The clinical data provenance management market was valued at USD 285.4 million in 2025. The sector is set to reach USD 318.5 million in 2026 at a CAGR of 11.72% during the forecast period. Sustained investment propels the total opportunity to USD 965.2 million through 2036 as automated cryptographic tracking of real-world evidence transformations becomes a hard prerequisite for FDA regulatory submissions.
Replacing fragmented proprietary audit logs with standardized W3C PROV-based metadata ledgers forces biopharma trial sponsors to completely overhaul their evidence pipelines. Sponsors who delay this implementation lose multi-million-dollar trial approval timelines when regulatory bodies question the integrity of sourced claims data. While observers frequently focus on securing the initial point of data capture, the actual regulatory failure point occurs during intermediate transformations applied by third-party aggregators, requiring sophisticated clinical interoperability engines to maintain unbroken custody chains.
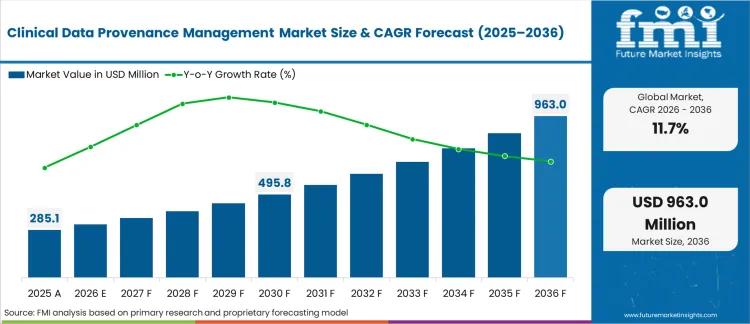
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 318.5 million |
| Industry Value (2036) | USD 965.2 million |
| CAGR (2026-2036) | 11.72% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Biopharma trial sponsors mandating cryptographic data lineage proofs from all tier-2 Contract Research Organizations before the 2028 operational cutoff forms the primary market inflection. CROs must embed automated data tracking directly into their clinical workflows to retain sponsor contracts, initiating a self-sustaining cycle of provenance standardization across the entire life sciences supply chain.
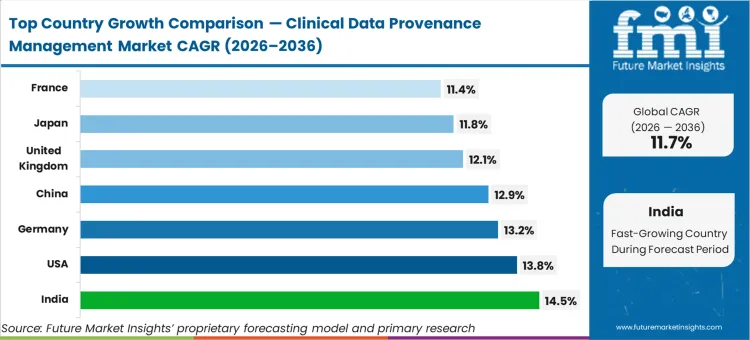
India tracks a 14.5% compound rate, followed by the United States expanding at 13.8%, Germany at 13.2%, China at 12.9%, the United Kingdom at 12.1%, Japan at 11.8%, and France recording 11.4%. The Central Drugs Standard Control Organisation, enforcing strict digital audit trail prerequisites for local sites attempting to participate in global multi-regional clinical trials, accelerates India's adoption relative to established Western markets. This spread exists because emerging regions are leapfrogging legacy systems to deploy modern cryptographic tracking as a baseline qualification for international trial revenue.
The Clinical Data Provenance Management Market encompasses the software platforms and integration services designed to mathematically and chronologically track the origin, ownership, and transformation history of healthcare data. This market distinguishes itself from standard cybersecurity or access control by focusing strictly on the unbroken chain of custody and lineage validation of medical information as it moves between disparate institutional silos, aggregators, and regulatory bodies.
