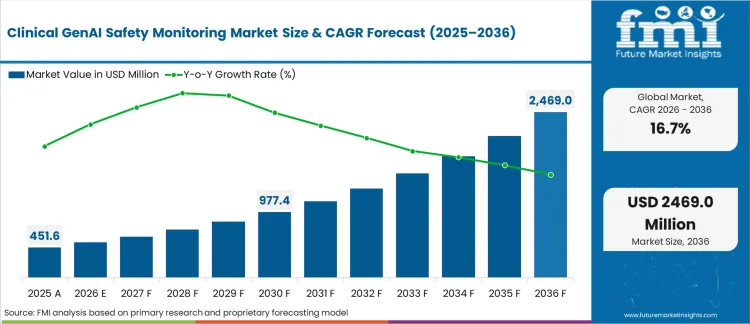
In 2025, the clinical GenAI safety monitoring market was valued at USD 450 million. Based on Future Market Insights' analysis, demand for clinical GenAI safety monitoring solutions is estimated to grow to USD 527 million in 2026 and USD 2,100 million by 2036. FMI projects a CAGR of 16.7% during the forecast period.
Absolute dollar growth of USD 1,650 million over the decade reflects rapid enterprise adoption as healthcare organizations transition from pilot deployments to production-scale AI operations. Growth acceleration stems from mounting regulatory pressure to demonstrate algorithmic accountability, rising awareness of GenAI failure modes in clinical workflows, and the urgent need to prevent patient harm from hallucinated diagnoses or treatment recommendations. Market expansion tracks directly with healthcare GenAI deployment velocity, creating mandatory observability infrastructure rather than optional compliance tooling.
India (18.2% CAGR) and the United States (17.4% CAGR) drive adoption through accelerated health system digitalization and stringent medical AI governance frameworks. The United Kingdom (16.9% CAGR) contributes via NHS AI deployment mandates and comprehensive algorithmic transparency requirements. Germany (16.3% CAGR) expands through Federal Ministry of Health digital health initiatives and hospital sector AI adoption. Canada (15.8% CAGR) grows via provincial health authority procurement standards for AI safety tools.
| Metric | Value |
|---|---|
| Market Size (2026) | USD 527 Million |
| Forecast Value (2036) | USD 2,100 Million |
| CAGR (2026-2036) | 16.7% |
The clinical GenAI safety monitoring market comprises software platforms, validation services, and managed monitoring solutions designed to detect, measure, and prevent harmful outputs from generative AI systems deployed in healthcare settings. These technologies continuously evaluate large language models and generative AI applications for clinical accuracy, bias, hallucination risk, privacy violations, and regulatory compliance across patient documentation, diagnostic support, treatment planning, and administrative workflows.
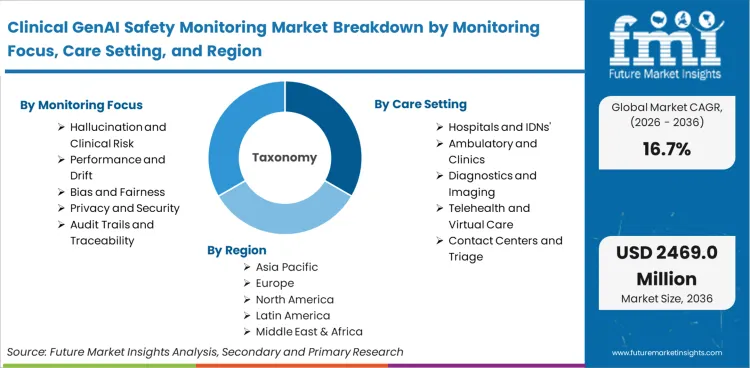
The report includes a comprehensive analysis of market dynamics, featuring Global and Regional Market Sizes (Value) and a 10-year Forecast (2026-2036). It covers segmental breakdowns by monitoring focus (Performance and Drift, Hallucination and Clinical Risk, Bias and Fairness, Privacy and Security, Audit Trails and Traceability), care setting (Hospitals and IDNs, Ambulatory and Clinics, Diagnostics and Imaging, Telehealth and Virtual Care, Contact Centers and Triage), and offering type (Software Platforms, Validation and Testing Services, Managed Monitoring Services, Compliance and Audit Support, Incident Response Services).
