Clinical Drainage Supplies Market
Clinical Drainage Supplies Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Clinical Drainage Supplies Market Forecast and Outlook 2026 to 2036
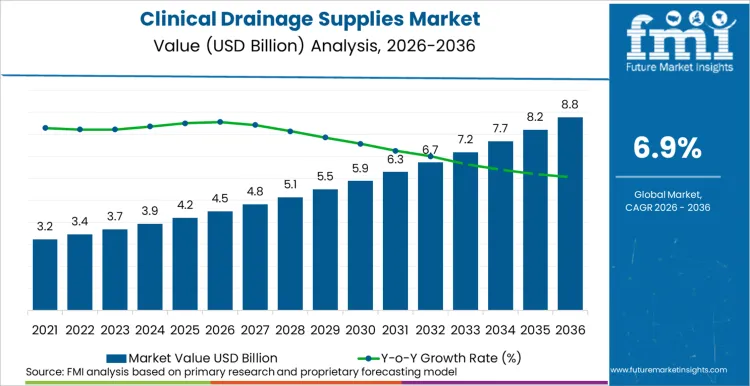
Global clinical drainage supplies market valuation is projected to reach USD 8.7 billion by 2036, rising from USD 4.5 billion in 2026 at a CAGR of 6.9%. Expansion is primarily driven by rising surgical site infection (SSI) rates, which necessitate advanced fluid management protocols to ensure positive patient outcomes. Data from the Centers for Disease Control and Prevention (CDC) in April 2024 indicates that SSIs remain a top cause of hospital readmissions, urging hospitals to adopt active drainage systems.
Manufacturers are responding by scaling production of closed-system solutions that minimize airborne contamination risks during post-operative recovery phases. This synergistic phase of growth combines regulatory pressure for safety with technological advancements in silicone materials to reduce tissue trauma. “The benefits of our diversified portfolio were evident as strong performances from Interventional and Surgical helped offset softness in Interventional Urology revenues. Palette Life Sciences revenues exceeded $75 million in 2025, which was above the $73-$75 million guidance.” - Liam Kelly, CEO, Teleflex Incorporated
Opportunities are consolidating around closed wound suction drains, which currently command a 24.3% market share due to widespread use in orthopedics. As per FMI's projection, this segment will expand rapidly as payers demand rigorous evidence of reduced hospital stays to justify reimbursement. Regulatory bodies like Anvisa (Brazil) validated this shift in July 2025 by launching a Unique Device Identification (UDI) system to enhance traceability for high-risk implantables and drains. Companies such as Getinge are capitalizing on this by integrating digital tracking, reporting strong order intake in January 2025. This competitive advantage rests on the ability to provide verifiable safety data to cost-conscious procurement departments in major health systems.
Quick Stats for Clinical Drainage Supplies Market
- Clinical Drainage Supplies Market Value (2026): USD 4.5 billion
- Clinical Drainage Supplies Market Value (2036): USD 8.7 billion
- Clinical Drainage Supplies Industry Forecast CAGR: 6.9%
- Clinical Drainage Supplies Industry Leading Product: Closed wound suction drains (24.3%)
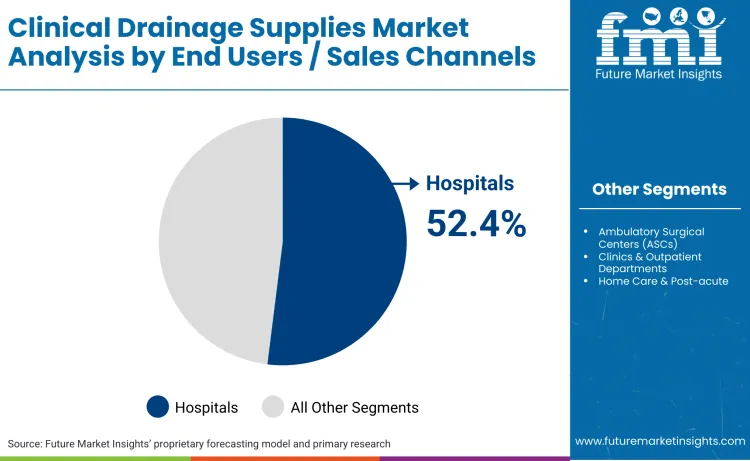
- Clinical Drainage Supplies Industry Leading Channel: Hospitals (52.4%)
- Key Growth Regions: India (9.0%), Brazil (8.1%)
- Key Players: BD, Medtronic, Teleflex, Getinge, Cardinal Health
Clinical Drainage Supplies Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 4.5 Billion |
| Industry Value (2036) | USD 8.7 Billion |
| CAGR (2026-2036) | 6.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
How do infection control mandates drive clinical drainage supplies adoption?
