Medical Plastics Market
The medical plastics market is segmented by Application Type (Medical Devices, Pharmaceutical Packaging, Disposable Supplies, Laboratory Equipment, Others), Material Type (Polyethylene, Polypropylene, Polystyrene, Polyvinyl Chloride, Others), and End-Use Industry (Healthcare, Pharmaceuticals, Laboratory, Biotechnology, Others). Forecast for 2026 to 2036.
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Medical Plastics Market Size, Market Forecast and Outlook By FMI
The medical plastics market is expected to expand from USD 42.8 billion in 2026 to USD 73.6 billion by 2036. It is anticipated to register a 5.6% CAGR during the forecast period across global healthcare material demand. Medical devices are likely to lead with a 38.4% share of application type demand in 2026. Polyethylene is projected to account for 29.7% of material type demand in 2026 within resin selection.
Summary of the Medical Plastics Market
- Demand and Growth Drivers
- Hospital device makers are expected to increase medical plastic use as biocompatibility rules intensify across regulated product programs.
- Sterilization compatibility is likely to guide resin selection across devices and drug packaging for safer product handling.
- Traceability and clinical safety rules are anticipated to lift demand for certified grades across regulated healthcare supply chains.
- Product and Segment View
- Medical devices are expected to lead application demand as surgical instruments and diagnostic equipment require stable biocompatible polymers.
- Polyethylene is likely to lead material selection through chemical resistance and processing flexibility in high-volume healthcare products.
- Pharmaceutical packaging and disposable supplies are anticipated to maintain steady adoption across drug handling and infection control uses.
- Geography and Competitive Outlook
- India is expected to record the fastest country-level expansion as device manufacturing capacity increases across healthcare clusters.
- China is likely to expand through hospital modernization and stronger domestic production of medical consumables.
- Suppliers with validated material portfolios and reliable technical service are anticipated to gain preference from regulated manufacturers.
- Analyst Opinion
- Nikhil Kaitwadec, Principal consultant for Chemicals and Materials at FMI, suggests, “Medical plastics demand is expected to shift toward suppliers able to combine biocompatibility evidence with reliable processing quality. Resin producers with validated grades and steady supply systems are likely to gain stronger preference as device makers reduce material risk across regulated programs.”
- Medical Plastics Market Value Analysis
- The medical plastics market is likely to advance as healthcare manufacturers prioritize patient safety and reliable material performance.
- Demand is expected to rise as certified polymers gain use across devices and pharmaceutical packaging.
- Adoption is anticipated to increase through sterilization-compatible grades used in clinical and laboratory products.
- Spending is likely to be guided by resin quality validation and long-term supplier qualification across healthcare supply chains.
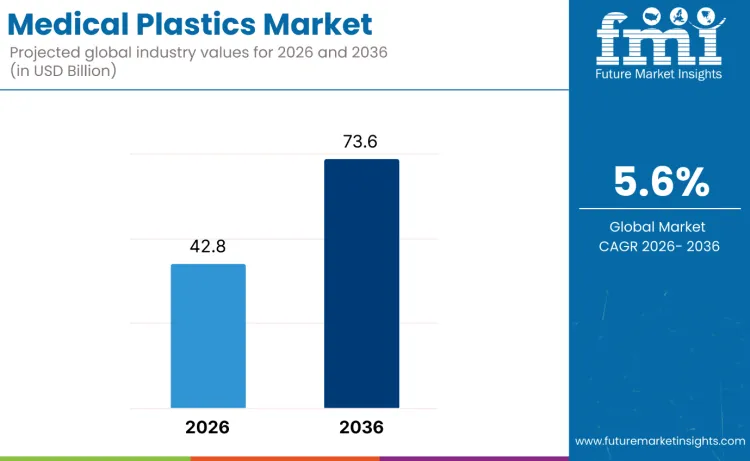
Medical Plastics Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 42.8 billion |
| Market Forecast Value (2036) | USD 73.6 billion |
| Forecast CAGR (2026 to 2036) | 5.6% |
What is the Trade Scenario for Medical Plastics?
