Clinical AI Model Governance Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
The Clinical AI Model Governance Market is segmented by Component (Solutions, Services), Deployment (On-Premises, Cloud), Functionality (Risk & Compliance, Model Lifecycle Management, Monitoring & Auditing, Ethics & Responsible AI), and Region. This forecast models demand from 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Clinical AI Model Governance Market Size, Market Forecast and Outlook By FMI
In 2025, the clinical AI model governance market was valued at USD 1.77 billion. Based on Future Market Insights’ analysis, demand for clinical AI model governance is estimated to increase to USD 2.48 billion in 2026 and USD 71.12 billion by 2036, reaching a CAGR of 39.9% during the forecast period.
Regulatory scrutiny over machine learning opacity fundamentally alters healthcare procurement strategies. Hospital IT architects refuse to authorise deep-learning diagnostic deployments without native tracing mechanisms that can explain individual patient predictions. Integrating enterprise ai governance and compliance platforms directly into the clinical data pipeline provides the necessary audit trails for external reviewers. Platform developers targeting these massive healthcare networks secure long-term recurring revenue by solving the exact validation bottleneck that previously stalled the adoption of artificial intelligence in the pilot phase.
Summary of Clinical AI Model Governance Market
- Clinical AI Model Governance Market Definition
- Clinical AI model governance represents the structured frameworks, software platforms, and validation protocols required to monitor, audit, and explain artificial intelligence algorithms deployed within healthcare settings.
- Demand Drivers in the Market
- Algorithmic drift within diagnostic models forces chief medical informatics officers to implement continuous performance monitoring systems.
- Evolving regulatory frameworks for medical devices compel hospital compliance teams to mandate auditable decision trees for all patient-facing applications.
- Liability concerns surrounding biased triage algorithms push healthcare network administrators to adopt independent validation software before clinical deployment.
- Key Segments Analysed in the FMI Report
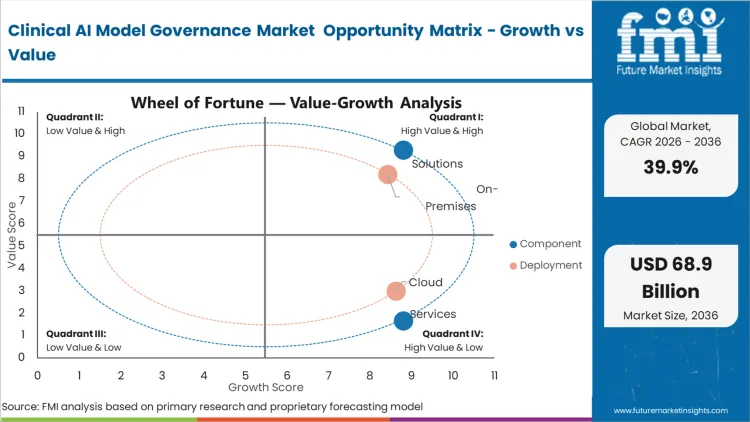
- Solutions: 67.48% share, reflecting the capital shift toward automated platform licenses rather than manual consulting engagements for algorithmic auditing.
- On-Premises: 51.4% share, resulting from strict patient data sovereignty laws that prevent hospital networks from exporting protected health information to public cloud evaluation environments.
- Risk & Compliance: 40% share, driven entirely by the necessity to meet federal medical device reporting standards before commercialising predictive healthcare applications.
- Analyst Opinion at FMI
- Sabyasachi Ghosh, Principal Consultant for Healthcare, opines: "Clinical informatics directors operate under a fundamental tension between innovation and medical liability. Algorithms demonstrating high accuracy in retrospective testing frequently exhibit dangerous performance drift when exposed to live, diverse patient populations. Hospital administrators who deploy predictive diagnostic models without automated governance frameworks face severe regulatory penalties when those algorithms inevitably degrade. Platforms that embed continuous statistical monitoring directly into the clinical workflow will capture the dominant share of hospital IT budgets over the next procurement cycle."
- Strategic Implications / Executive Takeaways
- Governance software developers must integrate native compliance reporting templates for global medical device regulatory bodies.
- Healthcare IT procurement directors require mandatory interoperability testing between governance platforms and legacy electronic health record systems.
- Clinical AI architects must prioritise local processing capabilities to satisfy data residency requirements while performing algorithmic auditing.
