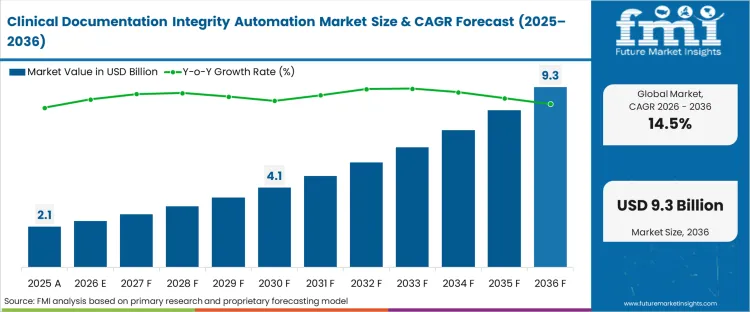
The Clinical Documentation Integrity Automation crossed a valuation of USD 2.1 billion in 2025. The sector is set to reach USD 2.4 billion in 2026 at a CAGR of 14.50% during the forecast period. Sustained investment propels the valuation to USD 9.3 billion through 2036 as narrowing margins under value-based care models force health systems to systematically eliminate undocumented patient acuity that results in reimbursement leakage.
Chief financial officers at major health systems are currently shifting their procurement strategy from retrospective auditing to concurrent clinical nudging. Facilities that wait until after a patient is discharged to correct missing specificity are structurally incapable of capturing their true case mix index. The commercial penalty for relying on retrospective queries is an unrecoverable margin loss on complex admissions. To prevent this leakage, administrators are integrating real-time natural language processing tools directly into the clinical interface. The paradox within this transition is that deploying an intelligent documentation tool often initially increases the manual review burden. It flags every missing specificity, creating a temporary bottleneck until the clinical rules engine is properly calibrated to ignore low-yield corrections.
Before growth becomes fully self-reinforcing across smaller provider networks, the technology must cross a specific operational threshold: passive ambient listening. Once the software can generate a compliant clinical note without requiring the physician to actively interface with a keyboard or dictate specific commands, the primary structural friction of physician burnout is neutralized. This shift transforms the system from an administrative burden into a clinical workflow accelerator.
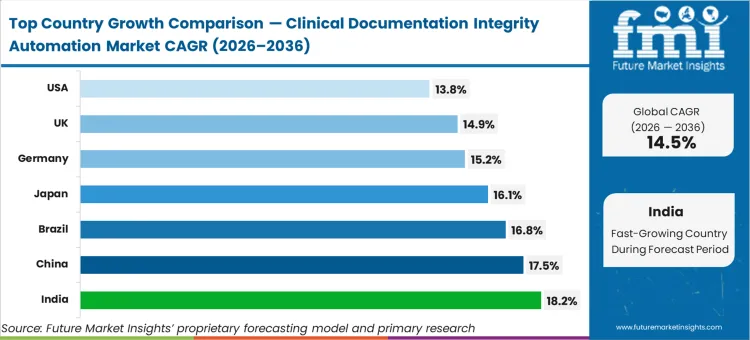
India is expected to track at 18.2% as corporate hospital chains centralize revenue cycle operations to bypass legacy manual coding frameworks. China is projected to advance at 17.5%, driven by the rapid digitization of provincial healthcare hubs. Brazil is likely to register 16.8% as large private insurers demand greater diagnostic specificity for claims processing. Japan is poised to garner 16.1% as an ageing demographic forces an emphasis on chronic disease documentation accuracy. Germany is estimated to expand at 15.2% due to rigorous national hospital reimbursement compliance audits. The United Kingdom follows at 14.9%, where national trust mandates prioritize standardizing patient data capture. The United States is anticipated to post 13.8%. The differences seen across global healthcare networks largely stem from how strongly regional authorities penalize vague diagnoses and how generously they reward accurate, coordinated care.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 2.4 billion |
| Industry Value (2036) | USD 9.3 billion |
| CAGR (2026-2036) | 14.50% |
Clinical Documentation Integrity Automation refers to the deployment of algorithmic software, particularly natural language processing and machine learning, to concurrently review, analyze, and correct patient health records. The technology functions as a real-time intermediary between clinical care delivery and medical coding. It is distinct from standard electronic health records because it actively prompts physicians for diagnostic specificity while the patient is still admitted, rather than passively storing text for retrospective human review.
