Synthetic Clinical Trial Data Market
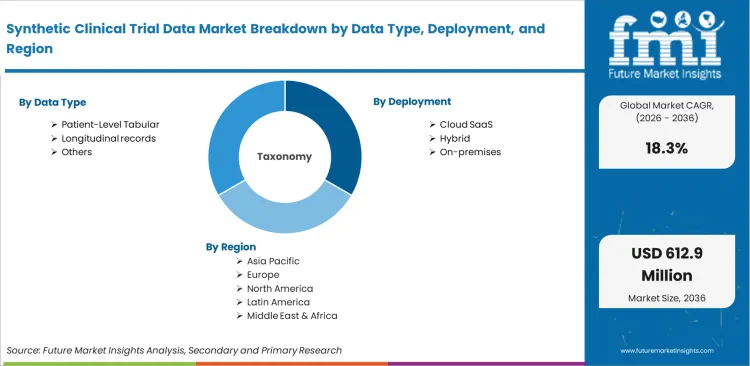
Synthetic clinical trial data market is segmented by Data type (Patient-level tabular, Longitudinal records, Multimodal records, Imaging-linked data), Deployment (Cloud SaaS, Hybrid, On-premises), Use case (Synthetic controls, Feasibility simulation, Privacy sharing, Model validation), End user (Pharma biotech, CROs, Academic centers, Regulators), Technology (Generative models, Digital twins, Bayesian synthesis, Rule-based engines), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Synthetic Clinical Trial Data Market Size, Market Forecast and Outlook By FMI
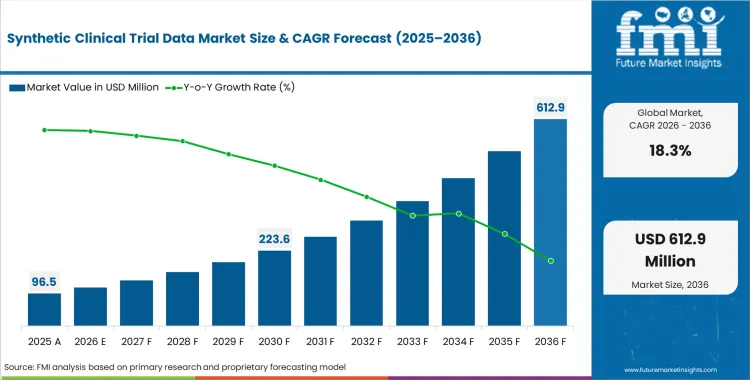
The synthetic clinical trial data market crossed a valuation of USD 81.6 million in 2025. The industry is expected to reach USD 96.5 million in 2026 at a CAGR of 18.3% during the forecast period. Demand outlook carries the market valuation to USD 518.1 million by 2036 as pharmaceutical sponsors increasingly adopt statistically generated patient baselines over traditional historical control groups.
Summary of Synthetic Clinical Trial Data Market
- Market Snapshot
- The Synthetic Clinical Trial Data Market is projected to expand from USD 81.6 million in 2025 to USD 518.1 million by 2036, signaling a steep scale‑up as synthetic controls and digital‑twin models become integral to evidence generation.
- The market is expected to grow at an 18.3% CAGR during 2026 to 2036, creating an incremental USD 421.6 million opportunity driven by rising expectations for privacy‑safe, regulator‑aligned trial datasets.
- This segment remains a regulation‑intensive clinical‑research data domain, where competitive differentiation depends on data fidelity, privacy guarantees, and methodological transparency rather than broad software distribution.
- Market architecture is now centered on synthetic controls, digital‑twin comparators, and high‑fidelity patient‑level synthetic datasets, marking a move away from general-purpose analytics toward purpose‑built regulatory‑grade data assets.
- Demand and Growth Drivers
- Demand is accelerating as the FDA formally recognizes AI‑generated synthetic controls as a legitimate innovation track in clinical trials, signaling sponsor‑level readiness beyond academic experimentation.
- The FDA’s emphasis that synthetic data used in submissions must be justified and demonstrated as fit‑for‑purpose is pushing buyers toward platforms with robust validation, auditability, and methodological traceability.
- European momentum is strengthening as the EMA’s data‑and‑AI agenda explicitly prioritizes patient‑level clinical study analytics, reinforcing institutional demand for reliable synthetic datasets.
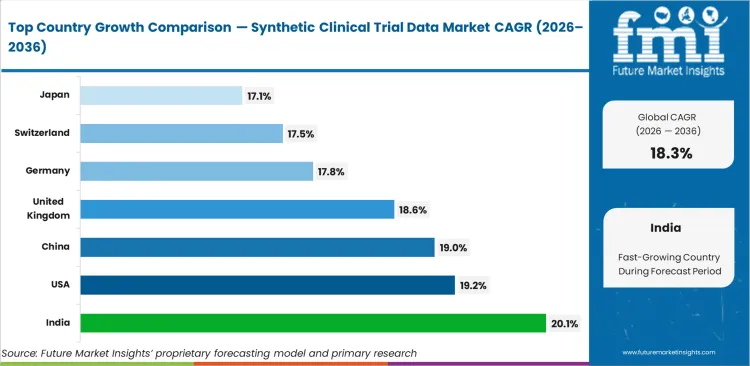
- India leads global growth at 20.1% CAGR, followed by the United States (19.2% CAGR), China (19.0% CAGR), United Kingdom (18.6% CAGR), Germany (17.8% CAGR), Switzerland (17.5%), and Japan (17.1% CAGR), reflecting a clear divergence between digitally progressive and conservative regulatory ecosystems.