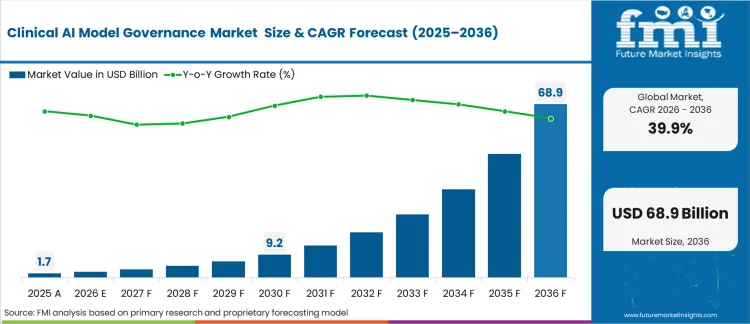
Clinical AI Model Governance Market Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 2.48 Billion |
| Industry Value (2036) | USD 71.12 Billion |
| CAGR (2026-2036) | 39.9% |
Positive growth trajectory mapped in the forecasting model stems directly from an impending regulatory cliff across major healthcare jurisdictions. Regional health authorities are rapidly transitioning from issuing voluntary artificial intelligence guidelines to enforcing mandatory algorithmic auditing requirements with strict financial penalties for non-compliance. Maturing clinical ecosystems now treat model oversight platforms as critical infrastructure rather than auxiliary software. This regulatory maturation establishes the foundation for explosive adoption rates across key global markets.
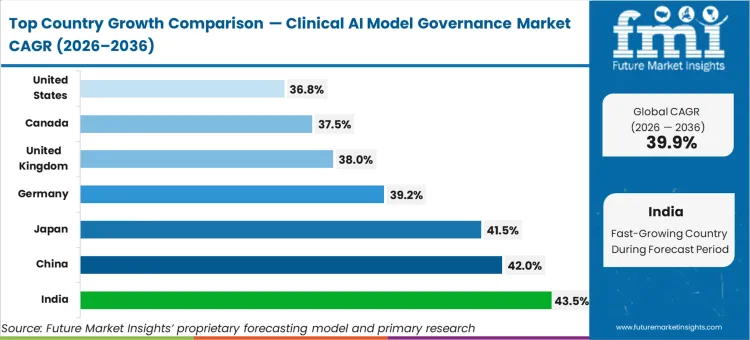
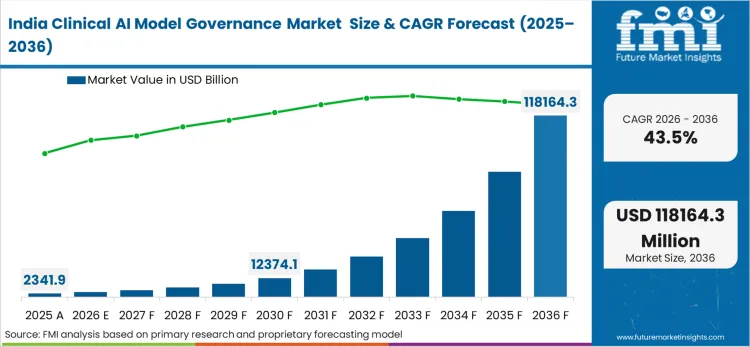
Geographic differences in data‑privacy legislation now shape the growth pace of clinical AI governance platforms, with Asia Pacific markets, led by India at 43.5%, China at 42.0%, and Japan at 41.5% CAGR, expanding fastest due to large‑scale digital‑health modernisation. Europe follows, anchored by Germany at 39.2% and the UK at 38.0% CAGR, where strict privacy mandates demand fully transparent auditing. In North America, Canada, at 37.5% and the United States, at 36.84% CAGR, prioritise certified compliance architectures to secure vendor eligibility across regulated healthcare networks. As these regional forces converge, hospitals increasingly view governance systems as foundational to future‑proof clinical innovation.
Clinical AI Model Governance Market Definition
Clinical AI model governance encompasses the dedicated software solutions, auditing tools, and operational frameworks utilised by healthcare organisations to monitor the performance, fairness, and regulatory compliance of artificial intelligence algorithms. Key functional areas include detecting data drift in diagnostic imaging models, tracking demographic bias in patient triage systems, and generating automated audit logs for medical device regulators. Independent algorithmic validation platforms that interface directly with electronic health records are fully included.
Clinical AI Model Governance Market Inclusions
The market scope includes dedicated algorithmic auditing platforms, bias detection software, model lifecycle management suites, and automated regulatory reporting tools designed specifically for healthcare environments. Software modules that monitor real-time inference data against baseline training datasets fall within the boundaries. Continuous validation tools applied to healthcare ai computer vision applications represent a core inclusion within the analytical framework.
Clinical AI Model Governance Market Exclusions
General-purpose enterprise governance platforms lacking specific healthcare regulatory compliance modules are excluded. Basic hospital IT security infrastructure, data storage hardware, and generalised clinical decision support systems that do not employ machine learning are omitted from the valuation. Manual consulting services that do not involve the deployment of automated software monitoring tools fall outside the defined parameters.
Clinical AI Model Governance Market Research Methodology
- Primary Research: FMI analysts engaged directly with clinical informatics directors and regulatory affairs leads at Tier-1 hospital networks to quantify internal budgets allocated to algorithmic oversight.
- Desk Research: The data collection phase aggregated compliance roadmaps from the FDA Digital Health Center of Excellence and the European Medicines Agency.
- Market-Sizing and Forecasting: The baseline value derives from scaling the broader healthcare artificial intelligence sector, isolating the specific software expenditure dedicated to model monitoring and compliance.