The scope includes standalone natural language processing engines, computer-assisted physician documentation modules, and cloud-hosted algorithmic rules engines designed specifically for acuity capture. It encompasses ambient clinical intelligence solutions that passively draft notes from patient encounters. Integrated platforms that combine severity of illness tracking with automated query generation to physicians fall directly within the analyzed boundary.
Basic dictation software and legacy transcription services without diagnostic rules engines are excluded because they merely digitize speech without analyzing it for clinical accuracy. General revenue cycle management tools that only sequence existing codes for billing are excluded because they do not interface directly with the physician workflow to alter the primary clinical narrative. Retrospective human auditing services utilizing legacy software are omitted because they fail the automation threshold.
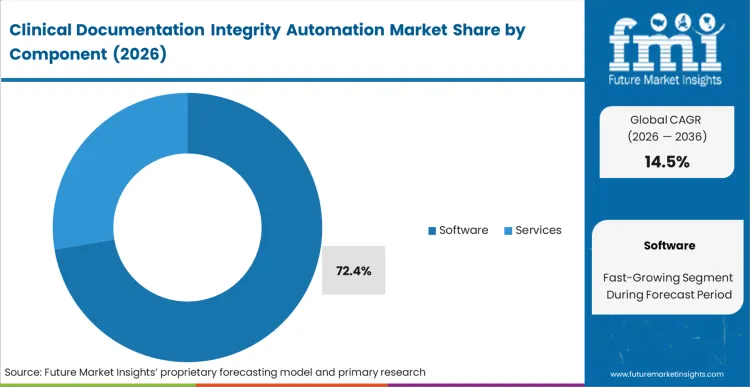
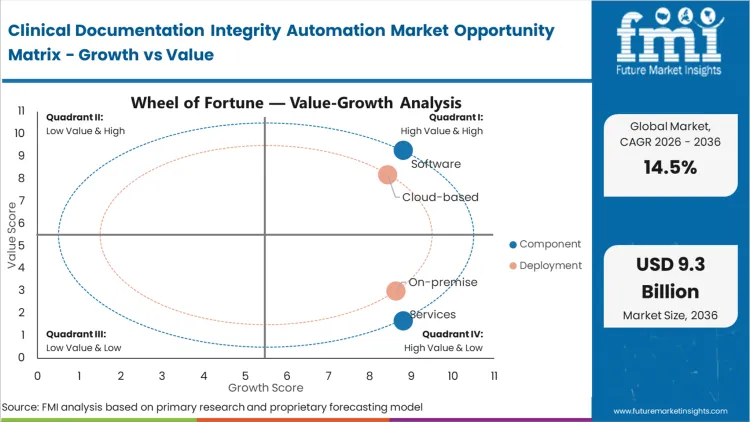
Retrospective human auditing fails to prevent revenue leakage because queries reach physicians days after the clinical context has faded. Software captures 72.4% of the industry in 2026 by shifting the intervention to the exact moment of care. As per FMI's projection, the algorithmic intelligence embedded within these platforms creates value by actively translating unstructured narrative into codable discrete data. Directors of clinical informatics prioritize software over augmented staffing services because the technology scales infinitely across different medical specialties without proportional cost increases. Once integrated, the software continuously scans for clinical indicators like abnormal lab values that lack a corresponding diagnosis in the physician's note. Networks that attempt to manage this volume with outsourced human review face permanent operational bottlenecks.
