Clinical Trial Data Management Service Market
Clinical Trial Data Management Service Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 to 2023 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2026 to 2035
Clinical Trial Data Management Service Market Forecast and Outlook 2025 to 2035
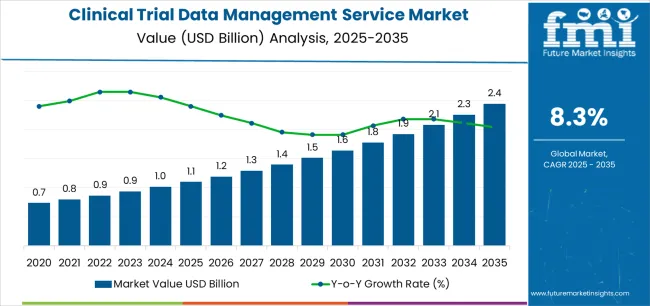
The clinical trial data management service market is valued at USD 1.1 billion in 2025 and is projected to reach USD 2.5 billion by 2035, advancing at a CAGR of 8.3%. Growth is supported by the expansion of global clinical research activity, increasing complexity of study protocols, and rising demand for accurate, compliant, and technology-enabled data management solutions. Pharmaceutical, biotechnology, and contract research organizations are adopting digital platforms to ensure data integrity, real-time access, and regulatory compliance across multi-site clinical operations.
Electronic Data Capture (EDC) services represent the leading segment, offering structured, automated systems that enhance accuracy and speed of data entry while reducing manual errors. The shift from paper-based processes to centralized digital workflows continues to improve operational efficiency, protocol adherence, and monitoring transparency. Integration of analytics tools, cloud-based architectures, and AI-assisted validation processes further enhances data accuracy and decision-making throughout trial lifecycles.
Quick Stats for Clinical Trial Data Management Service Market
- Clinical Trial Data Management Service Market Value (2025): USD 1.1 billion
- Clinical Trial Data Management Service Market Forecast Value (2035): USD 2.5 billion
- Clinical Trial Data Management Service Market Forecast CAGR: 8.3%
- Leading Type in Clinical Trial Data Management Service Market: Electronic Data Capture (EDC) Service
- Key Growth Regions in Clinical Trial Data Management Service Market: Asia Pacific, Europe, North America
- Top Players in Clinical Trial Data Management Service Market: Caidya, Cytel, PPD (Thermo Fisher Scientific Inc.), Veristat, Bioforum, Precision for Medicine, Emerald Clinical, Syneos Health, eClinical Solutions, Tata Consultancy Services (TCS)
Asia Pacific is the fastest-growing regional market, driven by expanding clinical research infrastructure and cost-efficient trial execution in emerging economies. Europe and North America maintain steady growth due to established regulatory frameworks and advanced technological adoption. Major market participants include Caidya, Cytel, PPD (Thermo Fisher Scientific Inc.), Veristat, and Bioforum, focusing on service quality, regulatory expertise, and scalable digital platforms.
What is the Growth Forecast for the Clinical Trial Data Management Service Market through 2035?
The clinical trial data management service market is projected to expand from USD 1.1 billion in 2025 to USD 2.5 billion by 2035, reflecting a strong CAGR of 8.3%. The 10-year growth comparison highlights significant acceleration in data-centric clinical operations as pharmaceutical and biotechnology firms increase outsourcing to specialized service providers. Between 2025 and 2030, market expansion will be driven by rising trial volumes, adoption of electronic data capture systems, and the integration of real-time monitoring tools that enhance data accuracy and regulatory compliance.
From 2030 to 2035, growth will intensify as decentralized and hybrid clinical trial models become more prevalent, supported by cloud-based platforms and advanced analytics. The market value will more than double over the decade, illustrating the sector’s transition from traditional data handling methods to automated and AI-enabled systems. Service providers offering integrated, compliant, and scalable data management frameworks will capture the most value during this phase. The 10-year comparison underscores a transformation toward digital efficiency and structured data governance, aligning with the industry’s emphasis on transparency, quality assurance, and faster drug development cycles.