- Data Validation and Update Cycle: Projections are rigorously tested against publicly reported capital expenditure guidance from leading clinical software platform providers.
Segmental Analysis
Clinical AI Model Governance Market Analysis by Component
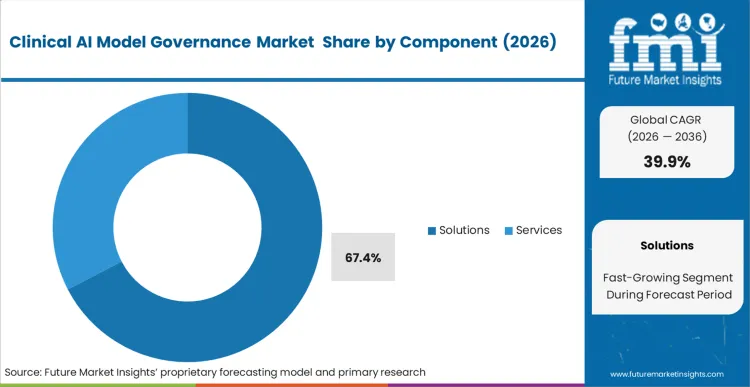
With algorithmic opacity threatening patient safety protocols, hospital network administrators execute full-scale integration of automated oversight platforms. Solutions command 67.48% share in 2026, reflecting the absolute requirement for continuous, software-driven monitoring over episodic manual audits. Clinical IT teams deploying these centralized platforms establish the foundation for scaling multiple diagnostic models safely. According to FMI's estimates, automated tracking reduces the administrative burden of regulatory reporting by digitizing the entire compliance workflow. Software vendors unable to supply certified, interoperable oversight platforms forfeit their position in early-stage vendor shortlisting processes.
- Continuous validation: Automated tracking algorithms constantly compare live patient data inferences against initial baseline training parameters to detect silent model degradation.
- Audit trail generation: Centralized reporting modules automatically compile comprehensive decision logs required by federal health authorities during unannounced regulatory inspections.
- Bias detection metrics: Statistical evaluation engines actively screen triage recommendations across diverse patient demographic groups to prevent systemic care disparities.
Clinical AI Model Governance Market Analysis by Deployment
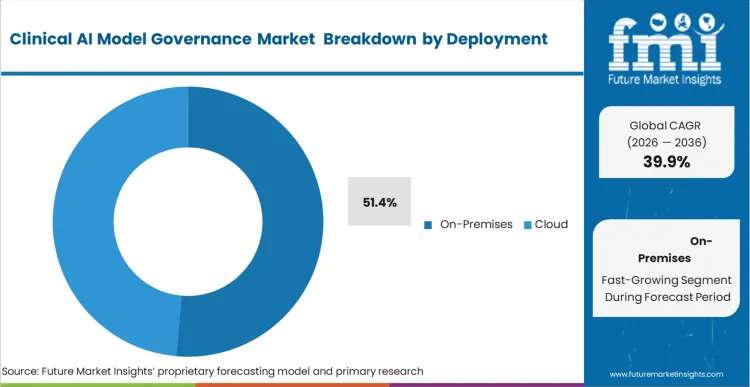
Every hospital IT director bidding on facility modernisation contracts now faces strict adherence criteria regarding patient data sovereignty. On-Premises deployment accounts for 51.4% segment share in 2026. Clinical compliance officers operating within highly regulated environments reject cloud-based evaluation designs that transmit protected health information to external servers. Incorporating localized validation infrastructure guarantees that sensitive patient telemetry remains secured behind the hospital firewall during algorithmic auditing. Software providers failing to demonstrate robust local processing capabilities lose priority status in critical clinical upgrade cycles.
- Data sovereignty: Keeping algorithm validation data on local servers ensures absolute compliance with strict national and regional health privacy legislation.
- Latency reduction: Processing inference checks directly at the hospital edge minimizes the delay in returning critical diagnostic support to the attending physician.
- Network isolation: Air-gapped monitoring environments protect the core hospital infrastructure from potential external vulnerabilities introduced through third-party cloud connections.
Clinical AI Model Governance Market Analysis by Functionality
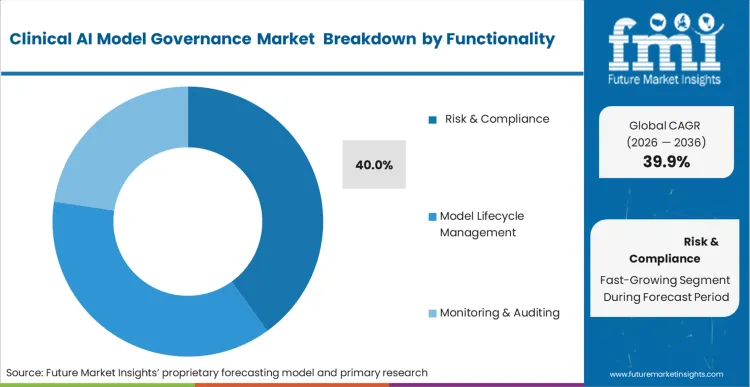
Evolving medical device frameworks from global health authorities dictate the structural priorities for healthcare technology investments. Risk & Compliance functionality captures 40% of the market share in 2026 by solving the exact reporting bottleneck that stalls algorithm commercialisation. Regulatory affairs directors use these compliance modules to automatically format the complex documentation needed for medical device approvals, eliminating manual data errors. Precise documentation of model performance parameters enables rapid clearance when clinical networks submit novel predictive tools for approval. Facilities operating without this foundational compliance architecture face systemic commercialization delays as regulatory bodies reject undocumented algorithmic submissions.
