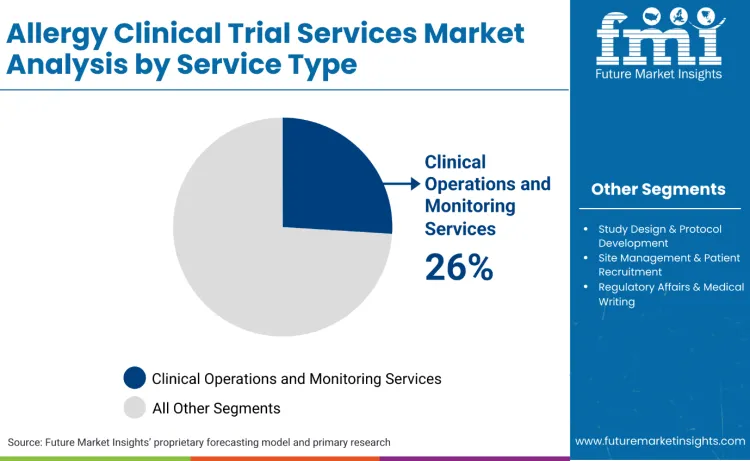
Clinical operations and monitoring services lead the market, accounting for 26.0% of the share. Allergy clinical trials require specialized monitoring capabilities due to the complex nature of allergen exposure protocols, safety considerations during challenge studies, and the need for real-time assessment of allergic reactions. These services are critical for ensuring patient safety during controlled allergen exposure, managing emergency response protocols, and maintaining data quality across multiple specialized endpoints. Companies, such as IQVIA provide operational excellence and clinical development support for allergy and respiratory therapeutics, demonstrating its involvement in clinical trial execution and site operations.
The dominance of clinical operations services shows the technical complexity of allergy trials, which often involve environmental exposure chambers, food challenge protocols, and specialized safety monitoring. Service providers must maintain expertise in allergy-specific adverse event assessment, have established relationships with specialized investigative sites, and possess capabilities for managing complex logistics around allergen sourcing and standardization.
How Do End User Requirements Influence Service Provider Selection?
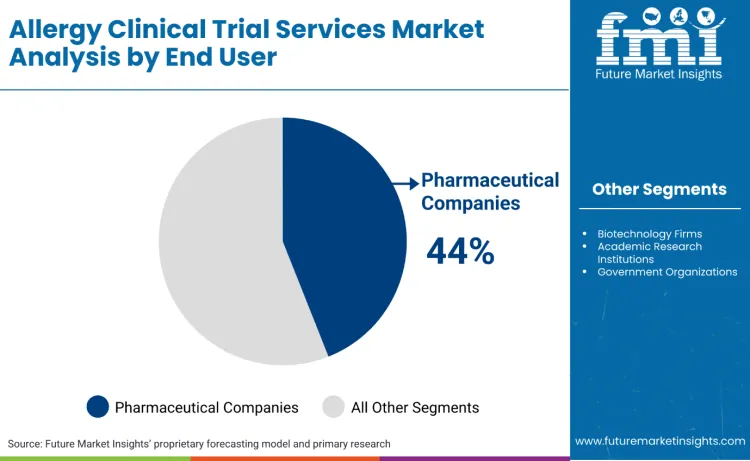
Pharmaceutical companies represent the dominant end user segment, accounting for 44.0% of the market share. Pharmaceutical sponsors drive demand for comprehensive allergy clinical trial services due to their focus on large-scale therapeutic development programs, regulatory submission requirements, and need for specialized expertise across multiple therapeutic indications. These companies require service providers with demonstrated experience in complex protocol designs, established regulatory relationships, and capabilities for managing multinational clinical trial programs.
The preference for pharmaceutical company partnerships reflects the industry's shift toward precision medicine approaches and biomarker-driven trial designs in allergy therapeutic development. Service providers must demonstrate expertise in novel therapeutic modalities including biologics, cell therapies, and allergen immunotherapies while maintaining capabilities for traditional small molecule development. Companies such as IQVIA support pharmaceutical companies in allergy clinical trials by delivering comprehensive clinical research services.
How Are Advanced Biomarker Platforms and Digital Endpoints Reshaping Allergy Clinical Trials?
