Preclinical Medical Device Testing Services Market
The Preclinical Medical Device Testing Services Market is segmented by Service Type (Biocompatibility Testing, Sterility Testing, Toxicology Testing, Performance Testing, Safety Testing, Efficacy Testing, Other Preclinical Testing Services), Testing Service (Mechanical Testing, Electrical Testing, Biomechanical Testing, Biological Testing, Other Testing Services), Application (Medical Device Manufacturers, Pharmaceutical Companies, Research Institutions, Contract Research Organizations, Others), Device Class (Class I Devices, Class II Devices, Class III Devices), Device Type (Implantable Devices, Diagnostic Devices, Surgical Instruments, Wearable Devices, Single-Use Devices), and Region. Forecast for 2026 to 2036.
Preclinical Medical Device Testing Services Market Size, Market Forecast and Outlook By FMI
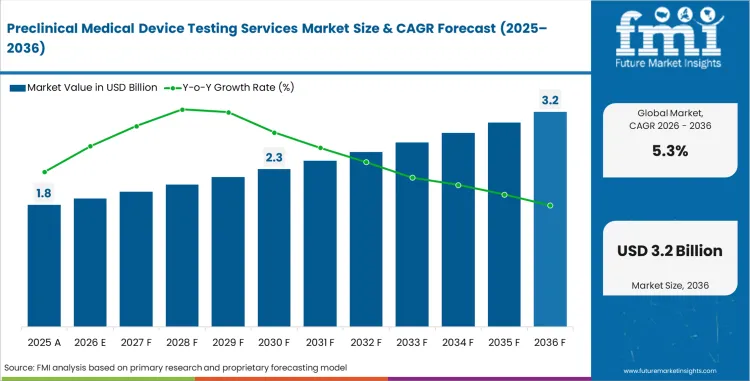
The preclinical medical device testing services market was valued at USD 1.7 billion in 2025. The market is set to reach USD 1.8 billion by 2026-end and expand at a CAGR of 5.3% between 2026 and 2036 to reach USD 3.0 billion by 2036. Medical device manufacturers will dominate with a 47.0% application share, and Class II devices will lead the device class segment with a 42.0% share.
Summary of the Preclinical Medical Device Testing Services Market
- Demand and Growth Drivers
- Regulatory evidence needs support the demand for preclinical testing services as device makers prepare safety records before clinical work.
- Complex device design boosts outsourced testing as implants and diagnostics need stronger performance validation.
- High-risk device programs reinforce specialist studies as biocompatibility and toxicology work carry higher technical depth.
- Product and Segment View
- Mechanical testing is expected to lead the testing service segment with 35.0% share in 2026, due to durability checks for surgical tools and implants.
- Medical device manufacturers are expected to lead application demand with 47.0% share in 2026, reflecting safety and performance filing needs.
- Biocompatibility testing is expected to hold high-value service demand as patient-contact materials need biological safety records.
- Geography and Competitive Outlook
- India is expected to record a CAGR of 8.1% through 2036, because of local medical device output and testing capacity.
- China is projected to expand at 7.6% CAGR through 2036, as it has high device standards and quality control needs.
- Companies combining deep regulatory expertise with global laboratory access are strengthening their market position during the forecast period.
- Analyst Opinion
- Sabyasachi Ghosh, Associate Vice President at FMI says, “Preclinical testing is becoming a evidence step for device makers seeking to avoid redesign and filing delays. Buyers want mechanical performance records and biological safety data in one study plan. Suppliers with regulatory skill and reliable reporting can reduce work.”
- Preclinical Medical Device Testing Services Market Value Analysis
- The preclinical medical device testing services market is moving from ad hoc validation work into a structured outsourced service base.
- Adoption is being due to the safety evidence needs for implants and active medical devices.
- The market is benefiting from deeper use of medical device analytical testing outsourcing across regulated device programs.
- Demand is supported by the need for test records meeting compliance requirements. These records ensure performance alignment and support efficient product launch timing.
Strategic Pointers: Preclinical Medical Device Testing Services Market
Preclinical Medical Device Testing Services Market is moving toward higher-value evidence planning as regulators tighten expectations after 2024. FDA’s 2025 ASCA annual report states that accredited testing improves consistency and predictability in premarket review, while FDA’s September 2025 ASCA database update showed that summary test reports from laboratories under “FDA Initiated Withdrawal” status are no longer accepted. FDA’s recognized standards database also added ISO 10993-4 Third edition 2017 with Amendment 1:2025 on December 22, 2025 for hemocompatibility test selection.
