Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market Size, Market Forecast and Outlook By FMI
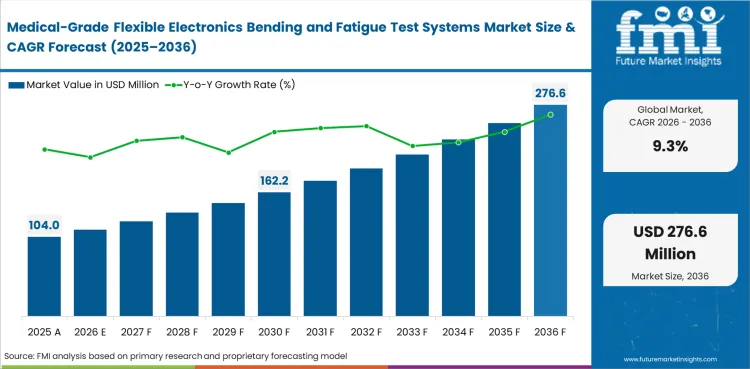
The medical-grade flexible electronics bending and fatigue test systems market was valued at USD 95.2 million in 2025. It is estimated at USD 104 million in 2026 and is projected to reach USD 253.1 million by 2036, expanding at a CAGR of 9.3% during 2026 to 2036. Cyclic bending is expected to lead with a 38% share, while Medtech OEMs are projected to account for 45% of demand.
Summary of Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market
- Demand and Growth Drivers
- Wearable patches, implantables need repeated-cycle testing before product release. This supports demand for bending and fatigue systems built for delicate assemblies.
- Medtech developers are placing more focus on mechanical reliability as flexible circuits, conductive traces, and laminated sensor layers move into longer-use medical applications.
- Laboratories are investing in configurable benchtop and low-force platforms as developers need controlled bend radius, cycle count, and failure-point analysis during qualification work.
- Product and Segment View
- Cyclic bending is expected to lead the test mode, supported by its direct relevance to repeated flexing in wearable and skin-applied medical electronics.
- Benchtop systems are likely to hold a strong position, since many medtech R&D and validation programs need compact setups for controlled low-force testing.
- Wearable patches represent a major application area, as these products combine thin materials and repeated motion during actual use.
- Geography and Competitive Outlook
- The United States is expected to remain a key demand center due to its strong base in medical device development and digital health product qualification.
- Western Europe stays important as medical electronics programs move toward home-use and body-worn formats that require stronger mechanical validation.
- Suppliers with strength in dynamic testing, fixture customization, and biomedical sample handling are likely to stay better placed as buyers look for repeatable results and application support.
- Analyst Opinion
- , Principal Consultant at FMI says, “Demand is rising as wearable patches and other flexible medical devices move into longer-use applications, which is pushing Medtech OEMs to invest earlier in cyclic bending, low-force fatigue testing, and fixture customization to improve qualification reliability.”
- Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market Value Analysis
- The market is moving from a narrow lab equipment category into a more defined testing niche for flexible medical electronics.
- Demand is being supported by wider use of wearable patches and other body-worn medical devices.
- Adoption is also rising as Medtech OEMs place greater focus on cyclic bending, fatigue validation, and repeatable qualification data.
- Growth is further backed by the need for testing systems that improve reliability checks and handle delicate flexible assemblies.
Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market Definition
The market covers test systems designed to evaluate the bending durability, cyclic fatigue response, and mechanical reliability of medical-grade flexible electronic assemblies and their subcomponents.
Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market Inclusions
Included in scope are low-force and mid-force systems configured for cyclic bending, dynamic fatigue, tension-compression or flexural work, and custom fixture arrangements used on flexible printed circuits, laminated sensor stacks, conductive films, stretchable traces, smart adhesive patches and implant leads.
Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market Exclusions
Excluded from scope are broad universal testing systems sold into general plastics, metals, packaging, or construction work without a clear medical flexible-electronics application. Also excluded are electrical-only testers, connectivity validation tools, cybersecurity systems and finished wearable devices themselves.
Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market Research Methodology
- Primary Research: Secondary evidence was organized into an analyst-built demand model that reflects medtech R&D intensity, digital-health reimbursement signals, patent activity, and the installed base for specialized mechanical qualification work.
- Desk Research: Non-commercial sources included FDA digital-health device listings, WIPO patent analytics, and MedTech Europe’s 2025 facts and figures publication, which provided medical technology scale and country structure.
- Market Sizing and Forecasting: FMI used a bottom-up addressable-lab and average selling price framework and cross-checked the result against the global medical device parent market and specialized material tester demand.
- Data Validation and Update Cycle: Forecast assumptions were stress-tested against current company product activity, regional medtech concentration, and a conservative adoption curve for cyclic qualification tools through 2036.
Why is the Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market Growing?
- Body-worn devices are shifting from short pilot runs to regulated product programs that need repeatable cycle-life evidence.
- Short medtech product refresh cycles keep laboratories investing in configurable fixtures and faster data capture tools.
- Digital reimbursement and home-monitoring adoption are raising the number of flexible assemblies that require mechanical validation.
Wearable and skin-interfaced medical devices are moving into more demanding service conditions. This changes the way developers qualify flexible circuitry. The FDA’s current list of sensor-based digital health devices reaches back to 2014 and explicitly covers smartwatches, rings, patches, and bands. As these products stay on the body longer, engineers need more than a single flex demonstration, since repeated body motion can shift signal stability long before a visible fracture appears. Spending moves toward dedicated rigs that control bend radius, cycle count, and failure thresholds with better repeatability. Demand therefore tracks adjacent adoption in remote patient monitoring devices.
Pressure-sensing lens programs are one of the clearest growth anchors for this niche. Glaucoma-related work continues to show how smart lenses rely on reliable readout and repeatable calibration, which supports steady demand for optical and telemetry validation platforms. Broader work in tear glucose sensing and therapeutic lens formats is adding testing needs across early development programs. As more smart lenses combine biosensing with wireless communication, suppliers with strong optical metrology and compact electronics validation are likely to stay in a better position.
Segmental Analysis
- Smart Contact Lens Optical and Telemetry Test Equipment is estimated at USD 78 million in 2025 and is projected to reach about USD 188 million by 2036.
- Cyclic bending is expected to account for 38.0% of the test mode segment in 2026, since repeated flexing remains the first reliability check for most flexible medical electronic assemblies.
- Benchtop systems are estimated to capture 42.0% share in 2026, wearable patches are set to hold 34.0% of application demand.
- Medtech OEMs are projected to account for 45.0% of end-user demand, while direct sales remain important due to the need for fixture customization and technical support.
- Direct sales are expected to contribute 63.0% of sales channel demand in 2026.
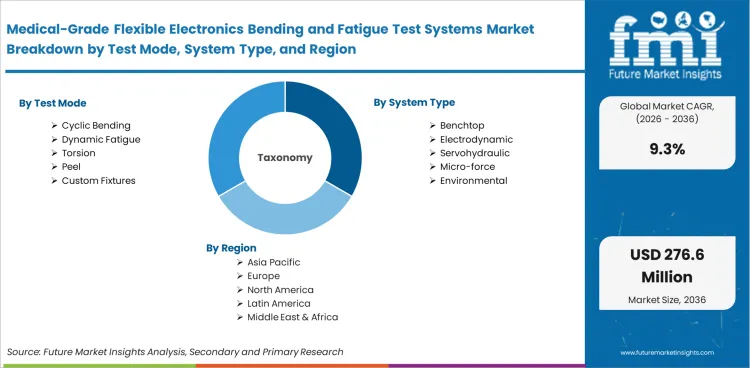
Segmentation in this market is shaped by test method, load profile, fixture adaptability, application fit, and end-user workflow. Manufacturers usually compare systems based on how well they can reproduce real flex conditions across delicate medical assemblies rather than on frame size. The market is segmented by test mode, system type, application, end user, sales channel, and region. Test mode includes cyclic bending, dynamic fatigue, torsion, peel, and custom fixtures. System type includes benchtop, electrodynamic, servohydraulic, micro-force, and environmental platforms. Application coverage includes wearable patches, implantables, smart catheters, neuro interfaces, and biosensor strips. End users include Medtech OEMs, contract labs, university labs, CDMOs, and hospitals. Sales channels include direct sales, distributors, integrators, service contracts, and retrofits.