Clinical Trial Data Management Service Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2025) | USD 1.1 billion |
| Market Forecast Value (2035) | USD 2.5 billion |
| Forecast CAGR (2025-2035) | 8.3% |
Why is the Clinical Trial Data Management Service Market Growing?
The clinical trial data management service market is expanding as pharmaceutical and biotechnology companies increase outsourcing of data handling to specialised service providers to improve trial efficiency and regulatory compliance. Data management services help manage vast volumes of patient-level data, e-clinical data capture, and trials with complex site networks. Stakeholders benefit from centralised electronic data management, quality-assured data cleaning, and integrated analytics tailored to clinical trial workflows.
Digitisation of trials, including cloud-based platforms and hybrid/decentralised trials, further drives demand for professional data management services. At the same time, regulatory requirements such as Good Clinical Practice (GCP) and 21 CFR Part 11 elevate the need for robust data handling processes. Constraints on growth include high complexity and cost of advanced data management operations, data privacy and cybersecurity risks, and variation in trial infrastructure across regions that may limit uptake of full-service data management outsourcing.
Which Classification and Application Segments Lead the Clinical Trial Data Management Service Market?
The clinical trial data management service market is segmented by classification and application. By classification, the market is divided into electronic data capture (EDC) service, paper and fax-based service, and hybrid services. Based on application, it is categorized into pharmaceutical and biotech clinical trials, medical device clinical trials, and other research-based applications. Regionally, the market is divided into Asia Pacific, Europe, North America, and other key regions.
By Classification, the Electronic Data Capture (EDC) Service Segment Accounts for the Largest Market Share
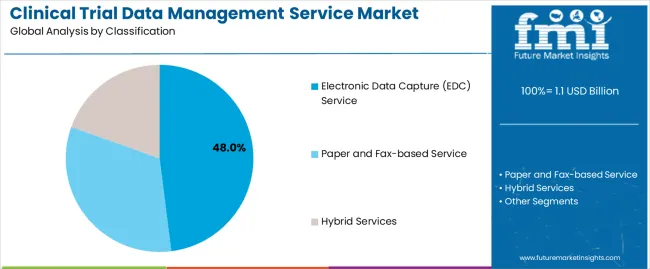
The electronic data capture (EDC) service segment holds the leading position in the clinical trial data management service market, representing approximately 48.0% of total market share in 2025. EDC systems are the preferred mode of data collection and management in modern clinical research, offering real-time data entry, validation, and monitoring capabilities that improve data integrity and operational efficiency. These platforms reduce manual entry errors, enable remote access for multi-site studies, and facilitate faster database lock times, which collectively enhance overall trial timelines.
The segment’s growth is supported by increasing adoption among contract research organizations (CROs), sponsors, and academic institutions conducting multi-phase, multi-country clinical trials. The transition from paper-based to digital systems reflects the broader movement toward standardized, regulatory-compliant, and audit-ready data handling solutions. Paper and fax-based services, while declining, remain in use for limited-scope or resource-constrained studies. Hybrid services combine electronic and manual approaches to accommodate trials in regions with inconsistent digital infrastructure.
Key factors supporting the EDC service segment include:
- Rising adoption of centralized digital trial management systems by CROs and sponsors.
- Enhanced data accuracy, security, and traceability through real-time electronic monitoring.
- Compliance with international clinical data standards such as CDISC and FDA 21 CFR Part 11.
- Ongoing digital transformation in clinical research operations and multi-site study coordination.
