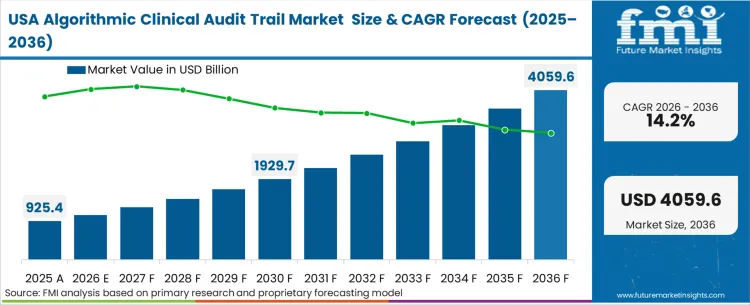
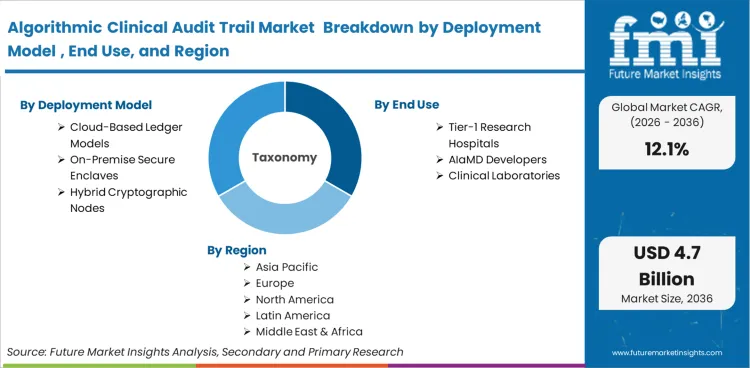
The algorithmic clinical audit trail market was valued at USD 1.2 billion in 2025. The sector is set to reach USD 1.5 billion in 2026 at a CAGR of 12.14% during the forecast period. Sustained investment propels the valuation to USD 4.7 billion through 2036 as regulatory mandates for immutable provenance in Software as a Medical Device compel clinical risk managers to replace centralized logs with cryptographic ledgers.
The shift toward autonomous clinical decision support immediately exposes Chief Medical Information Officers (CMIOs) at research hospitals to unprecedented liability gaps. Traditional IT logging architectures fail to capture the probabilistic inference paths of continuous-learning algorithms, leaving institutions unable to defend against algorithmic malpractice claims under the Bolam principle. CMIOs who delay the implementation of cryptographic provenance tools risk losing regulatory clearance for their in-house healthcare artificial intelligence systems. Outsiders often assume these audit mechanisms are merely post-incident investigation tools, but practitioners know they are the critical enabling technology that allows legal model retraining without triggering full FDA 510(k) resubmissions.
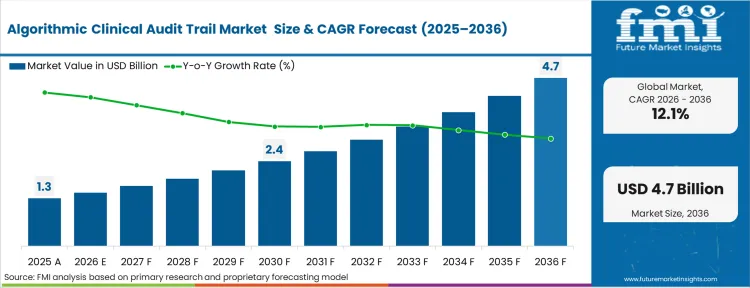
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.5 billion |
| Industry Value (2036) | USD 4.7 billion |
| CAGR (2026-2036) | 12.14% |
Source: FMI analysis based on primary research and proprietary forecasting model
Healthcare systems must transition their enterprise liability policies to explicitly cover algorithmic malpractice before widespread continuous-learning adoption can accelerate safely. Hospital legal departments must finalise shared accountability contracts delineating vendor versus provider liability by 2028. Once this liability framework is codified, algorithmic deployment will shift from experimental sandboxes to acute care mainframes.