Insights into the test mode segment
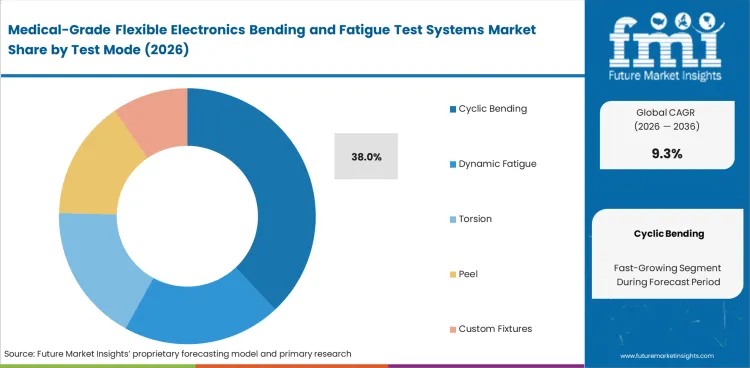
- Cyclic bending is expected to account for 38.0% of the test mode segment in 2026. Demand is strongest here since flexible medical electronics are exposed to repeated movement during handling and actual use. This makes controlled bend-cycle testing the first purchase for many labs.
- Dynamic fatigue and torsion systems matter as programs mature and manufacturers usually start with repeated-radius validation because it translates directly into wearable use conditions.
Insights into the system type segment
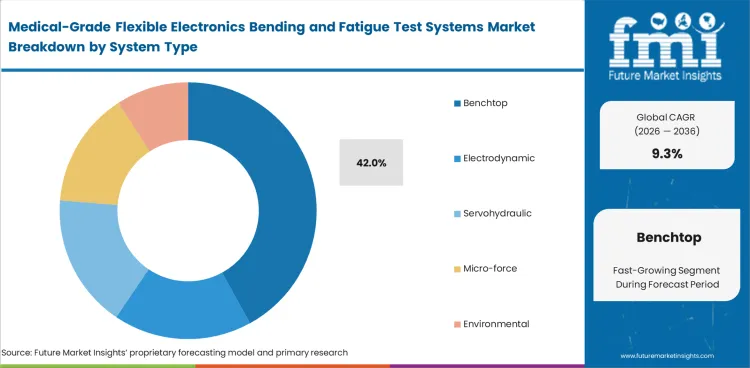
- Benchtop system’s 42% lead in 2026 is tied to laboratory reality rather than simple price. Most medtech development teams working on flexible electronics run low-force and mid-force protocols inside space-constrained R&D, biomaterials, or pilot manufacturing rooms.
- ZwickRoell continues to position electrodynamic platforms for flexural fatigue work where noise, maintenance, and space all matter alongside flexible substrate development.
Insights into the application segment
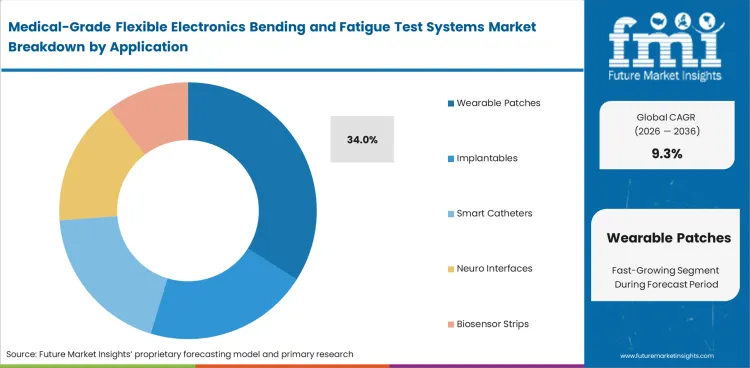
- Wearable patches are expected to represent 34.0% of application demand in 2026 due to repeated body movement and routine durability testing needs. Higher development volumes and faster design changes keep patch testing demand above other medical flexible applications.