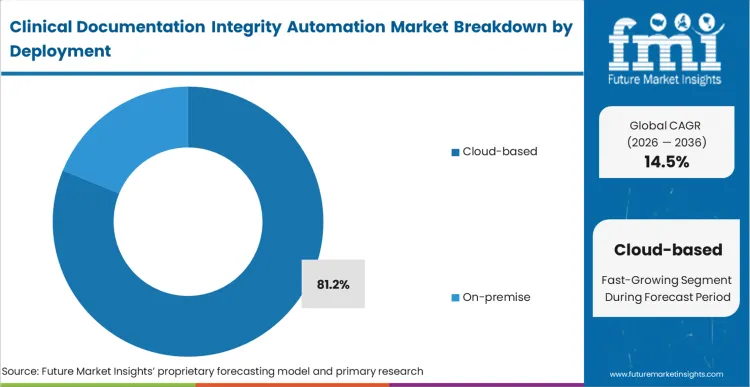
The architectural tension in deployment centers on the massive processing power required for natural language processing versus the strict data sovereignty rules governing patient health information. Cloud-based infrastructure secures 81.2% share in 2026 because on-premise servers simply cannot support the continuous machine learning model updates required for accurate clinical rules engines.
FMI analysts opine that enterprise architecture teams are abandoning local server deployments to avoid the hidden costs of maintaining specialized computing hardware. The cloud model allows vendors to aggregate anonymized clinical patterns across thousands of hospitals, instantly refining the artificial intelligence modules for all users simultaneously. Facilities clinging to local deployments restrict their algorithms from learning newly established medical terminology, degrading their automated query accuracy over time.
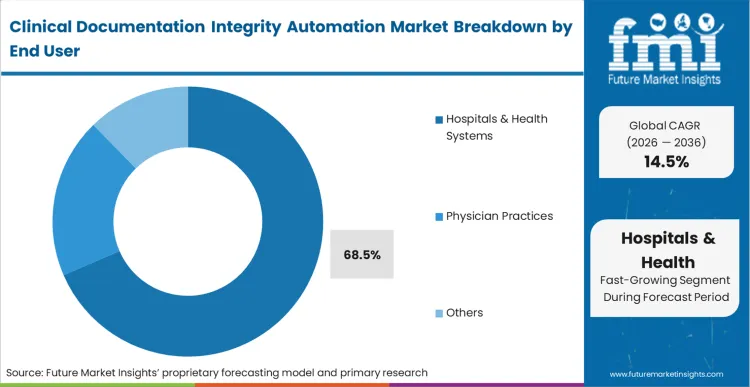
The commercial consequence of undocumented patient acuity falls disproportionately on complex inpatient facilities. Hospitals & Health Systems account for 68.5% share in 2026 due directly to their high exposure to value-based care penalties.
Based on FMI's assessment, a physician practice might lose a fraction of an evaluation fee due to poor documentation, but a major hospital loses the entire margin on a complex surgical admission if secondary comorbidities remain unrecorded. Chief financial officers at these institutions authorize enterprise-wide deployments because the automation prevents the structural failure of under-reporting severity of illness. Regional medical centers that delay implementing these concurrent intelligence layers forfeit critical reimbursement adjustments necessary to fund their high-acuity departments.
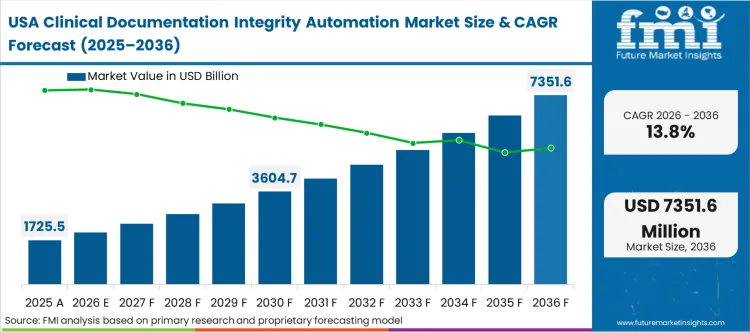
The shift toward value-based reimbursement forces hospital financial executives to prove the severity of their patient populations through meticulous data capture. Chief financial officers are deploying concurrent auditing tools because undocumented acuity translates directly into unrecoverable margin loss under fixed-payment contracts. A hospital treating highly complex cases cannot survive financially if its clinical notes reflect only basic, uncomplicated encounters. Delaying the adoption of real-time intelligent nudging means accepting a structural depression of the facility's case mix index, directly reducing future contracted payment rates from major insurers.