On the exporting side, the United States is a leading supplier due to its large medical device industry and advanced polymer processing capabilities. Germany is a major exporter supported by precision medical manufacturing and strong regulatory compliance expertise. Japan plays a key role through high quality medical components and specialty plastics used in devices and disposables. China is a significant exporter driven by large scale production of medical consumables and cost competitive plastic components. Switzerland is notable for exporting high value medical grade plastics integrated into precision devices. Belgium and the Netherlands also serve as important export hubs due to their role in European medical supply chains and logistics networks. South Korea is increasingly prominent through advanced polymer technologies and medical manufacturing exports.
On the importing side, the United States remains one of the largest importers because of high healthcare consumption and demand for specialized medical plastics. China imports medical grade plastics and components for advanced devices and domestic manufacturing upgrades. Germany imports materials and components to support its large medical technology sector. India is a growing importer due to expanding healthcare infrastructure and medical device production. Brazil imports medical plastics to support hospital demand and local device assembly. Mexico is an important importer linked to cross border medical manufacturing supply chains. Saudi Arabia and the United Arab Emirates also import medical plastics to meet healthcare expansion and hospital modernization needs
How Is the Medical Plastics Market Structured by Application Type and Material Type?
Medical plastics play a critical role in delivering biocompatibility assurance, chemical resistance, and manufacturing performance across medical device packaging, pharmaceutical packaging, and diagnostic equipment applications. Adoption is influenced by safety requirements, regulatory specifications, processing compatibility, and end-use performance criteria. Segmentation by application type, material type, and end-use industry highlights how manufacturers select specific medical plastic grades to meet biocompatibility targets, chemical properties, and performance standards across diverse healthcare processes.
Which Application Type Holds the Largest Share in Medical Plastics?
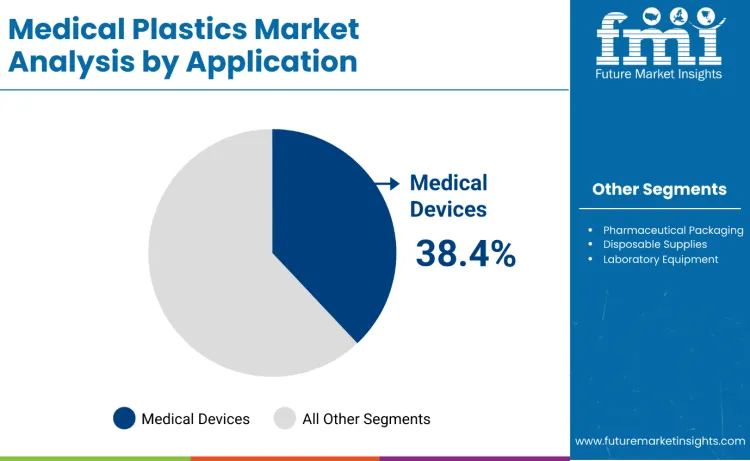
Medical devices accounts for 38.4%, driven by extensive use in surgical instruments, diagnostic equipment, and implantable devices requiring biocompatibility and chemical resistance. Pharmaceutical packaging holds 26.8%, supporting drug containers, delivery systems, and protective plastic healthcare packaging. Disposable supplies represents 19.3%, favored for syringes, catheters, and single-use medical products applications. Laboratory equipment contributes 10.7%, used in analytical instruments, sample containers, and testing apparatus. Other applications account for 4.8%.
Key Points:
- Medical devices dominate high-volume manufacturing applications.
- Pharmaceutical packaging supports drug safety and delivery requirements.
- Disposable supplies address infection control and convenience specifications.
How Do Material Type Requirements Influence Material Selection?
