Medical Device Packaging Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
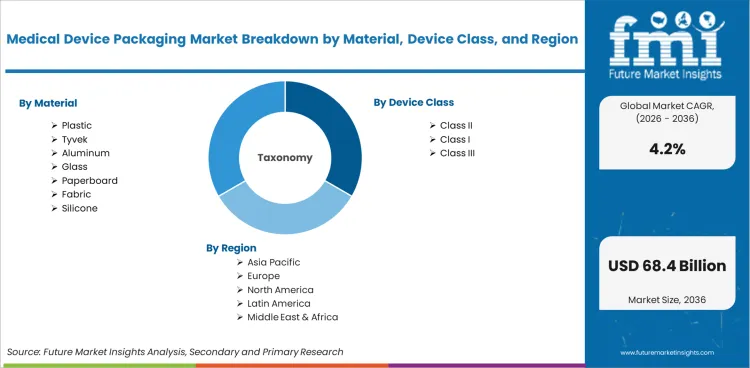
The Medical Device Packaging Market is segmented by Material (Plastic, Tyvek, Aluminum, Glass, Paperboard, Fabric, Silicone), Class (Class I, Class II, Class III), Application (Sterile Packaging, Non-Sterile Packaging, Implantable Device Packaging, Diagnostic and Monitoring Device Packaging), Packaging Format, Sterilization Route, and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Medical Device Packaging Market Forecast and Outlook By FMI
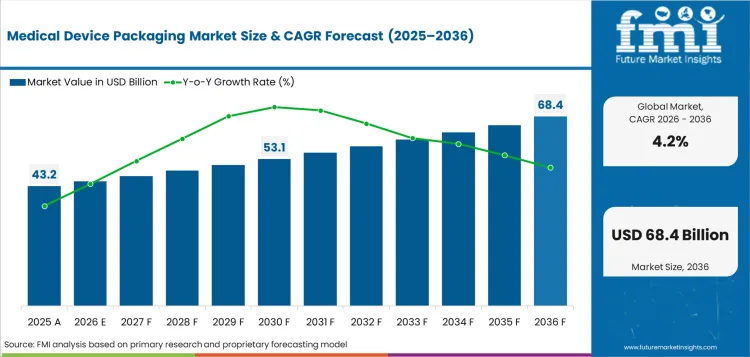
The medical device packaging market was valued at USD 43.2 billion in 2025 and is projected to reach USD 45.2 billion in 2026. It is forecast to reach USD 68.4 billion by 2036 at 4.2% CAGR. Plastic is projected to lead the material segment with 42.7% share, and bags and pouches are expected to hold 21.5% share by packaging format in 2026.
Summary of the Medical Device Packaging Market
- Demand and Growth Drivers
- Sterile barrier validation is lifting order value since device firms need proof across sealing and distribution tests.
- Growth reflects more single-use surgical devices and diagnostic kits that need individual protective packs.
- Ethylene oxide controls are changing material screening as breathable pouches must protect sterility and meet emissions limits.
- Medical device output in China and India is raising demand for export-ready packs with stronger technical files.
- Product and Segment View
- Bags and pouches are expected to account for 21.5% share in 2026 as flexible formats suit many sterile devices.
- Class II devices are estimated to account for 52.0% share in 2026 as moderate-risk products need repeat validation.
- Sterile packaging is estimated at 58.0% share in 2026 since instruments, implants and catheters need protected release.
- Demand is anticipated to be driven by sterile packaging and medical packaging films as barrier quality decides shelf life and clinical handling.
- Geography and Competitive Outlook
- China is expected to post 6.2% CAGR through 2036 as local device manufacturing and regulatory standards deepen.
- India is projected at 6.0% CAGR through 2036 as medical device production incentives expand packaging needs.
- Brazil is expected to record 5.3% CAGR through 2036 as device rules and healthcare access support sterile formats.
- Companies such as Amcor, DuPont and Nelipak shape the premium end through cleanroom converting and medical-grade material platforms.
- Analyst Opinion
- Ismail Sutaria, Principal Consultant for Packaging at FMI says, “Medical device packaging is becoming a risk-control system. Device makers now ask whether a supplier can prove sterility/sealing discipline and change control before they ask for the lowest unit price. The practical opportunity sits with converters that combine cleanroom capacity, sterilization compatibility and documentation support. Suppliers that reduce audit friction for device manufacturers will gain repeat contracts in Class II and Class III products.”
- Medical Device Packaging Market Value Analysis
- The market is moving from basic protection into compliance-led sterile barrier systems with recurring validation needs.
- Market value is increasing due to recurring validation needs such as seal integrity and distribution testing, which add ongoing service and material costs for suppliers.
- Growth is supported by rising production of single-use medical devices and diagnostic kits, each requiring individual sterile packaging solutions that raise overall packaging demand.
- Regulatory pressure and stricter sterilization controls are pushing companies toward higher-quality packaging materials and processes, increasing the overall value per packaged device.
Sterility assurance and documentation control are considerable pressures behind medical device packaging selection. The United States Environmental Protection Agency stated in March 2024 that ethylene oxide commercial sterilizers must cut emissions by more than 90%. The rule affects nearly 90 commercial sterilization facilities operated by about 50 companies. That policy pressure keeps packaging material choice tied to sterilization access and validated barrier performance. The International Organization for Standardization lists ISO 11607-2:2019 for validation of forming, sealing and assembly processes. The standard pushes device makers toward suppliers that can prove seal integrity through repeatable process records and aging studies.