- Regulatory alignment: Automated templates format performance data to match the exact submission criteria demanded by medical device oversight committees.
- Liability mitigation: Comprehensive risk scoring algorithms quantify the potential diagnostic error rates before clinical deployment, informing critical insurance and liability models.
- Change control tracking: Documentation engines precisely record every weight adjustment and parameter shift in continuously learning models to satisfy pre-determined change control requirements.
Clinical AI Model Governance Market Drivers, Restraints, and Opportunities
The convergence of predictive diagnostics and stringent medical device regulations forces chief medical informatics officers to deploy independent validation software. This architectural requirement renders manual spreadsheet-based auditing obsolete. Hospital administrators integrating artificial intelligence in healthcare applications face a strict binary choice between deploying comprehensive governance tools or accepting massive regulatory liability. Transitioning to a unified oversight backbone simplifies reporting topologies and enables safe clinical scaling. Clinical networks that fail to modernize their auditing layers risk severe financial penalties and immediate suspension of their predictive tools.
Security configuration parameters required to interface modern governance platforms with highly customized, legacy electronic health records create steep technical barriers for hospital IT teams. Extracting standardized inference data from fragmented clinical databases demands specialized data architecture expertise that most regional health networks lack internally. To mitigate this integration friction, clinical informatics directors increasingly rely on API-driven middleware solutions that standardize the data pipeline between the core health record and the independent algorithmic auditing platform.
- Predictive Maintenance Monitoring: Integrating governance tracking into imaging equipment allows clinical engineers to monitor the performance of embedded diagnostic algorithms alongside hardware degradation. Software vendors providing unified hardware and algorithm monitoring suites capture expanded hospital maintenance budgets.
- Distributed Clinical Trials: Regulatory sponsors require immutable proof of algorithmic integrity when deploying predictive patient selection tools across multiple geographic sites. Integrating ai based clinical trials solution provider governance modules ensures cross-site data consistency.
- Federated Learning Oversight: Privacy-preserving collaborative model training requires specialized governance tools to audit the weight updates shared between cooperating hospitals. Decentralized validation frameworks unlock massive data pooling opportunities without violating patient privacy laws.
Regional Analysis
Based on the regional analysis, the Clinical AI Model Governance market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania and the Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 43.5% |
| China | 42.0% |
| Japan | 41.5% |
| Germany | 39.2% |
| United Kingdom | 38.0% |
| Canada | 37.5% |
| United States | 36.84% |
Source: FMI analysis based on primary research and proprietary forecasting model
Asia Pacific Clinical AI Model Governance Market Analysis
Rapid digitisation of the national healthcare infrastructure across the Asia Pacific region accelerates the bypass of legacy manual auditing constraints. Capital projects directors constructing new digital-first hospital networks specify unified algorithmic governance platforms in their initial IT blueprints. By building native monitoring capabilities into the foundation, regional health authorities establish highly flexible data environments capable of safely deploying advanced predictive tools. This aggressive modernisation strategy directly fuels the demand for high-performance clinical software solutions. The specific healthcare priorities of individual nations dictate the precise implementation parameters for these oversight technologies.
- China: China's centralized healthcare strategy deploys massive predictive population health models requiring seamless performance tracking across thousands of provincial clinics. Clinical data architects bypass localized auditing protocols, specifying national-level governance platforms for all new diagnostic procurement contracts. The sheer volume of synthetic diagnostic data generated by urban research hospitals requires automated oversight engines to filter algorithmic anomalies before national deployment. This aggressive clinical modernization strategy drives a 42.0% CAGR for algorithmic governance infrastructure in China through 2036. Vendors unable to demonstrate verified high-volume processing capabilities lose access to the most lucrative state-backed hospital projects.
- India: India's rapidly expanding private hospital networks operate under increasing pressure to achieve international clinical accreditation standards to support medical tourism. This standardization pressure anchors India's 43.5% compound expansion of algorithmic oversight demand to 2036. As a result, chief medical informatics officers must deploy auditing networks capable of guaranteeing diagnostic consistency across multiple geographic locations. The push toward utilizing predictive triage algorithms in resource‑constrained clinics further triggers a wave of IT upgrades prioritizing bias‑detection mechanisms. Suppliers failing to provide platforms with built‑in demographic fairness metrics face disqualification from major private healthcare facility tenders.
