Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market
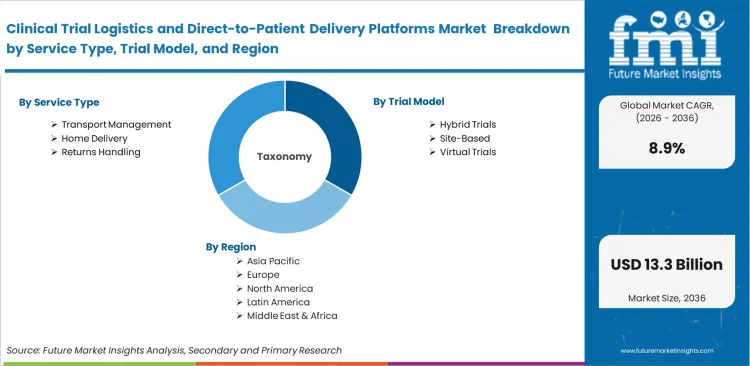
The clinical trial logistics and direct-to-patient delivery platforms are segmented by Service Type (Transport Management, Home Delivery, Returns Handling, Sample Pickup, Comparator Sourcing), Trial Model (Hybrid Trials, Site-Based, Virtual Trials), Delivery Mode (Depot-to-Patient, Site-to-Patient, Pharmacy-to-Patient, Nurse-Assisted), Temperature Profile (Cold Chain, Ambient, Frozen), End User (Biopharma Sponsors, CROs, Academic Centers, Medical Device Firms), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market Size, Market Forecast, and Outlook By FMI
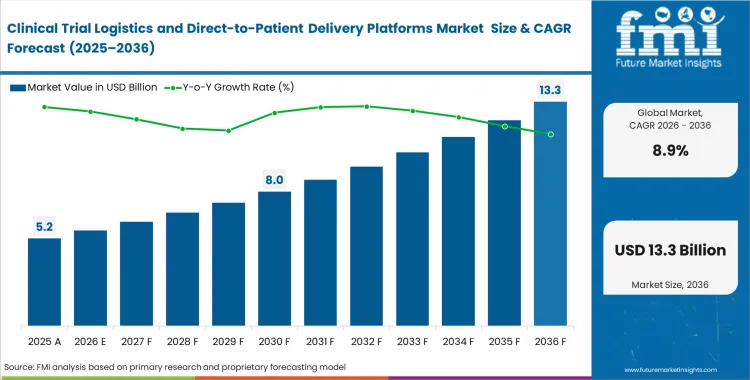
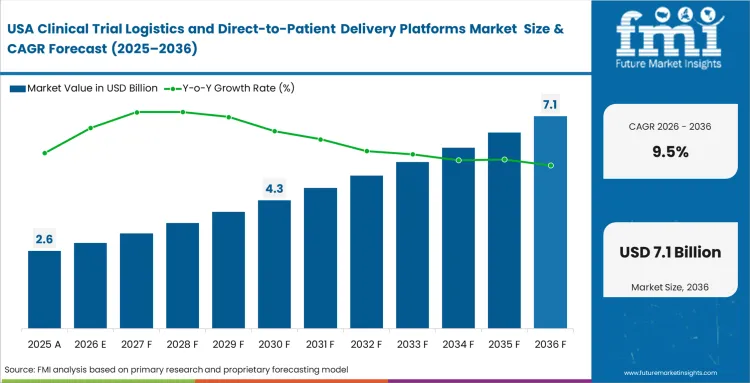
The clinical trial logistics and direct-to-patient delivery platforms market stood at USD 5.2 billion in 2025 and is projected to reach USD 5.5 billion in 2026, reflecting an annual growth rate of 8.9%. Total revenue is expected to reach USD 13.3 billion by 2036. This expansion is closely tied to a shift in how trials are conducted, with clinical operations teams gradually moving away from centralized site networks toward models that accommodate patients at home. The change is redefining how logistics networks are structured and managed across trial programs.
Summary of Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market
- Market Snapshot
- The clinical trial logistics and direct-to-patient delivery platforms market is poised to cross a valuation of USD 5.2 billion in 2025 and USD 13.3 billion in 2036.
- Industry is anticipated to garner a CAGR of 8.9% between 2026 and 2036, which is set to build an incremental opportunity of USD 5.5 billion through 2026.
- It is a clinical-operations compliance-based niche that encompasses temperature-controlled trial logistics, patient-home delivery, returns, pickup flows, and workflow platforms between depots, sites, pharmacies, and participants.
- This industry is a compliance-led clinical-operations niche covering temperature-controlled trial logistics, patient-home delivery, returns, pickup flows, and workflow platforms that connect depots, sites, pharmacies, and participants.
- Government recommends that investigators carefully consider the timing of drug testing, the timing of drug testing is another aspect that should be looked into by investigators.
- Demand and Growth Drivers
- Demand is increasing as government is finalizing decentralized-clinical-trial guidance, giving sponsors a clearer operating framework for remote and non-site trial activities.
- Adoption of direct-to-patient logistics is rising because it reduces participant travel burden and supports recruitment and retention in decentralized and hybrid studies.
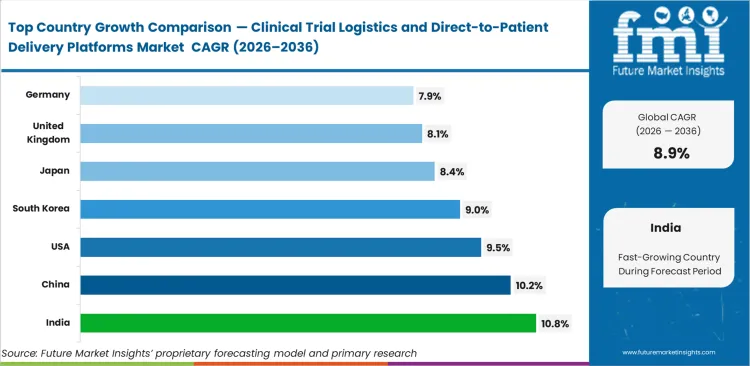
- India leads at 10.8% CAGR, followed by China at 10.2%, the United States at 9.5%, South Korea at 9.0%, Japan at 8.4%, the United Kingdom at 8.1%, and Germany at 7.9%.