The primary organizational friction slowing adoption is intense physician resistance to any software that introduces additional clicks into their daily workflow. Clinical informatics directors recognize that even highly accurate algorithms will be ignored if they trigger disruptive pop-up alerts during patient care. This friction is structural because physician burnout is already a critical operational liability. Vendors are attempting to solve this by developing passive background listening tools, but these early ambient systems still struggle with complex multi-speaker clinical environments.
Opportunities in the Clinical Documentation Integrity Automation Market
Based on the regional analysis, the Clinical Documentation Integrity Automation market is segmented into North America, Europe, Asia Pacific across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 18.2% |
| China | 17.5% |
| Brazil | 16.8% |
| Japan | 16.1% |
| Germany | 15.2% |
| United Kingdom | 14.9% |
| United States | 13.8% |
The regulatory environment surrounding Medicare reimbursement policies shapes the absolute baseline for buyer behaviour in this region. The Centres for Medicare & Medicaid Services consistently tightens the criteria for hierarchical condition category coding, making manual chart review financially unviable for large networks. According to FMI's estimates, hospital administrators are not purchasing these tools for general efficiency: they are defending their operating margins against aggressive federal recoupment audits. The intense scrutiny on billing accuracy forces institutions to embed population health management documentation rules directly into the physician interface.
According to FMI’s analysis of the Canadian market, strict national reimbursement policies, not hospital‑level choices, largely determine how and when clinical documentation automation tools are adopted.
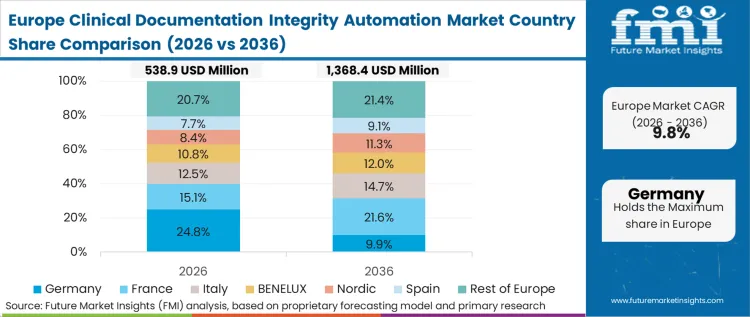
The condition of digital health infrastructure strictly limits how rapidly intelligent auditing can be deployed across the continent. Fragmented electronic health record deployments within nationalized health systems create massive interoperability barriers for third-party algorithmic engines. In FMI's view, informatics directors spend more time harmonizing legacy database architectures than they do training clinical staff on the new software. The focus remains heavily on standardizing the baseline patient narrative before complex predictive nudging can function properly.
FMI's report includes an analysis of France, Italy, and Spain. The common shift observed in these nations is the gradual replacement of fragmented, custom hospital databases with large‑scale interoperable systems.
Cost structures and the availability of capital exclusively drive procurement decisions across these rapidly modernizing networks. The extreme disparity between tier-1 corporate hospital chains and rural public facilities creates a highly polarised technology scenario. As per FMI's projection, corporate hospital directors bypass the legacy evolution of human transcription teams entirely, moving straight to advanced algorithmic deployment. This leapfrogging behavior is funded by the aggressive expansion of the private health insurance sector, which demands rigorous documentation to authorize premium procedural payouts.
FMI's report includes analysis of the South Korean and Australian networks. The defining structural pattern here is the prioritization of smart hospital infrastructure that natively includes documentation integrity layers at the architectural level.