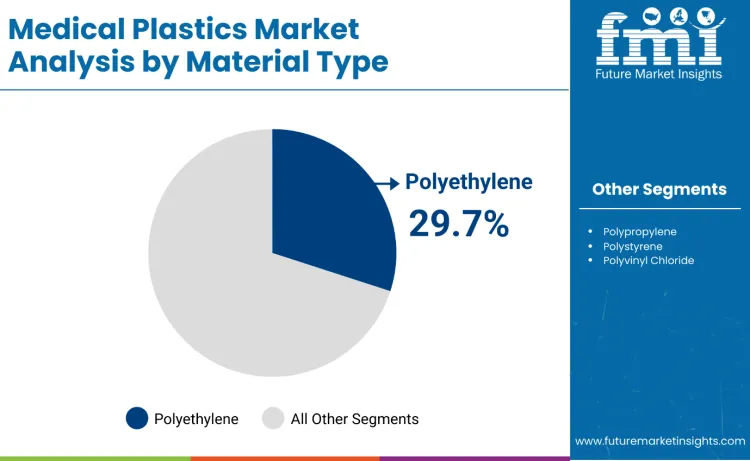
FMI is of the opinion that polyethylene represents 29.7%, reflecting priority on applications requiring chemical resistance and processing flexibility characteristics. Polypropylene accounts for 24.1%, suitable for sterilizable applications requiring heat resistance and cost effectiveness. Polystyrene contributes 18.6%, used in laboratory and diagnostic applications requiring clarity and dimensional stability. Polyvinyl chloride represents 15.2%, favored for flexible tubing and medical bags applications. Other materials account for 12.4%.
Key Points:
- Polyethylene grades remain the primary market segment.
- Polypropylene serves sterilization-compatible applications.
- Material selection depends on biocompatibility requirements and processing needs.
What are the Key Dynamics in the Medical Plastics Market?
Demand in the medical plastics market arises from manufacturers seeking to improve biocompatibility performance, chemical resistance, and material safety in applications where conventional plastics prove insufficient. Activity is notable in medical device packaging, pharmaceutical packaging operations, and laboratory equipment manufacturers where high-performance medical plastics address regulatory requirements, patient safety needs, and quality control standards. Material selection centers on chemical composition, biological compatibility, and sterilization resistance that support manufacturing performance without compromising patient safety efficiency.
How do Performance Requirements In Healthcare Affect Market Growth?
Market uptake grows where biocompatibility performance roles of medical plastics are critical to product acceptance. In healthcare applications, materials must maintain consistent biological compatibility and chemical resistance levels to achieve regulatory specifications and patient safety requirements. High-risk medical applications use premium medical plastic grades to ensure biocompatibility performance during service operations, preventing adverse reactions while maintaining efficient manufacturing characteristics. Healthcare developers depend on consistent medical plastic performance to deliver patient safety, regulatory compliance, and manufacturing efficiency comparable to established healthcare standards. Producers evaluate performance under sterilization exposure and biological contact to ensure stable properties through various service conditions.
What Supply Constraints Influence Medical Plastics Demand?
Broader deployment is shaped by the availability of certified raw materials and regulatory approval limitations. Some regions experience supply constraints due to limited specialty polymer availability or competing demand from industrial and consumer applications, requiring careful sourcing strategies and supplier qualification processes. Regional differences in regulatory standards and certification requirements affect where producers can access premium medical plastics without triggering complex validation challenges. Cost considerations arise when raw material prices and processing requirements are needed to balance material quality, regulatory compliance, and delivered cost outcomes in healthcare operations requiring consistent material specifications.
How Is the Medical Plastics Market Evolving Globally?
