About The Report
The industry is valued at USD 2.7 billion in 2026 and is expected to reach USD 7.6 billion by 2036, registering a strong value based CAGR of 11% during the forecast period. Growth is supported by rising adoption across core applications, increasing production activity, and steady demand from both established and developing economies. Expanding industrial usage and improved acceptance across multiple end use segments continue to create a favorable demand environment through the forecast timeline.
Consumption remains strong in mature regions where developed infrastructure, stable policy frameworks, and consistent industrial output support regular demand. Faster expansion is observed in emerging economies driven by manufacturing growth, urban development, and higher capital investment. These regions are contributing a growing share of incremental demand as industries scale operations and integrate advanced solutions to improve efficiency and output reliability.
Competitive dynamics are shaped by capacity expansion, process optimization, and focus on cost efficiency. Companies are strengthening supply networks, enhancing production quality, and securing long term customer relationships to stabilize volumes and margins. The industry also navigates challenges related to regulatory compliance and input price variability, encouraging players to refine sourcing strategies and improve operational control. Overall, strong demand fundamentals and ongoing investment activity are expected to support steady expansion and long term value creation through 2036.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 2.7 billion |
| Market Forecast Value (2036) | USD 7.6 billion |
| Forecast CAGR (2026 to 2036) | 11% |
The industry is classified by solution, company size, vertical, and region, reflecting how regulatory complexity varies across organizations and geographies. By solution, demand is divided between software and services, with software further split into cloud based and on premise platforms, while services include training and support along with consulting. By company size, adoption spans large enterprises with global operations and small and medium enterprises managing regional or export focused compliance. By vertical, demand covers pharmaceuticals, biologics, medical devices, cosmetics, and nutraceuticals. Regionally, the industry extends across North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia, and the Middle East and Africa.
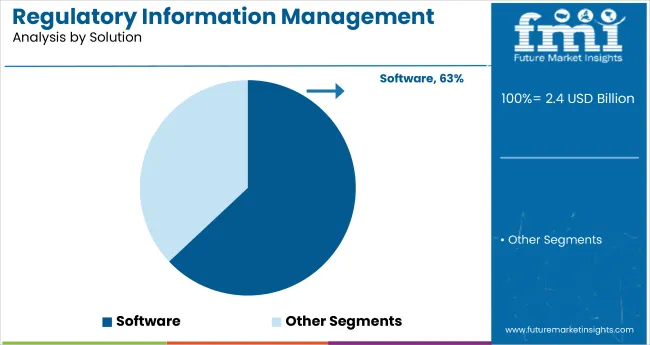
Software solutions account for roughly 70% of total global demand, making them the dominant solution segment across the industry. These platforms are widely used to manage regulatory submissions, product registrations, labeling updates, and lifecycle tracking across multiple authorities. Within software, cloud based deployments contribute close to 60% of software demand, supported by scalability, faster rollouts, and easier access for globally distributed regulatory teams. On premise solutions retain around 40% share within software, largely driven by organizations with strict internal data governance needs.
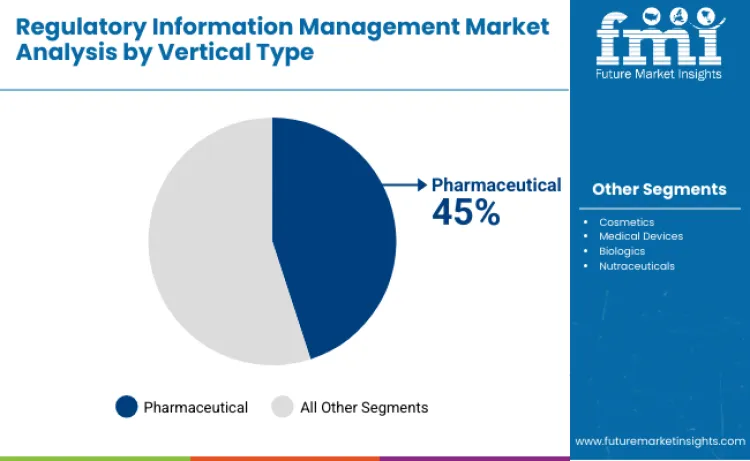
Pharmaceutical companies represent the largest share in the industry, contributing 45% of total demand. This dominance is fueled by the frequent filing of global regulatory submissions, ongoing post-approval modifications, and rigorous scrutiny by major regulatory bodies. Biopharmaceuticals account for nearly 18% of the share, with growth driven by the complex approval processes and comprehensive documentation requirements. Medical device manufacturers contribute about 14%, while nutraceuticals and cosmetics together account for 23%, supported by heightened compliance regulations and the increasing demand for cross-border trade.
The industry is expanding steadily as life sciences companies face rising regulatory complexity, higher submission volumes, and tighter global compliance timelines. Growth is driven by expanding pharmaceutical and biologics pipelines, frequent variations, and stricter oversight from authorities such as the FDA, EMA, and PMDA. Organizations increasingly rely on centralized digital platforms to manage regulatory data, reduce manual effort, and maintain consistency across markets. The industry gains momentum as firms prioritize faster approvals, improved data accuracy, and better visibility across the full product lifecycle.
How Are Compliance Pressures and Digital Workflows Shaping Industry Adoption?
Rising compliance pressure is accelerating adoption of structured, software driven regulatory workflows. Companies use regulatory information management solutions to track submissions, manage labeling changes, and respond quickly to regulatory updates across regions. Cloud based platforms support real time collaboration among regulatory, quality, and clinical teams, especially for global organizations operating across multiple jurisdictions. Increased use of automation, analytics, and artificial intelligence improves submission quality and reduces approval delays. Adoption reflects the need for operational efficiency, regulatory confidence, and scalable systems that support long term pipeline growth.