Scope includes cryptographic metadata tracking engines, W3C PROV-compliant lineage visualization tools, blockchain-anchored audit software, and API connectors designed to link electronic data capture systems with regulatory submission portals. Implementation consulting, system validation services aligned with 21 CFR Part 11, and managed provenance-as-a-service platforms targeting trial sponsors and CROs are formally incorporated within the boundary of this market.
General hospital information systems, basic electronic health records devoid of cryptographic lineage tracking, and perimeter cybersecurity tools like firewalls and endpoint protection are explicitly excluded. These adjacent technologies manage daily clinical workflows or defend network perimeters but lack the specific mathematical proof-of-transformation capabilities required to validate data integrity for formal regulatory evidence submissions.
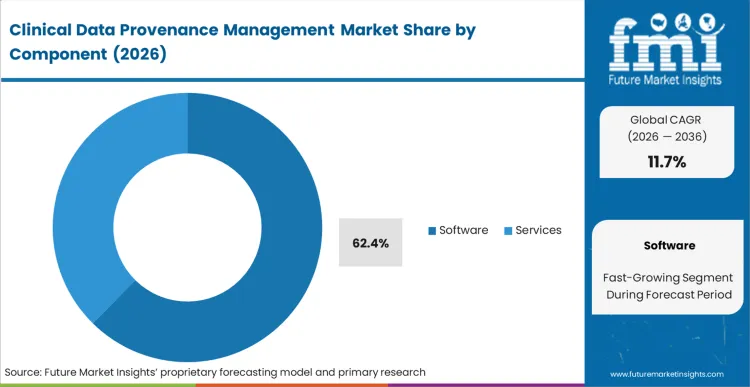
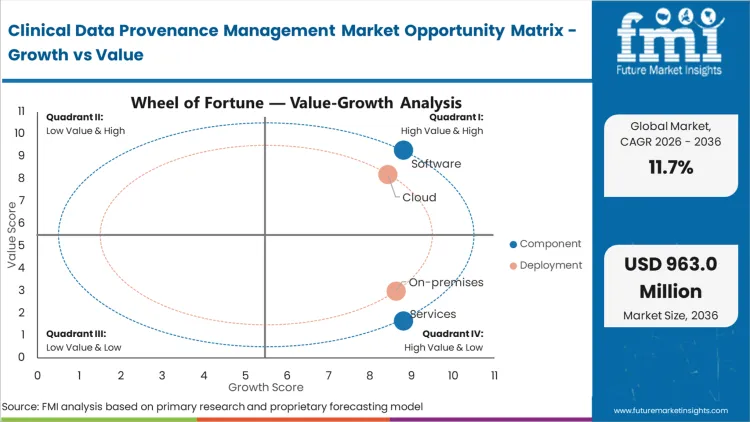
Replacing manual compliance reviews with algorithmic auditing tools exposes the severe scalability limits of service-based consulting. Software holds a dominant 62.4% share in 2026 because the sheer velocity of data transformations in modern trials physically exceeds human auditing capacity. According to FMI's estimates, biopharma data managers mandate standalone software engines to guarantee continuous, immutable tracking without constant consulting intervention. Service providers are increasingly pivoting to integration and validation support, acknowledging that the core tracking mechanism must be programmatically embedded via healthcare api integration rather than manually applied. Trial sponsors utilizing legacy consulting-led audits face unacceptable regulatory delays when attempting to map millions of concurrent data transformation events.