By Application, the Pharmaceutical and Biotech Clinical Trials Segment Represents the Dominant Market Category
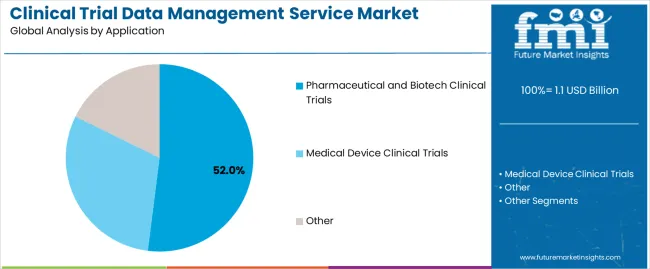
The pharmaceutical and biotech clinical trials segment accounts for approximately 52.0% of the clinical trial data management service market in 2025. This category dominates due to the high volume of ongoing drug development programs, biologics research, and vaccine trials requiring structured and regulatory-compliant data management frameworks. The increasing complexity of clinical studies, combined with a growing emphasis on patient safety and data transparency, has accelerated reliance on professional data management services in this segment.
Medical device clinical trials form a secondary category, driven by expanded global regulatory submissions and the need for precise device performance data documentation. The “other” segment includes investigator-initiated and academic trials where limited-scale data management systems are employed.
Primary dynamics driving demand from pharmaceutical and biotech clinical trials include:
- Expansion of multi-phase global clinical studies with increased patient recruitment.
- Stringent data governance requirements under regulatory bodies such as the FDA and EMA.
- Integration of digital platforms enabling real-time monitoring and cross-site coordination.
- Growth of outsourcing models among sponsors to enhance operational efficiency and compliance.
What are the Drivers, Restraints, and Key Trends of the Clinical Trial Data Management Service Market?
Increasing clinical trial volumes, growing outsourcing of data services, and adoption of digital data-management platforms are fostering service demand.
The clinical trial data management service market is being driven by an expanding global pipeline of drug and biologic development programmes, which elevates the volume of data needing management and processing. Life-science companies increasingly outsource data-collection and management functions to specialist service providers, allowing internal teams to focus on core R&D work. The adoption of cloud-based platforms, electronic data capture tools and analytics-enabled workflows further supports service uptake, as sponsors seek high-quality, compliant data-processing and endpoint-management capabilities.
Complex regulatory requirements, data-security concerns and integration challenges limit market growth.
Stringent requirements governing clinical-trial data integrity, audit trails, traceability and regulatory submission formats create significant service-provider burden and cost. Protecting sensitive participant data across multi-site, multi-country trials raises security and privacy risks that deter some organisations from outsourcing. Legacy systems, heterogeneous data-sources and the need to integrate disparate formats (such as EDC, eTMF, imaging and laboratory data) impose operational and technical constraints that slow adoption and raise service delivery cost.
Rise of decentralised trial models, analytics-driven workflows and regional expansion shape service-market evolution.
The market is trending toward decentralised and hybrid clinical-trial designs that generate higher-volume, real-time data from remote devices and wearables; data-management service providers are adapting by offering integrated analytics, remote monitoring and data-quality tools. Demand is increasing in emerging markets across Asia-Pacific and Latin America as trial-sponsors extend geography; service providers are establishing local capacity to meet regional regulatory and infrastructure demands. A shift toward outcome-driven service models and platform-as-a-service offerings signals growing maturity in data-management services within the broader clinical-trial ecosystem.
Analysis of the Clinical Trial Data Management Service Market by Key Country
The global clinical trial data management service market is expanding rapidly through 2035, supported by advancements in electronic data capture systems, decentralized trials, and regulatory digitalization. China leads with a 11.2% CAGR, followed by India at 10.4%, reflecting strong growth in pharmaceutical R&D and contract research outsourcing. Germany records 9.5%, supported by clinical technology innovation and regulatory harmonization, while Brazil’s 8.7% growth is driven by expanding healthcare trials and government-led research initiatives. The United States grows at 7.9%, maintaining leadership in cloud-based trial systems. The United Kingdom (7.1%) and Japan (6.2%) sustain steady progress through data integrity and regulatory modernization initiatives.