Medical Device Packaging Market Definition
The medical device packaging market covers sterile and non-sterile packaging formats used to protect medical devices during sterilization, storage, shipment and clinical opening. Scope includes pouches, rollstock, films, trays, lids, clamshells and device-specific protective formats. These systems must protect device function and maintain sterility until point of use. The scope connects with sterile medical packaging where medical-grade barriers are specified for surgical instruments, implants and in-vitro diagnostic products.
Medical Device Packaging Market Inclusions
Market scope includes primary medical device packaging materials, preformed sterile barrier systems and packaging systems used by device manufacturers or contract packagers. Material coverage includes plastic, Tyvek, aluminum, glass, paperboard, fabric and silicone. Format coverage includes bags and pouches, films and wraps, lids, rollstock, containers, trays, stick packs, clamshells, blister packs, boxes and flow wraps. Sterilization route coverage includes ethylene oxide, radiation, steam and vaporized hydrogen peroxide where packaging compatibility affects product release.
Medical Device Packaging Market Exclusions
Scope excludes finished medical devices and bulk logistics cartons that do not protect device function or sterility. Primary pharmaceutical containers are excluded unless the pack is part of a device or combination product. General consumer healthcare packaging and hospital storage bags are excluded unless they are sold as device-specific protective systems. Packaging machinery is excluded from the market value, although related demand is discussed where it affects sealing quality.
Medical Device Packaging Market Research Methodology
- Primary Research: FMI analysts reviewed device manufacturer needs, converter capacity, sterilization service feedback and cleanroom packaging qualification practices.
- Desk Research: The study used public sources from regulators, standards bodies, government health agencies and official company technical pages.
- Market sizing and forecasting: Market value was estimated through device output mapping, packaging format adoption and supplier capacity checks.
- Data validation: Forecast checks compared material share, country device production signals, sterilization requirements and recent supplier investments.
Why is the Medical Device Packaging Market Growing?
- Sterile barrier systems are gaining value as medical device makers need documented seal strength and aging proof.
- Single-use medical devices are widening the base for pouches, trays and diagnostic kit packaging.
- Supplier qualification is tightening as buyers want cleanroom output and ISO 13485-aligned documentation.
Medical device packaging is anticipated to gain demand where packaging failure can trigger product holds or recalls. The United States Food and Drug Administration recorded 4,515,331 electronic submissions to the Center for Devices and Radiological Health in 2024. The regulatory activity shows how much device work now moves through formal evidence systems. Packaging suppliers benefit when their validation records shorten review cycles for device firms.
Sterilization pressure is changing material evaluation. The United States Environmental Protection Agency final rule in March 2024 requires commercial sterilizers to reduce ethylene oxide emissions by more than 90%. This does not remove ethylene oxide from device supply chains. The rule makesdwell-time performance and alternative sterilization routes more important in early pack design. Demand is also estimated to increase in sterilized packaging and medical tray sealers where process control affects sterile release.
Market Segmentation Analysis
- Plastic is projected to account for 42.7% share of material demand in 2026 as converters need versatile medical-grade structures.
- Class II devices are estimated to account for 52.0% share in 2026 as common regulated devices need validated protective packs.
- Sterile packaging is estimated to hold 58.0% share of application demand in 2026 as instruments and implantable products.
- Bags and pouches are projected to account for 21.5% share of packaging format demand in 2026.
- Ethylene oxide-compatible packs are estimated to account for 50.0% share of sterilization-route demand in 2026.
The market is divided into five primary segment groups based on material, device class, application, packaging format and sterilization route. Material includes plastic, Tyvek, aluminum, glass, paperboard, fabric and silicone. Device class covers Class I, Class II and Class III medical devices. Application includes sterile packaging, non-sterile packaging, implantable device packaging and diagnostic packaging. Format demand connects with blister packaging when unit protection and opening control are central to product use.
Insights into the Plastic Material Segment
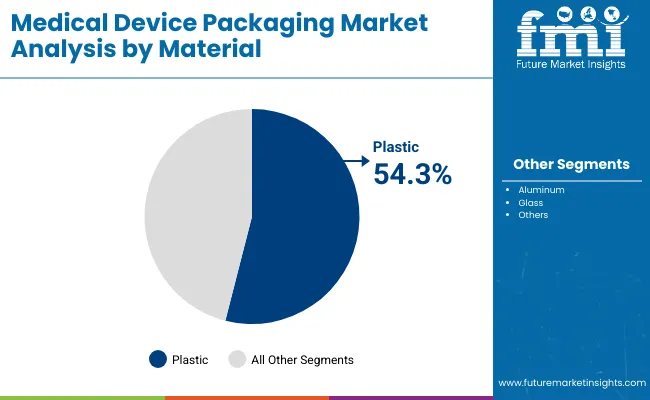
- Plastic is projected to account for 42.7% share of material demand in 2026. Medical-grade polyethylene, polypropylene and polyester structures are used in pouches and wrap formats because they combine process speed with sealing control.
Plastic formats remain practical where high-volume device lines need consistent forming and puncture resistance. Ethylene oxide and radiation-compatible films support broad device use when supplier data proves aging and microbial barrier performance.