The United States advances at 14.2% compound annual growth, driven by aggressive FDA Predetermined Change Control Plan frameworks. Germany records 13.5% as strict DiGA Fast-track evaluations mandate transparent algorithmic behaviour. The United Kingdom tracks 12.8% due to localised MHRA guidelines on Good Machine Learning Practice. Japan reaches 11.4%, while China posts 10.9% through National Medical Products Administration directives. France grows at 9.5%, and India follows at 8.2%. The structural spread across these geographies stems directly from the varying maturity of localized software-as-a-medical-device regulations rather than generalized healthcare spending differences.
The algorithmic clinical audit trail market encompasses software and middleware that record, encrypt, and store the inference pathways of artificial intelligence applications used in healthcare. These systems provide immutable cryptographic provenance for probabilistic clinical recommendations, bridging the gap between algorithmic outputs and regulatory compliance. The market explicitly addresses the need to map model versions, training datasets, and inference logic into auditable, human-readable ledger entries.
Inclusions cover blockchain-based inference ledgers, Predetermined Change Control Plan (PCCP) automation software, semantic disambiguation mapping tools for Retrieval-Augmented Generation (RAG) models, and API middleware that links autonomous decision algorithms to the broader digital health ecosystem. Software tools designed to conduct exploratory error analysis, subgroup testing, and adversarial testing against deployed medical algorithms are fully integrated into this scope.
The market excludes general-purpose IT system access logs, standard electronic health record (EHR) patient history logs, and basic network cybersecurity monitoring tools. These legacy systems track human access and data movement rather than capturing the internal mathematical weighting, dataset provenance, and probabilistic inference steps generated autonomously by machine learning models during clinical diagnostic workflows.
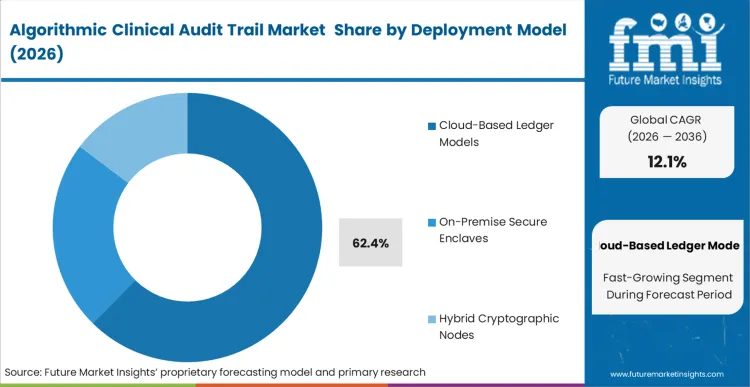
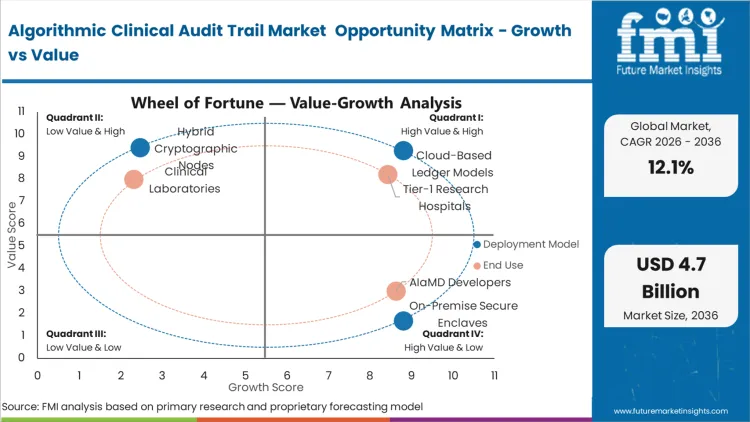
Centralized relational databases are actively failing to meet the strict immutability standards required for continuous-learning algorithms, forcing the displacement of legacy IT systems by decentralized ledgers. Cloud-Based Ledger Models command a 62.4% share in 2026 because their decentralized architecture prevents any single hospital node from altering a historical algorithmic decision.