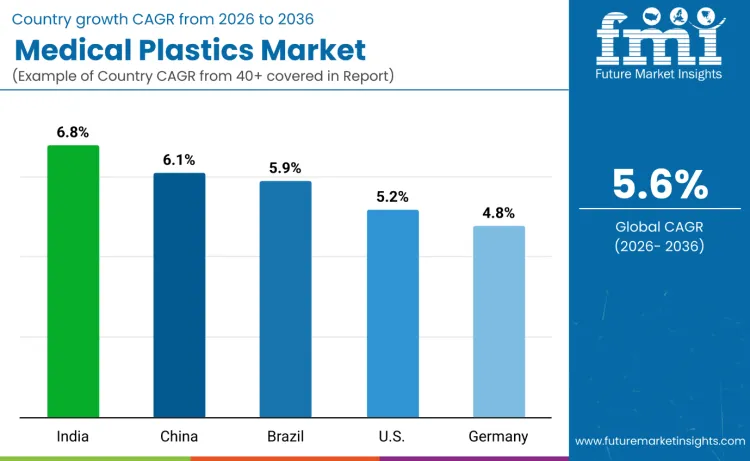
Global demand for medical plastic bags is increasing as manufacturers expand production capacity while addressing medical device, pharmaceutical packaging, and laboratory equipment application requirements. Growth reflects rising use of high-performance medical plastic grades, advanced processing methods, and quality-controlled supply chains across healthcare facilities, pharmaceutical manufacturers, and diagnostic laboratories. Material selection focuses on chemical composition, biological compatibility, and sterilization performance under various service scenarios. India records 6.8% CAGR, China records 6.1% CAGR, Brazil records 5.9% CAGR, USA records 5.2% CAGR, and Germany records 4.8% CAGR. Adoption remains driven by biocompatibility quality requirements and regulatory efficiency rather than volume expansion alone.
| Country | CAGR (%) |
|---|---|
| India | 6.8% |
| China | 6.1% |
| Brazil | 5.9% |
| USA | 5.2% |
| Germany | 4.8% |
What is Driving Growth of Medical Plastics Demand in India?
Demand for medical plastics in India is expanding as healthcare facilities develop manufacturing capabilities and quality improvement programs. Growth at 6.8% CAGR reflects rising use of high-performance medical plastic grades, medical device manufacturing applications, and pharmaceutical packaging systems addressing domestic and export market requirements. Material quality improvement under service conditions remains critical for regulatory efficiency and product acceptance. Cost considerations encourage medical plastic grades delivering biocompatibility performance and chemical resistance at competitive price points. Healthcare companies prioritize materials compatible with existing manufacturing infrastructure and sterilization capabilities. Demand concentrates within medical device clusters, pharmaceutical zones, and laboratory facilities serving domestic and international markets.
- Expansion of healthcare manufacturing capabilities
- Need for medical device and pharmaceutical packaging solutions
- Cost-effective high-performance medical plastic materials
- Growth of domestic and export healthcare manufacturing capacity
Why is China Seeing Expansion in Medical Plastics Adoption?
Medical plastics demand in China is rising as manufacturing systems expand healthcare programs and quality enhancement initiatives. Growth at 6.1% CAGR reflects strong activity in medical device manufacturing, pharmaceutical packaging, and laboratory equipment applications. Large-scale healthcare infrastructure development is driving demand for materials delivering biocompatibility stability, chemical performance, and processing efficiency. Healthcare facilities increase utilization of premium medical plastic grades due to regulatory requirements and export market specifications. Domestic healthcare policies support advanced manufacturing and material safety standards. Demand remains centered on major healthcare regions and integrated manufacturing complexes serving national and international markets.
- Expansion of healthcare and manufacturing programs
- Healthcare system support for advanced material processing
- Growth of medical device manufacturing and pharmaceutical packaging services
- Development of specialty processing capabilities
What Factors are Shaping Medical Plastics Sales in Brazil?