MDCG 2025-9 links breakthrough device review with stronger use of non-clinical and preclinical data. FDA’s 2026 Medical Device Development Tools page defines non-clinical assessment models as tools that measure or predict device safety, effectiveness, or performance. FDA’s July 2025 Medical Device Software Guidance Navigator also points to non-clinical testing needs for physiologic closed-loop control devices. Testing providers that combine active accreditation, updated ISO 10993 capability, toxicology review, model validation, and software-linked bench testing can shift from transactional lab work to managed preclinical evidence packages for device makers.
Preclinical Medical Device Testing Services Market Definition
The preclinical medical device testing services market includes laboratory testing services used before clinical evaluation or market filing. These services assess device safety and performance for implants, surgical tools, and diagnostic instruments. Scope includes biocompatibility testing, sterility testing, toxicology testing, and performance testing. Clinical trial operations and finished device sales are outside the scope.
Preclinical Medical Device Testing Services Market Inclusions
Market scope encompasses all commercially traded preclinical testing services categorized by service type, testing service, application, and region from 2026 to 2036.
Preclinical Medical Device Testing Services Market Exclusions
Scope does not include clinical trial management, patient diagnostic services, routine hospital equipment repair, or finished medical device sales.
Preclinical Medical Device Testing Services Market Research Methodology
- Primary Research: FMI analysts talked to medical device manufacturers, testing laboratories, and regulatory service specialists in important markets.
- Desk Research: Combined data from regulatory guidance, company service portfolios, and device testing requirements.
- Market sizing and forecasting: Added service use across testing services and application groups from the bottom up.
- Data validation: Cross-checked every three months against regulatory evidence needs and company service coverage.
Why is the Preclinical Medical Device Testing Services Market Growing?
- Device makers are using preclinical testing earlier to reduce redesign risk before clinical review begins.
- Testing partners are becoming important as device designs combine materials, electronics, and patient-contact functions.
- Manufacturers are outsourcing complex studies to laboratories with stronger methods and submission-ready documentation systems.
Medical device makers need stronger preclinical evidence, as their products carry greater patient safety exposure. FDA’s FY2026 standard user fee is USD 26,067 for 510(k) submissions and USD 579,272 for PMA submissions. This cost context makes early testing discipline important for device firms seeking to avoid repeat filing expense. Labs with reliable test design help manufacturers protect submission budgets and reduce preventable delays.
Global device variety is broad, and testing plans must match patient-contact risk. WHO notes an estimated 2 million different kinds of medical devices on the world market, categorized into more than 7,000 generic device groups. This broad device pool affects demand for specialized testing plans across materials and intended use. Testing providers gain importance as device makers need studies suited to each product format.
Market Segmentation Analysis
- Biocompatibility Testing is estimated to account for 31.0% of service type demand in 2026 with strong use across patient-contact devices.
- Mechanical Testing is expected to account for 35.0% of testing service demand in 2026 with higher use across implants and surgical tools.
- Medical Device Manufacturers are expected to account for 47.0% of application demand in 2026 with direct use across filing support.
- Class II Devices are estimated to account for 42.0% of device class demand in 2026 with broad testing needs across moderate-risk products.
- Implantable Devices are estimated to account for 38.0% of device type demand in 2026 with deeper need for safety and performance studies.
The market for preclinical medical device testing services is divided into five primary segment groups based on service type, testing service, application, device class, and device type. Service type includes Biocompatibility Testing, Sterility Testing, Toxicology Testing, Performance Testing, Safety Testing, Efficacy Testing, and Other Services. Testing service includes Mechanical Testing, Electrical Testing, Biomechanical Testing, Biological Testing, and Other Testing. Application includes Medical Device Manufacturers, Pharmaceutical Companies, Research Institutions, Contract Research Organizations, and Others. Device class includes Class I Devices, Class II Devices, and Class III Devices. Device type includes Implantable Devices, Diagnostic Devices, Surgical Instruments, Wearable Devices, and Single-Use Devices.
Insights into the Biocompatibility Testing Service Type Segment
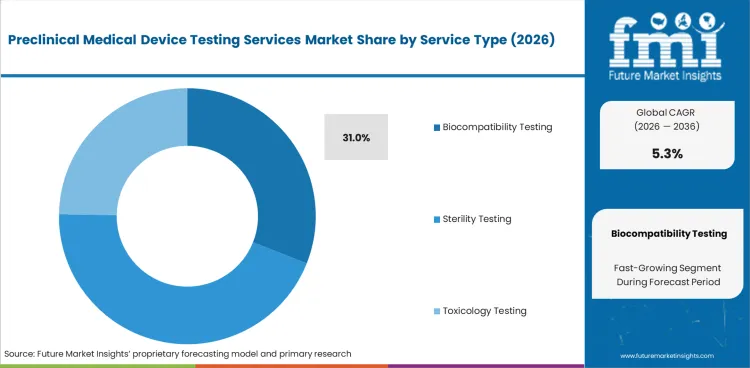
- In 2026, Biocompatibility Testing is estimated to make up 31.0% of the service type segment.