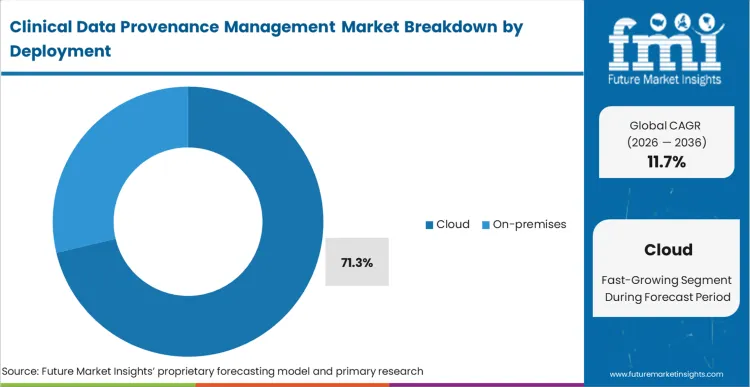
Cloud deployment holds a 71.3% share in 2026, fundamentally driven by the multi-institutional reality of modern clinical research. The necessity to securely track assets moving from hospital networks to CROs and ultimately to sponsors makes localized healthcare cloud infrastructure highly efficient for cross-boundary lineage tracking. Based on FMI's assessment, CRO IT directors prioritise cloud-native provenance engines because they provide a centralized, immutable ledger accessible to all authorised trial participants simultaneously. Organizations resisting cloud-based lineage tracking encounter massive friction when attempting to synchronize audit trails with external trial partners, jeopardizing timeline deliverables.
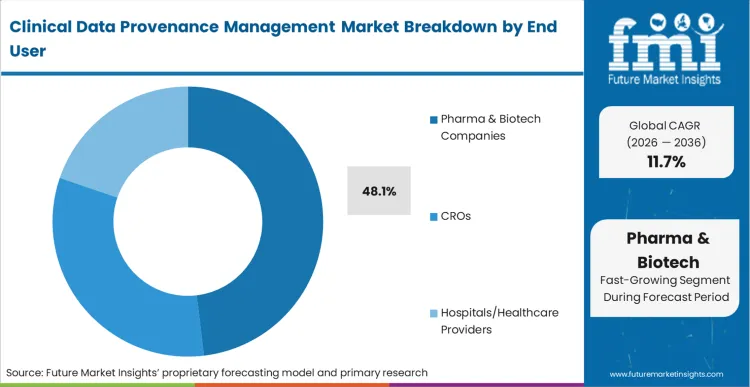
The regulatory burden of final drug approval submissions forces Pharma & Biotech Companies to assume ultimate liability for data integrity. This liability dynamic secures their position as the leading end user with 48.1% share in 2026. As the FDA increases scrutiny on clinical trial management utilizing real-world evidence, sponsor-level compliance directors cannot offshore the risk of data contamination to their vendors.
In FMI's view, sponsors must enforce provenance tracking from the top down, purchasing enterprise-wide software licenses that they require all subordinate CROs to utilize. Sponsors failing to implement sponsor-controlled lineage platforms risk catastrophic regulatory rejections if an outsourced vendor alters data without a verifiable audit trail.
The FDA enforcement of 21 CFR Part 11 and its finalizing of guidance for Real-World Data usage mandate mathematical proof of data integrity for regulatory submissions. Biopharma trial sponsors are forced to implement unbroken custody chains to prove that observational data extracted from hospital records has not been manipulated during aggregation. This direct regulatory pressure drives rapid market expansion as legacy, siloed audit logs fail to meet the new cross-institutional tracking standards. Sponsors who fail to upgrade their provenance infrastructure will see their clinical trial submissions outright rejected due to unverifiable evidence sources.
The primary operational friction involves integrating modern cryptographic ledgers with decades-old, proprietary hospital Electronic Health Record systems that lack native API export capabilities. Data engineers face massive technical debt when attempting to pull clean, time-stamped metadata from these legacy environments. While middleware workarounds are emerging, these temporary patches have structural limits regarding scalability and latency, creating significant integration bottlenecks for multiregional trial rollouts.