The imperative to reduce hospital-acquired infections (HAIs) is fundamentally reshaping operations, manifesting as a shift toward closed-system active drainage. FMI analysis indicates that updated CDC SSI prevention protocols (February 2025) now emphasize precise fluid management to prevent hematoma formation, directly influencing procurement criteria. Becton Dickinson (BD) validated this trend by completing its acquisition of Edwards Lifesciences' Critical Care unit in September 2024, signaling a strategic pivot toward advanced patient monitoring and fluid management technologies. This aligns with the broader industry adoption of enhanced recovery after surgery (ERAS) protocols, where negative pressure wound therapy plays a critical role in accelerating discharge timelines for complex cases.
What is the segmentation outlook for clinical drainage supplies?
The segmentation landscape is characterized by a distinct transition from passive gravity-based systems to active, vacuum-assisted solutions. FMI observes that closed wound suction drains currently dominate the market, driven by their standard-of-care status in high-volume orthopedic and general surgery procedures. Emerging segments like digital chest drainage are gaining momentum, fueled by the rising incidence of cardiothoracic conditions requiring precise pressure monitoring. Manufacturers are increasingly prioritizing material innovation, shifting from latex to silicone to improve biocompatibility and patient comfort. This strategic implication forces suppliers to diversify portfolios, balancing high-volume consumables with high-value, tech-enabled devices to capture varied hospital departmental budgets.
Why do closed wound suction drains hold a dominant share?
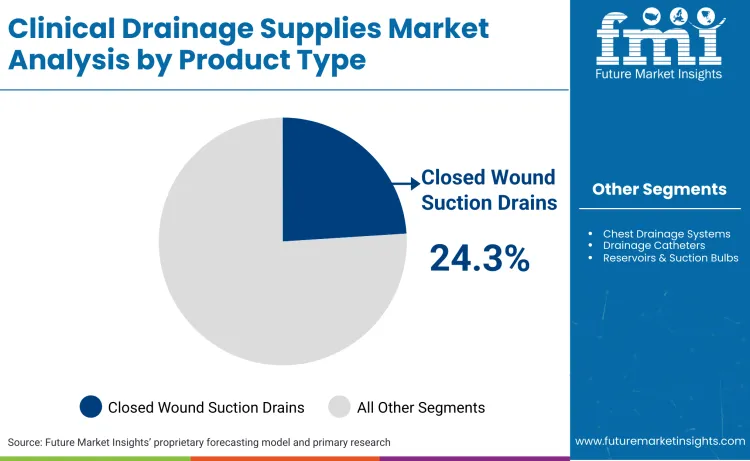
Closed wound suction drains command a significant 24.3% share, largely due to their indispensable role in preventing seroma formation after major surgeries. CDC guidelines (April 2024) specifically highlight the efficacy of fluid removal in preventing deep tissue infections, reinforcing their status as a clinical necessity. Surgeons prefer these wound evacuators for their ability to maintain consistent suction without damaging delicate tissue structures during removal. Market dominance is further cemented by the availability of various sizes and trocars, making them versatile for procedures ranging from mastectomy to joint replacement. Cost-effectiveness compared to complex digital systems ensures their continued volume leadership in cost-constrained public healthcare systems globally.