The allergy clinical trial services market is shifting toward integrated biomarker analysis and digital endpoint collection platforms that provide comprehensive immunological profiling alongside traditional clinical assessments. In February 2026, Medtronic announced to acquire CathWorks, a privately held medical device company for USD 585 million to boost its innovation strategy in diagnostics and treatment. This trend represents a transition from traditional symptom-based endpoints toward molecular and digital biomarker integration that enables more precise efficacy assessment.
Advanced biomarker platforms and digital endpoints are revolutionizing allergy clinical trials by enabling real-time, remote data collection and personalized treatment insights. These technologies enhance trial accuracy, accelerate patient recruitment, and improve monitoring of allergic responses. As a result, they increase trial efficiency, reduce costs, and support more targeted therapeutic development.
How Is the Allergy Clinical Trial Services Market Evolving Globally?
Global demand for allergy clinical trial services is increasing as pharmaceutical companies expand therapeutic development while addressing regulatory requirements, patient safety considerations, and specialized endpoint assessment across diverse indication areas. Growth demonstrates rising adoption of biomarker-driven trial designs, environmental exposure methodologies, and integrated service platforms across pharmaceutical companies, biotechnology firms, and academic research institutions. Service selection focuses on therapeutic expertise, regulatory track record, and specialized infrastructure capabilities across various allergy conditions.
China records 13.8% CAGR, India records 12.4% CAGR, Brazil records 10.7% CAGR, USA records 6.9% CAGR, and Germany records 5.3% CAGR. Market expansion remains driven by therapeutic innovation and regulatory expertise rather than cost considerations alone.
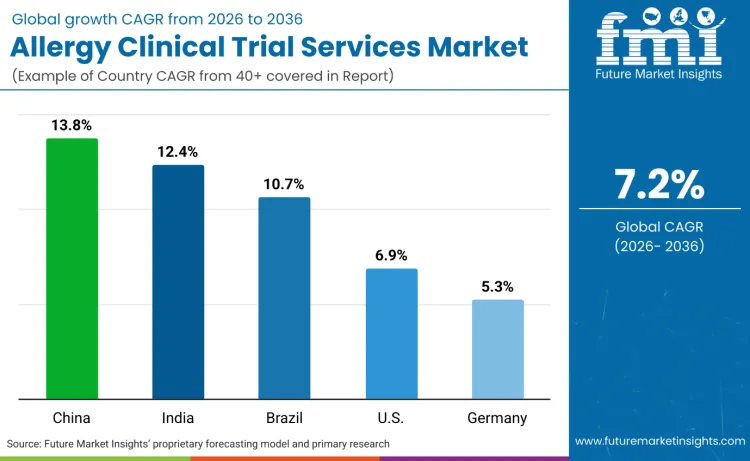
| Country | CAGR (2026-2036) |
|---|---|
| China | 13.8% |
| India | 12.4% |
| Brazil | 10.7% |
| USA | 6.9% |
| Germany | 5.3% |
Source: FMI historical analysis and forecast data.
China: How is Expanding Pharmaceutical Investment Driving Specialized Service Adoption?
The primary driver in China is the significant growth of domestic pharmaceutical investment in allergy drug development and international clinical trial participation. With the expansion of regulatory harmonization initiatives, Chinese clinical research sites are experiencing increased demand for allergy-specific trial capabilities, making traditional general clinical research approaches insufficient. This shift has promoted specialized service platform adoption for complex allergy protocols to ensure regulatory compliance and data quality. The Chinese allergy clinical trial services market is set to grow at a 13.8% CAGR during the study period, driven by pharmaceutical investment and international collaboration in allergy research.
China is transitioning toward becoming a global contributor to allergy clinical research, expanding beyond its traditional role as a cost-effective clinical trial location. Companies are establishing specialized allergy research capabilities to support both domestic drug development and international pharmaceutical partnerships. In February 2025, WuXi Clinical launched its Allergy Center of Excellence in Shanghai, featuring environmental exposure chambers and specialized bioanalytical capabilities for international allergy clinical trials.
India: Is the Growing CRO Infrastructure Elevating Allergy Research Standards?
In India, the market is driven by expanding contract research organization infrastructure and increasing pharmaceutical investment in allergy therapeutic development. The establishment of specialized allergy research capabilities is essential for Indian service providers seeking to participate in international clinical trial programs and support domestic pharmaceutical innovation. The Indian allergy clinical trial services market is projected to grow at a 12.4% CAGR during the study period.