Sales of medical plastics in Brazil are increasing as manufacturers expand healthcare facilities and quality improvement capabilities aligned with medical device development and pharmaceutical packaging needs. Growth at 5.9% CAGR reflects rising use in medical device manufacturing, pharmaceutical packaging, and laboratory equipment systems. Material performance under service conditions and supply chain reliability drives technology selection. Manufacturing financing mechanisms increase access to high-quality medical plastics for qualifying healthcare facilities. Healthcare programs focus on biocompatibility and regulatory compliance to demonstrate material value. Demand remains strongest within healthcare centers and manufacturing facilities serving regional markets.
- Expansion of healthcare and manufacturing facilities
- Manufacturing access to premium medical plastic materials
- Focus on biocompatibility and regulatory compliance
- Growth of medical device development and pharmaceutical packaging services
How is U.S. Supporting Growth of Medical Plastics Demand?
Medical plastics market demand in U.S. is advancing as manufacturing systems expand healthcare capabilities across medical device projects, pharmaceutical packaging, and laboratory equipment applications. Growth at 5.2% CAGR reflects strong use in surgical devices, drug delivery systems, and diagnostic equipment applications. Advanced manufacturing operations introduce complex regulatory scenarios requiring consistent medical plastic composition and processing reliability. Manufacturing strategies prioritize materials with proven biocompatibility characteristics and supply chain stability. Major healthcare facilities emphasize material specifications and quality control protocols to optimize processing efficiency. Demand is driven by performance evidence and regulatory effectiveness rather than raw material availability trends.
- Expansion of healthcare and pharmaceutical manufacturing programs
- Advanced medical facility management
- Focus on biocompatibility characteristics and supply reliability
- Growth of specialty processing centers
What is Driving Expansion of Medical Plastics Demand in Germany?
Demand for medical plastics in Germany is rising as healthcare programs expand material quality capabilities across medical device projects, pharmaceutical packaging, and specialty laboratory applications. Growth at 4.8% CAGR reflects utilization driven by regulatory standards and performance-based material protocols. Material consistency under long-term service scenarios influences technology selection and healthcare management approaches. Manufacturers prioritize systems supporting processing efficiency, biocompatibility, and chemical resistance. Quality acceptance standards elevate focus on chemical composition, biological safety, and supply reliability. Demand remains tied to healthcare program development and manufacturing volume rather than general industrial material expansion.
- Regulatory standard-driven material utilization
- Expansion of healthcare and manufacturing programs
- Focus on processing efficiency and biocompatibility
- Performance-based material supply protocols
Who are the Prominent Players Active in Medical Plastics Landscape Globally?
BASF SE holds leading positioning through integrated medical plastic operations designed to deliver chemical purity, biological consistency, and supply reliability capabilities. Dow Inc. competes by combining high-grade medical plastic materials with comprehensive quality control systems supporting biocompatibility performance across diverse healthcare applications. Covestro AG supports demand through specialized medical plastic grades aligned with medical device and pharmaceutical packaging requirements. SABIC maintains relevance by supplying high-performance medical plastic materials enhancing biocompatibility stability and chemical resistance potential.
Evonik Industries AG participates with medical plastic products emphasizing chemical composition and processing integration. Celanese Corporation provides materials focusing on sterilization resistance and mechanical properties. Solvay S.A. delivers specialty grades for demanding applications. Arkema S.A. supports market needs through advanced processing capabilities. Competitive differentiation centers on material safety, supply reliability, technical specifications, and depth of healthcare collaboration.
Quality assessment includes molecular weight, extractable compounds, processing compatibility, and integration with existing healthcare protocols. Procurement behavior reflects extensive biological validation, supply chain reliability requirements, and reliance on suppliers offering technical support and consistent material specifications. Trends in the medical plastics market reflect emphasis on material safety and processing efficiency in healthcare, pharmaceutical, and laboratory applications.
Key Players in the Medical Plastics Market
- BASF SE
- Dow Inc.
- Covestro AG
- SABIC
- Evonik Industries AG
- Celanese Corporation
- Solvay S.A.