Why do chest drainage systems lead in high-acuity care?
Chest drainage systems expand at a robust CAGR, reflecting the rising incidence of cardiothoracic procedures and trauma cases globally. NMPA (China) regulations on import approvals (November 2025) have streamlined access for these Class III devices, acknowledging their critical role in life-support contexts. High adoption in trauma centers and thoracic units drives demand for systems that offer real-time air leak monitoring. Teleflex and Getinge maintain dominance in this chest drainage system category by offering integrated digital solutions that reduce nursing burden. This technological edge allows clinicians to make faster decisions regarding tube removal, significantly reducing the average length of hospital stay for patients.
What impact does supply chain localization have on the industry?
Supply chain localization is significantly reshaping the medical device industry, especially in emerging markets where there is a shift from import dependence to local manufacturing. For example, Medtronic's recent appointment of Mandeep Singh Kumar as VP of India reflects a strategic move to indigenize complex innovations, effectively aligning with national initiatives like "Make in India." This trend not only boosts local economies and job creation but also enhances access to medical technologies. By fostering domestic production of medical consumables, companies can respond quicker to market demands and regulatory changes while adhering to national policies promoting self-sufficiency.
How are regulatory changes affecting the medical devices market?
Regulatory harmonization is driving substantial changes in the medical devices market as governments worldwide implement stricter traceability standards. A notable example is Brazil's Anvisa, which launched the Siud UDI system in July 2025 to align with global safety protocols. Such regulations require manufacturers to enhance their labeling and data management systems for compliance, particularly in their drainage catheter portfolios. This trend not only ensures improved patient safety and product accountability but also compels companies to invest in technology upgrades and training. As a result, manufacturers might face increased operational costs but can ultimately enhance their brand reputation and market trust.
What is the Regional Outlook for Clinical Drainage Supplies Market?
In the United States and Europe, the clinical drainage markets are grappling with significant reimbursement constraints that challenge healthcare providers. These developed nations are increasingly focusing on integrating high-value digital technologies to streamline operations and reduce labor costs. In contrast, countries in Asia and other emerging economies are experiencing a rapid expansion in access to healthcare services, driven by the growing needs of their aging populations. FMI anticipates that this divergence will shape distinct product strategies, with premium devices being tailored for the requirements of the US and European markets, while more cost-effective solutions will cater to the basic surgical needs in Asia. As a result, multinational companies will need to maintain a bifurcated portfolio to effectively address the varying demands of these diverse markets and maximize their global outreach.
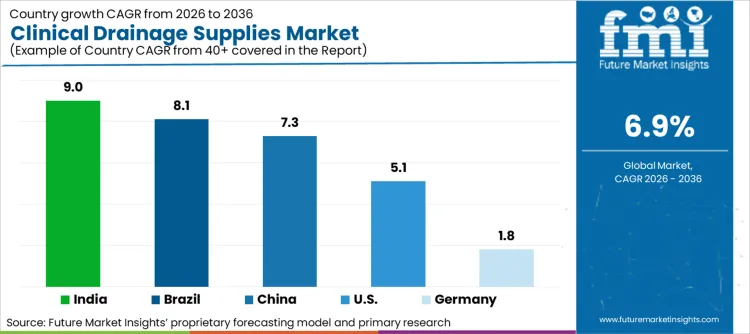
| Country | CAGR (2026–2036) |
|---|---|
| India | 9.0% |
| Brazil | 8.1% |
| China | 7.3% |
| United States | 5.1% |
| Germany | 1.8% |
Source: Future Market Insights (FMI) historical analysis and forecast data.
How is the 'Make in India' initiative catalyzing local growth?
