Medical Specialty Bag Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
The Medical Specialty Bag Market is segmented by Product Type (Ostomy Bags, Urinary Bags, IV Fluid Bags, Blood Bags, Drainage Bags, and Feeding Bags), Material Type (PVC, Polyolefin, Silicone, and Nonwoven), End User (Hospitals, Homecare, Ambulatory Centers, and Clinics), Usage Type (Disposable and Reusable), and Distribution Channel (Direct Sales, Distributors, Online Sales, and Retail Pharmacies ). Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Medical Specialty Bag Market Size, Market Forecast and Outlook By FMI
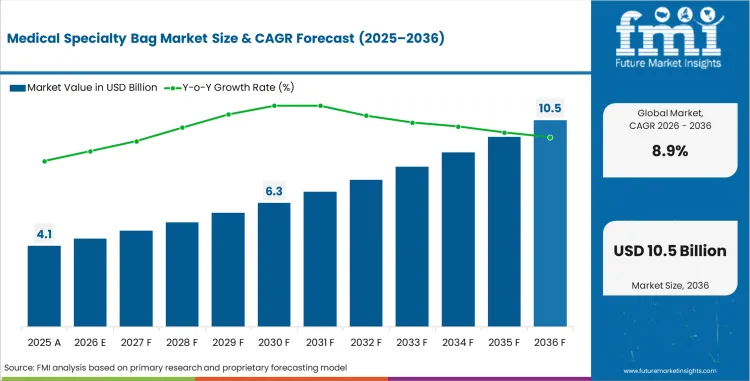
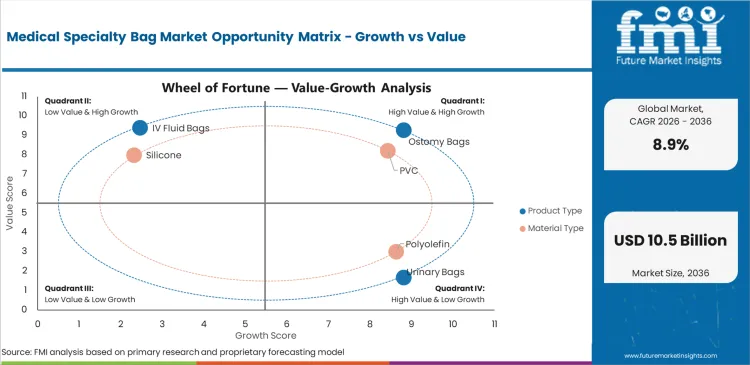
Medical specialty bags support fluid collection and fluid delivery across hospital care and home-based treatment. The market is valued at USD 3.8 billion in 2025 and is projected to reach USD 4.1 billion in 2026. Sector is forecast to reach USD 9.6 billion by 2036 at 8.9% CAGR. Ostomy bags are forecast to account for 34.0% share in 2026 and PVC is expected to represent 42.0% share.
Summary of the Medical Specialty Bag Market
- Demand Drivers
- Ostomy care supports repeat purchasing since many patients need pouch replacement throughout daily life.
- Hospital fluid handling supports demand for IV fluid bags across surgery and inpatient care.
- Urinary drainage use is expected to rise with older patient groups and post-surgical recovery needs.
- Homecare channels increase product movement as patients shift from hospital stays to supervised self-care.
- Product and Segment View
- Ostomy bags are projected to account for 34.0% share in 2026 with repeat use across colostomy and ileostomy care.
- PVC is expected to represent 42.0% share in 2026 as cost and processing ease support large-volume supply.
- Disposable bags are projected to account for 71.0% share in 2026 with infection control shaping hospital choice.
- Hospitals are expected to hold 45.0% share in 2026 as surgery and inpatient care use more than one bag type.
- Geography and Competitive Outlook
- India is projected at 10.7% CAGR through 2036 as hospital expansion and homecare access lift product need.
- China is expected to expand at 10.2% CAGR through 2036 with large patient volumes and domestic production scale.
- Leading companies include Coloplast and Convatec in ostomy care and B. Braun and Baxter offer fluid bags.
- Specialist suppliers are focusing on application design and local tenders across medical disposables.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Analyst at FMI, states “Medical specialty bags are no longer single‑use hospital consumables but a care‑continuity product category spanning surgery and home recovery. Demand is shaped by two forces: cost and sterility efficiency in hospitals, and comfort in homecare. Leak control reliability and medical‑grade material sourcing are now core drivers of buyer trust. Reimbursement rules and tender pricing increasingly act together to decide product flow across channels.”
- Medical Specialty Bag Market Value Analysis
- Market expansion is driven by ostomy bags, where long‑term patient use supports repeat purchasing and sustains revenue growth across care settings.
- Hospital‑based demand continues to anchor market growth, as rising surgery volumes and inpatient treatment expand fluid‑collection requirements.
- Distributor‑led channels support market expansion by enabling supplier access to public tenders and multi‑site hospital networks.
- Margin pressure in PVC bags shapes competitive expansion strategies, pushing suppliers to balance scale and supply reliability while protecting volume growth.