- Growth is moderated by uneven country rules for investigational medicinal product shipment to homes, privacy handling, pharmacist oversight, and reverse-logistics complexity for unused product and samples.
- Product and Segment View
- Clinical transport management, home delivery, sample pickup, returns, comparator sourcing, and digital routing/integration layers in decentralized and hybrid trials are all covered by the market.
- These services are applicable in an oncology, rare-disease, biologics, vaccine, device, and multi-country protocol setting where access to sites or retention of participants is challenging.
- Reflecting the current dominance of core distribution, temperature control, chain-of-custody, and time-definite movement, transport management leads the service type category with the majority of the holdings.
- Hybrid Trials dominate the Trial Model segment at 42.6% share, since most sponsors are incorporating decentralized features without undergoing a complete transition to virtual.
- Depot-to-Patient is the leader of the Delivery Mode segment with the majority of shares, which is facilitated by centralized inventory management and simpler standardization across geographies.
- With 47.2% share in 2026, the Temperature Profile category is dominated by Cold Chain, which indicates the proportion of the biologics, specialty medicines, and sample-stability conditions in contemporary trials.
- The End User segment is dominated by Biopharma Sponsors with a 49.5% share because they continue to be the primary purchasers of outsourced trial logistics and patient-delivery capacity.
- The scope includes dragging in investigation-product, patient-home, sample-pickup, returns, and operational platforms, related to trial execution, but not all CRO service revenue, extensive pharmaceutical logistics, not related to trial conduct, and generic telehealth platforms.
- Geography and Competitive Outlook
- India, China and the United States are the highest growth national markets in this model with Germany, the United Kingdom and Japan being stable regulated demand bases.
- Competition is defined by the depth of the global network, cold-chain validation, protocol flexibility, last-mile visibility, and integrations with trial-management systems as opposed to mere freight capacity.
- Some of the major players include Marken, World Courier, Thermo Fisher Scientific, DHL Group, Almac Group, PCI Pharma Services and Parexel and the market is moderately fragmented with the leading player owning a majority of shares.
- DTP functionality, patient-centric design, and orchestration via platforms are emerging into a more powerfully differentiating feature as sponsors switch to hybrid execution of trials instead of the site-only model.
Rising protocol complexity is pushing biopharma supply chain teams to build establish investigational product home delivery networks. Frequent site visits, especially for complex therapies, are proving difficult to sustain and often lead to lower patient retention. Delivering therapies to patients’ homes introduces stricter coordination requirements, as dosing schedules, handling conditions, and delivery timing must align precisely. This has increased reliance on specialized courier networks that can operate within tight clinical parameters. Without reliable direct-to-patient delivery platforms, trial timelines tend to stretch, costs escalate, and sponsors face greater pressure from limited patent lifecycles.
This transition is being facilitated by regulatory alignment. With the FDA giving more explicit guidance on the model of decentralized trials, institutional review boards are being more open to protocol amendments that involve remote delivery and monitoring. The key to this move is to develop uniform chain-of-custody standards in non-traditional clinical environments, which will guarantee data integrity and permit sponsors to develop residential delivery models with fewer regulatory issues.
The clinical trial logistics sector in India is set to rise at a CAGR of 10.8% to 2036, amidst the growing number of trials and the continuous digitalization of research activities. China comes next with an estimated CAGR of 10.2 %, where the policy emphasis is laid on speeding up trial processes and enhancing efficiency. The market is expected to grow by 9.5 % CAGR in the United States as the finalized FDA guidance and increased utilization of decentralized clinical trial logistics are adopted. South Korea will experience growth of 9.0% due to the well-structured infrastructure and the ability to coordinate the execution. Japan will grow with 8.4%, and the compliance requirements are still restricting the flexibility of operations. The UK is projected at 8.1 %, as compared to Germany at 7.9 %, which indicates stricter regulatory supervision throughout Europe.
Segmental Analysis
Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market Analysis by Service Type
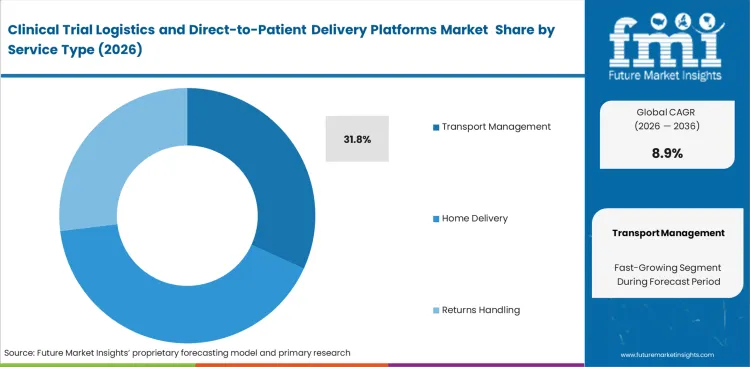
Core coordination workflows are at the center of early decentralized trial investments, as execution depends on how precisely movement is planned and controlled. Clinical operations teams focus on specialized routing rather than standard delivery, since protocol timelines leave little room for error. Moving away from site-based dispensing toward direct-to-patient distribution introduces additional variables, including patient availability, dosing schedules, and handling conditions. Transport Management is expected to hold for 31.8% share of the market in 2026, reflecting the weight placed on scheduling accuracy. Much of the operational differentiation now lies in reverse logistics, particularly in managing biological sample returns from patient homes, where timing is unpredictable. Conventional life sciences logistics models struggle to handle these unscheduled flows. Coordinated systems are required to ensure temperature-sensitive samples reach central laboratories within viable timeframes, as delays can compromise analytical results. Sponsors relying on fragmented courier networks face increased audit risk, including the possibility of entire patient cohorts being excluded due to gaps in traceability.