The intense concentration at the top of this sector is dictated entirely by the closed architectures of the dominant electronic health record providers. Companies like Microsoft (Nuance Communications), 3M Health Information Systems, and Oracle Health dictate the baseline because they control the physician's immediate screen space. Buyers utilise the depth of native integration as the primary variable to distinguish qualified vendors from generic algorithmic startups. A standalone natural language processing engine with superior clinical accuracy will be entirely disqualified if it requires the physician to log into a separate browser window.
Incumbents maintain their advantage through vast proprietary libraries of historical clinical narratives. 3M Health Information Systems and Optum leverage decades of claims denial data to refine their predictive nudging algorithms in ways that new entrants cannot synthetically replicate. A challenger must build highly specialised ontological mapping capabilities that target narrow, highly complex medical specialities like interventional cardiology or neurosurgery. Establishing this interoperability capability allows them to secure niche contracts where the brgeneralisedized engines provided by the incumbents fail to capture the necessary severity of illness.
Large hospital networks resist vendor lock-in by demanding modularity via application programming interfaces. They refuse to sign enterprise contracts that bind their clinical documentation engine to a single billing service provider. The structural tension between the hospital's desire for agile, swappable software components and the vendor's incentive to bundle services will define the competitive trajectory through 2036. The sector is moving toward deeper consolidation as major enterprise technology firms acquire specialized algorithmic startups to complete their ambient intelligence portfolios.
| Metric | Value |
|---|---|
| Quantitative Units | USD 2.4 billion to USD 9.3 billion, at a CAGR of 14.50% |
| Market Definition | Clinical Documentation Integrity Automation refers to algorithmic software that concurrently reviews and corrects patient health records to ensure diagnostic specificity. The technology bridges clinical care delivery and medical coding in real time. |
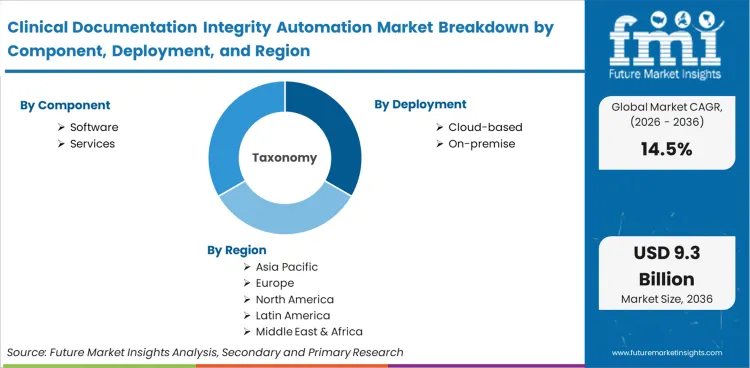
| Component Segmentation | Software, Services |
| Deployment Segmentation | Cloud-based, On-premise |
| End User Segmentation | Hospitals & Health Systems, Physician Practices, Others |
| Regions Covered | North America, Europe, Asia Pacific |
| Countries Covered | United States, United Kingdom, Germany, Japan, Brazil, China, India, and 40 plus countries |
| Key Companies Profiled | 3M Health Information Systems, Optum, Oracle Health, Iodine Software, Dolbey Systems, AGS Health, Microsoft (Nuance) |
| Forecast Period | 2026 to 2036 |
| Approach | Primary research targeted Chief Medical Information Officers and revenue cycle directors to gauge procurement sentiment. The baseline data anchored to annual enterprise software procurement budgets across tier-1 health systems. Forecasts were cross-validated using quarterly deployment rates from top-tier health record vendors. |
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the demand for Clinical Documentation Integrity Automation in the global market in 2026?
Demand for Clinical Documentation Integrity Automation in the global market is estimated to be valued at USD 2.4 billion in 2026.
What will be the market size of Clinical Documentation Integrity Automation in the global market by 2036?
Market size for Clinical Documentation Integrity Automation is projected to reach USD 9.3 billion by 2036.