- Growth is tempered by heterogeneous evaluation standards, gaps in privacy stress‑testing, and limited harmonization for longitudinal synthetic data, which collectively constrain enterprise‑scale adoption.
- Product and Segment View
- The market spans tabular, longitudinal, multimodal, and imaging‑linked synthetic datasets, delivered through cloud or hybrid environments for trial design, controlled data access, and evidence‑generation workflows.
- These datasets support synthetic control development, feasibility simulation, privacy‑safe collaboration, and AI‑model validation, aligning directly with regulated clinical‑research requirements.
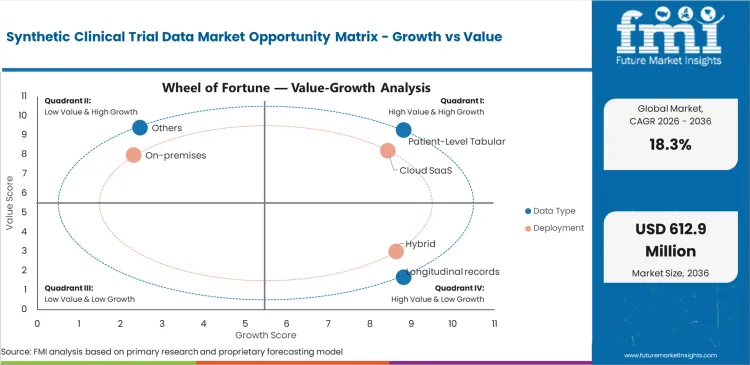
- Patient‑level tabular data leads at 42.0% share, as structured covariates and endpoint formats remain the most verifiable and broadly reusable across trial design and comparator construction.
- Cloud SaaS dominates with 58.0% share, driven by sponsor demand for faster provisioning, cross‑team collaboration, and standardized governance rather than fragmented on‑premise experimentation.
- Synthetic controls represent 34.0% of use cases, reflecting their clear linkage to cycle‑time compression and evidence‑generation efficiency.
- Pharma and biotech account for 46.0% of end‑user spending, given their ownership of protocol design, external comparator strategies, and submission‑oriented analytics.
- Generative models lead with 44.0% share, as GANs, flow‑based models, and related architectures continue to underpin the synthesis of regulator‑grade patient‑level data.
- The scope covers synthetic patient datasets, digital‑twin comparators, and synthetic‑control workflows, while excluding non‑healthcare synthetic‑data utilities, standard EDC tools, and general BI dashboards.
- Geography and Competitive Outlook
- The United States, India, and China remain the fastest‑growing markets, while Germany, Switzerland, and Japan represent stable, high‑trust adoption bases with more conservative procurement patterns.
- Competition is increasingly defined by historical‑trial‑data depth, external‑control methodology expertise, digital‑twin science, and privacy‑preserving data‑generation platforms, rather than general data‑hosting capacity.
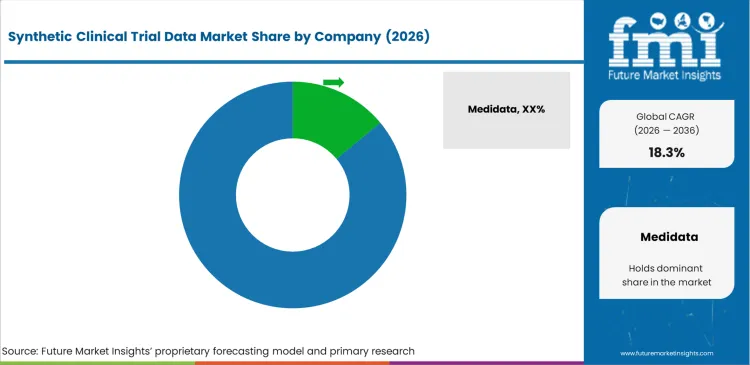
- Key participants include Medidata, Unlearn, Aetion, ConcertAI, and MDClone, with Medidata benefiting from its integrated synthetic‑control offering backed by a large proprietary clinical‑trial repository.
- The market remains fragmented, with the leading player at ~14.2% share, reflecting an ecosystem split across external‑control specialists, digital‑twin innovators, and broader synthetic‑health‑data platforms.
This is driven by intensifying operational pressures within clinical development, particularly among biostatistics leaders who face mandates to accelerate rare‑disease studies while navigating stringent data‑privacy constraints. Traditional methods of identifying and integrating physical historical control subjects not only add months to development timelines but also impose substantial costs and create compliance exposure. As a result, synthetic patient‑level datasets are moving from a conceptual innovation to a core enabler of Phase II program design.