India's allergy clinical trial services market is expanding as local contract research organizations develop specialized capabilities to support international pharmaceutical partnerships. Growing investigator expertise, expanding clinical research infrastructure, and increasing regulatory alignment with international standards are driving sustained adoption of allergy-specific trial methodologies across major metropolitan areas.
Brazil: Will Expanding Clinical Research Infrastructure Accelerate Service Integration?
Brazil's allergy clinical trial services market is shaped by expanding clinical research infrastructure and increasing participation in international pharmaceutical development programs. Brazilian service providers are developing comprehensive allergy research capabilities, combining established clinical operations expertise with specialized allergy trial methodologies. The Brazilian allergy clinical trial services market is expected to grow at a 10.7% CAGR during the study period, as international pharmaceutical companies increase clinical trial activity for allergy testers in Latin America.
Brazil presents opportunities centered on its expanding role in international clinical trial networks and growing domestic pharmaceutical innovation. The market is characterized by increasing investment in specialized research infrastructure and investigator training programs focused on allergy and immunology clinical research.
Germany: Is the Research Excellence Strategy Advancing Specialized Trial Capabilities?
In Germany, the market is driven by academic medical center excellence and pharmaceutical industry investment in specialized allergy research capabilities. German research institutions are developing integrated platforms that combine academic expertise with commercial clinical trial execution, creating comprehensive allergy research ecosystems. The German allergy clinical trial services market is set to grow at a 5.3% CAGR during the study period, focusing on research quality and regulatory expertise.
Germany offers opportunities rooted in academic research excellence and pharmaceutical industry collaboration. The market benefits from established relationships between academic medical centers and pharmaceutical companies, creating integrated research networks that combine scientific expertise with commercial clinical trial capabilities. The presence of leading companies such as Allergy Therapeutics PLC is further boosting market growth. Allergy Therapeutics PLC actively invests in allergy treatment development, focusing on research, production and commercialization of allergen‑specific immunotherapies targeting the underlying causes of allergic diseases.
USA: How is the Innovation Ecosystem Driving Specialized Service Development?
The USA market is focused on innovation in allergy clinical trial methodologies and regulatory leadership in specialized therapeutic areas. This transition aims at maintaining competitive advantages in pharmaceutical development while improving clinical trial efficiency and regulatory success rates. The USA allergy clinical trial services market is set to grow at a 6.9% CAGR during the study period.
The USA allergy clinical trial services market continues to expand as pharmaceutical investment in allergy therapeutics intersects with advancing clinical research methodologies. High prevalence of allergic conditions, established clinical research infrastructure, and regulatory leadership in specialized therapeutic areas sustain demand for comprehensive allergy clinical trial services. Service provider preference is shifting toward organizations with demonstrated expertise in biomarker-driven trial designs, environmental exposure methodologies, and regulatory affairs capabilities. Leading players are expanding their footprint in the country to boost their customer base. For instance, in 2024, Medpace invested to restructure the Cincinnati campus.
Who are the Major Players Active in the Allergy Clinical Trial Services Market?
The competitive landscape for the allergy clinical trial services market is defined by strategic investment in specialized infrastructure, drug discovery, and the integration of biomarker platforms to address comprehensive allergy research requirements. As pharmaceutical preferences prioritize regulatory success and therapeutic differentiation, the market features intensive competition focused on expanding specialized capabilities and therapeutic expertise. Market leaders are pursuing strategic partnerships with academic medical centers, acquiring specialized testing laboratories, and developing proprietary platforms that provide integrated research solutions from protocol development through regulatory submission.
Market leader IQVIA maintains dominance through successful expansion of specialized allergy research capabilities and advanced bioanalytical services, which provide comprehensive immunological profiling for diverse therapeutic approaches. ICON has strategically positioned its portfolio toward environmental exposure chamber studies, developing standardized protocols that meet international regulatory requirements. Parexel is gaining ground by integrating academic partnerships into its commercial service offerings to enhance scientific credibility and regulatory acceptance. Specialized players like Syneos Health and Labcorp Drug Development are competing on therapeutic expertise, with Syneos Health focusing on integrated drug development services and Labcorp emphasizing laboratory-based endpoint assessment capabilities.