- Patient-contact devices need biological safety checks before filing, which supports demand for biocompatible materials validation.
Insights into the Mechanical Testing Service Segment
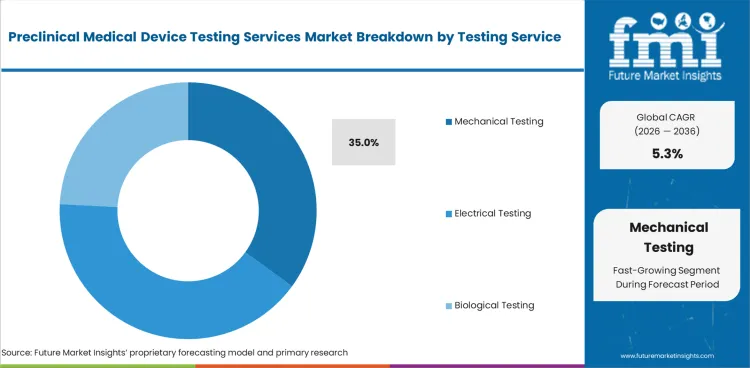
- Mechanical Testing is expected to hold 35.0% of the testing service segment in 2026.
- Load-bearing devices and surgical tools need strength evidence before clinical review and hospital use decisions.
Insights into the Medical Device Manufacturers Application Segment
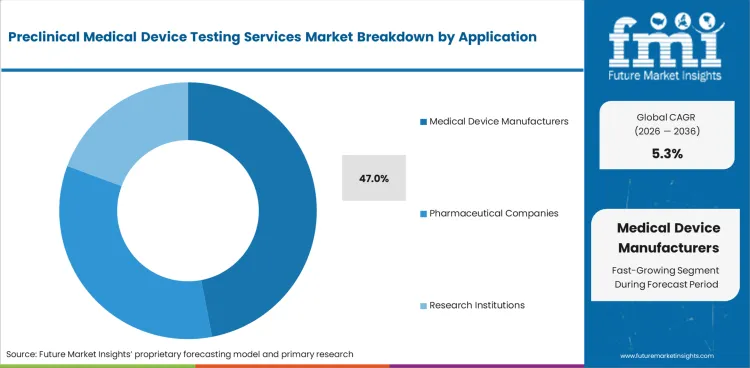
In 2026, Medical Device Manufacturers are poised to contribute to 47.0% share of the application segment.
Manufacturers use preclinical medical device testing services to prepare evidence packages before submission and launch planning.
Insights into the Class II Device Class Segment
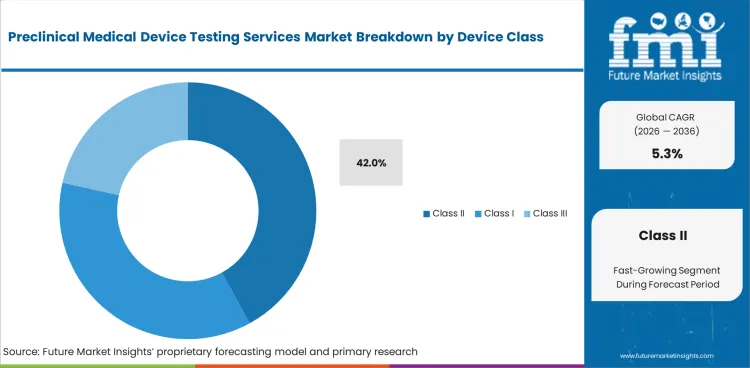
In 2026, Class II Devices are forecast to hold 42.0% of the device class segment.
Moderate-risk devices need wide verification work as electrical and biological checks often apply together.
Insights into the Implantable Device Type Segment
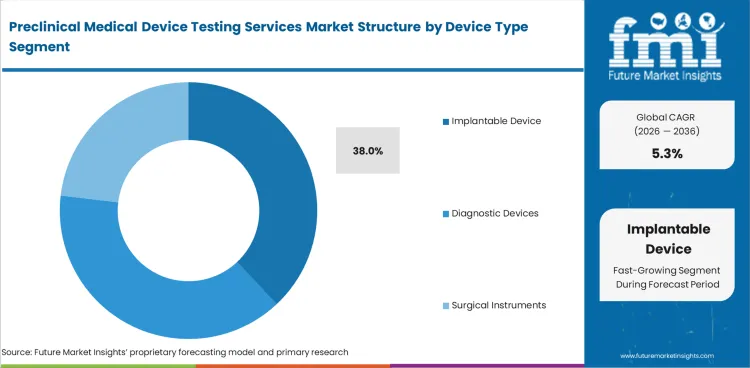
In 2026, Implantable Devices are projected to account 38.0% of the device type segment.