Medical Specialty Bag Market Definition
The medical specialty bag market includes ostomy bags and urinary drainage bags for fluid collection. It includes IV fluid bags and blood bags used for delivery and storage in medical care. Scope includes enteral feeding bags and wound drainage bags used in hospitals and homecare. The scope excludes general retail bags and non-medical storage pouches without clinical use approval.
Medical Specialty Bag Market Inclusions
The scope includes ostomy bags and urinary bags with IV fluid bags and blood bags across PVC and polyolefin materials for hospitals and homecare users.
Medical Specialty Bag Market Exclusions
The scope excludes non-medical plastic bags and general packaging pouches along with syringes and rigid containers outside fluid handling use.
Medical Specialty Bag Market Research Methodology
- Primary Research: FMI analysts reviewed product use patterns with hospital buyers and homecare channel participants. Discussions covered leak performance and sterility needs across ostomy care and fluid management.
- Desk Research: Desk research reviewed device classifications and company product portfolios. FDA guidance supported scope selection. European medical device guidance was reviewed separately.
- Market Sizing and Forecasting: The sizing model combined product use rates and average selling prices across key care settings. Country forecasts used hospital access and chronic care demand as adjustment factors.
- Data Validation: Forecasts were checked against product portfolios. Recent company launches were reviewed separately. Segment shares were reviewed against buyer patterns across hospitals and homecare channels.
Why is the Medical Specialty Bag Market Expanding?
- Ostomy procedures propels demand for medical specialty bags since pouch changes are part of long-term patient care.
- IV fluid handling raises product requirement as hospitals maintain ready-to-use sterile delivery systems.
- Homecare programs raise bag consumption as patients manage drainage and nutrition outside hospital settings.
- Infection control favors disposable bags as hospitals reduce cleaning steps and contamination risk.
Hospital use forms the first demand base for medical specialty bags since fluid handling appears across surgery and inpatient recovery. FDA shortage tracking in October 2024 brought national attention to IV fluid supply after hurricane disruption affected production. This pressure made IV bags and related sterile products part of supply-risk planning for hospitals. Buyers now compare supplier reliability and packaging integrity before price alone. Product makers with multi-site production and validated sterility documentation can retain contracts during supply stress.
Homecare use forms the second demand base as patients manage stoma care and urinary drainage away from hospitals. Convatec launched Esteem Body with Leak Defense in February 2024 with a pouch shape designed to sag less during filling. Product design improvements affect repeat selection as comfort and leakage control influence repeat buying. Suppliers combining skin-friendly materials with easy handling can serve patients and nurses better. This strengthens premium positions in ostomy care accessories and related pouch systems.
Market Segmentation Analysis
- Ostomy bags are projected to hold the largest product share with 34.0% share in 2026 as stoma care creates repeat need.
- PVC is expected to hold the largest material share with 42.0% share in 2026 with cost and sealing performance supporting scale.
- Hospitals are projected to account for 45.0% share in 2026 as inpatient care uses multiple bag formats.
- Disposable bags are expected to represent 71.0% share in 2026 as infection control guides buying standards.
- Direct sales are projected to hold 39.0% share in 2026 with tender contracts and hospital networks shaping volume.
Segmentation in this market follows clinical use and buyer control points. Product type defines the care pathway and material type affects price and leakage performance. End user groups separate institutional buying from patient-led repeat use. Disposable formats dominate as cleaning and reuse create compliance burden in many care settings. Distribution structure differs between hospital tenders and homecare refills. This keeps supplier strategy split between technical approval for hospitals and usability for patients. Demand for urinary drainage bags follows both surgery recovery and long-term bladder management.
Medical Specialty Bag Market Analysis By Product Type
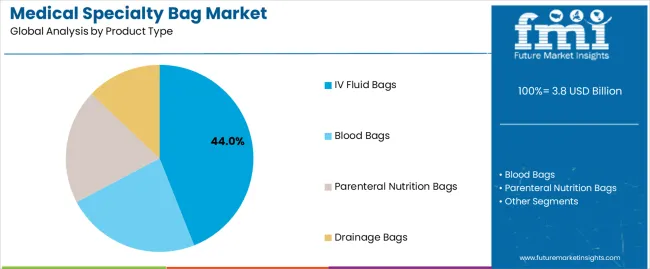
- Ostomy bags are projected to account for 34.0% in 2026 as pouch replacement supports repeat purchase across colostomy and ileostomy care.
- One-piece pouches often perform better in home settings as fewer assembly steps help users manage replacement without repeated nurse support.
Medical Specialty Bag Market Analysis By Material Type
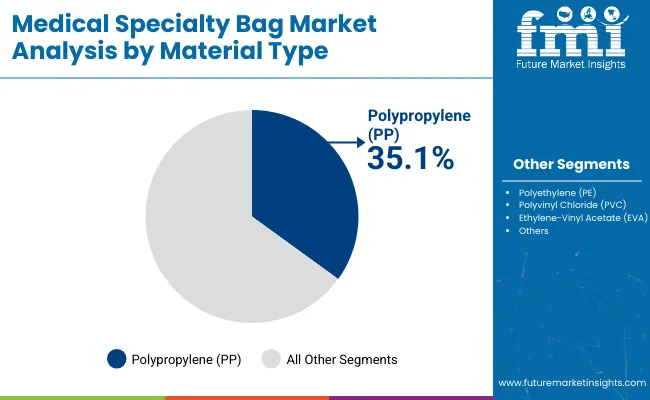
- PVC is expected to represent 42.0% share in 2026 as flexible forming and low unit cost support large-volume hospital supply.