The pace of adoption is likely to increase materially once regulatory authorities finalize formal FDA guidance on synthetic data usage in clinical research. Clear expectations around validation, evidentiary standards and methodological transparency will help mitigate perceived risks among principal investigators and institutional review bodies, paving the way for enterprise‑wide acceptance.
Global growth patterns reflect structural differences in digital readiness across clinical development ecosystems. India currently leads with a 20.1% compound annual growth rate, driven by expanding clinical‑delivery capacity and rapid adoption of AI‑based research tools. The United States follows at 19.2%, supported by active regulatory engagement on pathways for regulatory‑grade synthetic data. China’s 19.0% growth rate underscores its investment in biotech infrastructure and national data platforms. Development in the United Kingdom, Germany, and Switzerland, at a CAGR of 18.6%, 17.8%, and 17.5%, respectively, points to steady but more measured momentum, influenced by conservative evidence standards and slower institutional transformation. Japan, at 17.1% CAGR, continues to advance more gradually as procurement cycles and technology adoption processes evolve.
Segmental Analysis
Synthetic Clinical Trial Data Market Analysis by Data type
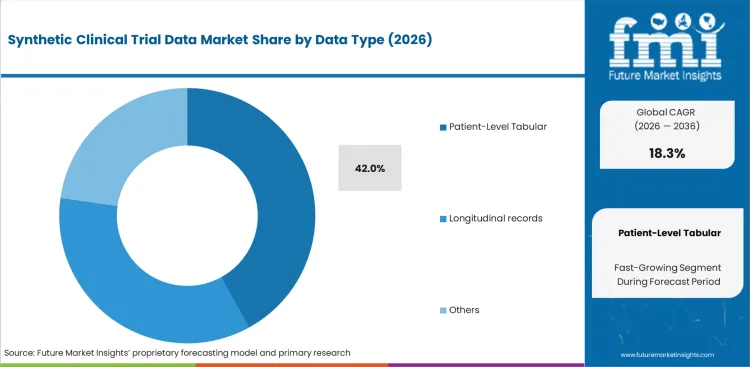
Legacy biostatistics workflows continue to mandate flat‑file structures for regulatory submissions, ensuring that simple tabular formats remain highly relevant across compliance‑driven review environments. Although many generalists expect advanced longitudinal or multimodal representations to replace flat files, patient‑level tabular data, anticipated to hold a 42.0% share in 2026, maintains its dominant position due to backward compatibility with the SAS programming ecosystems long used by major sponsors. Clinical data managers integrating simulated cohorts into existing trial‑management databases rely on familiar row‑and‑column architectures, while complex multimodal formats frequently break older visualization tools and require costly custom engineering. Regulatory agencies also continue to prefer straightforward tabular outputs during initial audits, meaning teams deploying unsupported or overly complex data structures risk submission rejection and the significant delays associated with forced data restructuring.
- Statistical compliance: Tabular structures map perfectly onto existing CDISC standards. Clinical data managers avoid format conversion errors during regulatory filing preparation.
- Audit transparency: Reviewers easily inspect flat rows for logic inconsistencies. Biostatistics leads secure faster preliminary approvals from agency data inspectors.
- Integration friction: Forcing multi-dimensional arrays into flat tables strips temporal relationships. Data engineers lose critical time-series insights when compressing complex patient histories.
Synthetic Clinical Trial Data Market Analysis by Deployment
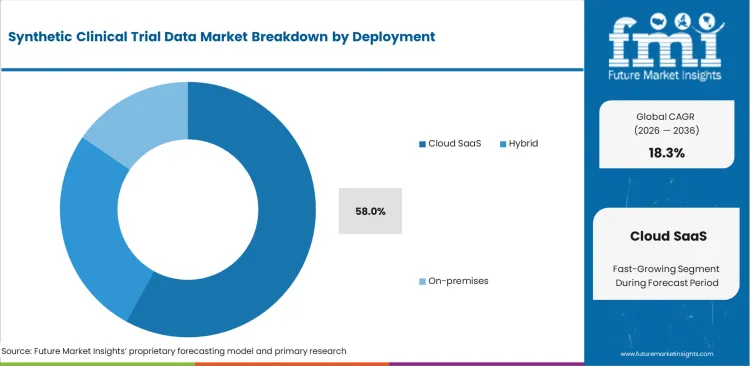
Computational demand continues to influence infrastructure choices for training generative models on millions of clinical records. Sponsors are moving toward environments that can support high-performance workloads at scale. Local servers often fall short because deep learning models require flexible GPU capacity during training. This is expected to help Cloud SaaS capture a 58.0% share of the deployment segment in 2026. Its position reflects the ability to scale processing without placing the full hardware burden on internal IT teams. Even so, deployment patterns are not fully cloud-based in practice. Model training often runs on cloud infrastructure, while patient-level record synthesis is carried out within air-gapped internal systems to meet privacy and security requirements. Vendors without strong edge-deployment support may face slower adoption from security teams that require strict network isolation. Buyers also face compliance risk when European patient cohorts are processed only through public cloud environments. The segment is expected to stay strong, though hybrid deployment capability remains a key factor in vendor selection.