Implantable products need detailed study planning as long duration body contact raises testing depth and documentation burden.
Preclinical Medical Device Testing Services Market Drivers, Restraints, and Opportunities
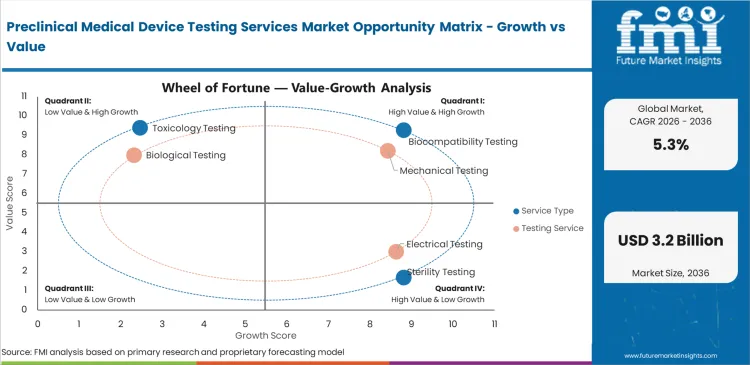
Regulatory filing pressure is creating structured demand for preclinical evidence across higher-risk device programs.
The requirement for advanced tools and skilled staff creates pricing pressure for smaller testing providers.
Connected devices create service opportunity through electrical safety and software validation needs.
The market for preclinical medical device testing services expands as device makers need stronger proof before clinical work. IMDRF’s June 2024 non-IVD submission guidance references pre-clinical and clinical evidence within regulatory submission content. This supports demand for study records connecting technical performance with biological safety. Testing providers with submission structure expertise can reduce gaps between lab results and reviewer expectations.
UK Testing Tariffs Are Still Firming
For preclinical medical device testing services, the clearest near-term price signal is not a sharp spike but steady service-cost firming. The Office for National Statistics’ April 2026 SPPI series for “M712 Technical testing and analysis services” moved from 132.1 in Q4 2025 to 133.4 in Q1 2026. This roughly 1.0% quarterly increase is important because biocompatibility and performance validation rely on qualified technical capacity. Safety testing and analytical review work are affected by specialized expertise beyond basic commodity inputs.
Lab Instrument Costs Keep Floor Elevated
Preclinical medical device testing prices are being held up by the cost of laboratory equipment used across analytical and safety-assessment workflows. FRED’s April 2026 BLS-linked series for “Laboratory Analytical Instruments” showed the index at 194.471 in December 2025 and 197.973 in March 2026, a rise of about 1.8% within the first quarter. This does not automatically translate one-for-one into higher test quotes, but it raises the operating-cost floor for laboratories running chemistry and materials characterization. It raises the cost floor for validation-heavy work. Device sponsors should treat quote inflation as a capacity-and-capex issue, not only a labor issue. They should avoid late-stage protocol changes, which lead to repeat instrument time.
Analysis of Preclinical Medical Device Testing Services Market By Key Countries
.webp)
| Country | CAGR |
|---|---|
| India | 8.1% |
| China | 7.6% |
| USA | 6.0% |
| Germany | 4.1% |
| UK | 3.1% |
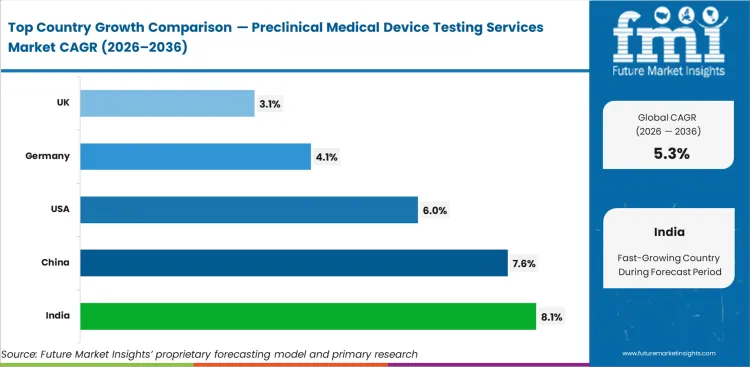
Source: Future Market Insights, 2026.
Preclinical Medical Device Testing Services Market CAGR Analysis By Country
India leads with an 8.1% CAGR through 2036 as medical device output and testing capacity expand.
China at 7.6% reflects device standards development and quality control needs across local manufacturers.