- Arkema S.A.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Application Type | Medical Devices; Pharmaceutical Packaging; Disposable Supplies; Laboratory Equipment; Others |
| Material Type | Polyethylene; Polypropylene; Polystyrene; Polyvinyl Chloride; Others |
| End-Use Industry | Healthcare; Pharmaceuticals; Laboratory; Biotechnology; Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, Brazil, USA, Germany, and 40+ countries |
| Key Companies Profiled | BASF SE; Dow Inc.; Covestro AG; SABIC; Evonik Industries AG; Celanese Corporation; Solvay S.A.; Arkema S.A.; Others |
| Additional Attributes | Dollar sales by application type, material type, and end-use industry; performance in biocompatibility and chemical resistance across medical device, pharmaceutical packaging, and laboratory equipment matrices; chemical composition improvement, processing efficiency enhancement, and safety benefit under manufacturing operations; impact on patient safety, regulatory compliance, and contamination reduction during service operations; compatibility with sterilization systems and quality control targets; procurement dynamics driven by healthcare expansion, quality improvement programs, and long-term material supply partnerships. |
Medical Plastics Market by Segment
Application Type:
- Medical Devices
- Pharmaceutical Packaging
- Disposable Supplies
- Laboratory Equipment
- Others
Material Type:
- Polyethylene
- Polypropylene
- Polystyrene
- Polyvinyl Chloride
- Others
End-Use Industry:
- Healthcare
- Pharmaceuticals
- Laboratory
- Biotechnology
- Others
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- International Medical Plastics Association. (2023). Medical Plastics Quality Standards for Healthcare Applications: Technical Guidelines. IMPA.
- Medical Device Manufacturing Institute. (2023). Material Specifications for Medical Grade Plastics: Chemical Composition and Biocompatibility Properties. MDMI.
- American Society for Testing and Materials. (2024). ASTM Guidelines for Medical Plastics Selection in Healthcare and Pharmaceutical Manufacturing (updated material specifications for biocompatibility and sterilization applications). ASTM.
- Thompson, M. L., et al. (2023). Chemical composition effects of high-performance medical plastics on healthcare efficiency: A comprehensive analysis of medical device and pharmaceutical packaging applications. Journal of Medical Materials Technology, 28, 112-129.
- Davis, P. R., et al. (2023). Seventh annual medical plastics performance report: Special focus on supply chain reliability and processing efficiency in healthcare manufacturing. International Healthcare Materials Review, 25, 1-21.
Frequently Asked Questions
How big is the medical plastics market in 2026?
The global medical plastics market is estimated to be valued at USD 42.8 billion in 2026.
What will be the size of the medical plastics market in 2036?
The market size for the medical plastics market is projected to reach USD 73.6 billion by 2036.
How much will the medical plastics market grow between 2026 and 2036?
The medical plastics market is expected to grow at a 5.6% CAGR between 2026 and 2036.
What are the key application types in the medical plastics market?
The key application types in the medical plastics market include medical devices, pharmaceutical packaging, disposable supplies, laboratory equipment, and other healthcare applications.
Which segment will contribute a significant share in the medical plastics market in 2026?