Momentum in the Indian market is accelerating toward a 9.0% CAGR through 2036, catalyzed by a dual engine of rising surgical volume and government incentives for local manufacturing. The National Medical Devices Policy (active 2024) aims to grow the sector to USD 50 billion, specifically targeting wound care and surgical consumables. Medtronic responded to this shift by appointing a new regional VP in May 2024 to drive the indigenization of complex innovations. Mandeep Singh Kumar stated: 'Medtronic's unwavering commitment to alleviating pain, restoring health, and extending lives resonates deeply with me, and I am elated to join the team in keeping the mission alive in India.' Such leadership appointments signify a permanent pivot toward regional self-sufficiency rather than temporary import reliance. The ecosystem is rapidly maturing, transforming India into an export-oriented hub that rivals traditional manufacturing centers in scale and capability.
What regulatory shifts are redefining the Brazil market?
The industry in Brazil is anticipated to grow at 8.1% CAGR. Brazil's demand is heavily influenced by its status as a global hub for plastic and general surgery, sectors that rely heavily on closed-suction drainage. Anvisa (July 2025) officially launched the Siud UDI system, aligning national traceability with global standards. Import data confirms this robustness, with the total medical device import market surging 18% to USD 9.33 billion in 2024. Anvisa officials stated: 'Starting July 2025, manufacturers must register devices in the database and apply standardized UDI labels because this national system aligns Brazil with the International Medical Device Regulators Forum standards.' Compliance with these stringent standards acts as a quality filter, effectively removing substandard low-cost competitors from the ecosystem. As regulatory barriers climb, established players are securing a fortified position that promises long-term revenue stability.
How is innovation transforming China's procurement models?
Modernization efforts in China are projecting the market to expand at a 7.3% CAGR, undergoing a structural shift from volume-based procurement to innovation-driven value. Market dynamics are defined by rapid modernization and an aging demographic requiring complex surgeries. The NMPA (October 2025) released announcements optimizing the review process for innovative medical devices, directly benefiting advanced drainage systems. Data from July 2024 indicates the approval of 59 AI-enabled medical devices, signaling a receptive environment for smart drainage technologies. Accelerating approval timelines for advanced devices forces domestic manufacturers to upgrade their R&D capabilities to compete with global standards. Domestic entities are consequently capturing market share once held by foreign firms, reshaping the competitive balance permanently.
Why is Germany focusing on value-based care?
The Germany market remains the European stronghold, poised to register a steady 1.8% CAGR. Germany's market is characterized by high saturation and intense regulatory scrutiny. The BfArM (December 2025) continues to maintain the DMIDS database as a prerequisite for market access until full EUDAMED integration. However, industry sentiment is cautious; a BVMed survey (late 2024) revealed that 30% of MedTech companies are reducing local investments due to rising costs. Dr. Marc-Pierre Möll, Managing Director of BVMed, warned: 'Germany as a medical technology location continues to become significantly less attractive primarily due to excessive bureaucracy and regulation.' This challenging environment compels manufacturers to abandon low-margin commodity products in favor of high-performance systems that offer undeniable clinical benefits. Manufacturers must therefore pivot their value proposition entirely toward efficiency gains to survive in this highly constrained fiscal landscape.
What sustains surgical demand in the United States?
A robust 5.1% CAGR is forecast for the United States, underpinned by high procedure volumes and adoption of minimally invasive techniques. The U.S. remains the largest single market, driven by the shift of surgeries to Ambulatory Surgical Centers (ASCs). According to FMI's estimates, the CDC (February 2025) updated SSI prevention strategies, placing renewed emphasis on effective wound drainage. Teleflex (February 2025) reported strong surgical revenue, validating the resilience of the elective surgery market. Liam Kelly emphasized: 'The benefits of our diversified portfolio were evident as strong performances from Interventional and Surgical helped offset softness in other areas.' Such financial resilience confirms that surgical consumables remain a stable revenue pillar even amidst broader economic fluctuations. Consequently, the region acts as a global proving ground where clinical drainage innovations are first commercialized before wider adoption.
What Strategic Moves Are Defining Competitive Landscape?