The USA at 6.0% reflects regulatory filing discipline and advanced device development.
Germany at 4.1% and the UK at 3.1% reflect mature service bases with controlled expansion.
Demand for preclinical medical device testing services is forecast to rise at 5.3% CAGR from 2026 to 2036. Country-level analysis covers the major markets contributing to this forecast, with the key markets listed below.
Demand Outlook for Preclinical Medical Device Testing Services Market in India
India’s preclinical medical device testing services industry is projected to record a CAGR of 8.1% through 2036. India’s medical devices sector was described in September 2025 as nearly USD 15 billion with over 800 MedTech start-ups. This manufacturing base supports demand for India preclinical medical device testing services. Local labs can gain from device makers who need credible studies before filing work.
Local device output supports demand for mechanical testing and biological safety studies.
The INR 500 crore scheme for strengthening medical device industry supports common infrastructure and clinical study support.
Domestic manufacturers need testing partners who can reduce rework across regulated submissions.
Sales Analysis of Preclinical Medical Device Testing Services Market in China
The preclinical medical device testing services sector in China is expected to expand at a CAGR of 7.6% during the assessment period. The standards-base in the country supports demand for China preclinical medical device testing services. Testing providers benefit from local manufacturers seeking quality records for domestic and export use.
Strong device standards support demand for structured validation and compliance testing.
Local device firms need reliable testing as Class II and Class III products carry higher review burden.
Demand Outlook for Preclinical Medical Device Testing Services Market in the United States
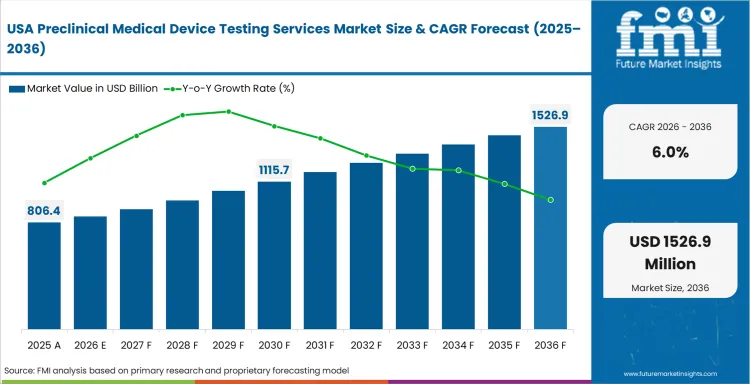
The USA preclinical medical device testing services industry is forecast to grow at a CAGR of 6.0% by 2036. Medical device manufacturers value testing partners with FDA evidence knowledge and study documentation expertise.
FDA aligned study design supports premium demand for complete device evidence packages.
Wearable devices and implants increase demand for electrical and biological validation.
Device complexity supports the use of biomedical tester systems in service laboratories.
Opportunity Analysis of Preclinical Medical Device Testing Services Market in Germany
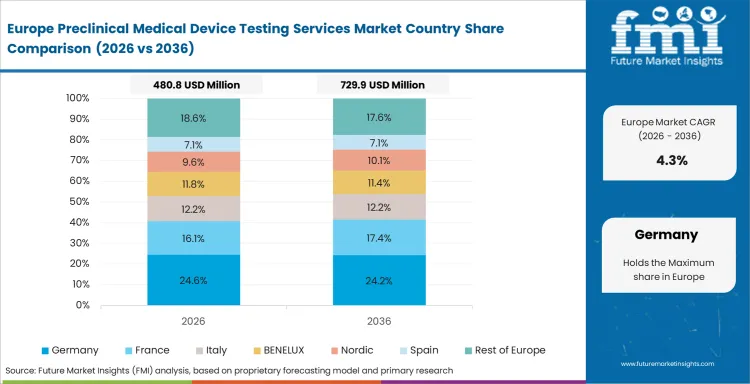
The preclinical medical device testing services segment in Germany is poised to advance at 4.1% CAGR over the forecast years. Established production base supports demand for Germany preclinical medical device testing services. Testing demand is strongest for precision devices, which need rigorous performance records.
Germany’s medtech production value reached EUR 46 billion in 2024 in the same official investment profile.
Precision device manufacturing supports demand for mechanical and biomechanical validation.
Advanced materials work supports testing needs across CF-PEEK for medical applications.
Future Outlook for Preclinical Medical Device Testing Services Market in the United Kingdom
The UK preclinical medical device testing services industry is expected to post 3.1% CAGR through 2036. MHRA approved 17% more clinical investigations in 2025 compared with 2024 for medical device testing. This favors demand for UK preclinical medical device testing services as developers prepare earlier evidence. Service providers with fast study planning and reliable records can support smaller innovators.
