Medical Cyclotron Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Medical Cyclotron Market The medical cyclotron market is segmented by Product Type (Ring Cyclotron, AVF Cyclotron), End User (Pharmaceutical Companies, Specialized Clinics, Hospitals), and Region. Forecast for 2026 to 2036.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Medical Cyclotron Market Size, Market Forecast and Outlook By FMI
The medical cyclotron market is projected to expand from USD 263.1 million in 2026 to USD 677.4 million by 2036. The market is anticipated to register a 9.9% CAGR during the forecast period. Ring cyclotron systems are likely to lead product type with a 56.0% share, while pharmaceutical companies are expected to account for 58.0% of end-user revenue in 2026.
Summary of the Medical Cyclotron Market
- Demand and Growth Drivers
- PET-aligned diagnostic capacity is expected to keep medical cyclotron demand active across radiopharmacy supply networks.
- Radiopharmaceutical manufacturing expansion is likely to increase the need for predictable isotope production capacity.
- Service-backed uptime requirements are anticipated to shape purchasing decisions as delivery windows remain narrow.
- Product and Segment View
- Ring cyclotron systems are expected to lead the product type segment due to standardized service routines and operating predictability.
- Pharmaceutical companies are likely to remain the primary purchasing group as controlled production networks scale.
- AVF cyclotron systems are anticipated to support specialized deployment needs where buyers require defined production profiles.
- Geography and Competitive Outlook
- India is expected to lead country-level expansion as diagnostic infrastructure and localized supply continuity receive stronger investment.
- China is likely to record steady gains as production networks and radiopharmaceutical logistics become more structured.
- Vendors with stronger qualification support and service coverage are anticipated to improve their position in regulated installations.
- Analyst Opinion
- Sabyasachi Ghosh, Principal Consultant for Healthcare at FMI, suggests, “The medical cyclotron market is likely to move toward service-backed and qualification-ready deployment models. Suppliers with strong documentation discipline, field engineering coverage, and restart reliability are expected to gain advantage as radiopharmaceutical networks protect production continuity.”
- Medical Cyclotron Market Value Analysis
- The medical cyclotron market is shifting from standalone equipment purchases toward lifecycle-backed production infrastructure.
- Demand is likely to rise as PET imaging volumes require stable isotope supply and reliable daily output.
- Equipment use is expected to increase as pharmaceutical companies expand radiopharmaceutical manufacturing networks.
- Spending is anticipated to be supported by theranostics momentum, localized production planning, and stronger uptime requirements.
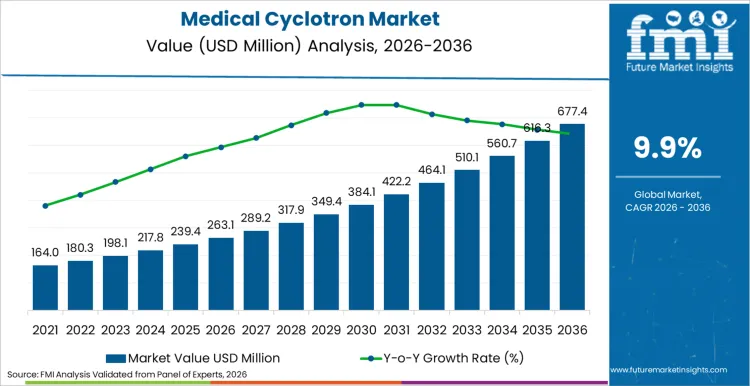
Medical Cyclotron Market Key Takeaways
| Metric | Value |
|---|---|
| Industry Size (2026) | USD 263.1 Million |
| Industry Value (2036) | USD 677.4 Million |
| CAGR (2026 to 2036) | 9.9% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
What Factors Contribute to the Growth of the Medical Cyclotron Market?
Demand is rising as healthcare systems scale PET-centric workflows and reinforce continuity requirements across radiopharmacy supply networks. Buyers are moving from opportunistic capacity additions toward structured planning driven by reliability, uptime performance, and repeatable daily output. This shift is expanding procurement emphasis on predictable commissioning outcomes and lifecycle service assurance, especially where production environments operate under controlled release timelines.
Supplier selection is also being shaped by governance maturity in regulated operational settings. Documentation discipline, qualification readiness, and vendor-led uptime support are being positioned as high-value differentiators. Operating models are frequently benchmarked against procurement frameworks associated with nuclear medicine equipment, where downtime exposure carries direct clinical and economic consequences.