FMI analysts suggest that this structure provides the exact mathematical certainty demanded by FDA investigators during adverse event reviews. Chief Information Officers operating multi-site acute care networks specify these cloud-ledgers to seamlessly synchronize vast quantities of inference data across their healthcare compliance software frameworks. Hospital legal departments that defer this upgrade risk having their entire evidentiary defense dismissed during algorithmic malpractice litigation due to theoretically manipulable logs.
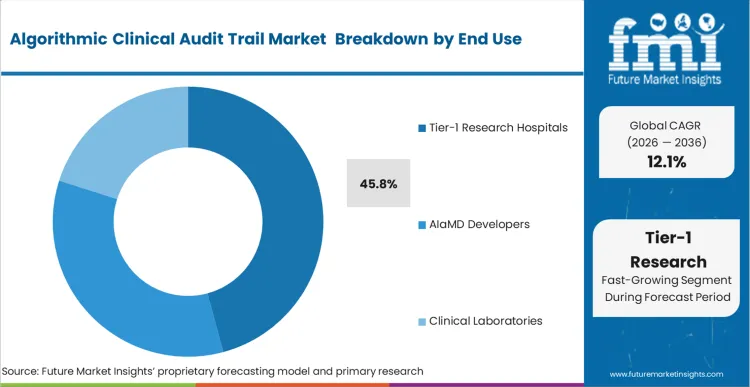
The concentration of Tier-1 Research Hospitals, holding a 45.8% share in 2026, is fused directly to their status as primary incubators for unproven, internally developed diagnostic algorithms. These institutions bear the brunt of early-adopter legal liability, forcing them to over-index on stringent provenance software compared to regional clinics.
Based on FMI's assessment, the sheer volume of experimental machine learning models running in these environments necessitates automated tracking to manage the Predetermined Change Control Plans. Chief Medical Information Officers within these academic hubs deploy massive medical image analysis overlays that update continuously based on new local data. Hospitals that fail to implement corresponding audit systems face immediate suspension of their clinical trial capabilities by federal oversight boards.
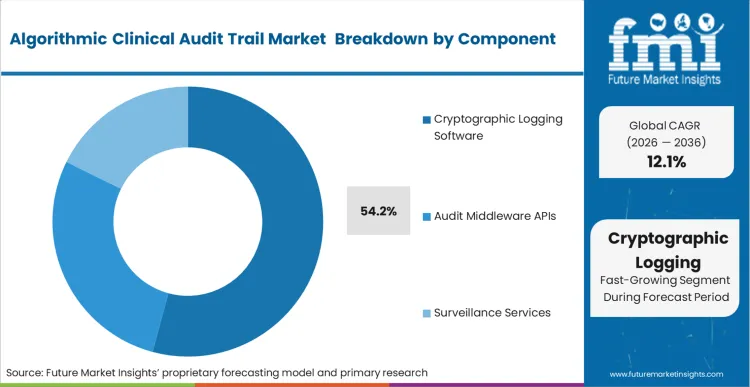
The strict compliance threshold of 21 CFR Part 11 dictates that the underlying architecture of any electronic record must be inherently secure and computer-generated without human intervention. This absolute requirement funnels buyers toward Cryptographic Logging Software, driving its 54.2% dominance in 2026.
According to FMI's estimates, this software layer is the non-negotiable prerequisite before any higher-level surveillance analytics can be deployed effectively. AIaMD developers embed these core clinical decision support logging tools directly into their product architectures to ensure out-of-the-box compliance. Developers who ignore this foundational layer find their products completely embargoed by enterprise hospital procurement committees.