Based on the regional analysis, the Clinical Data Provenance Management market is segmented into North America, Latin America, Europe, East Asia, South Asia & Pacific, and Middle East & Africa across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 14.5% |
| United States | 13.8% |
| Germany | 13.2% |
| China | 12.9% |
| United Kingdom | 12.1% |
| Japan | 11.8% |
| France | 11.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
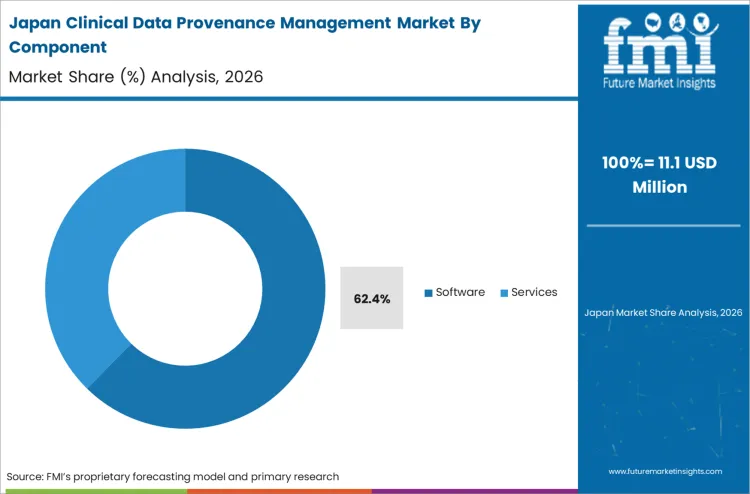
Specific cost structure pressures and the desire to capture high-margin international clinical trial revenue drive the Asia Pacific transition. The region is aggressively modernizing its clinical infrastructure to present itself as a compliant, cost-effective destination for global biopharma sponsors. By leapfrogging legacy systems and immediately deploying advanced cryptographic tracking, these markets are removing the primary barrier to Western trial investment, which relies entirely on verifiable data trust. As per FMI's projection, eclinical platform deployments across this region are structurally different because they are built explicitly to satisfy foreign FDA/EMA audits rather than internal domestic requirements.
FMI's report includes comprehensive evaluation of South Korea, Australia, and the broader ASEAN region. These markets exhibit a rapid consolidation of clinical IT vendors as smaller players fail to meet the new international data standards.
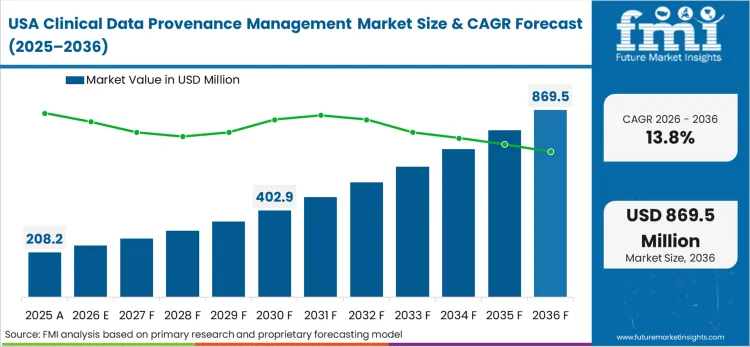
The FDA explicit guidance on Real-World Data acts as the primary forcing function in North America, completely redefining how observational evidence is evaluated. This policy-led environment shifts the burden of proof entirely onto the biopharma sponsors, demanding mathematical certainty regarding the origin and handling of every data point. FMI analysts opine that North America leads in overall volume because the majority of Tier-1 trial sponsors are headquartered here, dictating the technology standards for their global operations.
FMI's report includes detailed analysis of Canada. Canadian provincial health networks are increasingly deploying provenance tools to safely monetise their centralized patient databases for cross-border research.
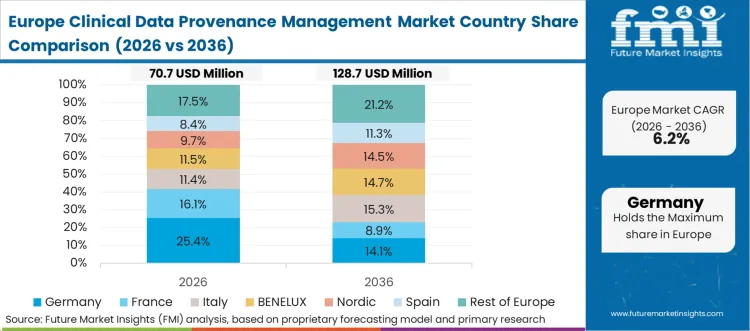
Specific physical and digital infrastructure constraints, namely the highly fragmented nature of European national health systems overlaid with strict GDPR requirements, mandate complex cross-border tracking solutions. The infrastructure must simultaneously prove data integrity for the European Medicines Agency while strictly masking patient identity across sovereign borders. Based on FMI's assessment, Europe's dynamic is heavily focused on privacy-preserving provenance protocols rather than raw data integration speed.