| Country | CAGR (%) |
|---|---|
| China | 11.2 |
| India | 10.4 |
| Germany | 9.5 |
| Brazil | 8.7 |
| USA | 7.9 |
| UK | 7.1 |
| Japan | 6.2 |
How is China leading the Clinical Trial Data Management Service Market?
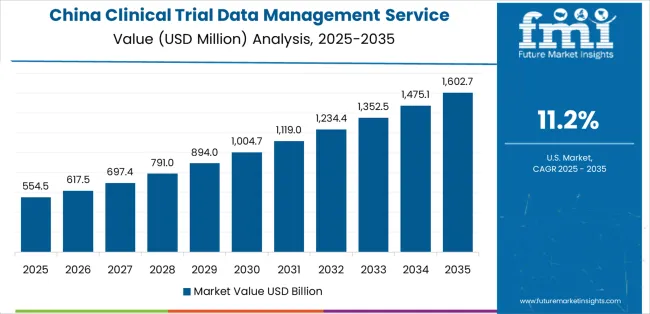
China’s market grows at 11.2% CAGR, driven by large-scale clinical research expansion and rising digitalization of trial operations. The government’s pharmaceutical innovation policies and reforms in clinical trial approval processes have encouraged outsourcing of data management to specialized service providers. Domestic contract research organizations (CROs) are investing in cloud-based electronic data capture (EDC) platforms and real-world data integration tools. Collaborations between hospitals, academic centers, and biopharma firms are enhancing data quality and compliance. The market also benefits from foreign CROs establishing regional data centers to support multi-country trials and local sponsor operations.
Key Market Factors:
- Expansion of clinical research infrastructure and CRO partnerships
- Implementation of electronic data capture and centralized data systems
- Policy reforms accelerating trial approvals and data digitalization
- Increased foreign investment in localized trial data management services
How is India strengthening its Clinical Trial Data Management Service Market?
India’s market grows at 10.4% CAGR, supported by its strong position as a global hub for clinical research outsourcing. The country’s contract research organizations are expanding capabilities in electronic trial master file (eTMF) management, data curation, and regulatory submission services. The National Digital Health Mission and evolving Good Clinical Practice (GCP) frameworks promote the use of digital platforms for trial documentation. India’s growing biopharmaceutical pipeline and cost-effective service ecosystem attract international sponsors seeking reliable data management solutions. Investments in clinical analytics, AI-assisted trial monitoring, and cloud integration are enhancing operational transparency and quality assurance.
Market Development Factors:
- Expansion of CRO services in data management and analytics
- Adoption of AI and automation in clinical data processing
- Government initiatives supporting digital health and clinical transparency
- Competitive outsourcing environment attracting global trial sponsors
How is Germany advancing the Clinical Trial Data Management Service Market?
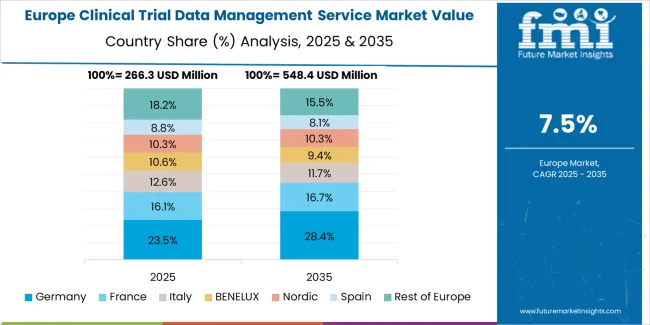
Germany’s market grows at 9.5% CAGR, driven by its strong regulatory environment, research infrastructure, and clinical technology integration. The country’s healthcare digitization initiatives under the European Health Data Space framework are improving interoperability and real-time data access. German CROs and technology vendors are developing secure EDC systems and audit-compliant databases. Pharmaceutical companies leverage hybrid clinical models combining traditional trials with remote data collection. Increased collaboration between biotech startups and IT providers supports continuous innovation in trial informatics. Germany’s focus on data privacy, cybersecurity, and system validation ensures high standards of data integrity across all phases of clinical research.