- Japan: This continuous qualification constraint sustains a 41.5% CAGR for governance software upgrades in Japan's healthcare corridor. Japan's aging demographic crisis forces rapid adoption of predictive care algorithms, driving strict national guidelines for artificial‑intelligence reliability in elder‑care facilities. Every new robotic or predictive care architecture must validate performance consistency before receiving authorization for clinical integration. The domestic medical technology sector embeds precision‑monitoring software directly into diagnostic hardware to maintain public trust in automated clinical decisions. Component suppliers lacking verified interoperability‑testing records forfeit access to the next generation of subsidized clinical‑automation contracts.
FMI's report includes extensive coverage of the Asia Pacific clinical governance landscape. It incorporates a detailed analysis of Australia, South Korea, and the broader ASEAN region. A primary trend shaping these nations is the rapid establishment of national artificial intelligence ethics committees, forcing hospital administrators to deploy transparent auditing architectures to satisfy stringent governmental oversight requirements before launching any clinical algorithms.
North America Clinical AI Model Governance Market Analysis
Healthcare modernization mandates across North America target the systematic eradication of opaque clinical decision support systems. Regulatory affairs leads guiding hospital IT overhauls face strict federal directives to validate algorithmic safety across diverse patient populations. Implementing a standardized auditing backbone enables clinical networks to deploy advanced predictive diagnostics without compromising patient safety protocols. This strategic shift requires significant capital allocation toward robust software platforms capable of isolating and explaining individual algorithmic inferences. National regulatory frameworks and medical liability standards govern the exact specifications required for these critical software deployments.
- United States: The United States medical device regulatory landscape enforces strict pre-determined change control plans for any continuously learning diagnostic algorithm. Healthcare compliance officers managing these deployments must document every model weight adjustment to avoid mandatory recall notices from federal health authorities. National hospital networks actively restrict predictive patient triage models to shadow-mode operation until automated drift detection platforms are fully integrated into the clinical workflow. This regulatory qualification barrier sustains a 36.84% compound annual growth rate for algorithmic oversight solutions in the United States through 2036. Software providers failing to integrate native FDA-compliant audit logging directly into their platforms lose their approved vendor status before the next major hospital procurement cycle opens.
- Canada: Canada expands at a 37.5% annual growth rate for governance software infrastructure through 2036 as provincial authorities systematically upgrade their clinical oversight capabilities. The provincial healthcare systems require immutable proof of algorithmic fairness before funding the deployment of artificial intelligence triage solutions across public clinics. Regional health IT directors must demonstrate that predictive models do not exhibit demographic bias against indigenous or remote populations. The integration of centralized health data repositories necessitates continuous independent validation to ensure model accuracy across vastly different geographic care settings. Systems integrators unable to demonstrate absolute demographic fairness tracking forfeit qualification for national digital health expansion projects.
FMI's report includes comprehensive evaluation of the North American clinical algorithmic oversight sector. It features specific analysis of the Mexican digital health market. A defining dynamic involves the integration of cross-border telemedicine platforms, which requires standardized governance protocols to coordinate diagnostic consistency and maintain synchronized algorithmic performance standards across multiple international clinical facilities.
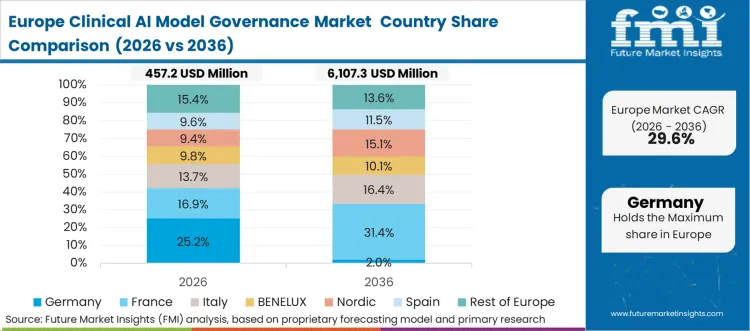
Europe Clinical AI Model Governance Market Analysis
European legislative frameworks actively penalize the deployment of opaque algorithms within critical civic and healthcare infrastructure. Clinical IT directors redesigning patient pathways must integrate continuous fairness monitoring data alongside standard diagnostic performance metrics. This dual-purpose reporting requirement forces the rapid adoption of advanced auditing protocols to guarantee model stability while adhering to strict continental data privacy laws. Upgrading the core software infrastructure provides the necessary transparency to support advanced predictive care practices. Hospital administrators recognize that maintaining undocumented algorithms severely limits their ability to operate in highly regulated European medical markets.