Insights into the Class II Device Class Segment
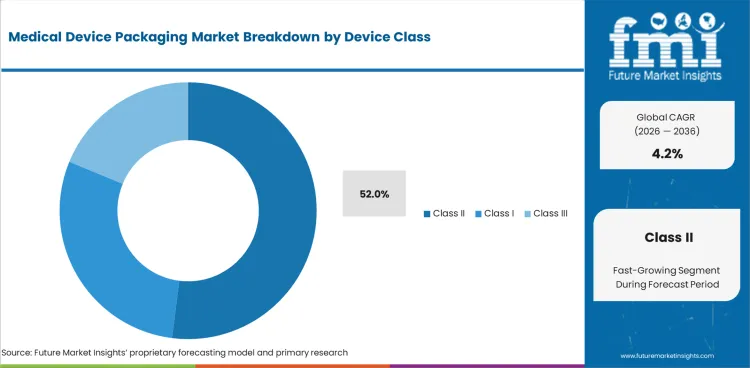
- Class II devices are estimated to account for 52.0% share of device-class demand in 2026. Moderate-risk devices create large packaging volumes because many products need sterile presentation without the lower volume profile of complex implants.
- Class II device makers need controlled packs that can support design history files and audit records. Syringes and diagnostic accessories support steady use of pouches/ trays and protective films.
Insights into the Sterile Packaging Application Segment
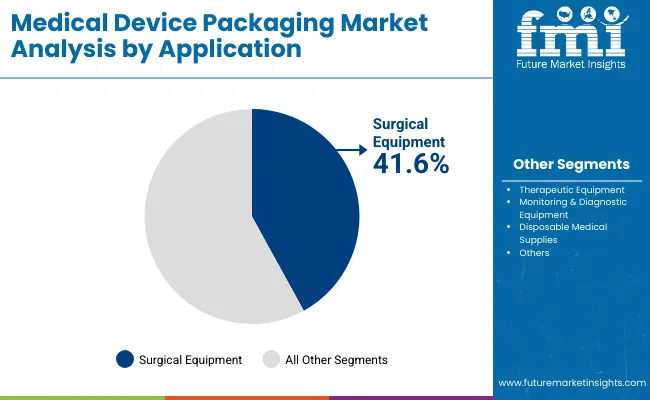
- Sterile packaging is estimated to account for 58.0% share of application demand in 2026. Hospitals and device firms use these formats to maintain sterility through storage and clinical opening.
- Peelable packs remain preferred where nurses need aseptic transfer without device contact.
Insights into the Bags and Pouches Packaging Format Segment
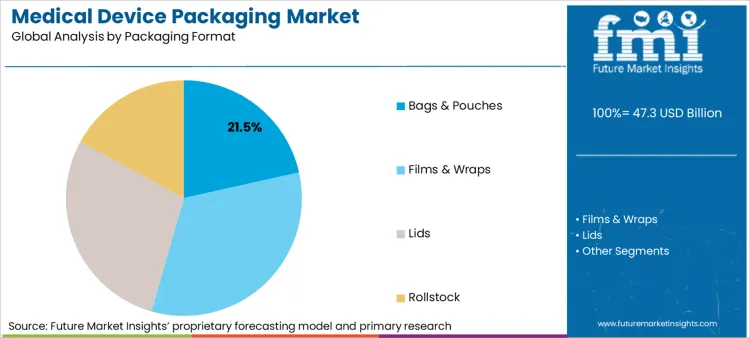
- Bags and pouches are projected to account for 21.5% share of packaging format demand in 2026. Flexible formats keep a clear role because they fit different device sizes with lower material weight than rigid trays.
- Roll-fed pouch systems support high-speed packing while keeping seal inspection manageable. Tyvek and film combinations let converters match porosity with microbial protection.
Insights into the Ethylene Oxide Sterilization Route Segment
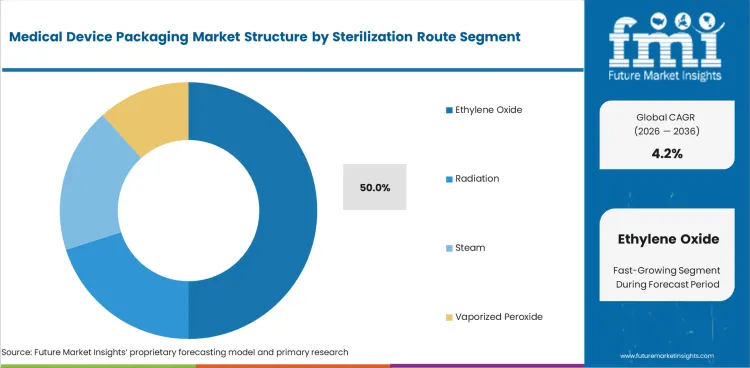
- Ethylene oxide-compatible packs are estimated to account for 50.0% share of sterilization-route demand in 2026. Porous packs remain important where complex devices cannot tolerate high heat or radiation.
- Pouch and lidding materials must allow gas entry while keeping microbes out after sterilization. Emissions controls push suppliers to prove cycle efficiency and explore alternative routes.