What is the expected demand growth for Clinical Documentation Integrity Automation in the global market between 2026 and 2036?
Demand for Clinical Documentation Integrity Automation is expected to grow at a CAGR of 14.50% between 2026 and 2036.
Which Component is poised to lead global sales by 2026?
Software accounts for 72.4% in 2026 as algorithmic intelligence converts unstructured clinical narratives into codable data without depending on retrospective human review.
How significant is the role of Cloud-based deployment in driving Clinical Documentation Integrity Automation adoption in 2026?
Cloud-based deployment represents 81.2% of segment share in 2026 as health systems require continuous machine learning updates and scalable processing capacity.
What is driving demand in China?
China’s rapid digitization of provincial healthcare hubs and large-scale investment in enterprise hospital software are driving demand for automated clinical documentation integrity platforms.
What compliance standards or regulations are referenced for Germany?
Germany’s market is shaped by rigorous hospital reimbursement compliance requirements and strict procedural documentation expectations within its financing system.
What is the China growth outlook in this report?
China is projected to grow at a CAGR of 17.5% during 2026 to 2036.
Why is North America described as a priority region in this report?
North America is described as a priority region because tightening Medicare reimbursement rules and risk adjustment scrutiny make manual chart review financially unsustainable for large health systems.
What type of demand dominates in North America?
Demand focuses on concurrent documentation review tools that improve acuity capture, strengthen reimbursement accuracy, and reduce exposure to federal audit risk.
What is India's growth outlook in this report?
India is projected to expand at a CAGR of 18.2% during 2026 to 2036.
Does the report cover USA in its regional analysis?
Yes, USA is included within North America under the regional scope of analysis.
What are the sources referred to for analyzing USA?
CMS reimbursement frameworks, risk adjustment policies, hospital revenue cycle requirements, and ambient documentation adoption patterns form the analytical basis.
What is the main demand theme linked to USA in its region coverage?
The main demand theme is the need to capture patient severity accurately under risk-bearing care models and federal reimbursement oversight.
Does the report cover Germany in its regional analysis?
Yes, Germany is included within Europe under the regional coverage framework.
What is the main Germany related demand theme in its region coverage?
Germany’s demand is tied to precise procedural and diagnostic documentation needed to support reimbursement integrity within a tightly regulated hospital financing environment.
Which product formats or configurations are strategically important for Asia Pacific healthcare networks?
Cloud-hosted NLP engines, ambient clinical intelligence tools, and centralized documentation automation platforms are strategically important for scaling across large hospital networks.
What is Clinical Documentation Integrity Automation and what is it mainly used for?
Clinical Documentation Integrity Automation uses algorithmic software to review and improve patient records in real time. It is mainly used to increase diagnostic specificity and support accurate medical reimbursement.
What does Clinical Documentation Integrity Automation mean in this report?
In this report, it refers to software platforms that bridge physician workflow and coding accuracy by analyzing unstructured notes and generating concurrent prompts for clarification.
What is included in the scope of this Clinical Documentation Integrity Automation report?
The market covers standalone NLP engines, computer-assisted physician documentation modules, cloud-hosted rules engines, ambient intelligence solutions, and integrated severity tracking platforms.
What is excluded from the scope of this report?
Basic dictation tools, transcription services without rules engines, general revenue cycle systems without physician workflow integration, and retrospective manual auditing services are excluded.
What does market forecast mean on this page?
The market forecast represents a model-based projection built on healthcare IT adoption patterns, enterprise software budgets, reimbursement pressure, and deployment trends.
How does FMI build and validate the Clinical Documentation Integrity Automation forecast?
FMI uses a baseline anchored to enterprise software procurement budgets for mid-revenue cycle optimization and validates projections through vendor deployment rates, primary interviews, and healthcare reimbursement frameworks.
What does zero reliance on speculative third-party market research mean here?
It means the analysis relies on primary interviews, hospital financial disclosures, interoperability registries, reimbursement guidelines, and verified vendor deployment data instead of unverified syndicated estimates.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.