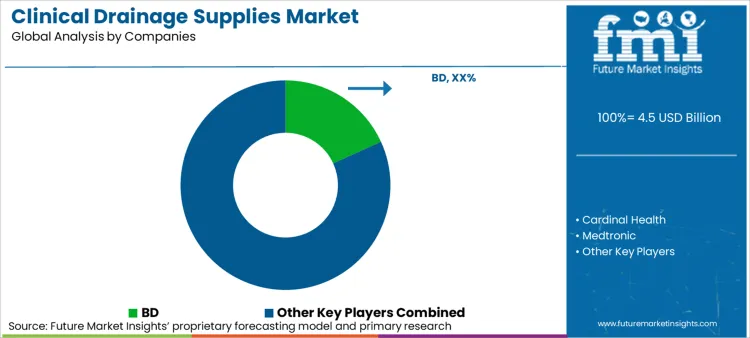
The current competitive environment is heavily influenced by the pursuit of leadership in disease-modifying therapies, alongside efforts to sustain traditional product lines. Analysis from FMI indicates a surge in mergers and acquisitions, as major pharmaceutical companies actively seek innovative pathways to enhance their amyloid treatment offerings. A notable instance of this trend is BD's acquisition of the Critical Care division of Edwards Lifesciences in September 2024, showcasing the industry's drive towards consolidation.
Key players such as BD, with an impressive 18.2% market share, alongside Medtronic and Teleflex, dominate the critical care sector. Their early entry into the market, combined with significant investments in research and development and well-established distribution networks, provides them with competitive advantages that are difficult for newer entrants to match. Companies like Getinge and Teleflex are leveraging early leads in immunotherapies to set standard-of-care, while competitors explore small molecules to establish differentiated positions. Patent expirations for legacy catheter products are forcing manufacturers to continuously innovate.
Key Players in Clinical Drainage Supplies Market
- BD (Becton, Dickinson)
- Medtronic
- Teleflex
- Getinge
- Cardinal Health
Market Definition
The clinical drainage supplies market represents revenue generated from medical devices and consumable systems used to remove fluids, air, or exudates from surgical sites or body cavities in clinical settings. As operationally defined in the article, the market measures commercial sales of drainage products deployed for post-operative care, infection control, and fluid management across surgical and critical care applications. Market sizing reflects product-level revenues from manufacturers supplying hospitals and other care settings, analysed by product type, clinical application, end user or sales channel, and region, and expressed in USD billion.
The market includes clinical drainage products explicitly segmented and quantified in the article, including closed wound suction drains, chest drainage systems, drainage catheters, reservoirs and suction bulbs, and related accessories. It covers use across general surgery, cardiothoracic surgery, orthopedic surgery, plastic and reconstructive surgery, and urology and gynecology procedures. Revenue generated through hospitals, ambulatory surgical centers (ASCs), clinics and outpatient departments, home care and post-acute settings, medical distributors, and e-commerce channels is included, with hospitals identified as the leading sales channel. The scope counts both passive and active drainage systems, including vacuum-assisted and digitally enabled drainage solutions, and encompasses sales across North America, Europe, East Asia, South Asia, Latin America, and the Middle East & Africa.