The scope excludes general AI observability tools not purpose-built for healthcare, traditional clinical decision support systems without generative AI components, and electronic health record platforms unless they incorporate GenAI monitoring capabilities. It also omits cybersecurity tools focused solely on network intrusion rather than algorithmic behavior, generic MLOps platforms lacking clinical validation features, and downstream medical devices or diagnostic equipment, focusing strictly on software-based monitoring infrastructure and services.
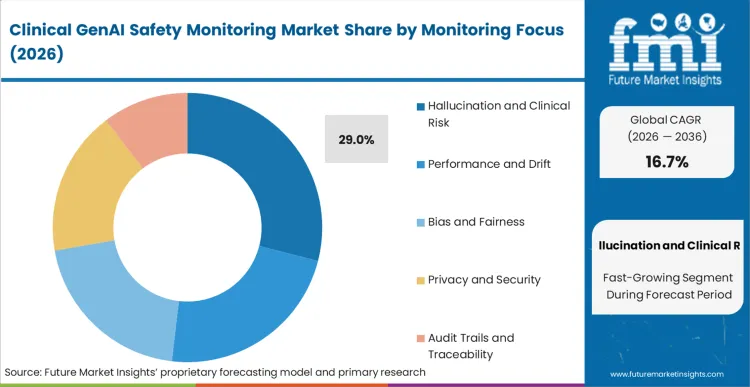
Based on FMI's clinical GenAI safety monitoring market report, hallucination and clinical risk monitoring are estimated to hold 29% share in 2026. Dominance stems from direct patient safety implications where factually incorrect AI outputs in clinical documentation, diagnostic recommendations, or treatment planning can trigger immediate harm, malpractice liability, and regulatory sanctions. This segment addresses the highest-consequence failure mode unique to generative AI, where models confidently generate plausible but medically inaccurate content that clinical staff may not catch before impacting patient care decisions.
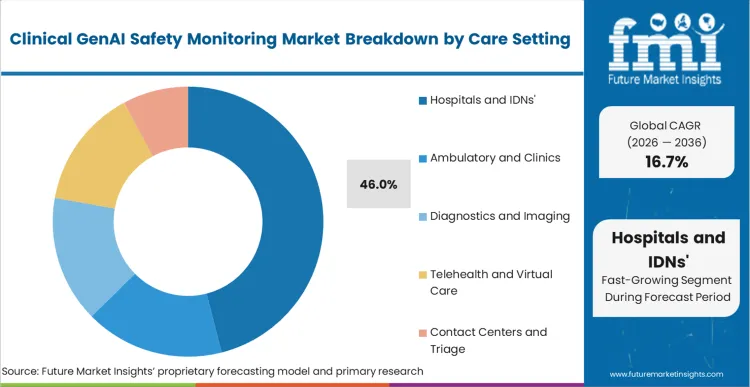
Hospitals and IDNs' 46% market share in 2026 derives from their role as primary adopters of enterprise GenAI platforms at scale, combined with heightened regulatory oversight and concentrated liability exposure. These organizations operate comprehensive AI governance programs that mandate monitoring infrastructure before deployment, possess dedicated IT budgets for safety tooling, and face direct Joint Commission scrutiny around algorithmic accountability. The segment's purchasing power and compliance requirements create natural demand concentration that outpaces smaller care settings still in pilot phases.
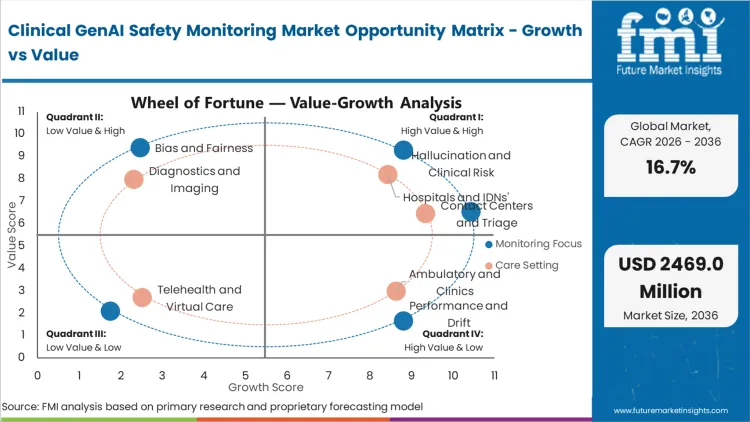
Future Market Insights analysis reveals that historical patterns point at an emerging, high-urgency market where safety monitoring infrastructure evolves from optional governance tool to mandatory deployment prerequisite. The market exists because generative AI in healthcare introduces novel failure modes including confident hallucination of medical facts, algorithmic bias amplification in diagnostic pathways, and privacy violations through training data leakage that traditional quality assurance cannot detect. Estimated 2026 valuation represents the initial enterprise wave as health systems recognize that clinical GenAI deployment without continuous monitoring creates unacceptable patient safety and liability risks.