How is Cyclotron Demand Structured by Architecture and Buyer Environment?
The market is segmented by product architecture and end-user profile to reflect how procurement requirements differ across manufacturing-led installations and care-delivery settings. Product type segmentation highlights where buyers standardize deployment to reduce operating variance, while end-user segmentation captures differences in qualification rigor, uptime sensitivity, and decision-making authority across production and clinical environments.
Segmentation is increasingly used to align supplier priorities with real-world operating constraints, including schedule certainty, service responsiveness, and maintenance predictability. As multi-site operating models expand, buyers are prioritizing deployment profiles that limit fleet complexity while supporting scalable training, standardized documentation, and consistent performance expectations.
Why do Ring Cyclotron systems hold a significant share?
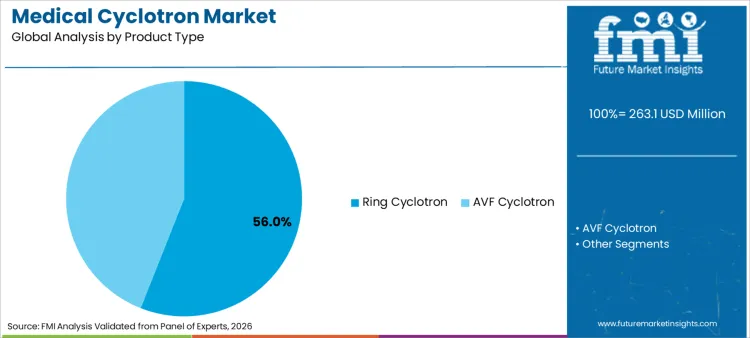
Ring cyclotron account for a 56.0% share in 2026, supported by fit with established deployment preferences that prioritize operating predictability and standardized service routines. This segment benefits from procurement consistency across networks that prefer repeatable implementation pathways and stable lifecycle support structures.
Why are Pharmaceutical Companies the primary purchasing group?
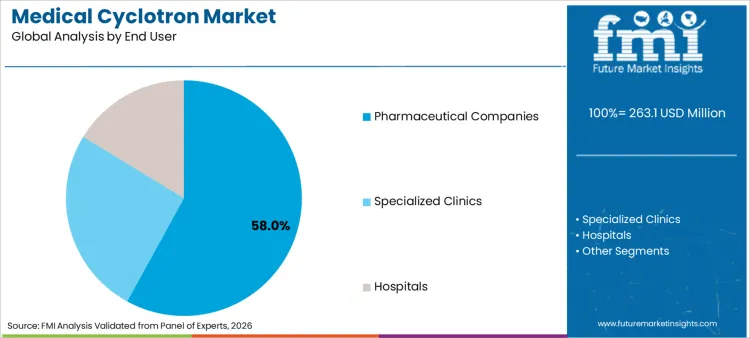
Pharmaceutical companies command a 58.0% share of end-user demand in 2026, reflecting their role in scaling controlled production networks under schedule-bound release models. Procurement is driven by uptime assurance, disciplined documentation requirements, and long-horizon service contracting practices. Planning is frequently aligned to capacity execution models associated with radiopharmaceuticals, where repeatable supply continuity directly influences commercial feasibility.
How Will Market Priorities Shift as Radiopharmaceutical Programs Scale?
How will Buyer Scorecards evolve as Delivery Windows Tighten Further?
Buyers are raising the weight of uptime performance, restart readiness, and predictable maintenance cycles as delivery windows narrow. Acceptance frameworks are shifting toward measurable operational stability that protects downstream scheduling and reduces disruption exposure under high utilization patterns.
What is Driving Stronger Preference for Standardized Multi-site Deployment Models?
Multi-site operators are limiting fleet complexity to reduce training variance, documentation fragmentation, and spares uncertainty. Procurement strategies are prioritizing replicable deployment frameworks aligned to stable execution practices common in nuclear medicine equipment environments where governance discipline influences performance consistency.
Which Supplier Capabilities are emerging as Differentiators beyond Equipment Availability?
Differentiation is strengthening around qualification support depth, service coverage density, and documentation readiness that reduces audit friction. Buyers are prioritizing vendors that can support consistent production cadence through structured engineering coverage and predictable lifecycle support.
Which National Ecosystems will shape Installed Base Strategy through 2036?