In terms of application type, the medical devices segment is set to command a 38.4% share in the medical plastics market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual Reports
- Industry Journals and Publications
- Medical and Healthcare Associations
- Government Statistics
- Trade and Association Data
- Academic Literature
- Press Releases
- Paid Databases
- Internal Knowledge Repository
- Expert Input and Fieldwork (Primary Evidence)
- Qualitative Interviews
- Quantitative Surveys
- Hybrid Research Approach
- Governance, Ethics, and Data Stewardship
- Desk Research Programme (Secondary Evidence)
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter's Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Medical Plastics Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Medical Plastics Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Y-o-Y Growth Trend Analysis 2021 to 2025
- Absolute $ Opportunity Analysis 2026 to 2036
- Global Medical Plastics Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Application Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Application Type, 2026 to 2036
- Medical Devices
- Pharmaceutical Packaging
- Disposable Supplies
- Laboratory Equipment
- Others
- Y-o-Y Growth Trend Analysis By Application Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Application Type, 2026 to 2036
- Global Medical Plastics Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Material Type, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Analysis and Forecast By Material Type, 2026 to 2036
- Polyethylene
- Polypropylene
- Polystyrene
- Polyvinyl Chloride
- Others
- Y-o-Y Growth Trend Analysis By Material Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Material Type, 2026 to 2036
- Global Medical Plastics Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Billion) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Billion) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Medical Plastics Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Latin America Medical Plastics Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Western Europe Medical Plastics Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Eastern Europe Medical Plastics Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- East Asia Medical Plastics Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- South Asia and Pacific Medical Plastics Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Middle East & Africa Medical Plastics Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- Competition Analysis
- BASF SE
- Dow Inc.
- Covestro AG
- SABIC
- Evonik Industries AG
- Celanese Corporation
- Solvay S.A.
- Arkema S.A.
- Assumptions & Acronyms Used
List of Tables
- Medical Plastics Market Size (USD Billion), 2021-2036
- Medical Plastics Market Y-o-Y Growth (%), 2021-2036
- Medical Plastics Market Absolute $ Opportunity Analysis (USD Billion), 2026-2036
- Medical Plastics Market Value Chain Analysis
- Medical Plastics Market Supply Chain Analysis
- Medical Plastics Market Investment Feasibility Matrix
- Medical Plastics Market PESTLE Analysis
- Medical Plastics Market Porter's Five Forces Analysis
- Medical Plastics Market Product Life Cycle Analysis
- Medical Plastics Market Opportunity Map Analysis
- Global Medical Plastics Market Share (%) By Application Type, 2026
- Global Medical Plastics Market Share (%) By Material Type, 2026
- Global Medical Plastics Market Share (%) By Region, 2026
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value Share and BPS Analysis by Plastic Type , 2025 and 2035
- Figure 4: Global Market Y to o to Y Growth Comparison by Plastic Type , 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Plastic Type
- Figure 6: Global Market Value Share and BPS Analysis by End Use, 2025 and 2035
- Figure 7: Global Market Y to o to Y Growth Comparison by End Use, 2025-2035
- Figure 8: Global Market Attractiveness Analysis by End Use
- Figure 9: Global Market Value Share and BPS Analysis by Processing Technology, 2025 and 2035
- Figure 10: Global Market Y to o to Y Growth Comparison by Processing Technology, 2025-2035
- Figure 11: Global Market Attractiveness Analysis by Processing Technology
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 13: Global Market Y to o to Y Growth Comparison by Region, 2025-2035
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2025-2035
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2025-2035
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2025-2035
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2025-2035
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2025-2035
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2025-2035
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 23: North America Market Value Share and BPS Analysis by Plastic Type , 2025 and 2035
- Figure 24: North America Market Y to o to Y Growth Comparison by Plastic Type , 2025-2035
- Figure 25: North America Market Attractiveness Analysis by Plastic Type
- Figure 26: North America Market Value Share and BPS Analysis by End Use, 2025 and 2035