- Germany: Germany's digital health application framework dictates stringent validation criteria for reimbursement approval of predictive diagnostic tools. Medical software engineers must demonstrate flawless execution of continuous performance tracking before receiving authorization to connect new algorithms to the national health infrastructure. The domestic medical device export sector operates under rigorous functional safety certifications that mandate unified diagnostic layers, creating a recurring compliance cycle that accelerates software replacement. This systemic qualification pressure drives a 39.2% CAGR for certified auditing platforms in Germany as the standard proliferates. Software infrastructure providers targeting domestic hospital retooling projects must guarantee comprehensive audit trail generation prior to the finalization of capital procurement budgets.
- United Kingdom: The United Kingdom's national health system demands precise clinical validation data to safely integrate diagnostic algorithms across regional trusts. Trust IT planners deploying advanced triage systems mandate software architectures capable of real-time bias detection across distributed demographic datasets. The shift toward decentralized diagnostic services forces health operators to abandon isolated evaluation methods in favor of highly resilient, continuous monitoring networks. This critical infrastructure transition anchors the UK's 38.0% compound trajectory to 2036 as regional authorities systematically upgrade their operational backbones. Developers unable to demonstrate absolute algorithmic transparency under severe clinical load conditions forfeit qualification for national diagnostic expansion programs.
FMI's report includes thorough investigation of the European clinical governance framework. The analysis encompasses France, Italy, Spain, and the Nordics. A prevailing structural condition across these nations is the mandatory compliance with the overarching continental artificial intelligence legislative act, forcing healthcare providers to specify governance platforms that automatically categorize clinical algorithms by risk tier and generate the mandated external compliance reports.
Competitive Aligners for Market Players
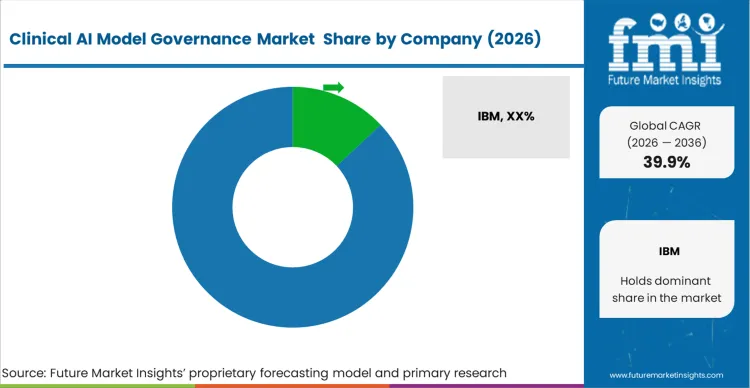
The introduction of rigorous algorithmic auditing requirements under emerging medical device frameworks forces fundamental shifts in how hospital networks evaluate ai enabled medical devices. Instead of accepting proprietary performance claims, chief medical informatics officers now demand certified, independent validation software alongside all new diagnostic procurement contracts. This regulatory barrier redefines vendor selection metrics. Predictive software developers who continue relying on black-box architectures face complete exclusion from major hospital modernisation programs.
Integrating continuous statistical monitoring directly into the clinical deployment pipeline drastically reshapes long‑term software‑support economics, especially as leading governance providers such as IBM, Microsoft, Securiti, Fiddler AI, 2021.AI, Monitaur, and Fairly AI push hospitals toward real‑time model validation. Algorithm developers proactively embedding API hooks for third‑party governance platforms, often aligned with the standards championed by these key vendors, minimise friction during the rigorous hospital IT‑security review process. Supplying native compatibility with established oversight tools accelerates the implementation timeline from initial procurement to live clinical deployment, while diagnostic vendors that rely on highly customised, manual evaluation frameworks increasingly risk losing their shortlist position as hospital administrators prioritise rapid, compliant deployment capabilities.
With standardized reporting metrics becoming the baseline requirement, clinical IT architects orchestrating complex digital health environments can source diagnostic algorithms from diverse providers without fearing validation‑integration failures. This architectural flexibility empowers hospital networks to optimise their predictive‑care tools for specific clinical specialties, while still maintaining a unified, centralised compliance dashboard supported by market‑leading governance technologies.
Key Players in Clinical AI Model Governance Market
- IBM
- Microsoft
- Securiti
- Fiddler AI
- 2021.AI
- Monitaur
- Fairly AI
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 1.4 billion to USD 6.3 billion, at a CAGR of 16.22% |
| Market Definition | The Clinical AI Model Governance Market represents the specific software infrastructure and compliance frameworks used to monitor, audit, and control artificial intelligence algorithms deployed in patient-facing medical environments to prevent concept drift. |
| Component Segmentation | Software Platforms, Services |
| Application Segmentation | Diagnostics & Imaging, Predictive Analytics, Drug Discovery, Operational AI |
| End User Segmentation | Hospitals & Clinics, Pharmaceutical Companies, Research Institutes |
| Deployment Segmentation | Cloud, On-Premises |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia, Oceania, Middle East & Africa |
| Countries Covered | USA, UK, Germany, China, Japan, South Korea, India, and 40 plus countries |
| Key Companies Profiled | IBM, Microsoft, Google Health, AWS, Siemens Healthineers, Philips Healthcare, GE HealthCare, Arthur AI |
| Forecast Period | 2026 to 2036 |
| Approach | The baseline value derives from a bottom-up aggregation of clinical AI governance software licenses, applying region-specific regulatory enforcement curves to project future adoption velocity. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Clinical AI Model Governance Market Analysis by Segments
Component:
- Solutions
- Services
Deployment:
- On-Premises
- Cloud
Functionality:
- Risk & Compliance
- Model Lifecycle Management
- Monitoring & Auditing
- Ethics & Responsible AI
Region:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- Medicines and Healthcare products Regulatory Agency. (2024, January 9). Regulatory roadmap points the way ahead for new measures to support safe access to medical technology including AI and diagnostics.