- Polyolefin use is rising in selected non-PVC IV bags as buyers review DEHP exposure and disposal concerns.
Medical Specialty Bag Market Analysis By End User
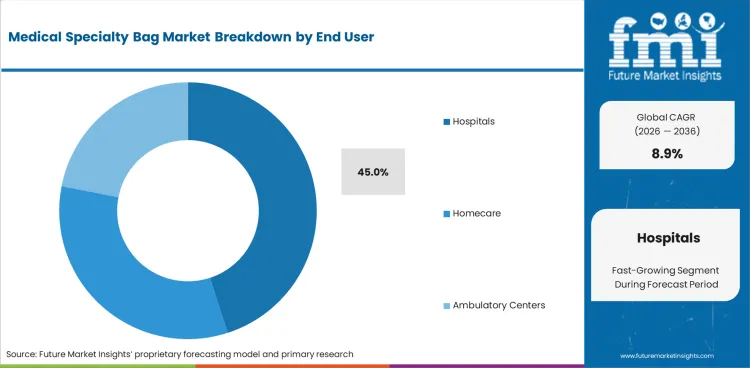
- Hospitals are anticipated to represent 45.0% share in 2026 with surgery and inpatient recovery driving multi-product use.
- Hospital buyers often test seal strength and drain security before wider listing as fluid leakage creates clinical risk and staff burden.
Medical Specialty Bag Market Analysis By Usage Type
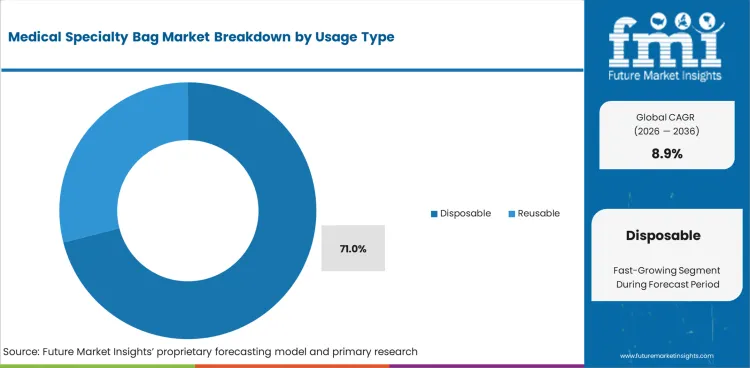
- Disposable bags are projected to account for 71.0% in 2026 as infection control favors single-use collection and delivery systems.
- Reusable options stay limited to selected cases as cleaning validation creates extra work for care teams and facility managers.
Medical Specialty Bag Market Analysis By Distribution Channel
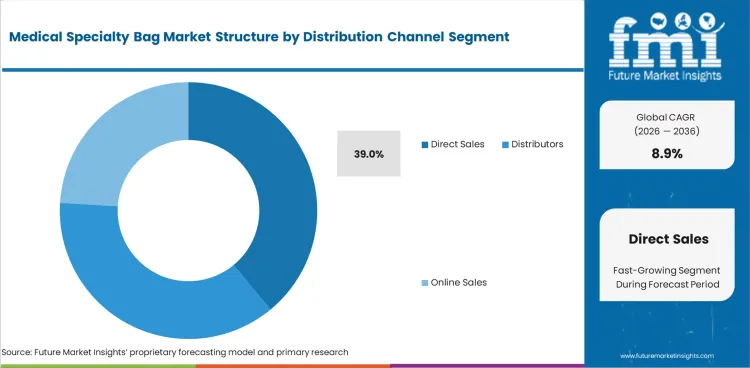
- Direct sales are expected to hold 39.0% in 2026 with hospital networks preferring supply contracts and technical support.
- Online channels advance faster in homecare refill ordering as patients seek repeat access to pouching and drainage supplies.
Medical Specialty Bag Market Drivers Restraints and Opportunities
- Stoma care demand creates repeat bag use as pouch replacement follows daily patient management needs.
- IV supply pressure increases buyer focus on backup suppliers and validated sterile bag production.
- Homecare expansion shifts demand toward simple handling and discreet bag design for patient use.
- Material regulation limits suppliers using legacy plastics without clear safety documentation.
- Premium design creates higher-value prospects in enteral feeding bags and ostomy pouch systems.
Stoma Replacement Frequency
Stoma care creates steady demand as bags are not a one-time hospital purchase. Patients need pouch systems after discharge. Homecare channels maintain recurring supply. Coloplast strengthened its SenSura Mio portfolio in May 2024 with black bags and convex pouch formats across European markets. The launch points to demand for comfort and discretion beyond basic collection. Suppliers must treat pouch design as a user-retention tool. Stronger skin protection can support premium pricing. Odor control affects repeat brand choice. Weak product comfort can push users toward alternate brands through nurse recommendations.