How Do Regulatory Complexity, Cost Barriers, and Data Security Concerns Affect Scalability?
Scalability is shaped by the complexity of aligning different regional regulatory frameworks and the high cost of system implementation and integration. Smaller organizations face budget constraints and limited in house expertise, slowing adoption despite strong compliance needs. Data security and privacy requirements further influence deployment decisions, particularly in regions with strict data protection rules. The industry responds through modular platforms, cloud deployment, and managed services that lower entry barriers. Long term scalability depends on balancing regulatory rigor, cost control, and secure digital infrastructure across global operations.
Demand across the industry is rising rapidly as pharmaceutical, biotech, and medical device companies shift toward structured digital compliance and lifecycle management. Increasing regulatory complexity, multi country filings, and mandatory electronic submissions are pushing organizations to replace manual workflows with centralized RIM platforms. Growth remains strongest in countries with active drug development pipelines, strong CRO presence, and evolving regulatory digitization frameworks. India leads growth with a CAGR of 12.8%, followed by China at 12.4% and South Korea at 11.9%, reflecting fast expanding life sciences ecosystems. Japan records 11.5% supported by global harmonization efforts, while Germany at 11.2% and the United Kingdom at 10.8% benefit from EU and post Brexit compliance needs. The United States grows steadily at 10.3% due to complex lifecycle requirements and high regulatory filing volumes.
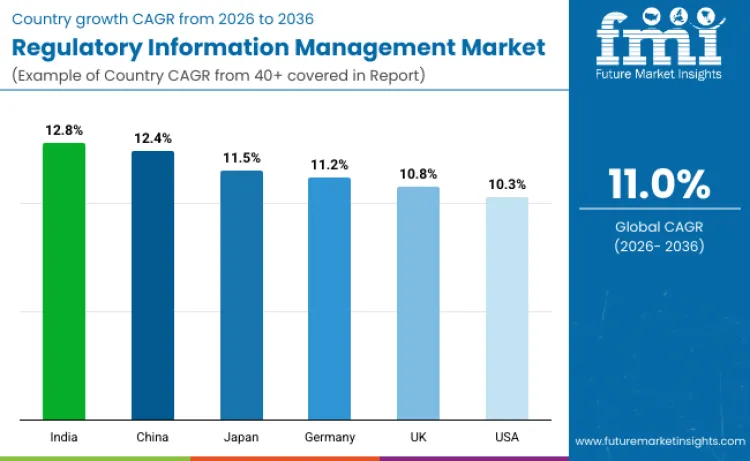
| Country | CAGR |
|---|---|
| India | 12.8% |
| China | 12.4% |
| Japan | 11.5% |
| United Kingdom | 10.8% |
| Germany | 11.2% |
| United States | 10.3% |
The United States records a CAGR of 10.3% as regulatory complexity and frequent FDA submissions continue to rise. The industry benefits from mandatory SPL labeling, eCTD filings, and ongoing post approval compliance across pharmaceuticals, biologics, and digital therapeutics. Large enterprises prioritize enterprise grade platforms that support real time dossier management and cross agency coordination. Cloud adoption among CROs and CDMOs further accelerates demand, while AI driven document processing and decentralized trial models increase reliance on automated regulatory systems across major life sciences hubs.
Germany grows at a CAGR of 11.2%, supported by strict EU compliance requirements and early adoption of digital regulatory standards. The industry benefits from implementation of eCTD v4.0 and IDMP frameworks, which require upgrades across regulatory operations. Pharmaceutical and biotech firms increasingly integrate RIM platforms with digital health systems. Preference for on premise and hybrid deployments reflects data sovereignty priorities, while mid sized firms begin adopting SaaS models to manage rising submission complexity and lifecycle tracking needs.
The United Kingdom records a CAGR of 10.8% driven by post Brexit regulatory divergence and the need for parallel compliance workflows. The industry benefits from MHRA digital modernization efforts and growing adoption among biotech and CRO communities. Companies managing both EU and UK submissions rely on centralized RIM platforms to track approvals, variations, and renewals. Increased use of structured digital labeling and AI based automation strengthens demand across mid sized and innovation focused firms.
Japan shows strong expansion with a CAGR of 11.5% as PMDA mandates eCTD submissions and aligns more closely with global regulatory frameworks. The industry gains from rising complexity linked to advanced therapies and global product launches. Japanese firms increasingly adopt hybrid RIM deployments to manage multilingual submissions and international coordination. Medical device manufacturers also contribute strongly due to rigorous premarket and postmarket compliance requirements.
China leads rapid expansion at a CAGR of 12.4%, driven by regulatory reform under the NMPA and alignment with international standards. The industry benefits from increased electronic submissions, structured data mandates, and expanding overseas product registrations. Domestic CROs and pharmaceutical exporters adopt cloud based RIM platforms to manage multi country compliance. Growth in biosimilars and traditional medicine exports further increases documentation complexity and system demand.
India records the highest CAGR at 12.8%, supported by rising pharmaceutical exports and accelerated regulatory digitization. The industry benefits from CDSCO initiatives such as SUGAM and growing alignment with global eCTD and IDMP standards. Contract manufacturers and CDMOs adopt enterprise platforms to support audit readiness and client compliance. Modular cloud solutions gain traction among mid sized exporters, positioning India as a major growth engine for RIM adoption through 2036.