MHRA-backed clinical investigation activity supports demand for better preclinical packages.
UK clinical research KPIs listed a 97% latest figure for approved studies receiving combined review approval within the target timeline, against a 99% target.
Digital device development supports validation work linked to digital twins technologies in testing workflows.
Competitive Landscape and Strategic Positioning
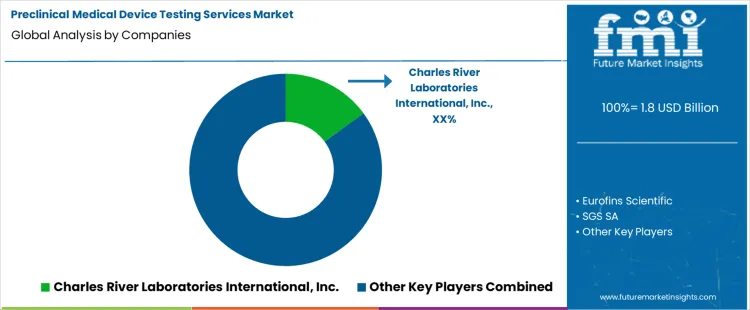
Preclinical Medical Device Testing Services Market Analysis By Company
- Charles River Laboratories International is a leading provider through broad preclinical service coverage and regulatory study support.
- Eurofins Scientific and SGS SA compete through global lab access and standardized testing systems.
- Specialist providers focus on biocompatibility and sterilization validation. They deliver device-specific study design.
Charles River Laboratories has strong positioning through integrated preclinical services across biocompatibility and toxicology work. Eurofins Scientific offers broad analytical and biological testing through global laboratory systems. SGS SA and Intertek Group Plc compete through mechanical testing and certification readiness. WUXI APPTEC adds integrated infrastructure and data systems for sponsor programs.
Barriers include high equipment cost and strict documentation needs across regulated device testing. Strategic priorities include method depth and faster reporting across long device development cycles. Buyers favor laboratories offering mechanical evidence and biological safety records while reducing filing uncertainty. Suppliers with broader testing menus can reduce coordination burden for manufacturers.
Key Companies in the Preclinical Medical Device Testing Services Market
Key global companies leading the preclinical medical device testing services market include:
- Charles River Laboratories International, Eurofins Scientific, and SGS SA have strong testing infrastructure for preclinical study design and global service delivery.
- Intertek Group Plc, WUXI APPTEC, and Laboratory Corporation of America Holdings serve manufacturers through multidisciplinary testing coverage and high-throughput reporting.
- Sotera Health, NAMSA, TÜV SÜD AG, and Pace Analytical Services LLC hold specialist positions across biocompatibility, sterilization validation, and chemical testing services.
Competitive Benchmarking: Preclinical Medical Device Testing Services Market
| Company | Regulatory Study Depth | Testing Portfolio Breadth | Global Lab Access |
|---|---|---|---|
| Charles River Laboratories International | High | High | Strong |
| Eurofins Scientific | High | High | Strong |
| SGS SA | High | High | Strong |
| Intertek Group Plc | High | High | Strong |
| WUXI APPTEC | High | High | Strong |
| Laboratory Corporation of America Holdings | Medium | Medium | Strong |
| Sotera Health | High | Medium | Moderate |
| NAMSA | High | Medium | Moderate |
| TÜV SÜD AG | High | Medium | Strong |
| Pace Analytical Services LLC | Medium | Medium | Moderate |
Source: Future Market Insights, 2026.
Key Developments in Preclinical Medical Device Testing Services Market
- January 2026, NAMSA completed the acquisition of Labcorp’s Early Development medical device testing business. The acquired portfolio includes biocompatibility testing, microbiological testing, analytical testing, and preclinical research, strengthening NAMSA’s medical device testing and preclinical service capacity in the United States.
- August 2025, Nelson Labs expanded its Wiesbaden, Germany facility with four new laboratories. The expansion added packaging testing services and strengthened microbiological testing capacity, including endotoxin, bioburden, EtO residual testing, and BI sterility, supporting medical device and pharmaceutical customers.