Sterile Fluid Supply Risk
Sterile fluid bags carry strategic value during hospital supply disruption. IV bags are used across surgery and emergency care B. Braun launched Midazolam in sodium chloride injection in October 2025 in IV bags not made with DEHP and PVC. This format points toward safer materials and clearer hospital handling. Buyers evaluate bag material first. Barcode readability adds a handling check. Suppliers with validated sterile capacity and traceable labels can gain access to large hospital systems.
Inspection Readiness Is Becoming a Bag-Supplier Filter
For the medical specialty bag market, regulatory compliance is now a records review and inspection-readiness issue. It is no longer only a product approval task. FDA states that the QMSR became effective on February 2, 2026. FDA also states that investigators may review manufacturer QMS records created before February 2, 2026. This affects suppliers of drainage bags and ostomy bags. It affects suppliers of blood bags and procedure bags. Buyer qualification will depend on design control evidence and supplier audit files. It can depend on complaint handling and lot traceability. Bag suppliers without inspection-ready records may face longer hospital qualification cycles. These suppliers may also face higher distributor risk reviews.
Field Malfunctions Are Raising Operating Scrutiny
Field malfunctions are increasing operating scrutiny for medical specialty bag suppliers. Operating standards are being shaped by failures reported through postmarket surveillance. One FDA MAUDE record updated March 31 2026 classified a COVIDIEN ENDO BAG event dated September 16 2025 as a malfunction. The record listed Medical Device Problem Code Material Split or Torn 4008. The same record showed one device and one patient involved. No patient injury was reported. For bag manufacturers, this record is crucial because buyers test sterility and labeling. Buyers review tear strength and seal integrity. A minor field failure can still increase documentation pressure. It can also create account-level caution.
Analysis of Medical Specialty Bag Market By Leading Countries
.webp)
| Country | CAGR |
|---|---|
| India | 10.7% |
| China | 10.2% |
| United States | 8.4% |
| Brazil | 8.6% |
| Germany | 7.7% |
| United Kingdom | 7.9% |
| Japan | 6.8% |
Source: FMI analysis based on primary research and proprietary forecasting model.
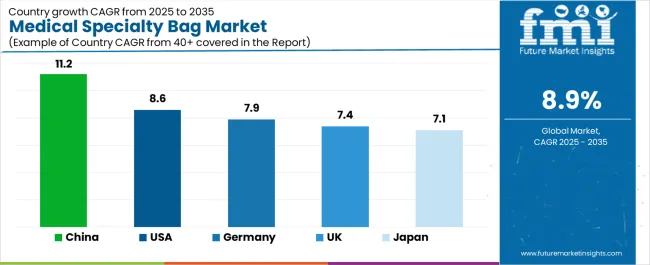
Medical Specialty Bag Market CAGR Analysis By Country
- India is likely to progress at a CAGR of 10.7% by 2036 supported by hospital capacity additions and homecare supply channels expanding across major cities.
- China is projected to record a CAGR of 10.2% during the forecast period owing to domestic medical device output and large hospital patient volumes.
- Brazil is set to record a CAGR of 8.6% during the assessment period supported by wider public healthcare access and private hospital concentration.
- The United States is anticipated to expand at a CAGR of 8.4% over the forecast period on account of hospital purchasing and homecare refill demand.
- The United Kingdom is likely to post a CAGR of 7.9% during the study period supported by NHS pathways and structured ostomy supply programs.
- Germany is expected to advance at a CAGR of 7.7% between 2026 and 2036 following mature reimbursement systems and strict product standards.
- Japan is expected to register a CAGR of 6.8% by 2036 because of aging care demand and mature product use.
Country expansion differs by care access and reimbursement strength. India and China track above the global average as patient access and local production expand together. Japan and Germany move below the global pace as established usage leaves less white space. The spread makes country strategy important for suppliers balancing price-sensitive volume with regulated premium products.
Demand Outlook for Medical Specialty Bag Market in India
India has a faster demand curve as hospital expansion and homecare supply improve across major states. The country is projected to record 10.7% CAGR by 2036. This pace reflects new patient access rather than only replacement purchases. Public hospital purchasing and private care chains both create demand for ostomy and urinary drainage supplies. Domestic distribution helps reduce delivery delays for routine bags. A small improvement in reimbursement coverage could expand repeat use across tier-two cities and homecare networks.
- Ayushman Bharat improves hospital access for lower-income patients and supports higher procedure-linked consumable use.
- Maharashtra and Tamil Nadu support large private hospital clusters with repeat demand for drainage bags.
- Delhi NCR homecare providers increase access to urinary and ostomy supplies for post-discharge patients.