- Elastic compute: Deep learning training phases require massive temporary processing power. Many teams avoid buying expensive hardware clusters that would remain underused outside peak training periods.
- Model updates: Centralized platforms push algorithmic improvements continuously. Data scientists access improved synthesis capabilities without manual software patching.
- Data gravity: Moving petabytes of historical trial records to external servers incurs massive transfer costs. Cloud architects must build localized synthesis nodes to prevent budget overruns.
Synthetic Clinical Trial Data Market Analysis by Use Case
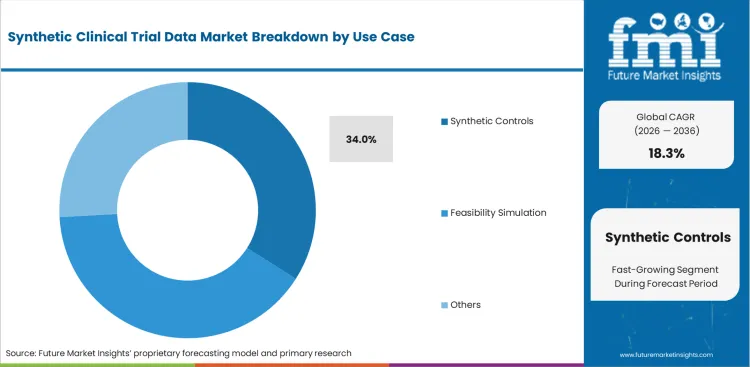
Oncology trials face strict ethical limits when placebo use involves terminal patients. This is pushing sponsors toward synthetic control arms as an alternative to physical comparator groups. Chief medical officers are using real-world evidence to build matched artificial patient profiles that help assess drug efficacy without denying treatment to enrolled participants. This shift is expected to help synthetic controls capture a 34.0% share of the application segment in 2026. Generated control arms cannot be used without early and detailed regulatory alignment. Statistical matching methods cannot fully address clinical variables that remain unmeasured in real patient populations. Sponsors that do not clearly document baseline variable selection may face delays during regulatory review. In some cases, weak documentation can affect final FDA assessment of trial validity. The segment is expected to expand further as sponsors balance ethical trial design with regulatory evidence standards.
- Placebo replacement: Generated cohorts eliminate physical control groups in rare disease studies. Chief medical officers accelerate total enrollment timelines by offering guaranteed active treatment.
- Confounding mitigation: Matching algorithms balance artificial patient characteristics against real active-arm subjects. Biostatisticians reduce observable bias impacting final efficacy calculations.
- Unmeasured risk: Algorithms only replicate documented historical variables. Trial directors face unexpected efficacy skews if unknown biological factors influenced original source data.
Synthetic Clinical Trial Data Market Analysis by End User
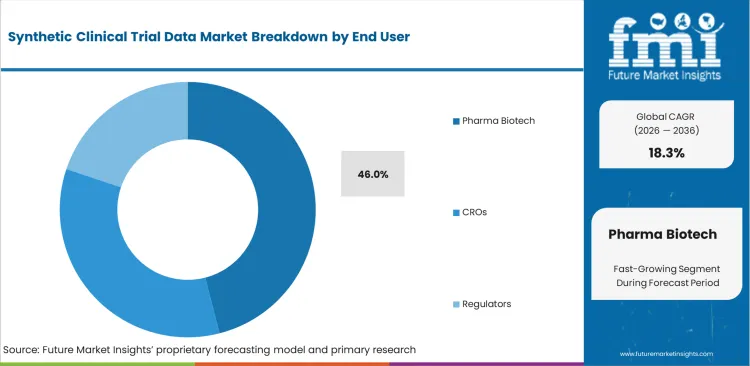
Massive proprietary data archives give large drug developers an insurmountable advantage in training custom simulation engines, enabling them to refine highly specialized synthetic patient models. Their R&D informatics teams leverage mature internal data‑governance frameworks to construct precise simulated populations for protocol optimization. These capabilities are further strengthened by decades of historical clinical trial repositories that support continuous algorithm tuning. Pharma biotech, holding 46.0% share in 2026, benefits disproportionately from this data depth, a dynamic that understates how thoroughly top‑tier sponsors dominate access to high‑quality baseline datasets. Smaller competitors lacking extensive patient‑record histories cannot generate accurate synthetic cohorts and are often forced to rely on generic off‑the‑shelf profiles with limited therapeutic specificity. Mid‑sized biotechs using synthetic cohorts for recruitment planning frequently over‑estimate the accuracy of their simulated control arms when operating without budgets for custom model development.
- Archive monetization: Decades of past trial results provide perfect training material. R&D informatics leads generate highly accurate disease progression models.
- Protocol stress-testing: Simulated patients test proposed inclusion criteria before real recruitment begins. Clinical operations managers identify potential enrollment bottlenecks early.