- Coordination scheduling: Precision routing aligns drug delivery with home healthcare visits, ensuring medication arrives when administration personnel are present and avoiding protocol deviations.
- Reverse integration: Biological sample collection depends on immediate and validated return transport, with pickup windows tightly managed to prevent degradation.
- Auditable documentation: Regulatory bodies require continuous chain-of-custody visibility for residential delivery, and any break in documented transit can lead to trial suspension.
Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market Analysis by Trial Model
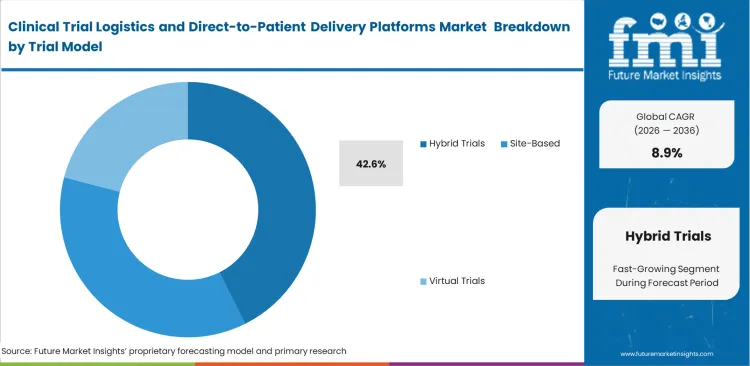
Protocols that combine in-clinic visits with remote treatment are now widely used, as fully virtual models remain difficult to execute for complex therapies. Hybrid Trials are estimated to account for 42.6% share of the market in 2026, reflecting their role in balancing clinical oversight with patient convenience. Investigators still rely on physical assessments during initial dosing to monitor patient response before shifting stable participants to home-based treatment. This transition depends on establishing local drug distribution points to maintain continuity once patients move out of clinical settings. Managing these models brings operational gaps that are often underestimated, particularly in healthcare cold chain logistics. Missed home deliveries or improper patient handling can lead to higher product wastage than standard metrics suggest. Clinical trial packaging is being redesigned to handle variable home environments, where temperature control and handling conditions are less predictable. Without these adjustments, sponsors face repeated losses from expired investigational products. Inventory planning also becomes less stable, as changing patient schedules make demand forecasting more difficult for procurement teams.
- Initial site qualification: Complex therapies require early clinical observation, allowing investigators to assess patient response before approving home-based treatment.
- Maintenance distribution: Ongoing dosing shifts to residential delivery, reducing reliance on site-level inventory and associated holding costs.
- Patient retention impact: Fewer clinic visits improve adherence, helping maintain trial cohorts and reach statistical targets faster.
Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market Analysis by Delivery Mode
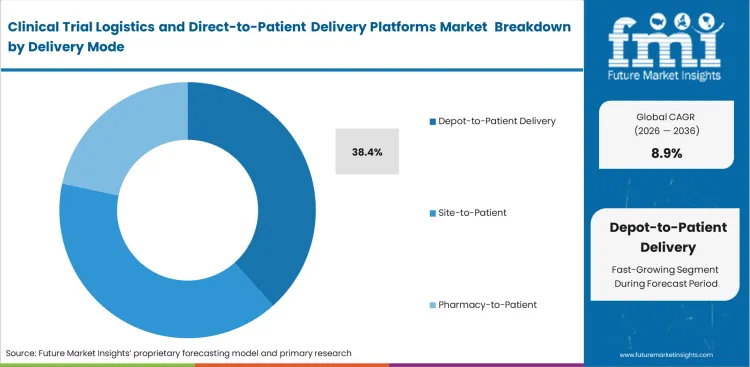
Direct distribution from centralized storage reduces variability linked to site-level handling and improves control over investigational product movement. Biopharma companies are increasingly standardizing inventory through regional depots, where contract packaging teams assemble patient-specific kits before dispatch through qualified courier networks. Linking clinical trial data management service with depot-to-patient trial delivery systems allows automated resupply based on patient dosing records, reducing manual intervention. Software triggers often do not align with depot working hours, leading to missed dispatches, particularly over weekends. Supply chain teams need to align system logic with actual operating schedules to avoid these gaps. Depot-to-Patient delivery is anticipated to account for 38.4% share of the sector in 2026, reflecting this shift toward centralized control. When depot activity and system triggers fall out of sync, patients can miss doses during critical treatment windows.
- Inventory centralization: Regional depots pool investigational products, helping procurement teams avoid excess production of high-cost biologics.
- Automated dispatching: Patient data inputs trigger follow-on shipments, reducing manual reordering and maintaining treatment continuity.
- Transit visibility: Specialized couriers provide real-time tracking, allowing quality teams to verify delivery conditions and product integrity.
Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market Analysis by Temperature Profile
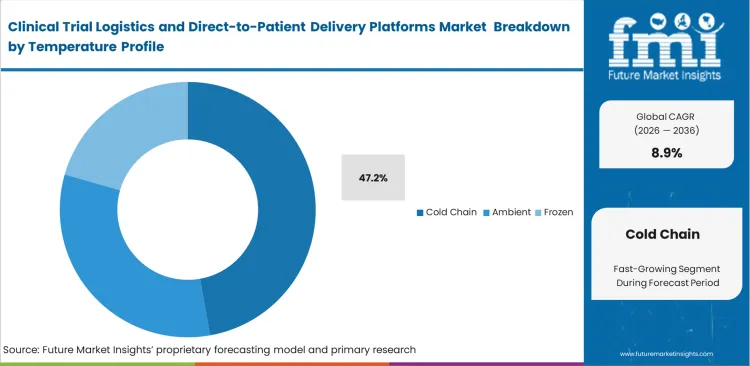
Biologic formulations require tight temperature control during transit, particularly as delivery shifts toward patient homes. This becomes more critical in cell and gene therapy trial logistics, where even minor temperature deviations can compromise product viability. Formulation teams define strict thermal limits that must be maintained from dispatch through final administration. Active cooling systems are now standard in last-mile delivery to prevent temperature excursions. Cold Chain is expected to account for 47.2% share of the market in 2026, reflecting the increasing share of advanced biologics in clinical pipelines. A recurring gap sits at the patient end. A healthcare contract research organization often does not fully account for variability in home refrigeration when setting stability parameters. Providing validated storage solutions at the patient level helps manage this risk. There is a clear risk of administering degraded therapies, which directly affects both patient safety and trial reliability.
- Active thermal packaging: Specialized shipping containers maintain stable temperatures over extended durations, reducing exposure during delays.
- Data logger integration: Continuous monitoring captures transit conditions, allowing quality teams to review temperature logs before approving administration.
- Return shipment control: Biological samples require separate thermal handling during return transit, with pre-conditioned packaging provided to maintain integrity.
Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market Analysis by End User
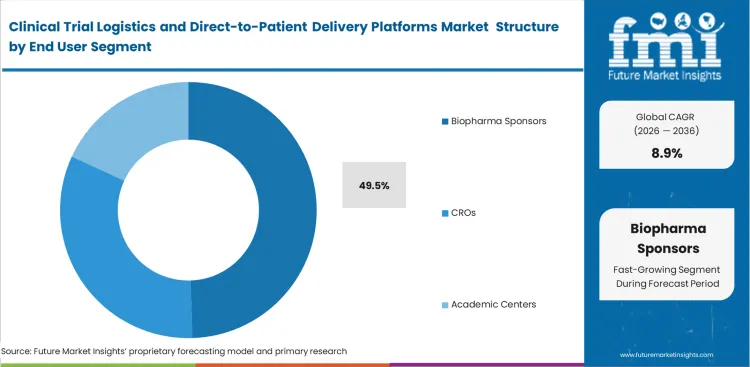
Drug developers remain the primary source of trial funding, as they carry the responsibility of moving therapies through regulatory approval. Biopharma sponsors retain full legal accountability for protocol execution, even when logistics operations are outsourced to external partners. Expanding early-phase clinical trial outsourcing is pushing sponsors to incorporate direct-to-patient delivery capabilities earlier in development cycles. Mid-sized biopharma companies often misjudge the complexity of residential delivery, particularly in rare disease clinic trials where patient dispersion is high, and handling requirements are strict. Local pharmacy networks are rarely equipped to manage investigational products under these conditions. Integrating specialized courier networks requires structured vendor qualification and ongoing compliance oversight. Biopharma Sponsors are anticipated to account for 49.5% share of the market in 2026, reflecting their central role in funding and regulatory responsibility. Sponsors that do not enforce rigorous logistics standards risk regulatory setbacks during final marketing authorization reviews.
- Protocol design influence: Biopharma teams incorporate decentralized elements early in study design, with medical leadership prioritizing patient access alongside clinical control.
- Vendor qualification: Procurement evaluates courier compliance capabilities, while quality teams audit depot operations to ensure adherence to Good Manufacturing Practice standards.
- Financial accountability: Sponsors carry the full cost of logistics failures, including product loss and delays, making courier reliability a key operational metric.
Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market Drivers, Restraints, and Opportunities
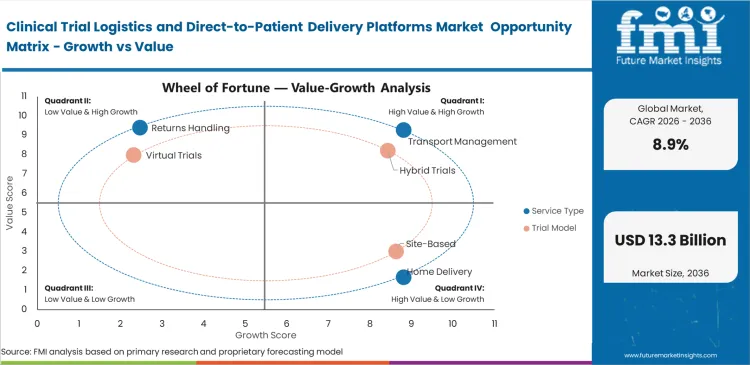
Patient recruitment challenges force clinical operations directors to remove geographical barriers from trial protocols. Requiring patients to travel long distances for site visits drastically reduces eligible enrollment pools. Implementing residential delivery expands catchment areas globally. This expansion requires immediate integration of electronic trial master file systems with DCT logistics platform suppliers to ensure compliance. Failing to establish this infrastructure stalls trial enrollment entirely. Sponsors must embrace direct distribution networks to achieve statistical powering within acceptable timeframes.
Cross-border regulatory fragmentation severely complicates international direct-to-patient deployment. Shipping investigational products across multiple jurisdictions requires navigating contradictory customs classifications and import restrictions. While patient engagement platforms streamline communication, physical goods face hard borders. Operations leads struggle to standardize global protocols when local health authorities prohibit residential dispensing of experimental compounds. This friction persists structurally because sovereign nations retain independent jurisdiction over unregistered drug distribution. Customs delays frequently destroy time-sensitive biological samples before they reach analytical laboratories.