Growth varies by installed base maturity, procurement discipline, and the scale of radiopharmaceutical production networks supporting clinical demand. Mature markets show a stronger bias toward upgrades and replacement planning tied to reliability, while emerging markets show higher momentum for infrastructure expansion and localized supply development. Buyers in scale-up regions are prioritizing deployment readiness and service-backed continuity to stabilize performance while capacity ramps.
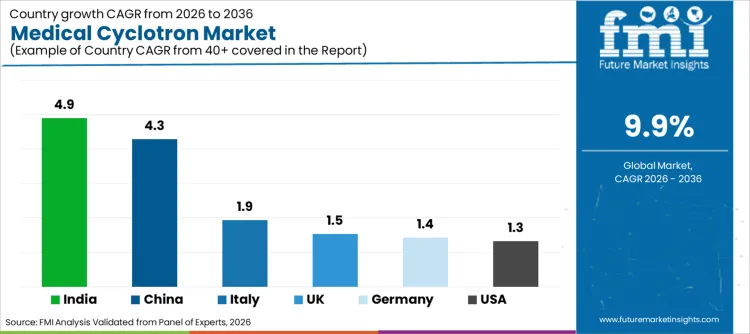
| Country | CAGR (2026 to 2036) |
|---|---|
| USA | 1.3% |
| Germany | 1.4% |
| India | 4.9% |
| Italy | 1.9% |
| UK | 1.5% |
| China | 4.3% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Why is the Medical Cyclotron Industry in USA expanding steadily?
Demand in USA is set to grow at a 1.3% CAGR through 2036, shaped by an established installed base and structured replacement planning focused on operational reliability. Buyers are prioritizing lifecycle service depth, predictable maintenance execution, and standardized operating baselines across multi-site production and imaging networks.
Why is Germany maintaining a Structured Growth Trajectory?
Germany is projected to expand at a 1.4% CAGR, supported by procurement discipline and strong compliance expectations across production environments. Buyer preference centers on documentation integrity, validated operating routines, and supplier capability to support qualification workflows with minimal variability.
Why is India emerging as a High-growth Market for Cyclotron Installations?
India is forecast to grow at a 4.9% CAGR, driven by expanding diagnostic infrastructure and stronger investment focus on localized supply continuity. Buyers are prioritizing schedule stability, service-backed uptime, and scalable deployment readiness aligned to rising imaging volumes linked to PET scanners adoption patterns.
Why is Italy showing Consistent Capacity Expansion?
Italy is expected to register a 1.9% CAGR, supported by measured capacity additions and upgrade cycles aligned to imaging demand planning. Procurement decisions are centered on predictable commissioning outcomes, stable uptime performance, and service responsiveness that reduces operating disruption during installation transitions.
Why is the UK’s Medical Cyclotron Market expanding at a measured pace?
The UK is projected to grow at a 1.5% CAGR, supported by structured procurement cycles and emphasis on operational continuity. Buyers are prioritizing reliability-backed contracting, documented service readiness, and deployment profiles that protect predictable output cadence.
Why is China positioned as a Scale-driven Growth Engine?
China is forecast to expand at a 4.3% CAGR, supported by rapid infrastructure scaling and stronger integration of production networks. Buyers are prioritizing deployment standardization, higher operational resilience, and long-term service coverage to sustain stable output as capacity expands, reflecting operating behaviors aligned to radiopharmaceutical logistics models where timing discipline directly influences delivery success.
What is the Competitive Outlook for the Medical Cyclotron Market?
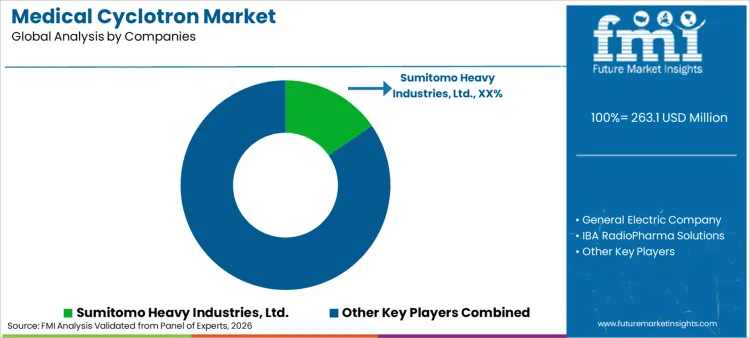
Competition is being shaped by execution credibility, service coverage strength, and qualification support maturity rather than narrow feature-level differentiation. Vendors are strengthening positioning through lifecycle contracting models, remote support readiness, and predictable spares availability aligned to uptime-driven procurement preferences.