FMI's report includes coverage of Italy, Spain, and the Nordics. The Nordic region specifically leads in integrating national civic registries directly with clinical trial tracking platforms.
The clinical data provenance management market is highly concentrated among a few major life sciences technology providers. This consolidation exists because Tier-1 biopharma sponsors require global scalability and deeply validated, pre-built connectors to existing Electronic Data Capture systems. Leading companies like Medidata and Oracle leverage their massive installed base of trial software to upsell provenance modules directly into existing workflows. Buyers utilize the breadth of a vendor's out-of-the-box API library as the primary competitive variable to distinguish qualified enterprise platforms from niche tracking startups.
Companies like Datavant and IQVIA possess distinct structural advantages due to their massive pre-existing networks of institutional data partnerships. A challenger attempting to replicate this advantage must spend years negotiating individual data-sharing and validation agreements with thousands of disparate hospital networks. Innovators embedding zero-knowledge proof verification into digital health records offer a technical edge, but challengers must partner with established data aggregators rather than attempt a direct rip-and-replace of incumbent EDC systems.
To prevent vendor lock-in, large biopharma buyers increasingly mandate adherence to open standards like HL7 FHIR and W3C PROV. This structural tension limits the pricing power of dominant vendors, as buyers refuse to store their critical regulatory audit trails in proprietary, closed-loop formats. As the market approaches 2036, the competitive trajectory points toward slight fragmentation in the aggregation layer, as specialised, standard-compliant interoperability startups carve out profitable niches linking legacy hospital systems to the major trial platforms.

| Metric | Value |
|---|---|
| Quantitative Units | USD 318.5 million to USD 965.2 million, at a CAGR of 11.72% |
| Market Definition | Software platforms and integration services designed to mathematically and chronologically track the origin, ownership, and transformation history of healthcare data for regulatory validation. |
| Component Segmentation | Software, Services |
| Deployment Segmentation | Cloud, On-premises |
| End User Segmentation | Pharma & Biotech Companies, CROs, Hospitals/Healthcare Providers |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia & Pacific, Middle East & Africa |
| Countries Covered | India, United States, Germany, China, United Kingdom, Japan, France, and 40 plus countries |
| Key Companies Profiled | IBM, Oracle, Veeva Systems, Medidata (Dassault Systemes), IQVIA, Datavant, BurstIQ, Guardtime |
| Forecast Period | 2026 to 2036 |
| Approach | Primary interviews with Clinical Data Managers and Biopharma IT Compliance Officers. Baseline volumes derived from global trial registrations. Financial projections validated through enterprise vendor licensing disclosures. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
How large is the Clinical Data Provenance Management Market in 2026?
Industry size is projected to reach USD 318.5 million in 2026, driven by biopharma trial sponsors moving away from manual audit logs to meet stringent FDA 21 CFR Part 11 requirements.
What will it be valued at by 2036?
The valuation is expected to reach USD 965.2 million by 2036 as automated cryptographic tracking of real-world evidence transformations becomes a hard prerequisite for FDA regulatory submissions.
What CAGR is projected?
A CAGR of 11.72% is projected, which remains defensible due to the structural liability trial sponsors face regarding data contamination from third-party aggregators.
Which Component segment leads?
Software holds a dominant 62.4% share in 2026 because the sheer velocity of data transformations in modern trials physically exceeds human auditing capacity, demanding algorithmic tracking tools.