The operational gap in managing millions of automated radiological scans requires a specialized tracking layer to catch silent model drift. Medical Imaging AI Oversight specifically exists to solve this scale problem, leading the Application dimension with a 38.6% share in 2026.
As per FMI's projection, algorithms evaluating X-rays and MRIs are highly susceptible to spurious correlates, such as learning the type of scanning machine rather than the pathology. Radiology department heads specify this oversight application to continuously monitor healthcare cybersecurity systems and imaging models for diagnostic degradation. Departments operating without continuous imaging oversight inevitably experience clustered misdiagnoses, resulting in massive malpractice exposure once the silent drift is finally discovered.
The US FDA's Good Machine Learning Practice (GMLP) principles and subsequent Predetermined Change Control Plan frameworks force AI-as-a-Medical-Device developers to integrate verifiable provenance layers. This regulatory pressure explicitly targets Chief Medical Information Officers, mandating that any continuous-learning algorithm deployed in acute care must document its evolving inference logic comprehensively. Hospitals are rapidly procuring cryptographic audit software to bridge the gap between static regulatory approval and dynamic software behaviour. Institutions that fail to implement these verifiable ledgers risk invalidating their 510(k) clearances, effectively shutting down their advanced autonomous care units.
Significant operational friction arises from the computational latency introduced by complex cryptographic logging. Generating an immutable, blockchain-anchored record for every split-second inference inside a high-frequency intensive care monitoring system can drastically slow down critical bedside alerts. While lightweight localized hashing presents an emerging workaround, it frequently lacks the cross-organizational traceability demanded by federal regulators.
Based on the regional analysis, the Algorithmic Clinical Audit Trail market is segmented into North America, Europe, East Asia, and Asia Pacific across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 14.2% |
| Germany | 13.5% |
| United Kingdom | 12.8% |
| Japan | 11.4% |
| China | 10.9% |
| France | 9.5% |
| India | 8.2% |
Source: FMI analysis based on primary research and proprietary forecasting model

The aggressive rollout of FDA Predetermined Change Control Plans (PCCPs) under the Software as a Medical Device framework forces a highly specific compliance burden on North American institutions. This policy-led environment dictates that algorithms capable of continuous learning cannot be deployed legally without a dynamic, unalterable ledger tracking every post-market modification. In FMI's view, this regulatory environment makes North America the primary testing ground for advanced cryptographic logging architectures. Vendors are leveraging cloud computing platforms to aggregate these vast inference logs securely across disparate state health networks.
FMI's report includes comprehensive evaluation of Canada and Mexico. The rapid harmonization of Health Canada's medical device directives with FDA standards creates a unified regulatory bloc requiring identical software provenance tools.
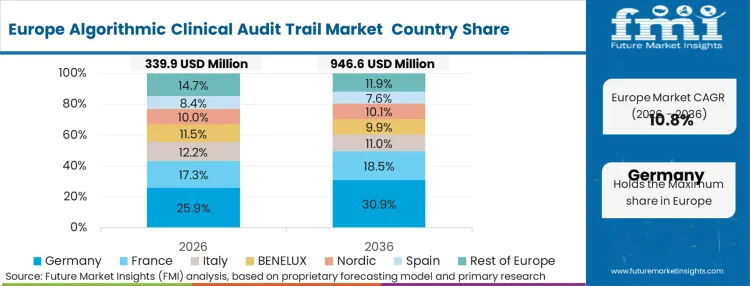
Strict infrastructure constraints surrounding the General Data Protection Regulation (GDPR) and the impending EU AI Act dictate how European hospitals handle algorithmic transparency. This unique regulatory interplay requires audit trails to simultaneously provide complete algorithmic explainability to regulators while fiercely protecting patient data from unauthorized exposure. According to FMI's estimates, European deployments rely heavily on localized secure enclaves rather than broad cloud networks.
FMI's report includes detailed analysis of Italy, Spain, and the Nordics. The emergence of unified European Health Data Space policies accelerates the adoption of cross-border cryptographic logging standards.