- National Institute for Health and Care Excellence. (2024, August 15). Use of AI in evidence generation: NICE position statement.
- Directorate-General for Health and Food Safety. (2025, June 19). MDCG 2025-6: FAQ on interplay between the Medical Devices Regulation & In vitro Diagnostic Medical Devices Regulation and the Artificial Intelligence Act. European Commission.
- National Academy of Medicine. (2025). An artificial intelligence code of conduct for health and medicine: Essential guidance for aligned action. National Academies Press. doi:10.17226/29087.
- Tighe, P., Mossburg, S. E., & Gale, B. (2024, March 27). Artificial intelligence and patient safety: Promise and challenges. PSNet, Agency for Healthcare Research and Quality.
This bibliography is provided for the reader's reference. The full FMI report contains the complete reference list with primary research documentation.
This Report Addresses
- Market intelligence to support strategic decision making across automated AI audit software and drift detection platforms unique to this market
- Market size estimation and 10-year revenue forecasts from 2026 to 2036, supported by bottom-up aggregation of clinical software licenses and hospital IT capital expenditure data
- Growth opportunity mapping across cloud deployments and diagnostic imaging applications with emphasis on the strict enforcement of malpractice liability standards by major medical insurers
- Segment and regional revenue forecasts covering predictive analytics governance and specialized MLOps platforms across strictly regulated North American and European healthcare jurisdictions
- Competition strategy assessment including the critical shift toward native automated compliance reporting, cross-vendor interoperability ecosystems, and embedded continuous monitoring capabilities
- Software capability development tracking including FDA Predetermined Change Control Plans (PCCP), EU AI Act compliance trackers, and demographic bias quantification metrics discussed in this article
- Market access analysis covering enterprise hospital procurement networks, medical device 510(k) regulatory clearance pathways, and national health service digitization grants
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive strategy, hospital IT risk management, and operational benchmarking use
Frequently Asked Questions
How large is the demand for Clinical AI Model Governance Market in the global market in 2026?
Demand for Clinical AI Model Governance Market in the global market is estimated to be valued at USD 2.48 billion in 2026.
What will be the market size of the Clinical AI Model Governance Market in the global market by 2036?
Market size for Clinical AI Model Governance Market is projected to reach USD 71.12 billion by 2036.
What is the expected demand growth for Clinical AI Model Governance Market in the global market between 2026 and 2036?
Demand for Clinical AI Model Governance Market is expected to grow at a CAGR of 39.9% between 2026 and 2036.
Which Component is poised to lead global sales by 2026?
Solutions commands 67.48% in 2026 as hospital network administrators execute full-scale integration of automated oversight platforms to replace manual auditing processes.
How significant is the role of Risk & Compliance in driving Clinical AI Model Governance Market adoption in 2026?
Risk & Compliance represents 40% of segment share as regulatory affairs leads coordinate complex medical device submission sequences to clear predictive algorithms for commercial use.
What is driving demand in China?
China's centralized healthcare strategy deploys massive predictive population health models, requiring clinical data architects to mandate high-volume automated oversight engines.
What compliance standards or regulations are referenced for United States?
The United States medical device regulatory landscape enforces strict pre-determined change control plans for any continuously learning diagnostic algorithm deployed within clinical workflows.
What is the China growth outlook in this report?
China is projected to grow at a CAGR of 42.0% during 2026 to 2036.
Why is North America described as a priority region in this report?
The North American clinical regulatory landscape actively drives the deployment of independent validation software to mitigate algorithmic liability across vast hospital networks.
What type of demand dominates in North America?
Demand heavily focuses on replacing manual spreadsheet-based auditing with automated oversight platforms that integrate directly with existing electronic health records.
What is India's growth outlook in this report?
India is projected to expand at a CAGR of 43.5% during 2026 to 2036.
Does the report cover United States in its regional analysis?
Yes, United States is included within North America under the regional scope of analysis.
What are the sources referred to for analyzing United States?
FDA algorithmic guidelines and mandatory software-as-a-medical-device reporting structures form the analytical basis.
What is the main demand theme linked to United States in its region coverage?
Strict compliance rules regarding model weight adjustments compel hospital administrators to restrict algorithms to shadow-mode until automated drift detection platforms are fully integrated.