- Europe remains an important medical device base, and development work is moving toward digital and home-use formats. Demand for flexible test equipment is rising in line with higher activity around sensor patches and related wearable medical electronics.
Insights into the end user segment
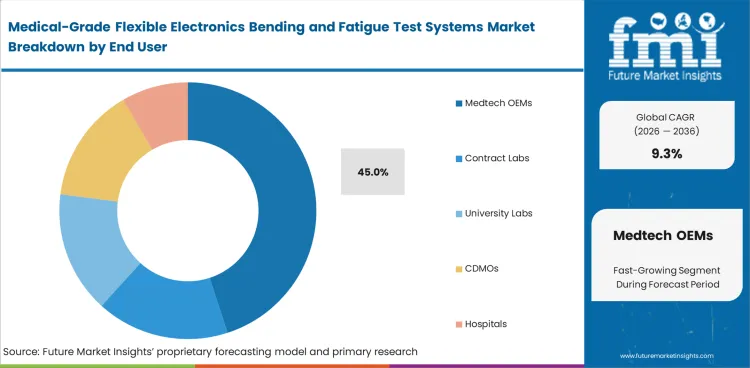
- Medtech OEMs are projected to account for 45.0% of end-user demand in 2026. This lead reflects their direct role in product qualification and pre-commercial reliability testing across flexible medical electronics programs.
- OEMs typically require tighter control over testing protocols since device performance and product consistency are linked closely to internal development timelines.
Insights into the sales channel segment
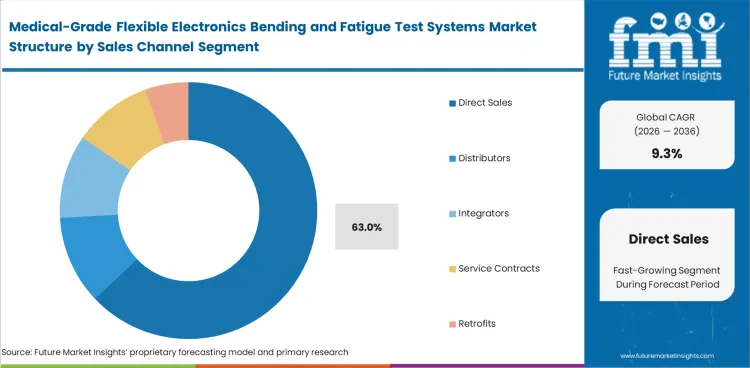
- Direct sales are expected to contribute 63.0% of sales channel demand. They lead in 2026 as buyers need supplier support for fixtures, setup, and testing requirements.
- Long qualification cycles keep direct supplier involvement important through evaluation, customization, and final purchase decisions.
Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market Drivers, Restraints, and Developments
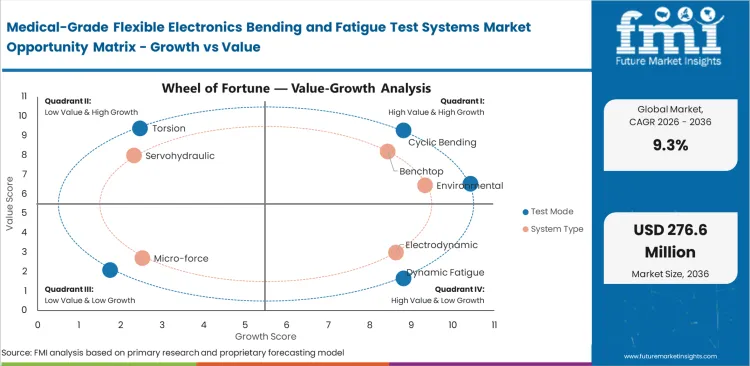
- Short-cycle validation for new wearable and implantable electronic formats is increasing the need for dedicated fatigue evidence.