Opportunities in the Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market
- Predictive routing: Artificial intelligence analyzes traffic patterns and weather disruptions. Logistics managers optimize delivery windows to ensure reliable patient access.
- Sensor integration: Embedding connected logistics sensors directly onto primary packaging provides unit-level visibility. Quality leads track individual doses rather than bulk shipments.
- Nurse network partnerships: Combining courier services with mobile healthcare providers streamlines administration. Clinical directors eliminate coordination errors by utilizing single-vendor solutions.
Regional Analysis
Based on regional analysis, Clinical Trial Logistics and Direct-to-Patient Delivery Platforms is segmented into Asia Pacific, North America, and Europe across 40 plus countries.
.webp)
| Country | CAGR (2026 to 2036) |
|---|---|
| India | 10.8% |
| China | 10.2% |
| United States | 9.5% |
| South Korea | 9.0% |
| Japan | 8.4% |
| United Kingdom | 8.1% |
| Germany | 7.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market Analysis
Cost advantages and access to large patient populations continue to draw biopharma investment toward Eastern clinical markets. Ongoing infrastructure upgrades are allowing sponsors to run complex hybrid trials beyond traditional Western locations. Expanding remote healthcare delivery across geographically dispersed populations requires a different logistics setup than what works in dense urban regions. Logistics teams are building hub-and-spoke depot networks to ensure consistent reach into rural areas. Regional regulators are also updating guidance frameworks to accommodate the growing volume of outsourced clinical trials and support broader adoption of decentralized models.
- India: Rising clinical trial volumes in India are pushing sponsors and CROs to modernize execution workflows, particularly across patient engagement, sample movement, and investigational product distribution. Procurement leaders are increasingly engaging specialized clinical couriers to manage regional infrastructure variability while maintaining protocol compliance. The sector in India is expanding at a compound annual growth rate of 10.8% CAGR, supported by sponsor efforts to reach large, treatment‑naïve patient populations beyond conventional site‑based trial models.
- China: Policy pressure to shorten research timelines has made China one of the most active domestic environments for clinical trials, reshaping how studies are operationalized on the ground. Clinical trial delivery and logistics services in the country are estimated to expand at a CAGR of 10.2%, reflecting rising protocol complexity and faster execution expectations. In response, clinical operations leaders are rapidly scaling hybrid trial designs to stay aligned with regulatory schedules. Strong local uptake has also pushed multinational sponsors to establish dedicated regional depots to support reliably paced, in‑country trial execution.
- South Korea: South Korea’s concentrated healthcare infrastructure supports relatively seamless direct‑to‑patient clinical trial execution, particularly across major metropolitan areas. Clinical trial logistics demand is expected to increase at a 9.0% CAGR, reflecting greater reliance on time‑critical home delivery models. Supply‑chain leaders focus heavily on route efficiency inside dense urban corridors, where execution precision directly impacts protocol adherence. Consistent cold‑chain performance has emerged as the key differentiator among domestic logistics providers.
- Japan: Policy initiatives aimed at strengthening healthcare innovation have encouraged the gradual adoption of decentralized trial approaches, while regulatory rigor remains unchanged. High compliance expectations continue to shape vendor selection, leading procurement directors to favor established global courier partners with deep experience managing complex import controls. This operating discipline supports steady expansion, even as the country sustains a compound annual growth rate of 8.4% in clinical trial logistics services. Strong oversight standards continue to translate into superior data quality, particularly for biologics and advanced‑therapy trials.
FMI's report includes Australia alongside the primary Asian healthcare economies. Fragmented archipelago geographies and diverse climate zones across this broader territory compel clinical operations directors to adopt highly localized secondary courier networks. The Philippines, where increasing clinical trial activity in Metro Manila and emerging provincial sites is raising demand for flexible, temperature‑controlled last‑mile delivery supported by local partners.
North America Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market Analysis
Well-established clinical infrastructure and strong research funding enables North America to lead in the decentralized trial execution. Many global logistics standards are developed here before being applied across other markets. Operational focus is shifting toward linking remote patient monitoring devices with physical drug distribution, as sponsors look for better visibility across the trial lifecycle. Coordinating biological sample collection with patient-reported data has become a priority for clinical teams. Courier networks in this region are increasingly evaluated on their ability to integrate with digital platforms, as software compatibility now carries as much weight as delivery performance.
- United States: In the United States, Formal clarification in the FDA’s final decentralized trial guidance removed lingering uncertainty around remote visits and the use of local healthcare providers, creating a more stable compliance environment for trial execution. Supply‑chain directors are responding by scaling residential delivery and sample‑return networks with greater confidence under this framework. Reflecting wider adoption of decentralized and hybrid study designs, demand for clinical trial logistics services is expected to expand at a 9.5% CAGR in the US. In this setting, global courier firms that build comprehensive national capabilities early are well-positioned to secure lasting competitive advantage.
FMI's report includes Canada within the North American assessment. Massive transcontinental distances combined with extreme winter temperature profiles force supply‑chain leads to over‑engineer thermal packaging configurations even for routine ambient shipments. Costa Rica is highlighted as well, supported by a growing life‑sciences manufacturing base that is driving demand for reliable, temperature‑controlled domestic and cross‑border clinical logistics services.