Medical Device Packaging Market Drivers, Restraints, and Opportunities
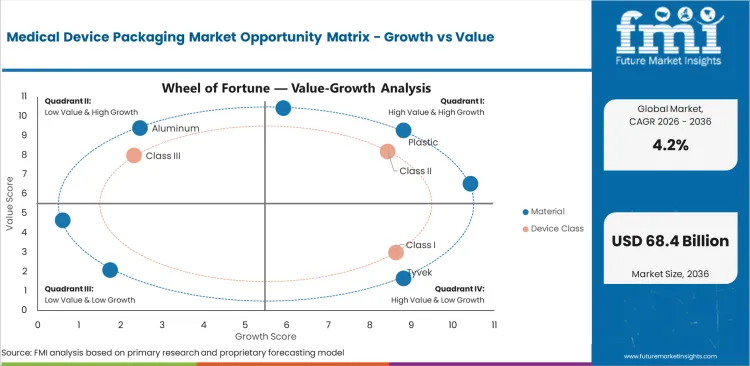
- Sterile validation needs are lifting demand for suppliers that can document sealing, aging and transport performance.
- Ethylene oxide rule pressure slows simple material switching because packs must work under tighter sterilization controls.
- Recyclable barrier development creates room for mono-material pouches where seal windows can be validated.
Sterile Validation Needs Are Raising Supplier Standards
Device makers treat packaging as part of product release because seal failure can stop a finished device from reaching hospitals. ISO 11607-2 covers validation of forming and assembly processes for sterile barrier systems. That requirement changes the buying question from material price to repeatable process evidence. Suppliers with cleanroom converting and test data are more likely to remain in approved vendor lists.
Ethylene Oxide Controls Are Reshaping Pack Design
One of the growth restraint is the cost and qualification burden created by sterilization rule changes. The Environmental Protection Agency rule applies to nearly 90 commercial sterilization facilities and requires stronger pollution controls. Device pack teams must now consider cycle efficiency and material porosity earlier in design. This keeps breathable pouches relevant but raises the burden on test data.
Recyclable Sterile Barriers Create A Selective Opportunity
The clearest opportunity is in formats that reduce plastic use without weakening sterile performance. DuPont reported that one B. Braun packaging redesign using Amcor Sureform Ultra and Tyvek 1059B eliminates 64 metric tons of plastic and 4,400 pounds of paper each year. The example shows why recyclable medical formats will grow first in projects with clear validation data. Demand for mono-PE medical device pouches will rise where seal-window control improves.
Analysis of Medical Device Packaging Market by Key Countries
.webp)
| Country | CAGR |
|---|---|
| United States | 4.1% |
| China | 6.2% |
| India | 6.0% |
| Germany | 3.9% |
| Japan | 3.5% |
| United Kingdom | 3.8% |
| Brazil | 5.3% |
Source: Future Market Insights, 2026.
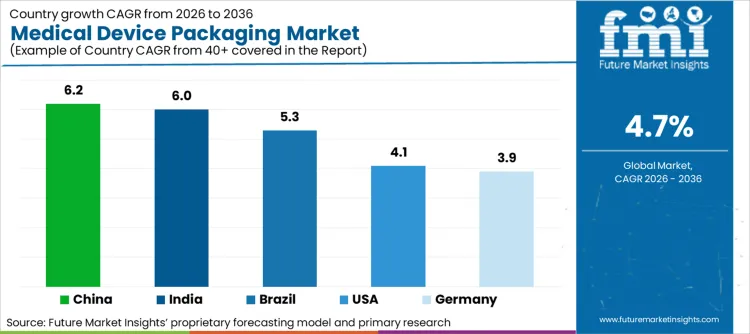
- China is projected to record 6.2% CAGR by 2036 owing to rising domestic device manufacturing and standards.
- India is forecast at 6.0% CAGR by 2036 as the Production Linked Incentive scheme supports local medical device output.
- Brazil is expected to advance at 5.3% CAGR by 2036 as regulatory updates and health access support packaged devices.
- The United States is projected at 4.1% CAGR by 2036 due to mature device production which creates steady replacement demand.
- Germany and the United Kingdom remain regulation-led markets where notified-body capacity and European data rules shape supplier choice.
Demand for medical device packaging is forecast to rise at 4.2% CAGR from 2026 to 2036. Country-level analysis covers the major markets where device output, regulatory oversight and sterilization routes directly affect packaging design.
Demand Outlook for Medical Device Packaging Market in the United States
The United States is set to grow at 4.1% CAGR through 2036, supported by stronger sterile barrier proof and traceability requirements among device firms. The Food and Drug Administration counted 4,515,331 electronic submissions to the Center for Devices and Radiological Health in 2024. The regulatory workload keeps technical documentation central to packaging selection. Wisconsin and Virginia remain important nodes for thermoforming and medical-grade materials.
- Wisconsin tray capacity supports medical device makers that need ISO Class 7 cleanroom output.
- Minnesota healthcare packaging plants support tray programs where design and tooling work stay close together.
- Virginia material supply supports Tyvek-based pouch systems used in ethylene oxide cycles.
Sales Analysis of Medical Device Packaging Market in China
China is likely to expand at 6.2% CAGR through 2036, driven by deeper local device output and standards activity. The National Medical Products Administration reported in May 2024 that China had 1,974 effective medical device standards at the end of 2023. That standards base gives packaging suppliers a clearer path for sterile barrier and labeling compliance. Suzhou and Beijing remain key demand centers for device-linked packaging.