- Figure 27: North America Market Y to o to Y Growth Comparison by End Use, 2025-2035
- Figure 28: North America Market Attractiveness Analysis by End Use
- Figure 29: North America Market Value Share and BPS Analysis by Processing Technology, 2025 and 2035
- Figure 30: North America Market Y to o to Y Growth Comparison by Processing Technology, 2025-2035
- Figure 31: North America Market Attractiveness Analysis by Processing Technology
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 33: Latin America Market Value Share and BPS Analysis by Plastic Type , 2025 and 2035
- Figure 34: Latin America Market Y to o to Y Growth Comparison by Plastic Type , 2025-2035
- Figure 35: Latin America Market Attractiveness Analysis by Plastic Type
- Figure 36: Latin America Market Value Share and BPS Analysis by End Use, 2025 and 2035
- Figure 37: Latin America Market Y to o to Y Growth Comparison by End Use, 2025-2035
- Figure 38: Latin America Market Attractiveness Analysis by End Use
- Figure 39: Latin America Market Value Share and BPS Analysis by Processing Technology, 2025 and 2035
- Figure 40: Latin America Market Y to o to Y Growth Comparison by Processing Technology, 2025-2035
- Figure 41: Latin America Market Attractiveness Analysis by Processing Technology
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 43: Western Europe Market Value Share and BPS Analysis by Plastic Type , 2025 and 2035
- Figure 44: Western Europe Market Y to o to Y Growth Comparison by Plastic Type , 2025-2035
- Figure 45: Western Europe Market Attractiveness Analysis by Plastic Type
- Figure 46: Western Europe Market Value Share and BPS Analysis by End Use, 2025 and 2035
- Figure 47: Western Europe Market Y to o to Y Growth Comparison by End Use, 2025-2035
- Figure 48: Western Europe Market Attractiveness Analysis by End Use
- Figure 49: Western Europe Market Value Share and BPS Analysis by Processing Technology, 2025 and 2035
- Figure 50: Western Europe Market Y to o to Y Growth Comparison by Processing Technology, 2025-2035
- Figure 51: Western Europe Market Attractiveness Analysis by Processing Technology
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Plastic Type , 2025 and 2035
- Figure 54: Eastern Europe Market Y to o to Y Growth Comparison by Plastic Type , 2025-2035
- Figure 55: Eastern Europe Market Attractiveness Analysis by Plastic Type
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by End Use, 2025 and 2035
- Figure 57: Eastern Europe Market Y to o to Y Growth Comparison by End Use, 2025-2035
- Figure 58: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Processing Technology, 2025 and 2035
- Figure 60: Eastern Europe Market Y to o to Y Growth Comparison by Processing Technology, 2025-2035
- Figure 61: Eastern Europe Market Attractiveness Analysis by Processing Technology
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 63: East Asia Market Value Share and BPS Analysis by Plastic Type , 2025 and 2035
- Figure 64: East Asia Market Y to o to Y Growth Comparison by Plastic Type , 2025-2035
- Figure 65: East Asia Market Attractiveness Analysis by Plastic Type
- Figure 66: East Asia Market Value Share and BPS Analysis by End Use, 2025 and 2035
- Figure 67: East Asia Market Y to o to Y Growth Comparison by End Use, 2025-2035
- Figure 68: East Asia Market Attractiveness Analysis by End Use
- Figure 69: East Asia Market Value Share and BPS Analysis by Processing Technology, 2025 and 2035
- Figure 70: East Asia Market Y to o to Y Growth Comparison by Processing Technology, 2025-2035
- Figure 71: East Asia Market Attractiveness Analysis by Processing Technology
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Plastic Type , 2025 and 2035
- Figure 74: South Asia and Pacific Market Y to o to Y Growth Comparison by Plastic Type , 2025-2035
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Plastic Type
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2025 and 2035
- Figure 77: South Asia and Pacific Market Y to o to Y Growth Comparison by End Use, 2025-2035
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Processing Technology, 2025 and 2035
- Figure 80: South Asia and Pacific Market Y to o to Y Growth Comparison by Processing Technology, 2025-2035
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Processing Technology
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Plastic Type , 2025 and 2035
- Figure 84: Middle East & Africa Market Y to o to Y Growth Comparison by Plastic Type , 2025-2035
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Plastic Type
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2025 and 2035
- Figure 87: Middle East & Africa Market Y to o to Y Growth Comparison by End Use, 2025-2035
- Figure 88: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Processing Technology, 2025 and 2035
- Figure 90: Middle East & Africa Market Y to o to Y Growth Comparison by Processing Technology, 2025-2035
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Processing Technology
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