Key Players in the Preclinical Medical Device Testing Services Market
Major Global Players:
- Charles River Laboratories International
- Eurofins Scientific
- SGS SA
- Intertek Group Plc
- WUXI APPTEC
- Laboratory Corporation of America Holdings
- Sotera Health
Specialist Players:
- NAMSA
- TÜV SÜD AG
- Pace Analytical Services LLC
Report Scope and Coverage

Preclinical Medical Device Testing Services Market Breakdown By Service Type, Testing Service, And Region
| Parameter | Details |
| Quantitative Units | USD 1.8 billion to USD 3.0 billion, at a CAGR of 5.3% |
| Market Definition | The preclinical medical device testing services market encompasses laboratory studies used to assess device safety and performance before clinical evaluation or filing. |
| Regions Covered | North America, Latin America, Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | USA, UK, Germany, China, India, Japan, South Korea, and 30 plus countries |
| Key Companies Profiled | Charles River Laboratories International, Eurofins Scientific, SGS SA, Intertek Group Plc, WUXI APPTEC, Laboratory Corporation of America Holdings, Sotera Health, NAMSA, TÜV SÜD AG, Pace Analytical Services LLC |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology starting with service demand and verified regulatory use patterns. |
Preclinical Medical Device Testing Services Market by Segments
Preclinical Medical Device Testing Services Market Segmented by Service Type:
- Biocompatibility Testing
- Sterility Testing
- Toxicology Testing
- Performance Testing
- Safety Testing
- Efficacy Testing
- Other Preclinical Testing Services
Preclinical Medical Device Testing Services Market Segmented by Testing Service:
- Mechanical Testing
- Electrical Testing
- Biomechanical Testing
- Biological Testing
- Other Testing Services
Preclinical Medical Device Testing Services Market Segmented by Application:
- Medical Device Manufacturers
- Pharmaceutical Companies
- Research Institutions
- Contract Research Organizations
- Others
Preclinical Medical Device Testing Services Market Segmented by Device Class:
- Class I Devices
- Class II Devices
- Class III Devices
Preclinical Medical Device Testing Services Market Segmented by Device Type:
- Implantable Devices
- Diagnostic Devices
- Surgical Instruments
- Wearable Devices
- Single-Use Devices
Preclinical Medical Device Testing Services Market by Region:
- North America
- U.S.
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Europe
- Germany
- UK
- Italy
- Spain
- France
- Rest of Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Saudi Arabia
- Other GCC Countries
- South Africa
- Rest of Middle East and Africa
Research Sources and Bibliography
- NAMSA. (2026, January 8). NAMSA announces strategic acquisition of select assets of the Early Development medical device testing business of Labcorp.
- Nelson Laboratories, LLC. (2025, August 26). Nelson Labs expands laboratory capacity and services in Wiesbaden, Germany to meet growing demand for packaging and microbiological testing. Nelson Labs.
- Office for National Statistics. (2026, April 22). SPPI INDEX OUTPUT DOMESTIC - M712 Technical testing and analysis services 2015=100.
- Federal Reserve Bank of St. Louis. (2026, April 14). Producer Price Index by Commodity: Machinery and Equipment: Laboratory Analytical Instruments (WPU118603). FRED.
- Medicines and Healthcare products Regulatory Agency. (2026, February). UK medical device testing hits record high as MHRA backs growth in brain and AI technology.
- Food and Drug Administration. (2025, September). Medical Device User Fee Amendments: Fees. USA Food and Drug Administration.
- Press Information Bureau. (2025, September). India MedTech Expo 2025 at Bharat Mandapam from 4th to 6th September to showcase India’s MedTech strength and innovation. Government of India.
- Press Information Bureau. (2026, March). Medical Devices Manufacturers. Government of India.
- Germany Trade & Invest. (2025, September). The Medical Technology Industry in Germany: Issue 2025/2026. GTAI.
- Department of Health and Social Care. (2026, March). UK Clinical Research Delivery key performance indicators: Data to February 2026. GOV.UK.
- International Medical Device Regulators Forum. (2024, June). Non-In Vitro Diagnostic Device Regulatory Submission Table of Contents (nIVD ToC). IMDRF.
This Report Answers
- What is the current and future size of the preclinical medical device testing services market?
- How fast is the preclinical medical device testing services market expected to expand between 2026 and 2036?
- Which testing service is expected to lead the market by 2026?
- Which application segment is expected to account for the highest demand by 2026?
- What factors are driving demand for preclinical medical device testing services globally?
- How are regulatory evidence needs influencing adoption of outsourced testing?
- Why are medical device manufacturers the main demand base for testing services?
- How are advanced devices creating demand for specialized preclinical validation?
- Which countries are projected to record faster expansion through 2036?
- What is driving market expansion in India and China?
- Who are the key companies active in the preclinical medical device testing services market?
- How does FMI estimate and validate the market forecast?
Frequently Asked Questions
What is the global market demand for Preclinical Medical Device Testing Services in 2026?
In 2026, the global market for preclinical medical device testing services is expected to be worth USD 1.8 billion.
How big will the market for Preclinical Medical Device Testing Services be in 2036?
By 2036, the market for preclinical medical device testing services is expected to be worth USD 3.0 billion.
How much is demand for Preclinical Medical Device Testing Services expected to expand between 2026 and 2036?