- Suzhou device clusters create steady demand for pouches and thermoformed trays.
- Shenzhen diagnostics producers need compact packs with stable printing and tamper evidence.
- Beijing standards activity gives converters clearer direction on medical packaging qualification.
Demand Outlook for Medical Device Packaging Market in India
India is poised to grow at 6.0% CAGR through 2036 as local medical device programs open new packaging demand. The Press Information Bureau reported in December 2024 that the medical device Production Linked Incentive scheme had a financial outlay of Rs. 3,420 crore. It also reported 19 greenfield projects and production of 44 products under the scheme.
- Gujarat stent and implant projects create demand for validated sterile pouches and trays.
- Tamil Nadu device manufacturing supports format demand across diagnostic and hospital supply packs.
- Noida device producers need suppliers that can combine scale with regulatory documentation.
Opportunity Analysis of Medical Device Packaging Market in Germany
Germany is forecast to advance at 3.9% CAGR through 2036, with European compliance rules keeping packaging evidence central. MedTech Europe valued the European medical technology market at about EUR 170 billion in 2024. Germany is among the leading European medical technology countries within that base.
- Stuttgart device engineering supports tray and pouch demand for surgical instruments.
- Bavaria medical technology firms need packaging evidence that aligns with European Medical Device Regulation reviews.
- North Rhine-Westphalia logistics links support distribution packs for regulated devices.
Future Outlook for Medical Device Packaging Market in Japan
Japan is expected to grow at 3.5% CAGR through 2036, shaped by mature device production and import activity. The Ministry of Health, Labour and Welfare reported 2024 medical device domestic production value near 2.6642 trillion yen and imports near 3.6056 trillion yen. This mix supports domestic packaging and imported device relabeling needs.
- Tokyo device firms need packaging partners with strong labeling and documentation control.
- Osaka medical technology clusters support flexible sterile formats for surgical and diagnostic products.
- Nagoya automotive-medical overlap supports specialty packaging for sensors and connected health devices.
Demand Outlook for Medical Device Packaging Market in the United Kingdom
The United Kingdom is on track to post 3.8% CAGR through 2036, helped by improving certification capacity and post-market controls. The Medicines and Healthcare products Regulatory Agency designated two new UK Approved Bodies in January 2024. The designation expanded the route for medical device certification in Great Britain.
- Cambridge medtech firms need sterile packs that support early design files and clinical studies.
- Midlands manufacturing sites create demand for trays and pouches used in surgical device supply.
- Welsh and Scottish life science clusters need smaller qualified suppliers for pilot device packs.
In-depth Analysis of Medical Device Packaging Market in Brazil
Brazil is likely to expand at 5.3% CAGR through 2036, as medical device regulation and health system demand continue to strengthen together. Agência Nacional de Vigilância Sanitária published five board resolutions and two normative instructions on medical devices in September 2024. The updates keep regulatory classification and technical files central to device market entry.
- São Paulo device distributors create demand for compliant packs and localized labeling.
- Minas Gerais hospitals support sterile consumable demand through public and private care networks.
- Paraná manufacturing links give regional converters a route into device packaging supply.
Competitive Landscape and Strategic Positioning
- Competition centers on cleanroom converting and supplier documentation strength.
- Material firms secure competitive edge if they can combine medical-grade resins, Tyvek or coated papers with qualified converting partners.
- Converters gain value when they reduce design changes and support repeated device audits.
- Entry barriers include cleanroom investment and long requalification cycles.
The market is moderately concentrated around suppliers that can handle medical-grade materials and regulated quality systems. Amcor is strengthening its position through tray and lidding capacity in Class 7 cleanroom settings. DuPont influences breathable sterile barriers through Tyvek materials used in ethylene oxide packaging. Nelipak and Oliver Healthcare Packaging compete by offering pouches, lids, rollstock and thermoformed solutions close to device qualification needs.
Competitive advantage extends beyond film conversion. Buyers want proof that a supplier can hold seal performance across sterilization, storage and transport. The shift favors companies with ISO 13485-aligned plants and technical teams that support design verification. Investment is also rising in pharmaceutical plastic packaging and high barrier packaging films where material data is becoming part of the buying decision.
Key Companies in the Medical Device Packaging Market
Competition is best read by role because material supply, converting depth and validation support differ sharply.
- Global Sterile Barrier and Tray Leaders: Amcor plc, DuPont de Nemours, Inc., Nelipak Corporation and Oliver Healthcare Packaging Company hold clear positions through medical-grade materials, cleanroom converting and device-specific technical support.
- Film and Pouch Specialists: Klöckner Pentaplast Group, PAXXUS, Inc., Wipak Group and Sterimed Group compete through films, pouches, coated materials and sterile barrier formats used by medical device firms.
- Regional and Adjacent Participants: Tekni-Plex, Inc., Huhtamaki Oyj and UFlex Limited support the wider healthcare packaging base through flexible packaging, specialty films and regional converting reach.