- The niche remains constrained by custom-fixture expense and a limited number of qualified high-volume manufacturers.
- Electrodynamic, software-led, and automated testing platforms are gaining visibility against heavier general-purpose frames.
Developers need proof that soft circuits and bonded sensing stacks remain mechanically stable through repeated use. The demand base is narrow, so each purchase tends to be technically reviewed and often delayed until a device platform reaches a formal validation gate. Third, suppliers are moving toward quieter electrodynamic systems, better data capture, and more automation, which improves reproducibility in a niche where results carry design and regulatory weight.
Input cost pressure
Custom grips, miniature fixtures, environmental add-ons, and application engineering keep system prices above what many university and early-stage programs can justify. Manufacturers rarely purchase a base frame, since thin laminates and soft substrates often need special clamping to avoid false failures. That raises project cost and extends sales cycles. A niche tool may be technically attractive, still procurement can still be slowed by budgeting rules and limited anticipated throughput.
Testing and validation needs
Mechanical qualification is becoming less optional as more sensor-rich devices move into home and ambulatory use. The FDA’s authorized sDHT list makes this visible, since the list explicitly includes rings, patches, and bands sold for medical use. Cyclic bending remains the first purchase in many labs. Companies that can combine tensile, flexural, and fatigue methods within one software environment gain an advantage. This technical need also connects directly with adjacent demand in biosensors.
Analysis of Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market by Key Countries
.webp)
| Country |
CAGR (2026 to 2036) |
| United States |
8.9% |
| South Korea |
8.5% |
| Japan |
8.1% |
| Germany |
7.8% |
| United Kingdom |
7.6% |
Source: FMI analysis based on primary research and proprietary forecasting model
Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market By Country
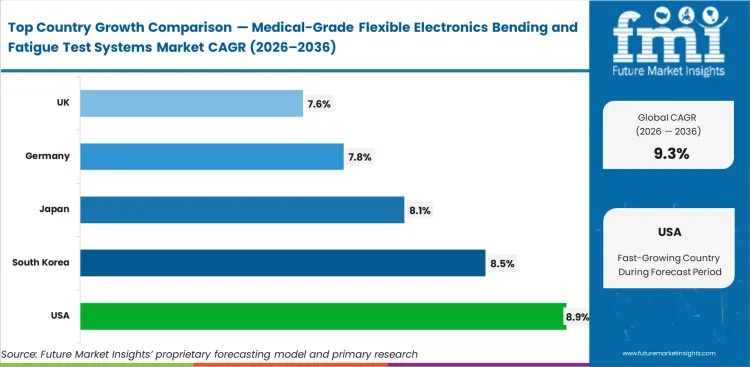
- The United States is expected to lead at an estimated 8.9% CAGR, supported by strong ophthalmic device research activity and continued work on smart lens platforms for intraocular pressure monitoring and biosensing.
- South Korea is projected to follow at around 8.5% CAGR, helped by its visible track record in smart contact lens development and wireless ocular sensing research.
- Japan at 8.1% and Germany at 7.8% are likely to post healthy expansion as precision optics, sensor integration, and medical electronics work continue to support specialized validation demand. This is an analyst inference based on the current direction of smart lens and wearable biosensor research.
- The United Kingdom is estimated at 7.6%, with demand supported by academic and clinical interest in noninvasive ocular monitoring technologies and connected medical sensing formats.
- The global Smart Contact Lens Optical and Telemetry Test Equipment Market is projected to expand at a CAGR of 8.3% from 2026 to 2036.