Strategic emphasis is moving toward reducing commissioning risk through structured documentation packages and repeatable deployment support, especially for operators standardizing multi-site performance expectations. Buyer evaluations increasingly reward suppliers that can sustain stable output cadence under real-world operating conditions where schedule integrity is non-negotiable.
Key Players in Medical Cyclotron Market
- Sumitomo Heavy Industries, Ltd.
- General Electric Company
- IBA RadioPharma Solutions
- Advanced Cyclotron Systems Inc.
- Best Medical International Inc.
- Siemens AG
- Varian Medical Systems Inc.
- Ionetix Corporation
- Alcen
- Isosolution Inc.
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD Million |
| Product Type | Ring Cyclotron, AVF Cyclotron |
| End User | Pharmaceutical Companies, Specialized Clinics, Hospitals |
| Key Countries | USA, Germany, India, Italy, China, UK |
| Companies | Sumitomo Heavy Industries, Ltd.; General Electric Company; IBA RadioPharma Solutions; Advanced Cyclotron Systems Inc.; Best Medical International Inc.; Siemens AG; Varian Medical Systems Inc.; Ionetix Corporation; Alcen; Isosolution Inc. |
| Regions | North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Medical Cyclotron Market by Segments
Product Type:
- Ring Cyclotron
- AVF Cyclotron
End User:
- Pharmaceutical Companies
- Specialized Clinics
- Hospitals
Region:
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia & Pacific
- Middle East & Africa
Bibliography
- International Atomic Energy Agency. (2009). Cyclotron produced radionuclides: Guidelines for setting up a facility (Technical Reports Series No. 471). IAEA.
- International Atomic Energy Agency. (2014). Radiation protection and safety of radiation sources: International basic safety standards (Safety Standards Series No. GSR Part 3). IAEA.
- USA Food and Drug Administration. (2018). PET drug products: Current good manufacturing practice (CGMP). FDA.
- USA Food and Drug Administration. (2019). PET drugs: Current good manufacturing practice (CGMP); Small entity compliance guide. FDA.
- Society of Nuclear Medicine and Molecular Imaging. (2025). Guideline for the use of radiopharmaceuticals in diagnostic nuclear medicine and PET. SNMMI.
Frequently Asked Questions
How large is the demand for Medical Cyclotron in the global market in 2026?
Demand for medical cyclotrons is estimated to be valued at USD 263.1 million in 2026.
What will be the market size of Medical Cyclotron in the global market by 2036?
The market size for medical cyclotron is projected to reach USD 677.4 million by 2036.
What is the expected demand growth for Medical Cyclotron in the global market between 2026 and 2036?
The medical cyclotron market is expected to grow at a CAGR of 9.9% between 2026 and 2036.
Which Product Type is poised to lead global sales by 2026?
Ring cyclotrons are expected to be the dominant product type, capturing 56.0% of the global market share in 2026.
How significant is the role of Pharmaceutical Companies End User in driving Medical Cyclotron adoption in 2026?
Pharmaceutical companies represent a critical end-user segment, projected to hold a substantial 58.0% share of the total market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Ring Cyclotron
- AVF Cyclotron
- Ring Cyclotron
- Y to o to Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2026 to 2036
- Pharmaceutical Companies
- Specialized Clinics
- Hospitals
- Pharmaceutical Companies
- Y to o to Y Growth Trend Analysis By End User, 2021 to 2025
- Absolute $ Opportunity Analysis By End User, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By End User
- Competition Analysis
- Competition Deep Dive
- Sumitomo Heavy Industries, Ltd.
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- General Electric Company
- IBA RadioPharma Solutions
- Advanced Cyclotron Systems Inc.
- Best Medical International Inc.
- Siemens AG
- Varian Medical Systems Inc.
- Ionetix Corporation
- Alcen
- Isosolution Inc.
- Sumitomo Heavy Industries, Ltd.
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End User, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End User, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021 to 2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 8: Global Market Attractiveness Analysis by End User
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026 to 2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026 to 2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 25: North America Market Attractiveness Analysis by End User
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 32: Latin America Market Attractiveness Analysis by End User
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 39: Western Europe Market Attractiveness Analysis by End User
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End User
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 53: East Asia Market Attractiveness Analysis by End User
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End User
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026 to 2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End User, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End User, 2026 to 2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End User
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