Which Deployment segment leads?
Cloud deployment captures 71.3% share because cross-institutional lineage tracking fundamentally requires decentralized, multi-party access to an immutable master ledger.
Which End User segment leads?
Pharma & Biotech Companies maintain 48.1% share as these entities bear the ultimate financial and regulatory liability for final drug approval submissions, forcing them to dictate technology standards top-down.
What drives rapid growth?
FDA real-world data guidances force trial sponsors to mathematically prove the integrity of external data, while hospital networks deploy tagging tools to legally monetize their de-identified patient data.
What is the primary restraint?
Integrating modern cryptographic ledgers with legacy, proprietary hospital electronic health records that lack native API export capabilities creates massive technical debt and slows multiregional trial deployments.
Which country grows fastest?
India tracks a 14.5% compound rate as the Central Drugs Standard Control Organisation enforces strict digital audit trail prerequisites for local sites attempting to participate in lucrative international trials.
How does FDA 21 CFR Part 11 impact vendor selection?
The regulation requires biopharma IT compliance officers to strictly evaluate vendors based on their ability to provide irrefutable, time-stamped, and tamper-evident digital records without human intervention.
Why are fragmented audit logs failing?
Legacy, siloed audit logs lose visibility the moment data crosses an institutional boundary, failing to track the intermediate transformations applied by third-party data aggregators before regulatory submission.
How do Medidata and Oracle maintain market dominance?
Top incumbents compete heavily on their massive installed base of trial software, upselling provenance modules directly into existing workflows using highly validated, pre-built API connectors.
How does the UK NHS infrastructure program affect local deployment?
The NHS centralized push to leverage patient databases for commercial research compels all downstream research partners to utilize specific, pre-approved tracking APIs to access the data.
Why is Germany's growth structurally different?
Germany's localized health data infrastructure requires highly specific cryptographic tools to connect regional hospital clusters without centralizing the data physically, favoring specialized regional software vendors.
What is the role of CROs in the adoption curve?
Contract research organizations act as the implementation layer, purchasing API-based provenance engines primarily to fulfill sponsor SLA demands and secure enterprise trial contracts.
How do self-describing data assets change the operational model?
Engineering data payloads to carry their own immutable metadata history eliminates the need for parallel audit databases, drastically reducing the computational overhead required for continuous lineage tracking.
What commercial risk do hospitals face if they ignore provenance standards?
Medical informatics directors who fail to deploy verifiable data lineage protocols face the total exclusion of their patient datasets from lucrative commercial real-world evidence licensing markets.
How does China NMPA alignment impact software procurement?
China's push to align NMPA standards with ICH guidelines requires local biopharma companies to prove data lineage for export applications, driving rapid enterprise software licensing among Chinese CROs.
Why are open standards like HL7 FHIR critical to buyers?
Large biopharma buyers increasingly mandate adherence to open standards to prevent vendor lock-in, refusing to store their critical regulatory audit trails in proprietary, closed-loop formats.
How do zero-knowledge proofs function in this space?
Advanced cryptographic protocols allow data transformation history to be verified by regulatory bodies without exposing the underlying Protected Health Information to unauthorized external auditors.
What role does consulting play in the future market?
Service providers are rapidly pivoting away from manual compliance reviews toward specialized integration and system validation support, ensuring the deployed software meets regulatory parameters.
Why is on-premises deployment losing share?
On-premises solutions struggle to securely synchronize metadata tracking across multiple external institutional boundaries, limiting their use to highly isolated, single-facility biobanks with extreme data sovereignty constraints.
How is the market validated by FMI?
Revenue models are built upon software-as-a-service pricing tiers mapped against global trial volumes, cross-referenced against enterprise software licensing contracts announced by top-tier biopharma sponsors.
Are perimeter cybersecurity tools included in this report?
General hospital information systems and perimeter cybersecurity tools like firewalls are explicitly excluded, as they lack the specific mathematical proof-of-transformation capabilities required for formal regulatory evidence.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.