Specific margin pressures and the urgent need to scale healthcare delivery across massive populations dominate the Asian dynamic. This economics-led environment pushes hospitals to adopt autonomous AI diagnostics to offset severe specialist shortages, which in turn dramatically escalates their institutional malpractice exposure. Based on FMI's assessment, Asian healthcare networks prioritize lightweight, high-speed audit middleware that will not disrupt the rapid patient throughput required in hyper-dense urban medical centers.
FMI's report includes coverage of South Korea and Australia. Strong investments by state health ministries in these nations underscore a rapid pivot toward standardized algorithmic accountability frameworks.
The algorithmic clinical audit trail market is moderately consolidated, dominated by major health IT incumbents and specialized regulatory software developers. This structure exists because navigating the complex intersection of FDA SaMD regulations, 21 CFR Part 11 compliance, and advanced cryptography requires massive upfront capital and specialized legal expertise. Companies like Merative and Microsoft Corporation leverage their existing hospital enterprise footholds to bundle audit capabilities into broader IT contracts. Buyers primarily distinguish qualified vendors based on their software's latency impact, specifically, the ability to generate an immutable cryptographic hash for a clinical decision without adding measurable delay to acute care diagnostic workflows.
Leading entities such as ProPharma Group and H2O.ai maintain distinct structural advantages through their deep integration with Good Machine Learning Practice frameworks and established regulatory consultancy practices. These companies possess proprietary semantic mapping algorithms that translate complex neural network weights into human-readable clinical logic instantly. A challenger entering this space must specifically build native Retrieval-Augmented Generation (RAG) provenance tracking to compete, proving exactly which medical journal an LLM utilized to generate a recommendation. By embedding ai in medical imaging overlays with these specific semantic translators, challengers can bypass established legacy IT vendors entirely.
To prevent vendor lock-in, large tier-1 research hospitals are increasingly mandating open-standard audit architectures. Buyers explicitly want their algorithmic ledgers physically and contractually separated from the vendors providing the actual AI diagnostic models to ensure unbiased performance reporting. This structural tension limits the pricing power of major AI vendors attempting to sell closed-loop, proprietary logging tools. The competitive trajectory to 2036 points toward slight fragmentation as specialized, third-party "black box" audit firms emerge to act as independent algorithmic arbitrators between healthcare providers and AI developers.
| Metric | Value |
|---|---|
| Quantitative Units | USD 1.5 billion to USD 4.7 billion, at a CAGR of 12.14% |
| Market Definition | Specialized software and middleware that securely record, encrypt, and store the inference pathways of medical artificial intelligence. It ensures immutable regulatory compliance and algorithmic accountability. |
| Deployment Model Segmentation | Cloud-Based Ledger Models, On-Premise Secure Enclaves, Hybrid Cryptographic Nodes |
| End User Segmentation | Tier-1 Research Hospitals, AIaMD Developers, Clinical Laboratories |
| Component Segmentation | Cryptographic Logging Software, Audit Middleware APIs, Surveillance Services |
| Application Segmentation | Medical Imaging AI Oversight, Predictive Analytics Tracking, Genomics Decision Support |
| Regions Covered | North America, Europe, East Asia, Asia Pacific |
| Countries Covered | United States, Germany, United Kingdom, Japan, China, France, India, and 40 plus countries |
| Key Companies Profiled | ProPharma Group, Merative, Microsoft Corporation, Oracle Cerner, Epic Systems Corporation, H2O.ai, Arthur AI, Enlitic Inc. |
| Forecast Period | 2026 to 2036 |
| Approach | FMI engaged Chief Medical Information Officers and regulatory compliance directors. The baseline was constructed from FDA 510(k) AI/ML medical device clearances and validated against enterprise software procurement expenditures. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Algorithmic Clinical Audit Trail Market in 2026?