Core market tension stems from collision between rapid GenAI adoption velocity in healthcare and lagging development of safety validation infrastructure. While standard medical AI focused on narrow prediction tasks with established validation protocols, generative models produce open-ended text outputs requiring fundamentally different monitoring approaches. Growth concentrates in platforms offering real-time hallucination detection and clinical accuracy scoring rather than generic MLOps observability, as hospitals need proof that AI-generated clinical content meets medical standards before physician reliance becomes institutionalized practice.
Based on the regional analysis, the clinical GenAI safety monitoring market is segmented into North America, Europe, Asia Pacific, and Rest of World across 20+ countries. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 18.2% |
| United States | 17.4% |
| United Kingdom | 16.9% |
| Germany | 16.3% |
| Canada | 15.8% |
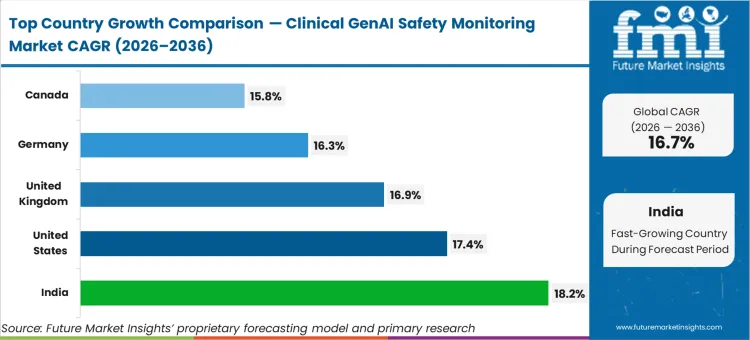
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
North America operates as the global regulatory pathfinder where FDA guidance on medical AI continuous monitoring establishes precedents that shape international standards. Leading regional players include Datadog, whose LLM Observability platform gained healthcare traction through EHR vendor partnerships, and Fiddler AI, specializing in model explainability for clinical applications. Arize AI maintains strong hospital penetration through integrated delivery network deployments.
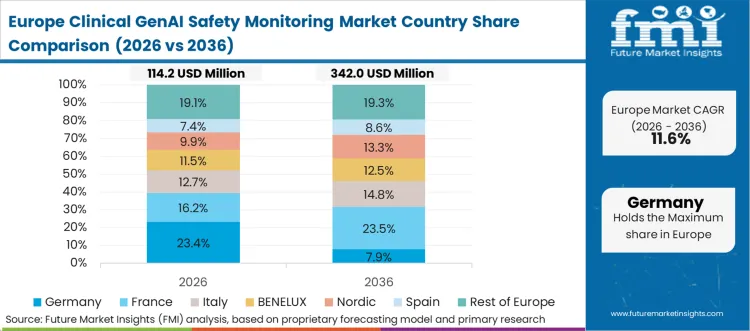
Europe serves as the compliance architecture laboratory where the EU AI Act's high-risk medical AI provisions effective August 2026 create comprehensive monitoring mandates that exceed North American requirements. Key regional vendors include Credo AI, which offers multi-framework AI governance with dedicated EU AI Act compliance documentation; TruEra, providing AI observability and bias monitoring tools applicable to regulated industries; and ValidMind, which maintains a strong presence in financial services AI model risk management with EU AI Act compliance tooling for MRM teams..
FMI's analysis of the clinical GenAI safety monitoring market in Europe consists of country-wise assessments that include the United Kingdom, Germany, France, Italy, and Spain. Readers can find detailed information about EU AI Act compliance requirements, national health system procurement standards, and GDPR intersection with algorithmic monitoring.