Key Industry Developments:
- In January 2026, Worldwide Clinical Trials unveiled to acquire Catalyst Clinical Research to boost its manufacturing capability.
- In June 2023, Charles River Laboratories completed the acquisition of SAMDI Tech, Inc, label-free high-throughput screening (HTS) solutions for drug discovery research provider.
Key Players in the Allergy Clinical Trial Services Market
- IQVIA Inc.
- ICON plc
- Parexel International Corporation
- Syneos Health Inc.
- Labcorp Drug Development
- PPD, Inc.
- Charles River Laboratories
- Medpace Holdings Inc.
- PRA Health Sciences
- Worldwide Clinical Trials
Market Definition
The allergy clinical trial services market refers to specialized research platforms designed to support clinical development of allergy therapeutics, encompassing protocol development, patient recruitment, clinical operations, bioanalytical services, and regulatory support. The market includes precision and standard service offerings across various therapeutic indications including allergic rhinitis, asthma, atopic dermatitis, food allergies, and drug hypersensitivity. Market size is measured in USD billion and analyzed over the 2026 to 2036 period.
The allergy clinical trial services market includes specialized methodologies such as environmental exposure chamber studies, controlled allergen challenge protocols, biomarker analysis, and digital endpoint collection, utilized by pharmaceutical companies, biotechnology firms, and academic research institutions. Market expansion is driven by increasing pharmaceutical investment in allergy therapeutics, advancing clinical trial methodologies, and evolving regulatory requirements for specialized therapeutic areas.
Market Inclusion
Included in the allergy clinical trial services market scope are specialized research services categorized by service type (protocol development, clinical operations, bioanalytical services, data management, regulatory affairs), therapeutic indication (allergic rhinitis, asthma, atopic dermatitis, food allergies), end user (pharmaceutical companies, biotech firms, academic institutions), and service complexity (standard protocols, specialized methodologies, integrated platforms). The scope encompasses allergy-specific trial capabilities with focus on regulatory compliance, patient safety, and therapeutic differentiation.
Geographically, the market encompasses key regions including North America, Europe, Asia Pacific, Latin America, and Middle East and Africa, with country-level analysis for significant markets including China, India, Brazil, USA, and Germany.
Market Exclusion
Excluded from the allergy clinical trial services market scope are general clinical research services not specifically designed for allergy therapeutic development, basic contract research activities without specialized allergy expertise, and non-clinical research services such as preclinical testing or regulatory consulting unrelated to clinical trial execution. Products and services not intended for allergy clinical development applications are not included.
The market excludes non-specialized clinical research platforms and services that do not incorporate allergy-specific methodologies, specialized endpoint assessment, or therapeutic expertise. Services with non-compliant regulatory specifications or those that do not meet international standards for specialized clinical research are outside the defined market scope.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Service Type | Study Design & Protocol Development; Site Management & Patient Recruitment; Clinical Operations & Monitoring; Bioanalytical & Laboratory Services; Data Management & Biostatistics; Regulatory Affairs & Medical Writing |
| Therapeutic Indication | Allergic Rhinitis; Allergic Asthma; Atopic Dermatitis; Food Allergies; Drug Hypersensitivity |
| End User | Pharmaceutical Companies; Biotechnology Firms; Academic Research Institutions; Government Organizations |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | China, India, Brazil, USA, Germany, and 40+ countries |
| Key Companies Profiled | IQVIA; ICON; Parexel; Syneos Health; Labcorp Drug Development; Others |
Allergy Clinical Trial Services Market by Segment
Service Type:
- Study Design & Protocol Development
- Site Management & Patient Recruitment
- Clinical Operations & Monitoring
- Bioanalytical & Laboratory Services
- Data Management & Biostatistics
- Regulatory Affairs & Medical Writing
Therapeutic Indication:
- Allergic Rhinitis
- Allergic Asthma
- Atopic Dermatitis
- Food Allergies
- Drug Hypersensitivity
End User:
- Pharmaceutical Companies
- Biotechnology Firms
- Academic Research Institutions
- Government Organizations
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- Thailand
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- Netherlands
- Rest of Europe
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Colombia
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- USA Food and Drug Administration. (2023). Guidance for industry: Clinical development programs for allergen immunotherapy products. USA Department of Health and Human Services.