Demand Outlook for Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market in the United States
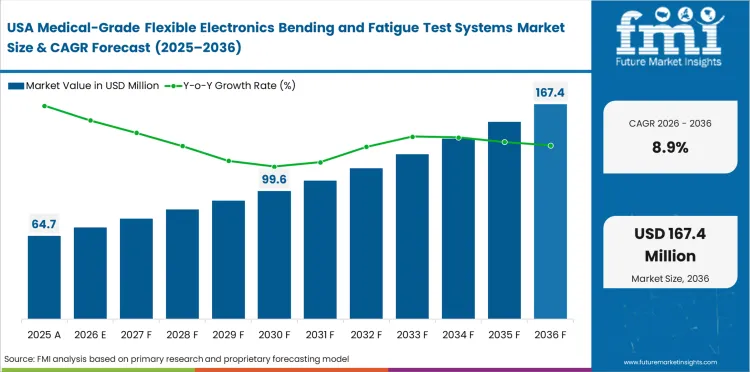
The USA medical-grade flexible electronics bending and fatigue test systems market is projected to grow at a CAGR of 8.9% through 2036. This is because of strong medical device development activity, rising work on wearable and skin-applied electronics, and steady demand for repeatable mechanical validation in regulated programs.
- Demand for controlled fatigue testing is supported by medical device development activity.
- Wearable and skin-applied electronics increase the need for repeated-cycle validation.
- Regulated product programs keep demand firm for repeatable test systems.
Future Outlook for Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market in the United Kingdom
The U.K. medical-grade flexible electronics bending and fatigue test systems market is projected to grow at a CAGR of 7.6% through 2036. This is due to academic research strength, clinical interest in connected medical devices, and wider development of flexible sensing formats.
- Academic research supports demand for specialized validation systems.
- Clinical interest keeps development work active in flexible medical electronics.
- Connected sensing formats raise the need for bending and fatigue testing.
Opportunity Analysis of Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market in Germany
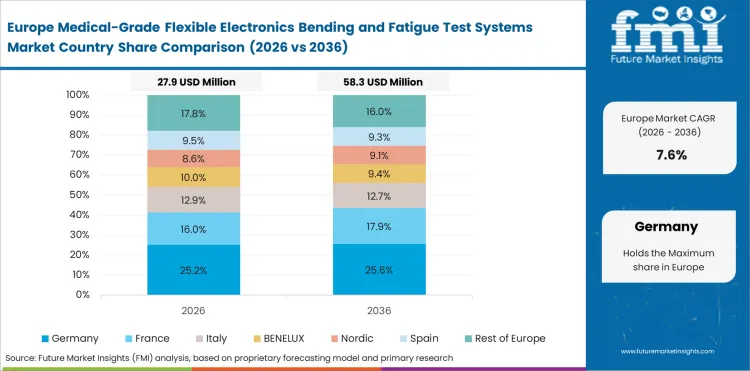
The Germany medical-grade flexible electronics bending and fatigue test systems market is projected to grow at a CAGR of 7.8% through 2036. This is because of strong medical engineering capability, advanced device quality requirements, and steady demand for controlled qualification work.
- Medical engineering depth supports demand for specialized testing systems.
- Device quality standards increase the need for repeatable validation.
- Qualification work stays active across flexible medical electronics programs.
In-depth Analysis of Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market in Japan
The Japan medical-grade flexible electronics bending and fatigue test systems market is projected to grow at a CAGR of 8.1% through 2036. This is due to precision manufacturing strength, advanced sensor integration work, and steady focus on reliable medical electronics validation.
- Precision manufacturing supports demand for accurate fatigue testing.
- Sensor integration work keeps controlled validation important in development.
- Reliability-focused product design supports use of repeatable test systems.
Sales Analysis of Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market in South Korea
The South Korea medical-grade flexible electronics bending and fatigue test systems market is projected to grow at a CAGR of 8.5% through 2036. This is because of active work in flexible electronics, strong innovation in connected medical devices, and rising interest in compact validation systems for new device formats.