Key Market Characteristics:
- Integration of clinical data systems under EU interoperability standards
- Expansion of hybrid and decentralized trial models
- Strong compliance culture ensuring audit-ready data management
- Collaboration between biotech firms and IT providers in clinical technology development
How is Brazil expanding its Clinical Trial Data Management Service Market?
Brazil’s market grows at 8.7% CAGR, supported by the rise in multinational clinical research, improving regulatory processes, and expanding healthcare digitalization. National health agencies are streamlining ethical and data review procedures, encouraging trial sponsors to conduct more studies locally. Domestic CROs are adopting electronic data capture and centralized monitoring systems for faster data turnaround. The country’s large and diverse patient population enables high-quality data generation for global drug development. Partnerships with North American and European sponsors are improving adherence to international data standards. Investment in bilingual data management teams strengthens Brazil’s role in global clinical operations.
Market Development Factors:
- Expansion of multinational clinical research partnerships
- Implementation of EDC and centralized trial monitoring systems
- Improved regulatory timelines enhancing trial feasibility
- Growth in trained clinical data management professionals
How is the United States advancing the Clinical Trial Data Management Service Market?
The United States grows at 7.9% CAGR, maintaining its leadership in digital clinical trial infrastructure and data compliance frameworks. Major CROs and technology firms are integrating real-world evidence platforms and AI-driven analytics into data management workflows. FDA guidance on electronic source data and decentralized trial operations has driven greater cloud adoption. The market benefits from the presence of major pharmaceutical firms and software providers offering end-to-end data lifecycle management. Increased collaboration between sponsors and IT vendors supports system standardization, interoperability, and regulatory audit readiness. Continued innovation ensures the USA remains central to global clinical data management.
Key Market Factors:
- Leadership in decentralized and hybrid clinical trial data systems
- Integration of AI and real-world data analytics for study optimization
- FDA-aligned frameworks promoting digital data management adoption
- Strong collaboration between CROs, sponsors, and technology providers
How is the United Kingdom advancing its Clinical Trial Data Management Service Market?
The United Kingdom’s market grows at 7.1% CAGR, supported by strong clinical research networks and digital health policies. NHS Digital initiatives and the Medicines and Healthcare products Regulatory Agency (MHRA) modernization programs promote the use of standardized digital systems for data collection and sharing. CROs and life science organizations are adopting AI-enabled quality control and automated data review tools. The country’s integration of healthcare and research data strengthens longitudinal study capabilities. Partnerships with European and USA technology firms support scalable, compliant data management frameworks that align with GCP and GDPR requirements.
Market Development Factors:
- NHS Digital integration enhancing interoperability and trial efficiency
- Expansion of automated data quality and validation systems
- Regulatory focus on data protection under GCP and GDPR standards
- Collaboration between clinical technology providers and academic research centers
How is Japan supporting growth in the Clinical Trial Data Management Service Market?

Japan’s market grows at 6.2% CAGR, reflecting modernization of its clinical data management ecosystem and alignment with global digital standards. The Pharmaceuticals and Medical Devices Agency (PMDA) promotes digital submissions and validation-ready data systems. Japanese CROs are investing in AI-supported case report management and cloud-based platforms to streamline data cleaning and verification. Collaboration between research hospitals and technology providers is expanding real-world data integration capabilities. The country’s emphasis on data quality, transparency, and regulatory alignment supports continuous improvement in trial data accuracy. Enhanced automation and language localization are strengthening Japan’s competitiveness in regional data management services.