The market excludes revenues from surgical procedures, physician services, nursing care, and hospital stay costs. Wound care products not designed for drainage, such as dressings, sutures, sealants, or topical therapies, are not included. General critical care equipment unrelated to fluid drainage, such as monitors or ventilators, is excluded unless sold as part of a drainage system. Reimbursement payments, government healthcare spending, regulatory fees, and policy initiatives referenced contextually are not counted as market revenue. Upstream raw materials, manufacturing equipment, and logistics services are also outside the scope of the clinical drainage supplies market.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 4.5 billion |
| Product Type | Closed Wound Suction Drains, Chest Drainage Systems, Drainage Catheters, Reservoirs & Suction Bulbs, Accessories |
| Clinical Applications | General Surgery, Cardiothoracic Surgery, Orthopedic Surgery, Plastic & Reconstructive Surgery, Urology & Gynecology |
| End Users / Sales Channels | Hospitals, Ambulatory Surgical Centers (ASCs), Clinics & Outpatient Departments, Home Care & Post-acute, Medical Distributors, E-commerce |
| Regions Covered | North America, Europe, East Asia, South Asia, Latin America, Middle East & Africa |
| Countries Covered | United States, Germany, China, India, Brazil, and 40+ countries |
| Key Companies Profiled | BD (Becton, Dickinson), Medtronic, Teleflex, Getinge, Cardinal Health |
| Additional Attributes | Dollar sales by product and application, regulatory-driven adoption analysis (CDC Guidelines, NMPA approvals, Anvisa UDI), digital drainage system evolution, supply chain localization (Make in India), and competitive positioning analysis |
Source: FMI historical analysis and forecast data.
Clinical Drainage Supplies Market by Segments
By Product Type
- Closed Wound Suction Drains
- Chest Drainage Systems
- Drainage Catheters
- Reservoirs & Suction Bulbs
- Accessories & Connectors
By Clinical Applications
- General Surgery
- Cardiothoracic Surgery
- Orthopedic Surgery
- Plastic & Reconstructive Surgery
- Urology & Gynecology
By End Users / Sales Channels
- Hospitals
- Ambulatory Surgical Centers (ASCs)
- Clinics & Outpatient Departments
- Home Care & Post-acute
- Medical Distributors
- E-commerce / Retail Medical Supply
By Region
- North America
- Europe
- East Asia
- South Asia
- Latin America
- Middle East & Africa
Bibliography
- Centers for Disease Control and Prevention. (2024, April). Surgical site infection basics. USA Department of Health and Human Services.
- Centers for Disease Control and Prevention. (2025, February). Surgical site infection prevention: Clinical guidance for healthcare professionals. USA Department of Health and Human Services.
- Press Information Bureau. (2023, April). National medical devices policy. Government of India.
- National Medical Products Administration. (2025, November). Regulatory announcement on importation of pre-approval medical products. Government of the People’s Republic of China.
- Agência Nacional de Vigilância Sanitária. (2025, July). Launch of the unique device identification (UDI) system. Government of Brazil.
- Federal Institute for Drugs and Medical Devices. (2025, December). DMIDS database requirements for medical devices. Government of Germany.
Frequently Asked Questions
What is the current global market size for Clinical Drainage Supplies?
The global market is valued at USD 4.5 billion in 2026, driven by a post-pandemic recovery in surgical volumes and new infection control mandates.
What is the projected Compound Annual Growth Rate (CAGR) for Clinical Drainage Supplies market over the next 10 years?
The market is projected to grow at 6.9% CAGR from 2026 to 2036.
Which therapeutic areas are the primary drivers of demand?
Rising prevalence of chronic diseases requiring surgery and strict CDC guidelines on surgical site infection (SSI) prevention drive demand for active drainage systems.
What are the primary market risks or regulatory hurdles?
Regulatory bodies like Anvisa (Brazil) and NMPA (China) are enforcing stricter traceability and approval standards, creating compliance barriers for new entrants.
Who are the leading suppliers?