- Data poverty: Emerging sponsors lack sufficient historical records for model training. Start-up clinical directors must rely on less accurate generalized external datasets.
Synthetic Clinical Trial Data Market Analysis by Technology
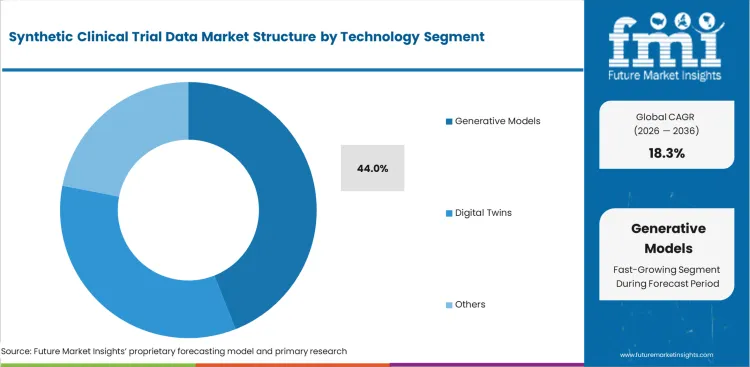
Deep neural networks are gaining wider use because they can identify hidden relationships across large sets of patient biomarkers. AI research teams favor these models when simulation workflows require a more realistic representation of disease behavior. Their strength lies in capturing feature interactions that rule-based systems often miss. This is expected to help generative models capture a 44.0% share of the model segment in 2026. Even so, higher mathematical performance does not remove concerns around regulatory review. Generative adversarial networks and related methods may outperform traditional approaches, yet their decision pathways are often harder to explain in formal audits.
- Correlation capture: Neural networks identify hidden variable relationships within source records. AI research scientists produce highly realistic simulated metabolic responses.
- Privacy preservation: Adversarial training techniques mathematically sever links to original patient identities. Compliance officers satisfy GDPR requirements while maintaining data utility.
- Interpretability failure: Opaque models obscure exact generation pathways. Regulatory affairs directors struggle to defend generated cohort validity during agency inspections.
Synthetic Clinical Trial Data Market Drivers, Restraints, and Opportunities
Extreme recruitment challenges in specialized therapeutic areas compel chief medical officers to seek alternative evidence generation strategies. Finding enough qualifying participants for orphan drug trials takes years and drains budgets completely. Delaying protocol launches while waiting for physical placebo cohorts threatens critical patent exclusivity windows. This urgency moves synthetic clinical data for rare disease trials from an experimental data science project into a mandatory clinical operations tool. Teams refusing to adopt artificial baselines face impossible enrollment targets and inevitable trial delays.
Algorithmic validation friction slows widespread adoption even when clinical teams desperately want simulated alternatives. Regulatory reviewers require exhaustive proof that generated patients exactly match physical human populations across all relevant biological markers. This proof requires complex mathematical justification that many software vendors cannot provide. Emerging clinical documentation platforms offer partial audit features, yet they lack standardized metrics acceptable to global health authorities.
Opportunities in the Synthetic Clinical Trial Data Market
- Pediatric trial simulation: Ethical restrictions limit physical testing on children. Clinical directors can generate juvenile cohorts using scaled adult data to model safety profiles safely.
- Cross-border data pooling: Strict localization laws prevent international record sharing. Privacy officers can synthesize regional datasets into compliant global healthcare intelligence dashboards for comprehensive analysis.
- Failed-trial rescue: Poorly designed historical studies contain valuable but unusable data. Biostatisticians can re-weight and synthesize these fragmented records to extract valid secondary endpoints.
Regional Analysis
The regional assessment divides the Synthetic Clinical Trial Data market into North America, East Asia, South Asia, and Europe, spanning more than 40 countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 20.1% |
| United States | 19.2% |
| China | 19.0% |
| United Kingdom | 18.6% |
| Germany | 17.8% |
| Switzerland | 17.5% |
| Japan | 17.1% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America Synthetic Clinical Trial Data Market Analysis
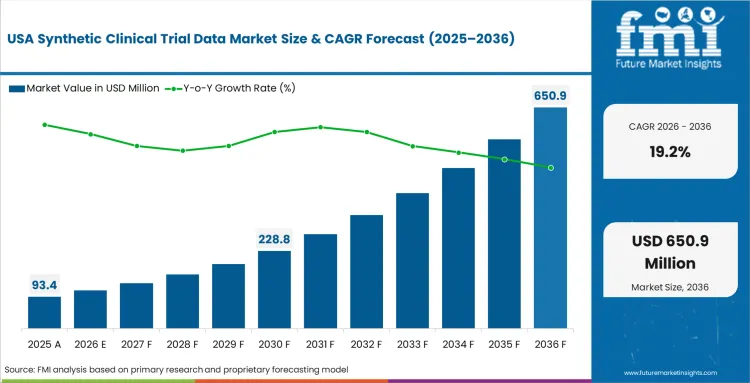
Regulatory agencies here actively publish guidance documents outlining acceptable methodologies for artificial cohort inclusion. This proactive stance eliminates guesswork for biopharma sponsors planning complex phase three studies. FMI analysts note that early alignment between software vendors and federal reviewers creates a highly permissive commercial environment. Sponsors confidently invest millions into generative data platforms knowing their final submissions have clear evaluation criteria. Startups providing transparent algorithmic validation documentation secure massive contracts from legacy drug makers.