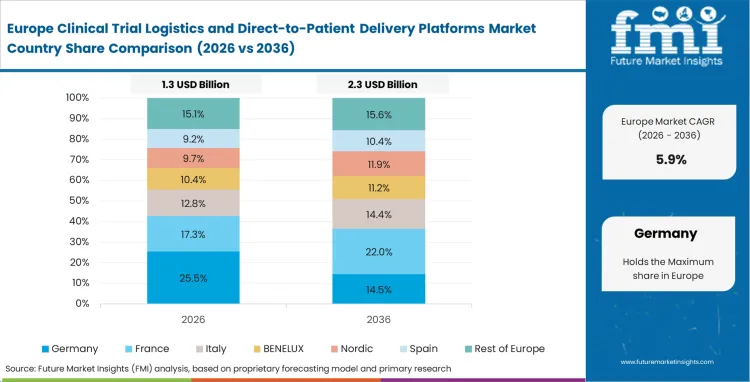
Europe Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market Analysis
Strict data privacy rules and complex cross-border transport requirements shape how clinical logistics operate across this continent. Quality teams require detailed chain-of-custody documentation before approving logistics partners, leaving little room for process gaps. Remote patient monitoring systems must align closely with General Data Protection Regulation requirements, adding another layer of compliance. Sponsors often centralize depot operations in select jurisdictions to manage distribution more efficiently across multiple countries. Differences in how national authorities interpret clinical trial regulations continue to create execution challenges for logistics providers operating across borders.
- United Kingdom: Regulatory support for decentralized research continues to strengthen, with the Health Research Authority playing an active role in expanding accepted operating models. Clinical trial logistics demand in the United Kingdom is expected to grow at a 8.1% CAGR, supported by the country’s compact geography and dense clinical site distribution. Operations teams take advantage of these conditions to execute rapid direct‑to‑patient shipping with minimal transit variability. A resilient domestic research ecosystem further limits exposure to broader European regulatory disruption, helping maintain execution continuity.
- Germany: Clinical trial execution in Germany remains defined by strict procedural controls and rigorous compliance oversight. Quality assurance teams place considerable emphasis on courier qualification, often requiring exhaustive thermal validation before investigational products are released into distribution. Against this backdrop, the clinical trial logistics segment is projected to expand at a CAGR of 7.9%, despite relatively conservative adoption of decentralized models. The market’s emphasis on accuracy and reproducibility continues to push logistics providers toward deploying their most advanced temperature‑control and packaging solutions.
FMI's report includes France, Italy, Spain, Russia, and Poland. Divergent national interpretations of centralized clinical trial directives force quality‑assurance directors to maintain country‑specific chain‑of‑custody documentation templates. The Netherlands, where high clinical trial density and advanced distribution infrastructure are encouraging wider adoption of standardized, audit‑ready logistics workflows.
Competitive Aligners for Market Players
Procurement teams assess clinical trial logistics providers primarily on their ability to maintain consistent temperature control and complete documentation across geographically dispersed operations. Companies such as Marken, World Courier, and Thermo Fisher Scientific have built strong positions by establishing globally validated depot networks that support standardized execution. Logistics managers are generally reluctant to engage regional providers that do not offer integrated pharmaceutical packaging, as splitting packaging and delivery across vendors increases the risk of protocol deviations. Sponsors expect clear, end-to-end accountability for investigational product handling, from origin through to patient delivery.
Established providers also benefit from extensive proprietary datasets that track thermal performance across different global shipping lanes. Quality assurance teams use this historical data to support vendor selection and demonstrate compliance during regulatory audits. Newer direct-to-patient delivery providers are working to build similar capabilities, but network validation takes time, often requiring several years before meeting Good Clinical Practice expectations. Leading players have also expanded into handling home infusion devices alongside drug delivery, allowing them to support more complex administration requirements within a single service model.
Large biopharma companies continue to avoid dependence on a single logistics provider by designing trial protocols that can operate across multiple courier networks. Operations teams require standardized data exchange formats, ensuring that logistics systems integrate with broader trial management platforms. Market consolidation is ongoing, with larger providers acquiring regional specialists to strengthen geographic coverage. Purchasing teams use this competitive environment to negotiate global contracts with strict performance clauses, particularly around temperature compliance and adherence to dosing schedules.
Key Players in Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market
- Marken
- World Courier
- Thermo Fisher Scientific
- DHL Group
- Almac Group
- PCI Pharma Services
- Parexel
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 5.5 Billion to USD 13.3 Billion, at a CAGR of 8.9% |
| Market Definition | Coordinating physical movement of investigational drugs, biological samples, and clinical equipment between sponsors, clinical sites, and patient residences. |
| Segmentation | Service Type, Trial Model, Delivery Mode, Temperature Profile, End User, Region |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, Asia Pacific, Middle East and Africa |
| Countries Covered | United States, Canada, Germany, United Kingdom, France, Italy, Spain, Russia, Poland, China, India, Japan, South Korea, Australia, Brazil, Mexico, GCC Countries, South Africa |
| Key Companies Profiled | Marken, World Courier, Thermo Fisher Scientific, DHL Group, Almac Group, PCI Pharma Services, Parexel |
| Forecast Period | 2026 to 2036 |
| Approach | Protocol amendment rates shifting from site-based to hybrid models anchor baseline valuation. |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Clinical Trial Logistics and Direct-to-Patient Delivery Platforms Market Analysis by Segments
Service Type
- Transport Management
- Home Delivery
- Returns Handling
- Sample Pickup
- Comparator Sourcing
Trial Model
- Hybrid Trials
- Site-Based
- Virtual Trials
Delivery Mode
- Depot-to-Patient
- Site-to-Patient
- Pharmacy-to-Patient
- Nurse-Assisted
Temperature Profile
- Cold Chain
- Ambient
- Frozen
End User
- Biopharma Sponsors
- CROs
- Academic Centers
- Medical Device Firms
Region
- North America
- United States
- Canada
- Latin America
- Brazil
- Mexico
- Western Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Eastern Europe
- Russia
- Poland
- Asia Pacific
- China
- India
- Japan
- South Korea
- Australia
- Middle East and Africa
- GCC Countries
- South Africa
Bibliography
- ClinicalTrials.gov. (2026, January). Clinical trial reporting requirements. ClinicalTrials.gov.