Competitive Benchmarking: Medical Device Packaging Market
| Company | Sterile Barrier Depth | Cleanroom Converting Support | Validation Documentation Support | Geographic Footprint |
|---|---|---|---|---|
| Amcor plc | High | Strong | Strong | Global with Europe and United States healthcare tray capacity |
| DuPont de Nemours, Inc. | High | Medium | Strong | Global with Tyvek material depth and technical support |
| Nelipak Corporation | High | Strong | Strong | North America and Europe with Asia-Pacific expansion |
| Oliver Healthcare Packaging Company | High | Strong | Strong | Global with pouch and lidstock focus |
| Klöckner Pentaplast Group | Medium | Strong | Medium | Global with rigid and flexible film supply |
| PAXXUS, Inc. | Medium | Medium | Strong | North America centered with specialty flexible barriers |
| Wipak Group | Medium | Strong | Medium | Europe centered with healthcare films and pouches |
| Sterimed Group | Medium | Medium | Strong | Europe centered with medical papers and sterile barriers |
| Tekni-Plex, Inc. | Medium | Strong | Medium | Global with healthcare materials and components |
Source: Future Market Insights competitive analysis, 2026. Ratings reflect relative positioning based on sterile barrier depth, cleanroom converting support and validation documentation support.
Key Developments in Medical Device Packaging Market
- In September 2025, Amcor expanded healthcare thermoformed tray capabilities in Sligo, Ireland and referenced 2024 additions in Oshkosh, Wisconsin and Mankato, Minnesota. The company stated that specialized healthcare tray sites operate under the ISO 13485 quality system.
- In 2025, DuPont named B. Braun among Tyvek Sustainable Healthcare Packaging Awards winners. The redesign eliminated 64 metric tons of plastic and 4,400 pounds of paper each year.
- In April 2025, Nelipak expanded its Asia-Pacific sterile barrier packaging presence through direct and partner channels. The move widened access to pouches, bags, lids and coated medical materials.
Key Players in the Medical Device Packaging Market
Major Global Players:
- Amcor plc
- DuPont de Nemours, Inc.
- Nelipak Corporation
- Oliver Healthcare Packaging Company
- Klöckner Pentaplast Group
- Wipak Group
Specialist and Regional Players:
- PAXXUS, Inc.
- Sterimed Group
- Tekni-Plex, Inc.
- Huhtamaki Oyj
- UFlex Limited
Report Scope and Coverage
| Parameter | Details |
|---|---|
| Quantitative Units | USD 45.2 billion to USD 68.4 billion, at 4.2% CAGR |
| Market Definition | Packaging materials and formats that protect medical devices and maintain sterility or integrity until clinical use |
| Material | Plastic, Tyvek, Aluminum, Glass, Paperboard, Fabric, Silicone |
| Class | Class I, Class II, Class III |
| Application | Sterile Packaging, Non-Sterile Packaging, Implantable Device Packaging, Diagnostic and Monitoring Device Packaging |
| Packaging Format | Bags and Pouches, Films and Wraps, Lids, Rollstock, Containers, Trays, Stick Packs, Clamshells, Blister Packs, Boxes, Flow Wraps |
| Sterilization Route | Ethylene Oxide, Radiation, Steam, Vaporized Peroxide |
| Regions Covered | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered | United States, China, India, Germany, Japan, United Kingdom, Brazil and 40+ countries |
| Key Companies Profiled | Amcor plc, DuPont de Nemours, Nelipak, Oliver Healthcare Packaging, Klöckner Pentaplast, PAXXUS, Wipak, Sterimed, Tekni-Plex |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid bottom-up and top-down methodology using device output, packaging format shares, supplier capacity and sterilization evidence |
Source: Future Market Insights, 2026.
Medical Device Packaging Market Breakdown by Material, Class, Application, Packaging Format, Sterilization Route, and Region
Medical Device Packaging Market Segmented by Material:
- Plastic
- Tyvek
- Aluminum
- Glass
- Paperboard
- Fabric
- Silicone
Medical Device Packaging Market Segmented by Class:
- Class I
- Class II
- Class III
Medical Device Packaging Market Segmented by Application:
- Sterile Packaging
- Non-Sterile Packaging
- Implantable Device Packaging
- Diagnostic and Monitoring Device Packaging
Medical Device Packaging Market Segmented by Packaging Format:
- Bags and Pouches
- Films and Wraps
- Lids
- Rollstock
- Containers
- Trays
- Stick Packs
- Clamshells
- Blister Packs
- Boxes
- Flow Wraps
Medical Device Packaging Market Segmented by Sterilization Route:
- Ethylene Oxide
- Radiation
- Steam
- Vaporized Peroxide
Medical Device Packaging Market by Region:
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- United Kingdom
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan and Baltic
- Rest of Eastern Europe
- East Asia
- China
- Japan
- South Korea
- South Asia and Pacific
- India
- ASEAN
- Australia and New Zealand
- Rest of South Asia and Pacific
- Middle East and Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East and Africa
Research Sources and Bibliography
- United States Environmental Protection Agency. (2024, March 14). EPA announces final rule to slash toxic emissions of ethylene oxide and reduce cancer risk. United States Environmental Protection Agency.
- United States Food and Drug Administration. (2026, May 6). Submission statistics. United States Food and Drug Administration.
- International Organization for Standardization. (2019). ISO 11607-2:2019 Packaging for terminally sterilized medical devices. International Organization for Standardization.
- National Medical Products Administration. (2024, May 11). China Medical Device Standards Management Annual Report (2023). National Medical Products Administration.