Does the report cover Germany in its regional analysis?
Yes, Germany is included within Europe under the regional coverage framework.
What is the main Germany related demand theme in its region coverage?
Rigorous functional safety certifications mandate comprehensive audit trail generation before predictive diagnostic tools receive national reimbursement approval.
Which product formats or configurations are strategically important for Europe supply chains?
Fully localized, on-premises algorithmic monitoring tools are critical for achieving compliance with stringent continental data residency and privacy legislation.
What is Clinical AI Model Governance Market and what is it mainly used for?
Clinical AI model governance provides automated monitoring and validation for healthcare algorithms. It is primarily used to ensure diagnostic models remain accurate, unbiased, and compliant with medical regulations.
What does Clinical AI Model Governance Market mean in this report?
The scope encompasses specialized software platforms and auditing tools designed to track performance drift and generate regulatory reports for healthcare artificial intelligence applications.
What is included in the scope of this Clinical AI Model Governance Market report?
The market covers dedicated algorithmic auditing platforms, bias detection software, model lifecycle management suites, and automated regulatory reporting tools designed for healthcare.
What is excluded from the scope of this report?
General-purpose enterprise governance platforms lacking specific healthcare regulatory modules and basic hospital IT security infrastructure are explicitly excluded.
What does market forecast mean on this page?
The market forecast represents a model-based projection built on defined technology and adoption assumptions for strategic planning purposes.
How does FMI build and validate the Clinical AI Model Governance Market forecast?
The model applies a top-down assessment of global healthcare artificial intelligence expenditure and cross-validates projections against software procurement budgets published by major hospital networks.
What does zero reliance on speculative third-party market research mean here?
Primary interviews verified regulatory drafts from health authorities, and official compliance registries are used exclusively instead of unverified syndicated estimates.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Capacity
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Capacity , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Capacity , 2026 to 2036
- Less than 100 ml
- 100 to 250 ml
- 251 to 500 ml
- More than 500 ml
- Less than 100 ml
- Y-o-Y Growth Trend Analysis By Capacity , 2021 to 2025
- Absolute $ Opportunity Analysis By Capacity , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type, 2026 to 2036
- Straight Wall Cans
- Necked-in Cans
- Shaped Cans
- Straight Wall Cans
- Y-o-Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Cosmetics & Personal Care
- Household Products
- Healthcare
- Automotive & Industrial
- Cosmetics & Personal Care
- Y-o-Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Propellant Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Propellant Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Propellant Type, 2026 to 2036
- Liquefied Gas Propellant
- Compressed Gas Propellant
- Liquefied Gas Propellant
- Y-o-Y Growth Trend Analysis By Propellant Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Propellant Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Capacity
- By Product Type
- By End Use
- By Propellant Type
- Competition Analysis
- Competition Deep Dive
- Ball Corporation
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Crown Holdings Inc.
- Ardagh Group S.A.
- Trivium Packaging
- CCL Container
- TUBEX GmbH
- Montebello Packaging
- Ball Corporation
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Capacity , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Propellant Type, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Capacity , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Propellant Type, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Capacity , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Propellant Type, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by Capacity , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Propellant Type, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by Capacity , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Propellant Type, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by Capacity , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Propellant Type, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by Capacity , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Propellant Type, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by Capacity , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Propellant Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Capacity , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Capacity , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Capacity
- Figure 6: Global Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Product Type
- Figure 9: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End Use
- Figure 12: Global Market Value Share and BPS Analysis by Propellant Type, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Propellant Type, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Propellant Type
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by Capacity , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Capacity , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Capacity
- Figure 29: North America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product Type
- Figure 32: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by End Use
- Figure 35: North America Market Value Share and BPS Analysis by Propellant Type, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Propellant Type, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Propellant Type
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by Capacity , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Capacity , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Capacity
- Figure 42: Latin America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Product Type
- Figure 45: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by End Use
- Figure 48: Latin America Market Value Share and BPS Analysis by Propellant Type, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Propellant Type, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Propellant Type
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by Capacity , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by Capacity , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by Capacity
- Figure 55: Western Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Product Type
- Figure 58: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by End Use
- Figure 61: Western Europe Market Value Share and BPS Analysis by Propellant Type, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Propellant Type, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Propellant Type
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by Capacity , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by Capacity , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by Capacity
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Propellant Type, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Propellant Type, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Propellant Type
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by Capacity , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by Capacity , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by Capacity
- Figure 81: East Asia Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Product Type
- Figure 84: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by End Use
- Figure 87: East Asia Market Value Share and BPS Analysis by Propellant Type, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Propellant Type, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Propellant Type
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by Capacity , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by Capacity , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by Capacity
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Propellant Type, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Propellant Type, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Propellant Type
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by Capacity , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by Capacity , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by Capacity
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Propellant Type, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Propellant Type, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Propellant Type
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis