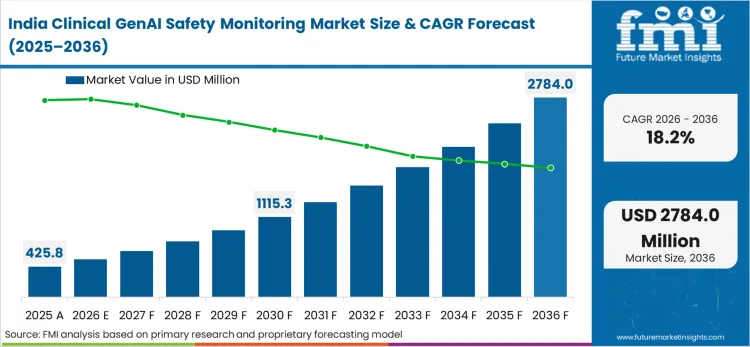
Asia Pacific emerges as the high-growth frontier where accelerating health system digitalization combines with government-led AI safety initiatives to create rapid monitoring adoption, with the region holding approximately 28% of the global healthcare AI market. Singapore's Healthtech Agency and the A*STAR-EVYD Joint Lab are actively building AI validation infrastructure, while Australia's private hospital networks are increasingly adopting clinical GenAI documentation tools. Global vendors including Nabla, Abridge, WhyLabs, and Arthur AI are positioned to serve this expanding market, though current documented deployments remain concentrated in North America and Europe
The full report analyzes the clinical GenAI safety monitoring market across Asia Pacific from 2026-2036, covering adoption patterns, regulatory frameworks, and growth drivers in India, China, Japan, South Korea, Australia, Singapore, and emerging Southeast Asian markets. The assessment highlights how government digitalization programs and public hospital procurement mandates shape regional demand.
Market structure remains highly fragmented across specialist monitoring vendors, EHR platform extensions, and general AI observability tools adapting to healthcare use cases. Primary competitive variables include not only technical capability but integration depth with existing hospital IT infrastructure, where vendors offering seamless EHR connectivity via standards-based APIs gain procurement advantage over standalone platforms.
Vendors like Fiddler AI and TruEra offer AI observability platforms applicable to healthcare, with Fiddler providing HIPAA-compliant monitoring for healthcare organizations and TruEra delivering ML quality monitoring that spans multiple regulated industries. Successful competitors integrate with EHR platforms including Epic, Cerner Oracle, and Meditech via standards such as FHIR and SMART on FHIR, embedding monitoring capabilities into clinical workflow platforms where hospital IT departments concentrate purchasing power.
Customer concentration among large integrated delivery networks creates buyer leverage that shapes pricing and feature roadmaps. Health systems increasingly demand proof that monitoring tools detect failure modes specific to clinical environments, such as medication dosing errors and diagnostic hallucinations, rather than accepting generic AI observability features. This selectivity drives vendors to build clinical validation partnerships with academic medical centers to maintain competitive positioningRecent Developments
| Metric | Value |
|---|---|
| Quantitative Units | USD 527 million (2026) to USD 2,100 million (2036), at a CAGR of 16.7% |
| Market Definition | The clinical GenAI safety monitoring market comprises software platforms, validation services, and managed monitoring solutions designed to detect, measure, and prevent harmful outputs from generative AI systems deployed in healthcare settings, with focus on clinical accuracy, bias detection, privacy protection, and regulatory compliance. |
| Monitoring Focus Segmentation | Performance and Drift, Hallucination and Clinical Risk, Bias and Fairness, Privacy and Security, Audit Trails and Traceability |
| Care Setting Segmentation | Hospitals and IDNs, Ambulatory and Clinics, Diagnostics and Imaging, Telehealth and Virtual Care, Contact Centers and Triage |
| Offering Segmentation | Software Platforms, Validation and Testing Services, Managed Monitoring Services, Compliance and Audit Support, Incident Response Services |
| Regions Covered | North America, Europe, Asia Pacific, Rest of World |
| Countries Covered | United States, Canada, United Kingdom, Germany, France, Italy, Spain, India, China, Japan, South Korea, Australia, Singapore, and 10 plus additional countries |
| Key Companies Profiled | Abridge, Nabla, Datadog, Arize AI, Fiddler AI, WhyLabs, Arthur AI, TruEra, Credo AI, ValidMind |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top-down and bottom-up market modeling validated through primary interviews with hospital AI governance officers and health system CIOs, supported by regulatory guidance analysis and vendor licensing data verification |
This bibliography is provided for the reader's reference and is not exhaustive. The full report contains the complete reference list and detailed citations.
How large is the demand for Clinical GenAI Safety Monitoring in the global market in 2026?