The industry is expected to cross USD 1.5 billion in 2026, driven directly by the aggressive rollout of FDA Predetermined Change Control Plan frameworks.
What will it be valued at by 2036?
Sustained investment propels the total valuation to USD 4.7 billion through 2036 as algorithmic malpractice liability forces a structural shift away from manual IT logging.
What CAGR is projected?
The sector advances at a 12.14% CAGR, anchored solidly by mandatory continuous monitoring requirements under international Software as a Medical Device (SaMD) directive.
Which Deployment Model segment leads?
Cloud-Based Ledger Models lead with a 62.4% share in 2026, due to their decentralized architecture which prevents any single node from altering historical algorithmic inference records.
Which End User segment leads?
Tier-1 Research Hospitals dictate this dimension, functioning as the primary incubators for high-risk autonomous diagnostic algorithms demanding extreme legal protection.
Which Component segment leads?
Cryptographic Logging Software holds dominance, forming the non-negotiable compliance foundation required under 21 CFR Part 11 before any higher-level surveillance can be implemented.
What drives rapid growth?
FDA mandates and Good Machine Learning Practice principles force AI developers to embed verifiable, immutable provenance into their software to maintain regulatory clearance.
What is the primary restraint?
Severe operational friction arises from the computational latency introduced by cryptographic logging, which can slow down high-frequency intensive care monitoring alerts.
Which country grows fastest?
The United States advances at 14.2% compound annual growth, driven by aggressive federal enforcement of transparent lifecycle management for software as a medical device.
How do 21 CFR Part 11 mandates affect adoption?
These mandates require that audit trails for electronic records be strictly computer-generated and unalterable, completely invalidating manual centralized legacy logs.
Why are cryptographic ledgers replacing legacy IT logs?
Blockchain-enabled ledgers lock AI decision provenance definitively, eliminating evidentiary disputes regarding probabilistic inference paths during medical malpractice litigation.
How do major vendors compete in this space?
Firms compete primarily on latency and semantic disambiguation mapping, ensuring complex algorithms are logged in milliseconds while remaining readable to compliance officers.
What is the role of Semantic Disambiguation?
It translates raw neural network outputs and mathematical weights into standardized medical ontologies like SNOMED-CT, making algorithmic logic instantly comprehensible.
How does the Bolam principle apply here?
The traditional Bolam principle protects human clinicians but struggles to accommodate non-human agency, forcing hospitals to acquire detailed audit logs to defend autonomous systems.
What constitutes Algorithmic Malpractice?
It is a novel liability concept where probabilistic outputs from AI systems cause or contribute directly to clinical errors, requiring specialized software to detect and document.
What are Predetermined Change Control Plans (PCCPs)?
PCCPs are FDA frameworks that allow AI models to learn and update continuously post-market, provided they possess a rigorous, automated audit trail justifying the changes.
Why do AIaMD Developers purchase audit middleware?
They embed these API modules directly into their core codebase to guarantee out-of-the-box compliance and secure rapid procurement from strict hospital legal departments.
How does Germany’s DiGA process impact adoption?
The DiGA fast-track evaluation mandates transparent algorithmic behavior, forcing digital health engineers to build deeply embedded cryptographic ledgers to qualify for reimbursement.
What role does Retrieval-Augmented Generation (RAG) provenance play?
It enables developers to track exactly which medical journal or consensus guideline a Large Language Model utilized to generate a specific clinical recommendation.
Why do hospitals want independent "black box" logging?
Institutions want their audit layers contractually separated from the AI vendors to prevent the manipulation of performance data and ensure completely unbiased quality reporting.
How does the NHS approach this technology?
The UK's MHRA enforces strict Good Machine Learning Practice rules, effectively barring system integrators from NHS procurement pipelines if they lack robust provenance logging.
What is the primary risk of Medical Imaging AI?
Radiological algorithms are highly susceptible to silent model drift and learning spurious correlates, requiring pixel-level continuous tracking to prevent clustered misdiagnoses.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.