How much is demand for Preclinical Medical Device Testing Services expected to expand between 2026 and 2036?
How much is demand for Preclinical Medical Device Testing Services expected to expand between 2026 and 2036?
Mechanical testing will make up 35.0% of the testing service segment in 2026.
What is causing demand to rise in India?
The India preclinical medical device testing services market is projected to record 8.1% CAGR through 2036, supported by medical device manufacturing and testing infrastructure.
What is causing demand to rise in China?
The China preclinical medical device testing services market is projected to expand at 7.6% CAGR through 2036, supported by standards development and quality control needs.
What does this report mean by Preclinical Medical Device Testing Services Market definition?
The preclinical medical device testing services market includes laboratory testing used to assess device safety and performance before clinical evaluation or filing.
How does FMI make the Preclinical Medical Device Testing Services forecast and check it?
Forecasting models use a hybrid bottom-up and top-down approach, starting with service demand and checking it against regulatory use patterns.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Service Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Service Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Service Type , 2026 to 2036
- Biocompatibility Testing
- Sterility Testing
- Toxicology Testing
- Biocompatibility Testing
- Y to o to Y Growth Trend Analysis By Service Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Service Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Testing Service
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Testing Service, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Testing Service, 2026 to 2036
- Mechanical Testing
- Electrical Testing
- Biological Testing
- Mechanical Testing
- Y to o to Y Growth Trend Analysis By Testing Service, 2021 to 2025
- Absolute $ Opportunity Analysis By Testing Service, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Medical Device Manufacturers
- Pharmaceutical Companies
- Research Institutions
- Medical Device Manufacturers
- Y to o to Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Device Class
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Device Class, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Device Class, 2026 to 2036
- Class II
- Class I
- Class III
- Class II
- Y to o to Y Growth Trend Analysis By Device Class, 2021 to 2025
- Absolute $ Opportunity Analysis By Device Class, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Device Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Device Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Device Type, 2026 to 2036
- Implantable Device
- Diagnostic Devices
- Surgical Instruments
- Implantable Device
- Y to o to Y Growth Trend Analysis By Device Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Device Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- By Country
- Market Attractiveness Analysis
- By Country
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Service Type
- By Testing Service
- By Application
- By Device Class
- By Device Type
- Competition Analysis
- Competition Deep Dive
- Charles River Laboratories International
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Eurofins Scientific
- SGS SA
- Intertek Group Plc
- WUXI APPTEC
- Charles River Laboratories International
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Testing Service, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Device Class, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Device Type, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Testing Service, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Device Class, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Device Type, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Testing Service, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Device Class, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Device Type, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Testing Service, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Device Class, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Device Type, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Testing Service, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Device Class, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Device Type, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Testing Service, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Device Class, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Device Type, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Testing Service, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Device Class, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Device Type, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Service Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Testing Service, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Device Class, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Device Type, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Service Type
- Figure 6: Global Market Value Share and BPS Analysis by Testing Service, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Testing Service, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Testing Service
- Figure 9: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Application
- Figure 12: Global Market Value Share and BPS Analysis by Device Class, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Device Class, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Device Class
- Figure 15: Global Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Device Type, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Device Type
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Service Type
- Figure 32: North America Market Value Share and BPS Analysis by Testing Service, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Testing Service, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Testing Service
- Figure 35: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Application
- Figure 38: North America Market Value Share and BPS Analysis by Device Class, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Device Class, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Device Class
- Figure 41: North America Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Device Type, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Device Type
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Service Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Testing Service, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Testing Service, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Testing Service
- Figure 51: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by Application
- Figure 54: Latin America Market Value Share and BPS Analysis by Device Class, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Device Class, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Device Class
- Figure 57: Latin America Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Device Type, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Device Type
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Service Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Testing Service, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Testing Service, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Testing Service
- Figure 67: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by Application
- Figure 70: Western Europe Market Value Share and BPS Analysis by Device Class, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Device Class, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Device Class
- Figure 73: Western Europe Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Device Type, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Device Type
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Service Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Testing Service, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Testing Service, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Testing Service
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by Application
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Device Class, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Device Class, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Device Class
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Device Type, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Device Type
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Service Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Testing Service, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Testing Service, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Testing Service
- Figure 99: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by Application
- Figure 102: East Asia Market Value Share and BPS Analysis by Device Class, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Device Class, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Device Class
- Figure 105: East Asia Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Device Type, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Device Type
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Service Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Testing Service, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Testing Service, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Testing Service
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Device Class, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Device Class, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Device Class
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Device Type, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Device Type
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Service Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Service Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Service Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Testing Service, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Testing Service, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Testing Service
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Device Class, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Device Class, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Device Class
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Device Type, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Device Type, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Device Type
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