The industry remains moderately consolidated, with competition shaped by depth of regulatory expertise, platform breadth, and ability to support complex global compliance workflows. Leading providers benefit from long standing relationships with pharmaceutical, biotech, and medical device companies, along with strong knowledge of regional regulatory frameworks. Competitive positioning is driven by platform scalability, cloud readiness, data security capabilities, and integration across submission management, labeling, and lifecycle tracking. Demand increasingly favors vendors that deliver reliable compliance outcomes while improving speed, visibility, and operational control across regulated product portfolios.
Aris Global and Veeva Systems hold strong positions through comprehensive cloud based platforms that support end to end regulatory operations for large life sciences organizations. Ennov and Amplexor strengthen their presence by combining software with deep regulatory consulting expertise, helping clients manage submissions, variations, and labeling across multiple regions. MasterControl and Calyx focus on structured data management, audit readiness, and traceability, appealing to companies operating under strict inspection and documentation requirements. Parexel and IQVIA differentiate through service led models that integrate regulatory technology with outsourcing and regulatory intelligence, supporting complex global pipelines.
Competitive dynamics reflect a balance between enterprise focused platforms and agile solutions targeting small and medium enterprises. Vendors compete on implementation speed, regulatory updates, customer support, and ability to adapt to evolving standards such as eCTD and IDMP. As regulatory expectations intensify, the industry rewards providers that combine technical reliability, regulatory credibility, and long term partnership value across global compliance programs.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Solutions Analyzed | Software, Services |
| Software Types | Cloud, On premise |
| Services Analyzed | Training and Support, Consulting |
| Company Size Analyzed | Small and Medium Enterprises, Large Enterprises |
| Verticals Analyzed | Pharmaceuticals, Cosmetics, Medical Devices, Biologics, Nutraceuticals |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, South Asia and Pacific, East Asia, Middle East and Africa |
| Countries Covered | United States, Germany, United Kingdom, Japan, China, Canada, France, South Korea, Australia, India |
| Key Companies Profiled | Veeva Systems, Kalypso , DDI, Körber AG, ArisGlobal , PhlexGlobal , AmpleLogic , Calyx, Amplexor Life Sciences, Ennov , MasterControl |
| Additional Attributes | Dollar sales by solution and vertical, regional and country level demand analysis, regulatory compliance impact assessment, competitive landscape and company share analysis, technology adoption and investment trends |
The industry is estimated to be valued at around USD 2.7 billion in 2026.
The industry is projected to reach USD 7.6 billion by 2036.
The industry is expected to expand at a value based CAGR of about 11% during the period.
Software solutions lead the industry due to strong adoption for digital compliance and submission management.
North America dominates the industry supported by high regulatory activity and advanced digital infrastructure.
Our Research Products