Market Characteristics:
- Regulatory reforms promoting digitalized clinical trial data management
- Integration of AI and cloud platforms in CRO operations
- Collaboration between academic hospitals and software providers
- Focus on data accuracy, transparency, and global compliance standards
What is the competitive landscape of the Clinical Trial Data Management Service Market?

The clinical trial data management service market is moderately fragmented, comprising over twenty-five contract research organizations (CROs) and technology providers operating across global clinical research networks. Caidya holds the leading position with an estimated 10.0% market share, supported by its integrated data management platform, end-to-end clinical operations capabilities, and strong regulatory compliance infrastructure across North America, Europe, and Asia-Pacific.
Cytel, PPD (Thermo Fisher Scientific Inc.), and Veristat follow as major players, leveraging advanced biostatistics, analytics-driven study design, and validated data systems to manage complex, multi-phase clinical trials. Their competitive edge arises from scalable cloud-based systems, adaptive trial methodologies, and strong sponsor relationships in biopharmaceutical and medical device sectors. Bioforum, Precision for Medicine, and Emerald Clinical serve as mid-tier specialists focusing on niche therapeutic areas and precision-medicine trials, emphasizing real-time data monitoring and audit readiness.
Larger CROs such as Syneos Health, Tata Consultancy Services (TCS), and Altasciences maintain competitiveness through global delivery models and integrated technology ecosystems that link electronic data capture (EDC) with clinical data repositories. Smaller firms, including Quanticate, ClinVigilant, and ProRelix Research, differentiate through regional expertise and flexible service offerings for small-to-midsized sponsors.
Competition centers on data integrity, regulatory compliance, and digital automation, as sponsors increasingly adopt AI-assisted data capture and decentralized clinical trial technologies. The market’s long-term trajectory favors providers capable of combining advanced analytics, secure data environments, and global regulatory expertise to improve study efficiency and submission timelines.
Key Players in the Clinical Trial Data Management Service Market
- Caidya
- Cytel
- PPD(Thermo Fisher Scientific Inc)
- Veristat
- Bioforum
- Precision For Medicine
- Emerald Clinical
- Syneos Health
- eClinical Solutions
- The Emmes Company, LLC
- BioPharma Services Inc.
- Quanticate
- Linical
- ethica CRO
- MED Institute
- CMIC
- IDDI
- Everest
- ProRelix Research
- Theradex
- ClinVigilant
- Altasciences
- Veramed
- GCP-Service
- Tata Consultancy Services (TCS)
- Atorus Research
- PharPoint
- ProTrials
- Phastar
- Jeeva Trials
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Classification | Electronic Data Capture (EDC) Service, Paper and Fax-based Service, Hybrid Services |
| Application | Pharmaceutical and Biotech Clinical Trials, Medical Device Clinical Trials, Other |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, USA, Germany, South Korea, Japan, Italy, and 40+ countries |
| Key Companies Profiled | Caidya, Cytel, PPD (Thermo Fisher Scientific Inc), Veristat, Bioforum, Precision For Medicine, Emerald Clinical, Syneos Health, eClinical Solutions, The Emmes Company LLC, BioPharma Services Inc., Quanticate, Linical, ethica CRO, MED Institute |
| Additional Attributes | Dollar sales by classification and application categories; regional adoption trends across Asia Pacific, Europe, and North America; competitive landscape of CROs and data management providers; advancements in EDC and hybrid data management systems; integration with regulatory compliance platforms and clinical trial digitization initiatives. |
Clinical Trial Data Management Service Market by Segmets
Classification:
- Electronic Data Capture (EDC) Service
- Paper and Fax-based Service
- Hybrid Services
Application:
- Pharmaceutical and Biotech Clinical Trials
- Medical Device Clinical Trials
- Other
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the clinical trial data management service market in 2025?
The global clinical trial data management service market is estimated to be valued at USD 1.1 billion in 2025.
What will be the size of clinical trial data management service market in 2035?
The market size for the clinical trial data management service market is projected to reach USD 2.4 billion by 2035.