- Flexible electronics development supports demand for bending test systems.
- Connected medical device innovation keeps fatigue validation relevant.
- Compact device formats increase the need for controlled low-force testing.
Competitive Landscape of Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market
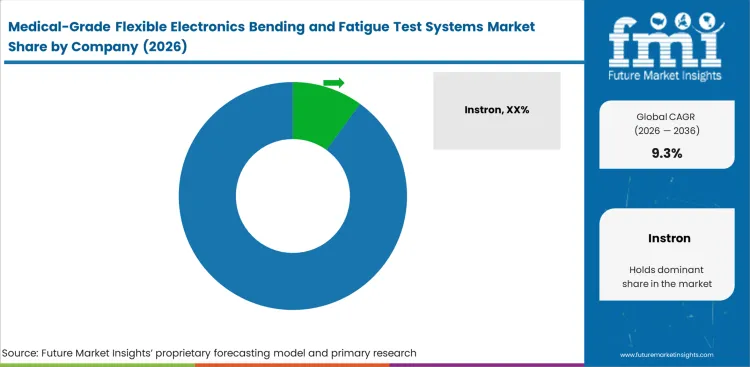
- The market is moderately fragmented with several established testing specialists and no single company dominating the full application space.
- Large incumbents compete on application support, software, service reach, and breadth across static and dynamic testing formats.
- Smaller specialists can still win where delicate samples, biomaterials handling, or custom fixture work are more important than global scale.
Keysight Technologies, Anritsu, and Rohde & Schwarz stand out through their capabilities in telemetry testing and high-frequency signal analysis, while optical metrology and imaging specialists support lens clarity, alignment, and calibration work. Smaller firms and custom system integrators continue to find openings in project-specific builds, especially where standard equipment needs fixture modification for soft curved lens formats.
Market entry remains difficult due to long qualification cycles, low-volume purchasing, and the need for application knowledge in ophthalmic devices. Strategic focus in this space is centered on measurement accuracy, customization, and the ability to support developers from prototype testing through preclinical validation.
Major Industry Players
The supplier base blends global materials-testing groups with smaller specialists that address delicate samples and customized workflows.
- Instron, ZwickRoell, Shimadzu, and TA Instruments fit the global player group because they combine broad mechanical testing portfolios with international service coverage, recognized software environments, and application support that can extend from static materials work into dynamic fatigue and biomedical validation.
- ADMET and AMETEK Lloyd fit the regional player group because they remain visible across materials testing and custom lab setups.
- CellScale and other soft-material specialists fit the emerging group because they address delicate biomaterials and low-force applications with equipment and methods that appeal to translational research labs and early-stage medical device developers.
Competitive benchmarking: Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market
| Company |
Dynamic Testing Breadth |
Fixture Adaptability |
Biomedical Relevance |
Geographic Footprint |
| Instron |
High |
High |
Strong |
Global |
| ZwickRoell |
High |
High |
Strong |
Global |
| Shimadzu |
High |
Medium |
Moderate |
Global |
| TA Instruments |
High |
Medium |
Strong |
Global |
| ADMET |
Medium |
High |
Moderate |
North America / Europe |
| AMETEK Lloyd |
Medium |
Medium |
Moderate |
Europe / North America |
| CellScale |
Low |
High |
Strong |
North America |
| TestResources |
Low |
Medium |
Moderate |
North America |
| Mark-10 |
Low |
Medium |
Low |
North America |
| Imada |
Low |
Medium |
Low |
Asia / North America |
Source: Future Market Insights competitive analysis, 2026.
Key Developments in Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market
- In June 2025, ADMET ADMET updated its materials research guidance to position the eXpert 3910, 3930, 5950 and 9900 tabletop dynamic systems for tension, compression and flexure durability studies.
- In December 2025, ZwickRoell ZwickRoell stated that electrodynamic drives are replacing conventional servohydraulic systems in many areas and positioned the approach as a low-maintenance option for modern fatigue testing.