- United States: Agency engagement on synthetic clinical trial data USA disclosure frameworks defines this jurisdiction's specific regulatory trajectory. Clinical operations heads deploy synthetic comparator data clinical trials confidently, accelerating overall drug development cycles significantly. Startups mastering these novel submission pathways capture dominant market share from hesitant incumbents. National synthetic cohort revenue is projected to expand at a 19.2% CAGR from 2026 to 2036. This solidifies the U.S. position as a leading growth market.
Asia Pacific Synthetic Clinical Trial Data Market Analysis
Domestic infrastructure buildouts prioritize massive computing clusters capable of processing national health repositories. This raw processing power allows regional tech giants to train unusually large and accurate patient simulation models. In FMI's view, aggressive national AI strategies subsidize generative research that would be cost-prohibitive elsewhere. Healthcare institutions eager to monetize their massive patient archives partner aggressively with algorithms developers.
China: Demand for the Synthetic Clinical Trial Data Market in China is anticipated to rise at a 19.0% CAGR from 2026 to 2036, supported by the country’s expanding clinical development capacity and large-scale data infrastructure investment. R&D teams are moving more quickly toward AI-enabled research tools as they look to reduce dependence on traditional placebo-based trial structures. This is helping local developers address recruitment constraints more efficiently in selected study settings. Trial sponsors outside China are also paying closer attention to domestic sites where simulated cohort integration is becoming more feasible. China is expected to remain a major market as data scale and research capacity continue to support synthetic trial adoption.
Japan: Sales of the Synthetic Clinical Trial Data Market in Japan are being shaped by conservative evidence requirements and careful approval standards. Biostatistics teams often request bridging studies that compare artificial records with real Japanese patient populations before broader use is accepted. This measured adoption pattern is expected to help the market record a 17.1% CAGR through 2036. Procurement cycles are also moving more slowly than in some other Asian markets, which keeps validation quality central to vendor selection. Local CROs that build Japan-specific validation frameworks are likely to gain stronger acceptance from cautious domestic sponsors. Japan is set to remain a market where evidence quality carries more weight than adoption speed.
India: Demand for the Synthetic Clinical Trial Data Market in India is set to expand at a 20.1% CAGR during the forecast period, driven by the growing role of outsourced biostatistics and protocol support services. Service providers are using artificial patient generation to deliver faster feasibility assessments for global pharmaceutical clients. AI-enabled research tools and broader clinical development activity are also supporting stronger local adoption. Organizations that build expertise in these generative workflows are better placed to secure long-term work in trial design and analytics support. India is likely to remain a high-growth market as sponsors seek faster and more scalable research execution models.
Europe Synthetic Clinical Trial Data Market Analysis
Strict privacy frameworks regarding patient records force data scientists to utilize mathematically anonymous datasets for cross-border research. Physical patient records cannot move between member states easily, making simulated cohorts highly valuable for multi-national studies. Based on FMI's assessment, local health technology assessment bodies remain deeply skeptical of efficacy claims backed purely by synthesized health data.
- United Kingdom: Strong biopharma research heritage conflicts with extremely conservative reimbursement evaluation criteria. Health economics directors use generated patient profiles for internal modelling but hesitate submitting them for final payer negotiations. Vendors demonstrating perfect alignment with local health authority standards capture premium pricing power. This cautious validation environment restrains the national synthetic trial data market to an 18.6% CAGR through 2036.
- Germany: Regulated trial environments demand absolute algorithmic transparency before permitting any artificial cohort integration. Clinical informatics leads scrutinize generation methodologies continuously to ensure compliance with stringent national medical data protection laws. This strict oversight shapes a steady 17.8% revenue CAGR for the German synthetic clinical trial data industry over the 2026 to 2036 period. Perfect compliance documentation becomes an absolute requirement for any software vendor attempting localized market entry.
- Switzerland: In the Swiss synthetic clinical trial data segment, deep concentrations of legacy pharmaceutical headquarters create massive demand for secure internal protocol simulation tools, maintaining a 17.5% revenue CAGR from 2026 to 2036. R&D heads prefer running generative models on highly secure local clusters rather than public clouds. Generalist assumptions miss that Swiss buyers prioritize strict data localization architectures completely over pure algorithmic generation speed or external cloud scaling.
FMI's report includes France, Italy, Spain, South Korea, and Australia. Emerging regional guidelines regarding algorithmic transparency dictate how quickly secondary jurisdictions approve completely simulated evidence submissions.