How much will be the clinical trial data management service market growth between 2025 and 2035?
The clinical trial data management service market is expected to grow at a 8.3% CAGR between 2025 and 2035.
What are the key product types in the clinical trial data management service market?
The key product types in clinical trial data management service market are electronic data capture (edc) service, paper and fax-based service and hybrid services.
Which application segment to contribute significant share in the clinical trial data management service market in 2025?
In terms of application, pharmaceutical and biotech clinical trials segment to command 52.0% share in the clinical trial data management service market in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Classification
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Classification , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Classification , 2025 to 2035
- Electronic Data Capture (EDC) Service
- Paper and Fax-based Service
- Hybrid Services
- Y to o to Y Growth Trend Analysis By Classification , 2020 to 2024
- Absolute $ Opportunity Analysis By Classification , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2025 to 2035
- Pharmaceutical and Biotech Clinical Trials
- Medical Device Clinical Trials
- Other
- Y to o to Y Growth Trend Analysis By Application, 2020 to 2024
- Absolute $ Opportunity Analysis By Application, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Classification
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Classification
- By Application
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Classification
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Classification
- By Application
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Classification
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Classification
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Classification
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Classification
- By Application
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Classification
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Classification
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Classification
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Classification
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Classification
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Classification
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Classification
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Classification
- By Application
- Competition Analysis
- Competition Deep Dive
- Caidya
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Cytel
- PPD(Thermo Fisher Scientific Inc)
- Veristat
- Bioforum
- Precision For Medicine
- Emerald Clinical
- Syneos Health
- eClinical Solutions
- The Emmes Company, LLC
- BioPharma Services Inc.
- Quanticate
- Linical
- ethica CRO
- MED Institute
- CMIC
- IDDI
- Everest
- ProRelix Research
- Theradex
- Caidya
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: Global Market Value (USD Million) Forecast by Classification , 2020 to 2035
- Table 3: Global Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 4: North America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 5: North America Market Value (USD Million) Forecast by Classification , 2020 to 2035
- Table 6: North America Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 8: Latin America Market Value (USD Million) Forecast by Classification , 2020 to 2035
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 11: Western Europe Market Value (USD Million) Forecast by Classification , 2020 to 2035
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Classification , 2020 to 2035
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 17: East Asia Market Value (USD Million) Forecast by Classification , 2020 to 2035
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Classification , 2020 to 2035
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2020 to 2035
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Classification , 2020 to 2035
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2020 to 2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value Share and BPS Analysis by Classification , 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Classification , 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Classification
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2025-2035
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2025-2035
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2025-2035
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2025-2035
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2025-2035
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: North America Market Value Share and BPS Analysis by Classification , 2025 and 2035
- Figure 21: North America Market Y-o-Y Growth Comparison by Classification , 2025-2035
- Figure 22: North America Market Attractiveness Analysis by Classification
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2025-2035
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 27: Latin America Market Value Share and BPS Analysis by Classification , 2025 and 2035
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Classification , 2025-2035
- Figure 29: Latin America Market Attractiveness Analysis by Classification
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2025-2035
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 34: Western Europe Market Value Share and BPS Analysis by Classification , 2025 and 2035
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Classification , 2025-2035
- Figure 36: Western Europe Market Attractiveness Analysis by Classification
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2025-2035
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Classification , 2025 and 2035
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Classification , 2025-2035
- Figure 43: Eastern Europe Market Attractiveness Analysis by Classification
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2025-2035
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 48: East Asia Market Value Share and BPS Analysis by Classification , 2025 and 2035
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Classification , 2025-2035
- Figure 50: East Asia Market Attractiveness Analysis by Classification
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2025-2035
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Classification , 2025 and 2035
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Classification , 2025-2035
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Classification
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2025-2035
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Classification , 2025 and 2035
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Classification , 2025-2035
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Classification
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2025 and 2035
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2025-2035
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