Key Players in the Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market
Major global players
- Instron
- ZwickRoell
- Shimadzu
- TA Instruments
Key emerging players/startups
- ADMET
- AMETEK Lloyd
- CellScale
Scope of the Report
| Parameter |
Details |
| Quantitative Units |
USD 95.2 million to USD 253.1 million, at a CAGR of 9.3% |
| Market Definition |
The Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market covers equipment used to measure bending durability, cyclic fatigue, torsion, peel resistance, and related reliability performance in flexible medical electronic components and devices. |
| Regions Covered |
North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia and Pacific, Middle East and Africa |
| Countries Covered |
United States, Germany, Japan, South Korea, United Kingdom, and additional countries covered across the study |
| Key Companies Profiled |
ADMET, Instron, ZwickRoell, MTS Systems, Shimadzu Corporation, Bose, and Hegewald & Peschke |
| Forecast Period |
2026 to 2036 |
| Approach |
Hybrid market sizing based on equipment demand across test mode, system type, application, end user, and regional adoption, supported by bottom-up validation of medical flexible electronics testing requirements. |
Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market by Segments
Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market segmented as Test Mode
- Cyclic Bending
- Dynamic Fatigue
- Torsion
- Peel
- Custom Fixtures
Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market segmented as System Type
- Benchtop
- Electrodynamic
- Servohydraulic
- Micro-force
- Environmental
Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market segmented as Application
- Wearable Patches
- Implantables
- Smart Catheters
- Neuro Interfaces
- Biosensor Strips
Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market segmented as End User
- Medtech OEMs
- Contract Labs
- University Labs
- CDMOs
- Hospitals
Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market segmented as Sales Channel
- Direct Sales
- Distributors
- Integrators
- Service Contracts
- Retrofits
Medical-Grade Flexible Electronics Bending and Fatigue Test Systems Market segmented by Region
- North America
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Western Europe
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- Eastern Europe
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- East Asia
- South Asia and Pacific
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- ADMET. (2025, June 27). Materials research and development.
- Food and Drug Administration. (2026, March 4). Medical Devices that Incorporate Sensor-based Digital Health Technology.
- MedTech Europe. (2025). Facts & figures 2025: The European medical technology industry in figures.
- World Intellectual Property Organization. (2025). Patent landscape report - Occupational health and safety (OHS): 6 Protection technologies: Mitigating workplace risks.
The bibliography is provided for reader reference.
This Report Addresses
- Market sizing for 2025, 2026, and 2036 with a conservative analyst-built forecast.
- Test mode, system type, application, end-user, and sales-channel segmentation.
- Country analysis for seven key national markets with individual CAGRs.
- Competitive assessment of leading testing system suppliers and niche specialists.
- Demand drivers linked to digital health, wearable medical devices, and qualification needs.
- Commercial implications of automation, electrodynamic systems, and custom fixtures.
- Scope boundaries that separate this niche from broad materials testing or device sales.
- Transparent data mapping through a companion checklist and source file.
Frequently Asked Questions
What is the 2026 market size for medical-grade flexible electronics bending and fatigue test systems?
The market is estimated at USD 104.0 million in 2026 based on analyst-built demand from medtech development, validation, and pilot manufacturing programs.
What will be the market size in 2036?
The market is projected to reach USD 253.1 million by 2036 as flexible medical devices require more repeated-cycle durability evidence.
What is the expected CAGR from 2026 to 2036?
FMI estimates a CAGR of 9.3% through 2036 for this specialized testing systems category.
Which segment leads the market?
Cyclic bending leads test mode demand with an estimated 38.0% share in 2026.
Which application area is largest?
Wearable patches are the leading application segment with an estimated 34.0% share in 2026.
Which sales channel is most important?
Direct sales lead the market with an estimated 63.0% share in 2026 because buyers need application and fixture support.







.webp)