- European Academy of Allergy and Clinical Immunology. (2024). EAACI guidelines for allergen challenge studies and biomarker endpoints in clinical trials. EAACI.
- World Allergy Organization. (2024). Clinical practice resources for allergic rhinitis, asthma, food allergy, and urticaria.
Frequently Asked Questions
How big is the allergy clinical trial services market in 2026?
The global allergy clinical trial services market is estimated to be valued at USD 1.4 billion in 2026.
What will be the size of the allergy clinical trial services market in 2036?
The market size for the allergy clinical trial services market is projected to reach USD 2.8 billion by 2036.
How much will the allergy clinical trial services market grow between 2026 and 2036?
The allergy clinical trial services market is expected to grow at a 7.2% CAGR between 2026 and 2036.
What are the key service types in the allergy clinical trial services market?
The key service types include clinical operations and monitoring, site management and patient recruitment, bioanalytical and laboratory services, and study design and protocol development.
Which end user segment will contribute a significant share in the allergy clinical trial services market in 2026?
In terms of end users, pharmaceutical companies are set to command a 44.0% share in the allergy clinical trial services market in 2026.
Table of Content
- Allergy Clinical Trial Services Market Size and Share Forecast Outlook (2026 to 2036)
- Allergy Clinical Trial Services Market Forecast and Outlook (2026-2036)
- Allergy Clinical Trial Services Market Key Takeaways
- Key Shifts in the Allergy Clinical Trial Services Market from 2026 to 2036
- Allergy Clinical Trial Services Market Segmentation Analysis
- By Service Type
- Study Design & Protocol Development
- Site Management & Patient Recruitment
- Clinical Operations & Monitoring
- Bioanalytical & Laboratory Services
- Data Management & Biostatistics
- Regulatory Affairs & Medical Writing
- By Therapeutic Indication
- Allergic Rhinitis
- Allergic Asthma
- Atopic Dermatitis
- Food Allergies
- Drug Hypersensitivity
- By End User
- Pharmaceutical Companies
- Biotechnology Firms
- Academic Research Institutions
- Government Organizations
- By Region
- By Service Type
- Market Dynamics and Growth Analysis
- Growth Drivers
- Market Restraints
- Strategic Trends
- Allergy Clinical Trial Services Market Global Analysis
- Asia Pacific
- North America
- Europe
- Latin America
- Middle East & Africa
- Competitive Landscape Analysis
- Key Players in the Allergy Clinical Trial Services Market
- Market Definition, Inclusion, and Exclusion Criteria
- Scope of the Allergy Clinical Trial Services Market Report
- Allergy Clinical Trial Services Market by Segment
- Bibliography
- Frequently Asked Questions (FAQs)
List of Tables
- Global Allergy Clinical Trial Services Market Size (USD Billion), 2021-2036
- Allergy Clinical Trial Services Market Y-o-Y Growth (%), 2021-2036
- Global Market Share by Treatment Service Type, 2026
- Global Market Share by Target Therapeutic Indication, 2026
- Global Market Share by End User, 2026
- Allergy Clinical Trial Services Market Share by Region, 2026
- Market Share by Treatment Service Type (USD Billion), 2026-2036
- Market Share by End User (USD Billion), 2026-2036
- Key Player Market Share in the Allergy Clinical Trial Services Market
- Allergy Clinical Trial Services Market Absolute $ Opportunity (USD Billion), 2026-2036
- Allergy Clinical Trial Services Market CAGR (%) by Region, 2026-2036
List of Figures
- Global Allergy Clinical Trial Services Market Size (USD Billion), 2021-2036
- Allergy Clinical Trial Services Market Y-o-Y Growth (%) from 2021-2036
- Allergy Clinical Trial Services Market Share by Treatment Service Type (%) in 2026
- Allergy Clinical Trial Services Market Share by Target Therapeutic Indication (%) in 2026
- Global Allergy Clinical Trial Services Market by Region (%) 2026-2036
- Global Allergy Clinical Trial Services Market by End User
- Competitive Landscape of Key Allergy Clinical Trial Services Market Players
- Market Trends in Photoaging Treatment
- Photoaging Treatment Adoption by Region
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