- European Medicines Agency. (2025, January). ICH E6 (R3) guideline on good clinical practice (GCP), Step 5. European Medicines Agency.
- Food and Drug Administration. (2024, September). Conducting clinical trials with decentralized elements: Guidance for industry, investigators, and other interested parties. FDA.
- Hanke, S., et al. (2025). Operationalizing decentralized clinical trials. Clinical and Translational Science.
- Heath, M., et al. (2025). The supply of investigational medicinal product and trial logistics in decentralized clinical research. British Journal of Clinical Pharmacology.
- Ministry of Health, Labour and Welfare. (2024, April). Policy recommendations to bolster Japan’s healthcare startup ecosystem. MHLW.
- UK Health Research Authority. (2025, July). Annual report and accounts 2024-2025. UK Government.
This bibliography is provided for reader reference. The full FMI report contains the complete reference list with primary source documentation.
This Report Addresses
- Protocol amendment rates shifting from site-based to hybrid models driving specialized courier demand.
- Reverse logistics friction in biological sample collection from patient residences.
- Temperature control requirements for complex biologic and cell therapy investigational products.
- Depot-to-patient direct distribution architecture adoption by global biopharma sponsors.
- Vendor qualification metrics utilized by clinical operations and quality assurance directors.
- Software integration challenges between clinical data systems and physical dispatch networks.
- Geographic divergence between Asian trial expansion and European process constraints.
- Patient retention impacts derived from minimizing physical clinic visit requirements.
Frequently Asked Questions
What is the clinical trial logistics and direct-to-patient delivery platforms market size?
FMI values the sector at USD 5.5 billion for 2026. This figure reflects the initial capital surge required to establish localized depot networks capable of serving decentralized trial protocols across fragmented global geographies.
What is the clinical trial logistics and direct-to-patient delivery platforms market forecast 2036?
Revenue builds to USD 13.3 billion by 2036. Sustained investment supports the complete re-architecture of investigational product supply chains from centralized bulk shipping to unit-level residential distribution.
What is the clinical trial logistics CAGR?
The valuation expands at a 8.9% CAGR. Complex biologic formulations driving research pipelines demand expensive, active-cooling courier services that significantly raise the revenue per shipped unit.
Why does transport management lead the service type segment?
Core coordination workflows dictate initial decentralized trial investments. Operations directors prioritize specialized routing platforms to ensure investigational drugs synchronize perfectly with scheduled home health nurse administration visits.
Why are hybrid trials increasing demand for patient delivery platforms?
Principal investigators require physical assessment for initial dosing before transitioning patients to home delivery for maintenance phases. This hybrid structure requires distinct logistical flows capable of supporting both bulk site delivery and individual residential dispatch.
What is the difference between depot-to-patient and site-to-patient delivery?
Depot distribution centralizes inventory to prevent overproduction of expensive biologics. Site distribution forces individual clinics to manage patient shipments, transferring logistical burden to healthcare workers untrained in transit protocols.
Who are the leading vendors in clinical trial home delivery logistics?
Marken, World Courier, Thermo Fisher Scientific, and DHL Group dominate the global landscape. These incumbents possess vast proprietary data libraries mapping thermal performance across millions of global transit lanes.
Why does cold chain matter in clinical trial home delivery?
Biologic formulations and mRNA therapeutics dictate stringent thermal control requirements. Specialized active cooling systems prevent temperature excursions during last-mile transit, ensuring vulnerable patients do not receive degraded experimental compounds.
Which countries are growing fastest in decentralized clinical trial logistics?
India and China outpace mature markets by expanding rapidly to capture outsourced trial volume. Digitalizing research execution allows sponsors to access enormous treatment-naive patient populations across these vast geographies.
What are the main regulatory constraints on investigational product home delivery?
Cross-border regulatory fragmentation complicates deployment. Shipping experimental compounds across jurisdictions requires navigating contradictory customs classifications, which frequently delay or destroy time-sensitive biological shipments.
How do direct-to-patient models improve recruitment and retention?
Removing geographical barriers drastically expands eligible enrollment pools. Patients facing complex disease burdens drop out of studies when protocols demand exhausting travel, making residential delivery critical for achieving statistical powering.
What is included and excluded in this market definition?
Scope captures residential transport management, specialized courier networks, and sample pickup coordination. General commercial drug distribution networks fall outside the analysis because they lack patient-specific protocol blinding requirements.
How does reverse logistics function in this environment?
Biological sample collection requires immediate return transit under specific thermal profiles. Operations leads must supply pre-conditioned thermal packaging to patients to prevent sample degradation before reaching central analytical laboratories.
Why do contract research organizations struggle with remote data capture?
Validating remote data systems alongside physical delivery pipelines creates massive compliance friction. Quality assurance teams must ensure electronic diaries synchronize perfectly with physical drug receipt documentation.
What role does predictive routing play?
Artificial intelligence analyzes traffic patterns and weather disruptions to optimize delivery windows. Logistics managers utilize these tools to ensure investigational drugs reach patients before their specific administration deadlines expire.
How do biopharma sponsors handle vendor qualification?
Procurement teams assess courier compliance capabilities against Good Clinical Practice standards. Quality assurance managers audit depot facilities intensely because sponsors bear complete financial and regulatory accountability for logistical failures.
What impact does the FDA decentralized trial guidance have?