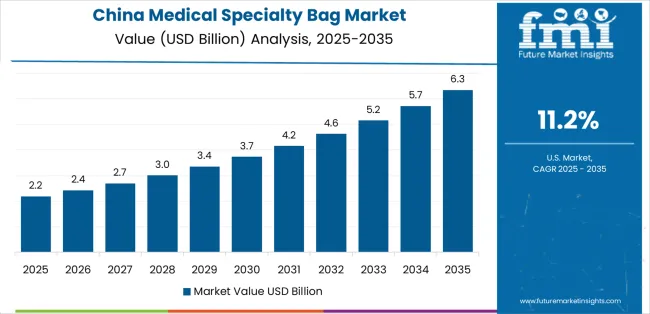
Sales Analysis of Medical Specialty Bag Market in China
China benefits from domestic device manufacturing and large hospital patient volumes. The country is expected to expand at 10.2% CAGR during the forecast period as local supply supports scale and price control. Public hospital purchasing favors suppliers able to meet volume and documentation needs. Local producers compete directly in standard bags while global brands hold stronger positions in premium ostomy systems. A shift toward home recovery could increase repeat bag purchasing through pharmacies and online care channels.
- National Medical Products Administration rules guide medical device registration and post-market control.
- Guangdong and Jiangsu support device production clusters with polymer processing and packaging capacity.
- Shanghai hospitals create demand for premium pouch systems through specialist colorectal care.
Demand Analysis of Medical Specialty Bag Market in the United States
The United States is shaped by hospital purchasing discipline and home medical supply reimbursement. The country is forecast to expand at 8.4% CAGR over the forecast period as chronic care and post-acute treatment support repeat product use. IV fluid shortages in 2024 raised attention on production continuity and backup sourcing. Supplier resilience becomes a buying factor for sterile bags. Homecare dealers and national distributors improve access for ostomy and urinary products. Better inventory planning could protect hospitals from future fluid-management disruptions.
- Medicare DME rules influence access to ostomy supplies and urinary collection products.
- North Carolina fluid supply disruption in 2024 raised hospital concern around IV bag sourcing.
- California and Florida manufacturing sites support domestic sterile fluid supply planning.
Opportunity Analysis of Medical Specialty Bag Market in Brazil
Brazil offers demand potential through public healthcare access and private hospital concentration. The country is expected to advance at 8.6% CAGR during the assessment period as procedure access and chronic care expand. São Paulo and Rio de Janeiro concentrate private hospital purchasing for higher-value products. Public channels create larger tenders for standard drainage and fluid bags. Imported premium ostomy systems face price sensitivity in broader public use. Local distribution partnerships could improve availability across regional hospitals and homecare suppliers.
- SUS purchasing influences standard bag volumes across public hospitals and regional care networks.
- São Paulo private hospitals support demand for higher-specification pouch and fluid systems.
- ANVISA device rules shape registration timing for imported blood bags.
Future Outlook for Medical Specialty Bag Market in Germany
Germany has mature demand but strict product review keeps supplier quality expectations high. The country is projected to rise at 7.7% CAGR between 2026 and 2036 as aging care and hospital use support stable volumes. Reimbursement structures create steady access for ostomy care products. Buyers review product evidence and patient comfort before switching brands. Local standards favor suppliers with documentation depth and reliable service. Faster use of alternative materials could support premium positions despite slower volume expansion.
- German sickness funds shape access to ostomy and continence supplies through reimbursement pathways.
- North Rhine-Westphalia hospital networks support large tender volumes for sterile fluid products.
- Berlin colorectal care centers influence specialist product choice for stoma patients.
Demand Outlook for Medical Specialty Bag Market in the United Kingdom
The United Kingdom market reflects structured public purchasing and nurse-guided product selection. The country is forecast to register 7.9% CAGR during the study period as NHS care pathways support use across ostomy and drainage products. Specialist nurses influence pouch selection through patient education and follow-up support. Product comfort and leakage control affect repeat ordering through prescription supply channels. Suppliers must support training and patient literature to maintain listing strength. Faster community care discharge could increase home supply needs for urinary incontinence products.
- NHS Supply Chain influences hospital purchasing for fluid handling and drainage consumables.
- England community stoma nurses guide pouch selection during post-discharge care.
- Scotland home delivery programs support repeat access to ostomy products outside hospital settings.
In-depth Analysis of Medical Specialty Bag Market in Japan
Japan has strong aging care demand but slower volume expansion due to mature product use. The country is expected to post 6.8% CAGR by 2036 as elderly care needs support steady purchasing. Hospitals and long-term care providers focus on comfort and low leakage risk. Domestic buyers value compact packaging and easy disposal. Premium pouch features can gain acceptance when they reduce nurse workload. Supplier strategy should focus on user comfort and care-setting efficiency rather than only price.
- Tokyo and Osaka hospital systems support demand for premium ostomy and urinary drainage products.
- Japan’s long-term care insurance system supports home and facility-based elder care needs.
- Local device rules under PMDA review strengthen documentation needs for imported medical bags.