- Press Information Bureau, Government of India. (2024, December 17). Production Linked Incentive Scheme for Promoting Domestic Manufacturing of Medical Devices. Press Information Bureau.
- Ministry of Health, Labour and Welfare, Japan. (2025, December 24). Annual report on dynamic statistics of pharmaceutical industry production in 2024. Ministry of Health, Labour and Welfare.
- Medicines and Healthcare products Regulatory Agency. (2024, January 31). MHRA announces two new UK Approved Bodies to certify medical devices. Government of the United Kingdom.
- Agência Nacional de Vigilância Sanitária. (2024, October 1). Anvisa revisa e consolida normas da área de Dispositivos Médicos. Agência Nacional de Vigilância Sanitária.
- DuPont de Nemours, Inc. (2025, October 8). Tyvek sustainable healthcare packaging awards. DuPont de Nemours, Inc.
- Amcor plc. (2025, September 23). Amcor expands thermoformed tray capabilities for healthcare. Amcor plc.
- Nelipak Corporation. (2025, April 7). Nelipak Healthcare Packaging expands commitment to serve customers in Asia-Pacific Region with its sterile barrier medical packaging products. Nelipak Corporation.
- PAXXUS, Inc. (2024, March 21). FPA awards StreamTwo®. PAXXUS, Inc.
- Klöckner Pentaplast Group. (2024, October 30). Klöckner Pentaplast launches kpNext® MDR1: sustainable medical device packaging film. Klöckner Pentaplast Group.
This bibliography is provided for reader reference and uses primary government, standards-body, regulatory and official company publications.
This Report Answers
- What is the expected value of the Medical Device Packaging Market in 2026 and by 2036?
- Which packaging formats will hold the strongest role in medical device sterile barrier systems?
- Why does plastic remain the leading material in the Medical Device Packaging Market?
- How do ethylene oxide rules affect medical device packaging material selection?
- Which countries are expected to record stronger growth in medical device packaging demand?
- How do ISO 11607 requirements influence supplier qualification and pack validation?
- Which companies are most relevant across sterile barrier materials and cleanroom conversion?
- What does the report include and exclude from medical device packaging scope?
- How was the 2026 to 2036 forecast built and checked against market evidence?
Frequently Asked Questions
What is the global Medical Device Packaging Market demand in 2026?
In 2026, the global Medical Device Packaging Market is expected to be worth USD 45.2 billion as sterile device output rises.
What will the global Medical Device Packaging Market be worth by 2036?
By 2036, the global Medical Device Packaging Market is projected to reach USD 68.4 billion as validation-led packaging value increases.
What is the expected CAGR for the Medical Device Packaging Market?
The Medical Device Packaging Market is projected to expand at 4.2% CAGR from 2026 to 2036 across global demand.
Which material leads the Medical Device Packaging Market?
Plastic is projected to account for 42.7% share in 2026 since films, trays and pouches need sterilization compatibility.
Which packaging format leads the Medical Device Packaging Market?
Bags and pouches are expected to hold 21.5% share in 2026 due to flexible sterile barrier use.
Which country has the stronger Medical Device Packaging Market outlook?
China is projected at 6.2% CAGR through 2036 as device production and standards activity expand together.
What does the Medical Device Packaging Market include?
The market includes sterile and non-sterile packaging materials that protect devices during sterilization, shipment and clinical opening.
How is the Medical Device Packaging Market forecast developed?
The forecast uses device output, packaging format shares, supplier capacity and sterilization-route checks across major regulated countries.
Why is sterile barrier validation important in Medical Device Packaging Market selection?
Sterile barrier validation proves that packaging can keep devices protected through sealing, aging, sterilization and distribution stress.
How are sustainability goals changing the Medical Device Packaging Market?
Sustainability goals are pushing mono-material pouches and lower-plastic designs, but every change still needs sterile barrier proof.
What should buyers check before changing a Medical Device Packaging supplier?
Device firms should check cleanroom controls, ISO 13485 alignment, seal data, aging results and sterilization compatibility before approving a switch.
How do medical device packaging suppliers compete?