Formalizing legal acceptance for remote visits and local healthcare provider utilization allows supply chain directors to confidently scale residential delivery networks without fearing retroactive data invalidation.
Why do local pharmacies struggle with investigational products?
Mid-size biopharma companies frequently assume local pharmacies can manage experimental compounds. However, pharmacies lack the specialized blinding protocols and thermal tracking infrastructure required by rigorous clinical trial regulations.
How do sensors improve chain-of-custody tracking?
Embedding connected logistics sensors onto primary packaging provides unit-level visibility. Quality leads track individual doses in real-time, verifying transit conditions immediately rather than waiting for post-delivery data logger downloads.
What happens if a patient misses a home delivery window?
Unsuccessful deliveries threaten the viability of temperature-sensitive biologics. Supply chain directors must implement robust contingency plans, including immediate return-to-depot protocols, to prevent drug spoilage and costly lot replacements.
Why is academic clinical trial logistics growing slower?
Academic centers manage smaller patient cohorts with concentrated regional footprints. These localized studies require less complex international courier coordination compared to massive global Phase III biopharma sponsor protocols.
How does cell and gene therapy alter logistics requirements?
Advanced therapies introduce extreme thermal constraints, often requiring cryogenic shipping. Furthermore, autologous therapies demand perfect chain-of-identity tracking to ensure patients receive their own modified cells safely.
How does the United Kingdom maintain strong trial growth?
The Health Research Authority actively modernizes decentralized research capabilities. Operations directors leverage concentrated geographic profiles to execute rapid direct-to-patient shipping protocols efficiently without cross-border friction.
Why do operations directors avoid fragmented local couriers?
Relying on multiple local providers shatters chain-of-custody documentation. Sponsors utilizing fragmented networks risk complete cohort data exclusion during regulatory audits due to inconsistent handling procedures and missing transit records.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Service Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Service Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Service Type , 2026 to 2036
- Transport Management
- Home Delivery
- Returns Handling
- Transport Management
- Y to o to Y Growth Trend Analysis By Service Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Service Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Trial Model
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Trial Model, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Trial Model, 2026 to 2036
- Hybrid Trials
- Site-Based
- Virtual Trials
- Hybrid Trials
- Y to o to Y Growth Trend Analysis By Trial Model, 2021 to 2025
- Absolute $ Opportunity Analysis By Trial Model, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Delivery Mode
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Delivery Mode, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Delivery Mode, 2026 to 2036
- Depot-to-Patient Delivery
- Site-to-Patient
- Pharmacy-to-Patient
- Depot-to-Patient Delivery
- Y to o to Y Growth Trend Analysis By Delivery Mode, 2021 to 2025
- Absolute $ Opportunity Analysis By Delivery Mode, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Temperature Profile
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Temperature Profile, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Temperature Profile, 2026 to 2036
- Cold Chain
- Ambient
- Frozen
- Cold Chain
- Y to o to Y Growth Trend Analysis By Temperature Profile, 2021 to 2025
- Absolute $ Opportunity Analysis By Temperature Profile, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Biopharma Sponsors
- CROs
- Academic Centers
- Biopharma Sponsors
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Service Type
- By Trial Model
- By Delivery Mode
- By Temperature Profile
- By End User
- Competition Analysis
- Competition Deep Dive
- Marken
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- World Courier
- Thermo Fisher Scientific
- DHL Group
- Almac Group
- PCI Pharma Services
- Parexel
- Marken
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Trial Model, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Temperature Profile, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Trial Model, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Temperature Profile, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Trial Model, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Temperature Profile, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Trial Model, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Temperature Profile, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Trial Model, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Temperature Profile, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Trial Model, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Temperature Profile, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Trial Model, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Temperature Profile, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Trial Model, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Delivery Mode, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Temperature Profile, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Service Type
- Figure 6: Global Market Value Share and BPS Analysis by Trial Model, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Trial Model, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Trial Model
- Figure 9: Global Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Delivery Mode
- Figure 12: Global Market Value Share and BPS Analysis by Temperature Profile, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Temperature Profile, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Temperature Profile
- Figure 15: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by End User
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Service Type
- Figure 32: North America Market Value Share and BPS Analysis by Trial Model, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Trial Model, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Trial Model
- Figure 35: North America Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Delivery Mode
- Figure 38: North America Market Value Share and BPS Analysis by Temperature Profile, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Temperature Profile, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Temperature Profile
- Figure 41: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by End User
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Service Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Trial Model, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Trial Model, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Trial Model
- Figure 51: Latin America Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Delivery Mode
- Figure 54: Latin America Market Value Share and BPS Analysis by Temperature Profile, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Temperature Profile, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Temperature Profile
- Figure 57: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by End User
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Service Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Trial Model, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Trial Model, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Trial Model
- Figure 67: Western Europe Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Delivery Mode
- Figure 70: Western Europe Market Value Share and BPS Analysis by Temperature Profile, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Temperature Profile, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Temperature Profile
- Figure 73: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by End User
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Service Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Trial Model, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Trial Model, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Trial Model
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Delivery Mode
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Temperature Profile, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Temperature Profile, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Temperature Profile
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by End User
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Service Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Trial Model, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Trial Model, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Trial Model
- Figure 99: East Asia Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Delivery Mode
- Figure 102: East Asia Market Value Share and BPS Analysis by Temperature Profile, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Temperature Profile, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Temperature Profile
- Figure 105: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by End User
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Service Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Trial Model, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Trial Model, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Trial Model
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Delivery Mode
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Temperature Profile, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Temperature Profile, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Temperature Profile
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Service Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Trial Model, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Trial Model, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Trial Model
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Delivery Mode, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Delivery Mode, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Delivery Mode
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Temperature Profile, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Temperature Profile, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Temperature Profile
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