Competitive Landscape and Strategic Positioning
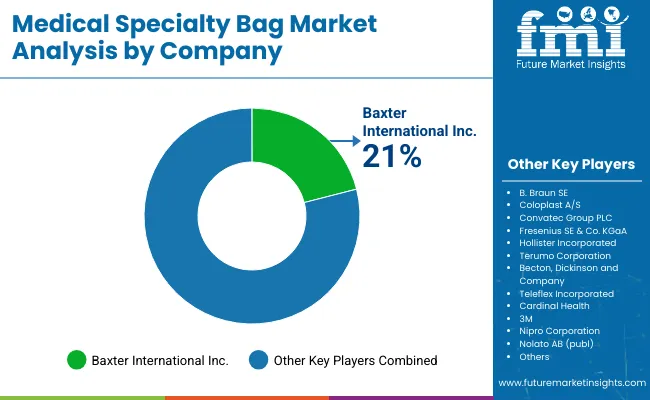
- Competition is moderately concentrated as global brands control premium ostomy care and sterile fluid bag positions.
- Smaller suppliers compete through tender pricing and regional distributor access in standard drainage products.
- Entry barriers come from sterility validation and leakage performance testing across regulated care settings.
- Product switching is difficult as nurses and hospital buyers need confidence before approving new bag formats.
Competitive strength in this market starts with product reliability during fluid handling. Buyers test leakage performance and skin comfort before wider approval. Convatec’s February 2024 Esteem Body launch used an 8-shape pouch and Leak Defense technology for better wear experience. This reflects competition based on fit and user confidence rather than basic containment. Suppliers with proven pouch design can secure orders in long-term care.
Established companies hold advantage through product breadth and hospital access. Coloplast’s May 2024 ostomy launches strengthened the SenSura Mio range with new formats and color options. The launch helped Coloplast address patient discretion and clinical fit in the same product family. B. Braun competes through sterile fluid bag capability and hospital injectable formats. Baxter holds a major role in IV solutions even after supply disruption concerns. Broad portfolios help firms bundle products across hospital supplies and chronic care channels.
Specialist companies can secure orders in regional tenders and focused applications. Urinary drainage and wound drainage products often allow local suppliers to sustain through delivery speed and lower pricing. Entry stays difficult in premium ostomy care as skin contact and leakage history shape brand trust. New entrants need user trials and nurse acceptance before gaining repeat orders. Technical files and post-market evidence therefore become sales tools. Suppliers with stable service records can retain accounts even under price pressure.
Main Companies in the Medical Specialty Bag Market
Competitive structure combines global ostomy specialists with sterile fluid bag suppliers and regional medical consumable producers.
- Global Leaders: Coloplast and Convatec hold established positions in ostomy bags through pouch design and skin-contact expertise. B. Braun and Baxter hold established positions in IV fluid bags through sterile manufacturing and hospital access. .
- Regional Players: Hollister and Cardinal Health dominate through product portfolios and distribution access in North America and Europe. Medline supports hospitals through broad consumable supply and contract relationships. Terumo supports blood bag and infusion-related systems across Asia and selected global markets.
- Emerging Players: Selected Asian suppliers compete in urinary drainage and feeding bag formats through cost control and faster regional supply. These companies often gain share in tenders before entering premium ostomy care.
Competitive Benchmarking: Medical Specialty Bag Market
| Company | Leak Control Depth | Sterile Bag Capacity | Homecare Channel Reach | Geographic Footprint |
|---|---|---|---|---|
| Coloplast | High | Medium | High | Global |
| Convatec | High | Medium | High | Global |
| B. Braun | Medium | High | Medium | Global |
| Baxter | Medium | High | Medium | Global |
| Hollister | High | Low | High | North America and Europe |
| Cardinal Health | Medium | Medium | High | North America |
| Medline | Medium | Medium | High | North America |
| Terumo | Medium | High | Medium | Asia and Global |
Source: Future Market Insights competitive analysis, 2026. Ratings reflect relative positioning based on leak control depth, sterile bag capacity, and homecare channel reach.
Recent Developments in Medical Specialty Bag Market
- In February 2024, Convatec launched Esteem Body with Leak Defense for ostomy care. The pouch design supports better wear experience and user confidence.
- In May 2024, Coloplast strengthened its ostomy care portfolio with three SenSura Mio product launches. The launches widened choice for pouch users.
- In July 2025, B. Braun expanded its heparin sodium injection portfolio in IV bags. The 250 mL and 500 mL formats included enhanced barcodes.
In October 2025, B. Braun launched midazolam in sodium chloride injection in IV bags not made with DEHP and PVC. The launch supports safer hospital handling.