Suppliers compete through material reliability, cleanroom converting, validation support and the ability to reduce audit risk for device makers.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Source
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Source , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Source , 2026 to 2036
- Soy
- Almond
- Rice
- Coconut
- Oats
- Others
- Soy
- Y-o-Y Growth Trend Analysis By Source , 2021 to 2025
- Absolute $ Opportunity Analysis By Source , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type, 2026 to 2036
- Non-Dairy Milk
- Butter
- Cheeses
- Yogurts
- Ice Cream
- Others
- Non-Dairy Milk
- Y-o-Y Growth Trend Analysis By Product Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Channel
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Channel, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Channel, 2026 to 2036
- Supermarkets and Hypermarkets
- Convenience Stores
- Online Retail
- Others
- Supermarkets and Hypermarkets
- Y-o-Y Growth Trend Analysis By Channel, 2021 to 2025
- Absolute $ Opportunity Analysis By Channel, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Source
- By Product Type
- By Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Source
- By Product Type
- By Channel
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Source
- By Product Type
- By Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Source
- By Product Type
- By Channel
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Source
- By Product Type
- By Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Source
- By Product Type
- By Channel
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Source
- By Product Type
- By Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Source
- By Product Type
- By Channel
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Source
- By Product Type
- By Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Source
- By Product Type
- By Channel
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Source
- By Product Type
- By Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Source
- By Product Type
- By Channel
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Source
- By Product Type
- By Channel
- By Country
- Market Attractiveness Analysis
- By Country
- By Source
- By Product Type
- By Channel
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Source
- By Product Type
- By Channel
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Source
- By Product Type
- By Channel
- Competition Analysis
- Competition Deep Dive
- Danone
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Oatly Group AB
- Blue Diamond Growers
- Califia Farms
- Daiya Foods
- SunOpta
- Vitasoy
- Ripple Foods
- Forager Project
- Danone
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Source , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Source , 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 10: Latin America Market Value (USD Million) Forecast by Source , 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 13: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Western Europe Market Value (USD Million) Forecast by Source , 2021 to 2036
- Table 15: Western Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 17: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 18: Eastern Europe Market Value (USD Million) Forecast by Source , 2021 to 2036
- Table 19: Eastern Europe Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 20: Eastern Europe Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 21: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: East Asia Market Value (USD Million) Forecast by Source , 2021 to 2036
- Table 23: East Asia Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 24: East Asia Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 25: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 26: South Asia and Pacific Market Value (USD Million) Forecast by Source , 2021 to 2036
- Table 27: South Asia and Pacific Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 28: South Asia and Pacific Market Value (USD Million) Forecast by Channel, 2021 to 2036
- Table 29: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 30: Middle East & Africa Market Value (USD Million) Forecast by Source , 2021 to 2036
- Table 31: Middle East & Africa Market Value (USD Million) Forecast by Product Type, 2021 to 2036
- Table 32: Middle East & Africa Market Value (USD Million) Forecast by Channel, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Source , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Source , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Source
- Figure 6: Global Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Product Type
- Figure 9: Global Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Channel
- Figure 12: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Region
- Figure 15: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 23: North America Market Value Share and BPS Analysis by Source , 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Source , 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Source
- Figure 26: North America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 28: North America Market Attractiveness Analysis by Product Type
- Figure 29: North America Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Channel
- Figure 32: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 33: Latin America Market Value Share and BPS Analysis by Source , 2026 and 2036
- Figure 34: Latin America Market Y-o-Y Growth Comparison by Source , 2026-2036
- Figure 35: Latin America Market Attractiveness Analysis by Source
- Figure 36: Latin America Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 37: Latin America Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 38: Latin America Market Attractiveness Analysis by Product Type
- Figure 39: Latin America Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by Channel
- Figure 42: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 43: Western Europe Market Value Share and BPS Analysis by Source , 2026 and 2036
- Figure 44: Western Europe Market Y-o-Y Growth Comparison by Source , 2026-2036
- Figure 45: Western Europe Market Attractiveness Analysis by Source
- Figure 46: Western Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 47: Western Europe Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 48: Western Europe Market Attractiveness Analysis by Product Type
- Figure 49: Western Europe Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 50: Western Europe Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 51: Western Europe Market Attractiveness Analysis by Channel
- Figure 52: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 53: Eastern Europe Market Value Share and BPS Analysis by Source , 2026 and 2036
- Figure 54: Eastern Europe Market Y-o-Y Growth Comparison by Source , 2026-2036
- Figure 55: Eastern Europe Market Attractiveness Analysis by Source
- Figure 56: Eastern Europe Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 57: Eastern Europe Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 58: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 59: Eastern Europe Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 60: Eastern Europe Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 61: Eastern Europe Market Attractiveness Analysis by Channel
- Figure 62: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 63: East Asia Market Value Share and BPS Analysis by Source , 2026 and 2036
- Figure 64: East Asia Market Y-o-Y Growth Comparison by Source , 2026-2036
- Figure 65: East Asia Market Attractiveness Analysis by Source
- Figure 66: East Asia Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 67: East Asia Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 68: East Asia Market Attractiveness Analysis by Product Type
- Figure 69: East Asia Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 70: East Asia Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 71: East Asia Market Attractiveness Analysis by Channel
- Figure 72: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 73: South Asia and Pacific Market Value Share and BPS Analysis by Source , 2026 and 2036
- Figure 74: South Asia and Pacific Market Y-o-Y Growth Comparison by Source , 2026-2036
- Figure 75: South Asia and Pacific Market Attractiveness Analysis by Source
- Figure 76: South Asia and Pacific Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 77: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 78: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 79: South Asia and Pacific Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 80: South Asia and Pacific Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 81: South Asia and Pacific Market Attractiveness Analysis by Channel
- Figure 82: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 83: Middle East & Africa Market Value Share and BPS Analysis by Source , 2026 and 2036
- Figure 84: Middle East & Africa Market Y-o-Y Growth Comparison by Source , 2026-2036
- Figure 85: Middle East & Africa Market Attractiveness Analysis by Source
- Figure 86: Middle East & Africa Market Value Share and BPS Analysis by Product Type, 2026 and 2036
- Figure 87: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type, 2026-2036
- Figure 88: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 89: Middle East & Africa Market Value Share and BPS Analysis by Channel, 2026 and 2036
- Figure 90: Middle East & Africa Market Y-o-Y Growth Comparison by Channel, 2026-2036
- Figure 91: Middle East & Africa Market Attractiveness Analysis by Channel
- Figure 92: Global Market - Tier Structure Analysis
- Figure 93: Global Market - Company Share Analysis