Competitive Aligners for Market Players
Rivalry dynamics diverge sharply from traditional healthcare software because algorithmic accuracy matters less than regulatory credibility. Medidata and ConcertAI dominate not through superior deep learning architectures, but by possessing documented histories of successful FDA and EMA submissions. Biopharma sponsors choose synthetic clinical trial data vendors based almost entirely on whether their generated datasets have previously survived hostile regulatory audits. This dynamic forces pure technology startups offering external control arm services to partner with established clinical research organizations to borrow their regulatory prestige.
Incumbent data providers possess massive proprietary libraries of historical patient records gathered from decades of previous trials. Aetion and MDClone leverage these closed datasets to train highly specific disease progression models that new entrants simply cannot replicate. Startups must rely on fragmented public registries or purchase expensive commercial datasets. This training data asymmetry creates a massive barrier, ensuring incumbents maintain superior simulation fidelity for complex therapeutic areas like the oncology synthetic control arm. Vendors lacking unique healthcare data streams essentially sell generic algorithms that sponsors find insufficient for pivotal trial work.
Buyers are increasingly resisting vendor control built around proprietary data ecosystems. Large trial sponsors do not want their entire simulation workflow tied to one provider. Many now prefer modular setups that let them use different algorithms for different disease areas. In comparisons such as Medidata and Unlearn for synthetic controls, buyers are separating data access from simulation software more often. Some license training datasets from one company and use another vendor’s generative engine for execution. This is pushing technology vendors to prove that their algorithms can work well even with third-party data inputs. The market is moving toward generation platforms that fit more easily into open and flexible research environments.
Key Players in Synthetic Clinical Trial Data Market
- Medidata
- Unlearn
- Aetion
- ConcertAI
- MDClone
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 96.5 million in 2026 to USD 518.1 million by 2036, at a CAGR of 18.30% |
| Market Definition | Synthetic clinical trial data involves computationally generated patient records matching statistical distributions of actual trial participants. These digital cohorts enable control arm simulation and protocol feasibility testing while maintaining absolute patient privacy. |
| Segmentation | Data type, Deployment, Use case, End user, Technology, and Region |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East and Africa |
| Countries Covered | United States, China, India, United Kingdom, Germany, Switzerland, Japan |
| Key Companies Profiled | Medidata, Unlearn, Aetion, ConcertAI, and MDClone |
| Forecast Period | 2026 to 2036 |
| Approach | Paid enterprise license volumes for generative trial software across top fifty biopharma sponsors. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Synthetic Clinical Trial Data Market Analysis by Segments
By Data type:
- Patient-level tabular
- Longitudinal records
- Multimodal records
- Imaging-linked data
By Deployment:
- Cloud SaaS
- Hybrid
- On-premises
By Use case:
- Synthetic controls
- Feasibility simulation
- Privacy sharing
- Model validation
By End user:
- Pharma biotech
- CROs
- Academic centers
- Regulators
By Technology:
- Generative models
- Digital twins
- Bayesian synthesis
- Rule-based engines
Region:
- North America
- United States
- Canada
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Asia Pacific
- China
- Japan
- South Korea
- Taiwan
- Singapore
- Latin America
- Brazil
- Mexico
- Argentina
- Middle East & Africa
- GCC Countries
- South Africa
Bibliography
- European Medicines Agency. (2025, July 10). Review of artificial intelligence and machine learning applications in the medicines lifecycle: 2024 horizon scanning short report.
- European Medicines Agency. (2026, February). Network Data Steering Group workplan 2026-2028: Data and artificial intelligence in medicines regulation.
- U.S. Food and Drug Administration. (2025, January 7). Artificial intelligence-enabled device software functions: Lifecycle management and marketing submission recommendations.
- Perkonoja, K., Auranen, K., & Virta, J. (2025). Methods for generating and evaluating synthetic longitudinal patient data: A systematic review. Journal of Healthcare Informatics Research, 1-39.
- Rujas, M., Hernández, A., et al. (2025). Synthetic data generation in healthcare: A scoping review of reviews on domains, motivations, and future applications. Journal of Biomedical Informatics, 164, 104826.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Friction slowing regulatory acceptance for generated patient cohorts globally.
- Vendor qualification requirements prioritized by biostatistics leads evaluating a synthetic health data platform for pharma.
- Specific validation architectures separating deep neural networks from legacy rules-based simulation engines.
- Geographic divergence shaping the synthetic clinical trial data usa trajectory alongside European growth.
- Core infrastructure barriers preventing rapid deployment within air-gapped internal clinical research environments.
- Specialized algorithms balancing absolute patient privacy against synthetic data for endpoint simulation utility.
- Baseline data monopolies giving incumbent providers unmatched advantages over pure technology startups.
- Actionable strategies enabling procurement directors to decouple raw training data from simulation models.
Frequently Asked Questions
What is synthetic clinical trial data?
Synthetic clinical trial data represents artificially generated patient profiles mirroring statistical properties of real human subjects without containing traceable personal health information. These digital cohorts enable control arm simulation and protocol feasibility testing while maintaining absolute patient privacy during medical research.
How is synthetic clinical trial data used in drug development?