The "Full Research Suite" delivers actionable market intel, deep dives on markets or technologies, so clients act faster, cut risk, and unlock growth.

The Leaderboard benchmarks and ranks top vendors, classifying them as Established Leaders, Leading Challengers, or Disruptors & Challengers.

Locates where complements amplify value and substitutes erode it, forecasting net impact by horizon

We deliver granular, decision-grade intel: market sizing, 5-year forecasts, pricing, adoption, usage, revenue, and operational KPIs—plus competitor tracking, regulation, and value chains—across 60 countries broadly.

Spot the shifts before they hit your P&L. We track inflection points, adoption curves, pricing moves, and ecosystem plays to show where demand is heading, why it is changing, and what to do next across high-growth markets and disruptive tech

Real-time reads of user behavior. We track shifting priorities, perceptions of today’s and next-gen services, and provider experience, then pace how fast tech moves from trial to adoption, blending buyer, consumer, and channel inputs with social signals (#WhySwitch, #UX).
Partner with our analyst team to build a custom report designed around your business priorities. From analysing market trends to assessing competitors or crafting bespoke datasets, we tailor insights to your needs.
Supplier Intelligence
Discovery & Profiling
Capacity & Footprint
Performance & Risk
Compliance & Governance
Commercial Readiness
Who Supplies Whom
Scorecards & Shortlists
Playbooks & Docs
Category Intelligence
Definition & Scope
Demand & Use Cases
Cost Drivers
Market Structure
Supply Chain Map
Trade & Policy
Operating Norms
Deliverables
Buyer Intelligence
Account Basics
Spend & Scope
Procurement Model
Vendor Requirements
Terms & Policies
Entry Strategy
Pain Points & Triggers
Outputs
Pricing Analysis
Benchmarks
Trends
Should-Cost
Indexation
Landed Cost
Commercial Terms
Deliverables
Brand Analysis
Positioning & Value Prop
Share & Presence
Customer Evidence
Go-to-Market
Digital & Reputation
Compliance & Trust
KPIs & Gaps
Outputs
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE
Product Information Management Market Growth – Trends & Forecast 2024-2034
Building Information Management (BIM) Market Analysis – Trends & Forecast 2024-2034
Security Information and Event Management Software Market
Physical Security Information Management (PSIM) Market Size and Share Forecast Outlook 2025 to 2035
Regulatory-Compliant Medical Device Plastics Recycling Platforms Market Size and Share Forecast Outlook 2026 to 2036
Western Europe Regulatory Reporting Solution Market Growth – Trends & Forecast 2025 to 2035
Regulatory Reporting Solution Market Growth – Trends & Forecast 2023-2033
Regulatory Reporting Solution Market Growth – Trends & Forecast 2023-2033
Regulatory Reporting Solutions Market
Information Security Consulting Market
Tax Management Market Size and Share Forecast Outlook 2025 to 2035
Key Management as a Service Market
The Cash Management Supplies Packaging Market is segmented by Material (Plastic, Paper, Fabric, Metal), Product Type (Bags, Seals, Wrappers, Tags), End User (Financial Institutions, Retail, Casinos, Government), and Region. Forecast for 2026 to 2036.
Bank Regulatory & Governance Consulting Market Size and Share Forecast Outlook 2025 to 2035
Fuel Management Software Market Size and Share Forecast Outlook 2025 to 2035
Risk Management Market Size and Share Forecast Outlook 2025 to 2035
SBOM Management and Software Supply Chain Compliance Market Analysis - Size, Share, and Forecast Outlook 2025 to 2035
Case Management Software (CMS) Market Size and Share Forecast Outlook 2025 to 2035
Farm Management Software Market Size and Share Forecast Outlook 2025 to 2035
Lead Management Market Size and Share Forecast Outlook 2025 to 2035

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.