Demand for Clinical GenAI Safety Monitoring in the global market is estimated to be valued at USD 527 million in 2026.
What will be the market size of Clinical GenAI Safety Monitoring in the global market by 2036?
Market size for Clinical GenAI Safety Monitoring is projected to reach USD 2,100 million by 2036.
What is the expected demand growth for Clinical GenAI Safety Monitoring in the global market between 2026 and 2036?
Demand for Clinical GenAI Safety Monitoring in the global market is expected to grow at a CAGR of 16.7% between 2026 and 2036.
Which monitoring focus is poised to lead global adoption by 2026?
Hallucination and clinical risk monitoring is expected to be the dominant focus area, capturing approximately 29% of global market share in 2026 due to direct patient safety implications.
How significant is the role of hospitals and IDNs in driving Clinical GenAI Safety Monitoring adoption in 2026?
Hospitals and IDNs represent the critical deployment segment, projected to hold a substantial 46% share of the total market in 2026 as concentrated AI adoption and regulatory scrutiny drive monitoring infrastructure requirements.
What is driving Clinical GenAI Safety Monitoring demand in the United States?
FDA guidance requiring continuous performance monitoring for medical AI devices, federal AI safety grant programs, and CMS reimbursement requirements linking payment to documented quality assurance are driving adoption.
What compliance standards are referenced for the United States?
FDA draft guidance on continuous monitoring for medical AI devices (June 2024), CMS reimbursement quality assurance requirements, and HIPAA compliance for algorithmic oversight are key regulatory references.
What is the United States growth outlook in this report?
The United States is projected to grow at a CAGR of 17.4% during 2026 to 2036.
Why is Europe described as an important regulatory framework region in this report?
Europe's EU AI Act establishes comprehensive high-risk medical AI monitoring mandates that exceed North American requirements, creating regulatory architecture that shapes global standards for algorithmic accountability.
What type of demand dominates in Europe?
Compliance-driven demand for monitoring infrastructure that satisfies EU AI Act algorithmic transparency and ongoing validation requirements dominates regional adoption patterns.
What is Germany's growth outlook in this report?
Germany is projected to expand at a CAGR of 16.3% during 2026 to 2036.
Does the report cover India in its regional analysis?
India is included within Asia Pacific under the regional scope of analysis.
What are the sources referred to for analyzing the market in India?
National Health Authority AI Safety Framework documentation, Ministry of Health and Family Welfare digital health allocations, Medical Council of India telemedicine guidelines, and major hospital network procurement announcements are cited as primary reference sources.
What is the main demand theme linked to India in Asia coverage?
Government-mandated AI governance frameworks and public hospital digitalization programs requiring monitoring infrastructure before GenAI deployment authorization drive accelerated adoption.
What is Clinical GenAI Safety Monitoring and what is it mainly used for?
Clinical GenAI Safety Monitoring comprises software and services that continuously detect, measure, and prevent harmful outputs from generative AI systems in healthcare, primarily used to ensure clinical accuracy, prevent hallucinated medical content, detect algorithmic bias, and maintain regulatory compliance.
What does Clinical GenAI Safety Monitoring Market mean in this report?
Clinical GenAI Safety Monitoring Market refers to global production, procurement, and deployment of monitoring platforms, validation services, and managed solutions designed to oversee generative AI systems operating in clinical environments.
What is included in the scope of this Clinical GenAI Safety Monitoring Market Report?
Scope covers monitoring solutions by focus area (performance, hallucination, bias, privacy, audit), care setting deployment (hospitals, ambulatory, diagnostics, telehealth, triage), and offering type (software platforms, validation services, managed monitoring, compliance support, incident response).
What is excluded from the scope of this report?
General AI observability tools not designed for healthcare, traditional clinical decision support without GenAI components, cybersecurity tools focused on network intrusion rather than algorithmic behavior, and downstream medical devices are excluded unless they incorporate GenAI monitoring capabilities.
What does market forecast mean on this page?
Market forecast represents model-based projections built on defined regulatory assumptions, hospital deployment patterns, and vendor adoption trajectories for strategic planning purposes.
How does FMI build and validate the Clinical GenAI Safety Monitoring Market forecast?
Forecast is developed using hybrid top-down and bottom-up modeling validated through primary interviews with hospital AI governance officers, health system CIOs, regulatory compliance directors, and cross-validation with hospital procurement data and regulatory filing trends.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.