Leading Players in the Medical Specialty Bag Market
Global Leaders
- Coloplast
- Convatec
- B. Braun
- Baxter
Regional Players
- Hollister
- Cardinal Health
- Medline
- Terumo
Emerging Players
- Well Lead Medical
- Romsons
- Flexicare
Report Scope and Coverage

| Parameter | Details |
|---|---|
| Quantitative Units | USD 4.1 billion in 2026 to USD 9.6 billion by 2036 at 8.9% CAGR |
| Market Definition | Medical specialty bags include ostomy bags and drainage bags with IV fluid bags and blood bags used in regulated care. |
| Regions Covered | North America and Latin America. Europe and East Asia. South Asia and Pacific. Middle East and Africa |
| Countries Covered | India and China. United States and Brazil. Germany and United Kingdom. Japan and 30+ countries |
| Leading Companies Profiled | Coloplast and Convatec. B. Braun and Baxter. Hollister and Cardinal Health. Medline and Terumo |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology based on product use rates and pricing checks. Regional demand assessment completed the validation |
Medical Specialty Bag Market by Segments
Medical Specialty Bag Market Segmented by Product Type:
- Ostomy Bags
- Urinary Bags
- IV Fluid Bags
- Blood Bags
- Drainage Bags
- Feeding Bags
Medical Specialty Bag Market Segmented by Material Type:
- PVC
- Polyolefin
- Silicone
- Nonwoven
Medical Specialty Bag Market Segmented by End User:
- Hospitals
- Homecare
- Ambulatory Centers
- Clinics
Medical Specialty Bag Market Segmented by Usage Type:
- Disposable
- Reusable
Medical Specialty Bag Market Segmented by Distribution Channel:
- Direct Sales
- Distributors
- Online Sales
- Retail Pharmacies
Medical Specialty Bag Market by Region:
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan and Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia and New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East and Africa
Research Sources and Bibliography
- USA Food and Drug Administration. (2026, February 2). Quality Management System Regulation – Frequently Asked Questions.
- USA Food and Drug Administration. (2026, March 31). MAUDE Adverse Event Report: COVIDIEN ENDO BAG; LAPAROSCOPE, GENERAL & PLASTIC SURGERY.
- Convatec Group. (2024, February 29). Convatec launches new Esteem Body™ with Leak Defense™. Convatec.
- B. Braun Medical Inc. (2025, July 18). B. Braun expands heparin sodium injection portfolio to seven heparin premixed sodium injection products. B. Braun.
- B. Braun Medical Inc. (2025, October 15). B. Braun launches midazolam in 0.8% sodium chloride injection with enhanced safety and usability features. B. Braun.
- European Commission. (2024). Medical devices sector new regulations. European Commission.
- USA Food and Drug Administration. (2024). Medical device and IV fluid supply disruption notices. USA Food and Drug Administration.
- Centers for Medicare and Medicaid Services. (2024). Durable medical equipment data and coverage references. Centers for Medicare and Medicaid Services.
- Coloplast. (2025, May 2). Roadshow presentation H1 2024/25. Coloplast.
- United Ostomy Associations of America. (2024, March 28). Ostomy awareness resources. United Ostomy Associations of America.
The bibliography is provided for reader reference. It includes official company sources and regulatory references.
This Report Answers
- What is the projected size of the medical specialty bag market in 2026 and 2036?
- Which product type is expected to hold the largest share in medical specialty bags?
- What CAGR is forecast for the medical specialty bag market from 2026 to 2036?
- Which material type is expected to lead demand across regulated medical bag applications?
- Which countries are projected to advance faster in medical specialty bag demand through 2036?
- What role do hospitals and homecare channels play in shaping medical specialty bag sales?
- Which companies compete across ostomy bags and sterile fluid bag products?
- What product types are included within the medical specialty bag market scope?
- Which factors could limit supplier access to hospital and homecare contracts?
Frequently Asked Questions
What is the global market demand for Medical Specialty Bag Market in 2026?
In 2026, the global Medical Specialty Bag Market is expected to be worth USD 4.1 billion across regulated care uses.
What is the expected market value by 2036?
The Medical Specialty Bag Market is projected to reach USD 9.6 billion by 2036 as chronic care and hospital use expand.
What CAGR is expected from 2026 to 2036?
The Medical Specialty Bag Market is forecast to expand at 8.9% CAGR during the 2026 to 2036 period.
Which product type is expected to hold the leading position?
Ostomy bags are forecast to account for 34.0% share in 2026 as repeat pouch use supports demand.
Which end user is expected to hold the largest share?
Hospitals are projected to account for 45.0% share in 2026 as inpatient care needs more than one bag format.
Which country is expected to grow fastest?
India is forecast to record 10.7% CAGR through 2036 as hospital access and homecare distribution expand.
What products are included in the market definition?
The market includes ostomy bags and urinary bags with IV fluid bags and drainage bags used in clinical care.
What methodology supports the forecast?