Clinical operations managers use synthetic data for protocol feasibility to test inclusion criteria before recruitment begins. Biostatistics teams use these simulated patient records to create mathematical control arms, replacing physical placebo groups in specialized therapeutic areas like rare diseases and oncology.
Can synthetic trial data replace control arms?
Generated cohorts serve as mathematically rigorous alternatives to physical comparator groups when navigating ethical constraints in terminal studies. Chief medical officers leverage these profiles to demonstrate drug efficacy without denying active care to living participants, reducing overall enrollment burdens completely.
Is synthetic clinical data accepted by regulators?
Algorithmic validation friction slows widespread usage because regulatory reviewers require exhaustive proof that generated patients exactly match physical human populations. Opaque generative adversarial networks frequently fail audits despite perfect statistical outputs because sponsors cannot explain exact generation pathways.
What is the difference between RWD and synthetic trial data?
Evaluating real-world data vs synthetic clinical data requires understanding origin methodologies. Real-world data involves actual anonymized patient records scraped from electronic health systems, while synthetic data generates entirely new, mathematically constructed patients that retain zero one-to-one mapping with living individuals.
What companies provide synthetic clinical trial data?
Organizations evaluating the digital twin clinical trials market frequently encounter Medidata, Unlearn, Aetion, ConcertAI, and MDClone. These synthetic clinical trial data vendors compete heavily based on their documented histories of surviving hostile regulatory audits rather than pure algorithmic speed.
How big is the synthetic clinical trial data market?
The synthetic trial data market size reached USD 81.6 million in 2025. Revenue expansion propels overall opportunity to USD 518.1 million by 2036. This trajectory signals deep reliance on computationally generated patient records, shifting clinical operations away from purely physical participant recruitment.
Why does Patient-level tabular format capture 42.0% share?
Legacy biostatistics workflows mandate flat-file structures for regulatory submissions. Reviewers prefer simple rows and columns during initial audits, forcing sponsors to generate outputs mapping perfectly onto existing CDISC programming standards.
How does Cloud SaaS maintain its 58.0% position?
Deep learning models demand elastic GPU scaling impossible on local servers. IT directors offload hardware maintenance to specialized virtual environments, though actual patient record synthesis frequently executes within air-gapped internal systems eventually.
Why do Pharma biotech users retain 46.0% dominance?
Massive proprietary data archives provide unmatched training material for custom simulation engines. R&D informatics leads utilize these historical trial repositories to build highly specific simulated populations, creating insurmountable advantages over smaller competitors.
How do Generative models secure 44.0% share?
Deep neural networks capture hidden correlations across thousands of distinct patient biomarkers automatically. AI research scientists prefer these architectures because they provide unmatched fidelity when simulating complex multi-system disease progressions compared to rigid rule engines.
Why does India grow faster than United States?
India expands at 20.1% while United States hits 19.2% because outsourced biostatistics teams adopt AI-enabled research tooling aggressively. Local organizations leverage generative tools to deliver faster feasibility analyses for global sponsors, driving rapid regional modernization.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Data Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Data Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Data Type , 2026 to 2036
- Patient‑Level Tabular
- Longitudinal records
- Others
- Patient‑Level Tabular
- Y to o to Y Growth Trend Analysis By Data Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Data Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Deployment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Deployment, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Deployment, 2026 to 2036
- Cloud SaaS
- Hybrid
- On-premises
- Cloud SaaS
- Y to o to Y Growth Trend Analysis By Deployment, 2021 to 2025
- Absolute $ Opportunity Analysis By Deployment, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Use Case
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Use Case, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Use Case, 2026 to 2036
- Synthetic Controls
- Feasibility Simulation
- Others
- Synthetic Controls
- Y to o to Y Growth Trend Analysis By Use Case, 2021 to 2025
- Absolute $ Opportunity Analysis By Use Case, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Pharma Biotech
- CROs
- Regulators
- Pharma Biotech
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Generative Models
- Digital Twins
- Others
- Generative Models
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Data Type
- By Deployment
- By Use Case
- By End User
- By Technology
- Competition Analysis
- Competition Deep Dive
- Medidata
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Unlearn
- Aetion
- ConcertAI
- MDClone
- Medidata
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Data Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Deployment, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Use Case, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Data Type
- Figure 6: Global Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Deployment
- Figure 9: Global Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Use Case
- Figure 12: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by End User
- Figure 15: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Technology
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Data Type
- Figure 32: North America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Deployment
- Figure 35: North America Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Use Case
- Figure 38: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by End User
- Figure 41: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Technology
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Data Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Deployment
- Figure 51: Latin America Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Use Case
- Figure 54: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by End User
- Figure 57: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Technology
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Data Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Deployment
- Figure 67: Western Europe Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Use Case
- Figure 70: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by End User
- Figure 73: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Technology
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Data Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Deployment
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Use Case
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by End User
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Data Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Deployment
- Figure 99: East Asia Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Use Case
- Figure 102: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by End User
- Figure 105: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Technology
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Data Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Deployment
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Use Case
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Data Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Data Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Data Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Deployment, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Deployment, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Deployment
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Use Case, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Use Case, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Use Case
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