BD leads with an estimated 18.2% share, followed by Medtronic, Teleflex, Getinge, and Cardinal Health.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Clinical Drainage Supply Types
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Clinical Drainage Supply Types , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Clinical Drainage Supply Types , 2026 to 2036
- Closed wound suction drains (JP-type)
- Chest drainage systems
- Surgical wound drains (flat/round)
- Drainage catheters (IR/specialty)
- Reservoirs & suction bulbs
- Accessories & connectors
- Closed wound suction drains (JP-type)
- Y to o to Y Growth Trend Analysis By Clinical Drainage Supply Types , 2021 to 2025
- Absolute $ Opportunity Analysis By Clinical Drainage Supply Types , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Clinical Applications
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Clinical Applications, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Clinical Applications, 2026 to 2036
- General surgery
- Cardiothoracic surgery
- Orthopedic surgery
- Plastic & reconstructive surgery
- Urology & gynecology
- Other procedures
- General surgery
- Y to o to Y Growth Trend Analysis By Clinical Applications, 2021 to 2025
- Absolute $ Opportunity Analysis By Clinical Applications, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Users / Sales Channels
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Users / Sales Channels, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Users / Sales Channels, 2026 to 2036
- Hospitals
- ASCs
- Clinics & outpatient departments
- Home care & post-acute
- Medical distributors
- E-commerce / retail medical supply
- Hospitals
- Y to o to Y Growth Trend Analysis By End Users / Sales Channels, 2021 to 2025
- Absolute $ Opportunity Analysis By End Users / Sales Channels, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- By Country
- Market Attractiveness Analysis
- By Country
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Clinical Drainage Supply Types
- By Clinical Applications
- By End Users / Sales Channels
- Competition Analysis
- Competition Deep Dive
- BD
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Cardinal Health
- Medtronic
- Teleflex
- Getinge
- Others
- BD
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Clinical Drainage Supply Types , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Clinical Applications, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Clinical Drainage Supply Types , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Clinical Applications, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Clinical Drainage Supply Types , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Clinical Applications, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Clinical Drainage Supply Types , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Clinical Applications, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Clinical Drainage Supply Types , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Clinical Applications, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Clinical Drainage Supply Types , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Clinical Applications, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Clinical Drainage Supply Types , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Clinical Applications, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Clinical Drainage Supply Types , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Clinical Applications, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by End Users / Sales Channels, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Clinical Drainage Supply Types , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Clinical Drainage Supply Types , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Clinical Drainage Supply Types
- Figure 6: Global Market Value Share and BPS Analysis by Clinical Applications, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Clinical Applications, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Clinical Applications
- Figure 9: Global Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End Users / Sales Channels
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Clinical Drainage Supply Types , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Clinical Drainage Supply Types , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Clinical Drainage Supply Types
- Figure 26: North America Market Value Share and BPS Analysis by Clinical Applications, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Clinical Applications, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Clinical Applications
- Figure 29: North America Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by End Users / Sales Channels
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Clinical Drainage Supply Types , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Clinical Drainage Supply Types , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Clinical Drainage Supply Types
- Figure 36: Latin America Market Value Share and BPS Analysis by Clinical Applications, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Clinical Applications, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Clinical Applications
- Figure 39: Latin America Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End Users / Sales Channels
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Clinical Drainage Supply Types , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Clinical Drainage Supply Types , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Clinical Drainage Supply Types
- Figure 46: Western Europe Market Value Share and BPS Analysis by Clinical Applications, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Clinical Applications, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Clinical Applications
- Figure 49: Western Europe Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by End Users / Sales Channels
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Clinical Drainage Supply Types , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Clinical Drainage Supply Types , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Clinical Drainage Supply Types
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Clinical Applications, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Clinical Applications, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Clinical Applications
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by End Users / Sales Channels
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Clinical Drainage Supply Types , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Clinical Drainage Supply Types , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Clinical Drainage Supply Types
- Figure 66: East Asia Market Value Share and BPS Analysis by Clinical Applications, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Clinical Applications, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Clinical Applications
- Figure 69: East Asia Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by End Users / Sales Channels
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Clinical Drainage Supply Types , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Clinical Drainage Supply Types , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Clinical Drainage Supply Types
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Clinical Applications, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Clinical Applications, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Clinical Applications
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by End Users / Sales Channels
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Clinical Drainage Supply Types , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Clinical Drainage Supply Types , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Clinical Drainage Supply Types
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Clinical Applications, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Clinical Applications, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Clinical Applications
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by End Users / Sales Channels, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by End Users / Sales Channels, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by End Users / Sales Channels
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