The forecast combines product use rates and regional pricing with hospital demand checks and company portfolio review.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Ostomy Bags
- Urinary Bags
- IV Fluid Bags
- Ostomy Bags
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material Type, 2026 to 2036
- PVC
- Polyolefin
- Silicone
- PVC
- Y to o to Y Growth Trend Analysis By Material Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Material Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Hospitals
- Homecare
- Ambulatory Centers
- Hospitals
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Usage Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Usage Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Usage Type, 2026 to 2036
- Disposable
- Reusable
- Disposable
- Y to o to Y Growth Trend Analysis By Usage Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Usage Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Distribution Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Distribution Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Distribution Channel, 2026 to 2036
- Direct Sales
- Distributors
- Online Sales
- Direct Sales
- Y to o to Y Growth Trend Analysis By Distribution Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Distribution Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Material Type
- By End User
- By Usage Type
- By Distribution Channel
- Competition Analysis
- Competition Deep Dive
- Coloplast
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Convatec
- B. Braun
- Baxter
- Hollister
- Cardinal Health
- Coloplast
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 6: Global Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 11: North America Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 12: North America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 16: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 17: Latin America Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 18: Latin America Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: Western Europe Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 22: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 23: Western Europe Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 24: Western Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 27: Eastern Europe Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 28: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 29: Eastern Europe Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 30: Eastern Europe Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 31: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 33: East Asia Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 34: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 35: East Asia Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 36: East Asia Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 37: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 38: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 39: South Asia and Pacific Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 40: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 41: South Asia and Pacific Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 42: South Asia and Pacific Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
- Table 43: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 44: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 45: Middle East & Africa Market Value (USD Million) Forecast by Material Type, 2021 to 2036
- Table 46: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 47: Middle East & Africa Market Value (USD Million) Forecast by Usage Type, 2021 to 2036
- Table 48: Middle East & Africa Market Value (USD Million) Forecast by Distribution Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Material Type
- Figure 9: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by End User
- Figure 12: Global Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Usage Type
- Figure 15: Global Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Distribution Channel
- Figure 18: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 19: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 20: Global Market Attractiveness Analysis by Region
- Figure 21: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 26: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 27: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 28: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 29: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Product Type
- Figure 32: North America Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Material Type
- Figure 35: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by End User
- Figure 38: North America Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 39: North America Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 40: North America Market Attractiveness Analysis by Usage Type
- Figure 41: North America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 42: North America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 43: North America Market Attractiveness Analysis by Distribution Channel
- Figure 44: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 45: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Product Type
- Figure 48: Latin America Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Material Type
- Figure 51: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 52: Latin America Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 53: Latin America Market Attractiveness Analysis by End User
- Figure 54: Latin America Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 55: Latin America Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 56: Latin America Market Attractiveness Analysis by Usage Type
- Figure 57: Latin America Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 58: Latin America Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 59: Latin America Market Attractiveness Analysis by Distribution Channel
- Figure 60: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 61: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Product Type
- Figure 64: Western Europe Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 65: Western Europe Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 66: Western Europe Market Attractiveness Analysis by Material Type
- Figure 67: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 68: Western Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 69: Western Europe Market Attractiveness Analysis by End User
- Figure 70: Western Europe Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 71: Western Europe Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 72: Western Europe Market Attractiveness Analysis by Usage Type
- Figure 73: Western Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 74: Western Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 75: Western Europe Market Attractiveness Analysis by Distribution Channel
- Figure 76: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 77: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 78: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 79: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 80: Eastern Europe Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 81: Eastern Europe Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 82: Eastern Europe Market Attractiveness Analysis by Material Type
- Figure 83: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 84: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 85: Eastern Europe Market Attractiveness Analysis by End User
- Figure 86: Eastern Europe Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 87: Eastern Europe Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 88: Eastern Europe Market Attractiveness Analysis by Usage Type
- Figure 89: Eastern Europe Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 90: Eastern Europe Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 91: Eastern Europe Market Attractiveness Analysis by Distribution Channel
- Figure 92: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 93: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 94: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 95: East Asia Market Attractiveness Analysis by Product Type
- Figure 96: East Asia Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 97: East Asia Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 98: East Asia Market Attractiveness Analysis by Material Type
- Figure 99: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 100: East Asia Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 101: East Asia Market Attractiveness Analysis by End User
- Figure 102: East Asia Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 103: East Asia Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 104: East Asia Market Attractiveness Analysis by Usage Type
- Figure 105: East Asia Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 106: East Asia Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 107: East Asia Market Attractiveness Analysis by Distribution Channel
- Figure 108: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 109: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 110: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 111: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 112: South Asia and Pacific Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 113: South Asia and Pacific Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 114: South Asia and Pacific Market Attractiveness Analysis by Material Type
- Figure 115: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 116: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 117: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 118: South Asia and Pacific Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 119: South Asia and Pacific Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 120: South Asia and Pacific Market Attractiveness Analysis by Usage Type
- Figure 121: South Asia and Pacific Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 122: South Asia and Pacific Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 123: South Asia and Pacific Market Attractiveness Analysis by Distribution Channel
- Figure 124: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 125: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 126: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 127: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 128: Middle East & Africa Market Value Share and BPS Analysis by Material Type, 2026 and 2036
- Figure 129: Middle East & Africa Market Y-o-Y Growth Comparison by Material Type, 2026-2036
- Figure 130: Middle East & Africa Market Attractiveness Analysis by Material Type
- Figure 131: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 132: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026-2036
- Figure 133: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 134: Middle East & Africa Market Value Share and BPS Analysis by Usage Type, 2026 and 2036
- Figure 135: Middle East & Africa Market Y-o-Y Growth Comparison by Usage Type, 2026-2036
- Figure 136: Middle East & Africa Market Attractiveness Analysis by Usage Type
- Figure 137: Middle East & Africa Market Value Share and BPS Analysis by Distribution Channel, 2026 and 2036
- Figure 138: Middle East & Africa Market Y-o-Y Growth Comparison by Distribution Channel, 2026-2036
- Figure 139: Middle East & Africa Market Attractiveness Analysis by Distribution Channel
- Figure 140: Global Market - Tier Structure Analysis
- Figure 141: Global Market - Company Share Analysis

