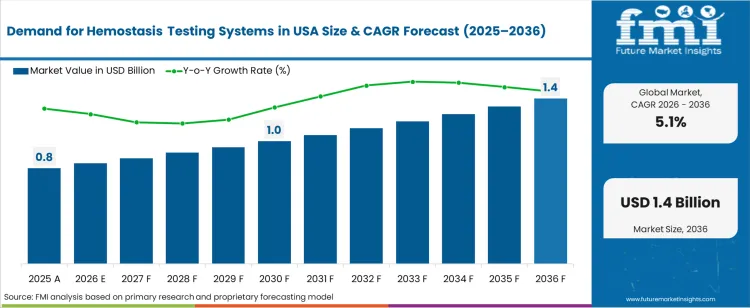
The demand for hemostasis testing systems in usa was valued at USD 0.80 billion in 2025, projected to reach USD 0.84 billion in 2026, and is forecast to expand to USD 1.38 billion by 2036 at a 5.1% CAGR. The expanding anticoagulant therapy patient base across Medicare and commercially insured populations is driving sustained coagulation testing volumes in hospital central laboratories and point-of-care settings. FDA clearances for next-generation multi-channel coagulation analysers are accelerating the installed base replacement cycle, while the growing complexity of perioperative bleeding risk assessment protocols is broadening the test menu per patient encounter.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 0.84 billion |
| Industry Value (2036) | USD 1.38 billion |
| CAGR (2026 to 2036) | 5.1% |
Source: Future Market Insights, 2026
The incremental opportunity between 2026 and 2036 amounts to USD 0.54 billion. FMI analysts observe that the transition from single-assay coagulation testing toward comprehensive hemostasis panels is increasing reagent consumption per analyser. Direct oral anticoagulant monitoring requirements are generating demand for drug-specific assay kits that command higher per-test pricing than conventional PT/INR and aPTT reagents. The shift toward outpatient anticoagulation management is expanding point-of-care coagulation testing device placements in physician offices and ambulatory clinics.
Sub-national growth trajectories reflect healthcare infrastructure density and anticoagulant prescribing patterns. West at 5.9%, South at 5.3%, Northeast at 4.7%, Midwest at 4.1%. The West leads, driven by California's concentration of large health system networks with centralised coagulation reference laboratories and high direct oral anticoagulant prescribing rates. The South follows, where expanding hospital construction across the sunbelt states is adding new laboratory capacity. The Northeast benefits from dense academic medical centre networks with high-complexity testing portfolios, while the Midwest reflects steady growth from rural hospital laboratory automation investments and telemedicine-supported anticoagulation management programmes.
Hemostasis testing systems are diagnostic instruments and consumables used to evaluate blood coagulation function, detect bleeding disorders, and monitor anticoagulant therapy. The USA market covers laboratory analysers, point-of-care testing systems, and associated consumables (reagent kits, calibrators, quality controls) used for hemophilia assessment, Von Willebrand disease diagnostics, and acquired bleeding disease evaluation. These systems are deployed across hospital central laboratories, ambulatory clinics, surgical centres, and physician office settings.
Market scope includes automated coagulation analysers, semi-automated coagulation instruments, point-of-care coagulation testing devices, and associated consumables including reagent kits, calibrators, and quality control materials. Replacement parts, software upgrades, and service contracts for installed analyser platforms are included.
General clinical chemistry analysers, hematology cell counters, blood banking equipment, blood gas analysers, and molecular diagnostic platforms are excluded. Surgical hemostatic agents, topical wound sealants, and transfusion medicine products are outside the analytical scope.
Demand for hemostasis testing systems in the USA is increasing because hospitals and diagnostic laboratories require faster and more accurate monitoring of coagulation disorders, especially among patients receiving anticoagulant therapy. Cardiovascular disease, atrial fibrillation and venous thromboembolism remain significant health concerns, and clinical teams rely on coagulation testing to manage bleeding and clotting risks during treatment. The growing number of surgeries and emergency care procedures also strengthens the need for reliable testing within operating rooms and intensive care units.
Point of care systems expand USAge in outpatient clinics and home health programs, allowing rapid decisions in settings where immediate results reduce hospital admissions. Blood banks adopt hemostasis testing to support safer transfusions and compatibility assessments. The increase in cancer therapies and chronic liver diseases contributes to higher demand for coagulation monitoring throughout complex care pathways. Constraints include capital cost for advanced analyzers and ongoing expenses for reagents, which may challenge smaller facilities. Skilled staff are required for accurate interpretation, and integration with electronic laboratory systems can extend adoption timelines. Some providers continue using older analyzers until replacement cycles align with budget planning.
Demand for hemostasis testing systems in the United States is influenced by the increasing prevalence of bleeding disorders, expanded surgical volumes, and routine coagulation monitoring in hospitals and specialized labs. The U.S. healthcare facilities prioritize timely diagnostics, automation, and integration with electronic lab systems to enhance clinical decision-making. Adoption is also driven by aging demographics and patients receiving anticoagulant therapy who require continuous coagulation screening.
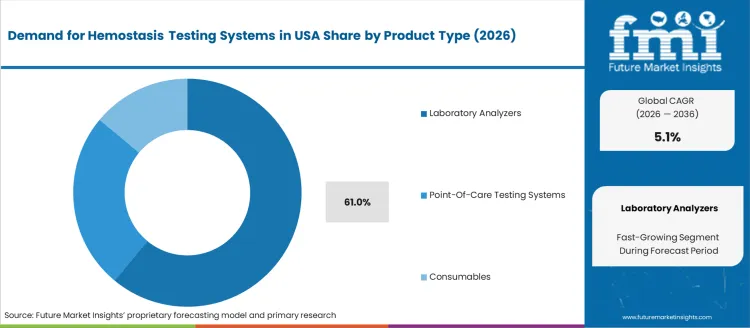
Laboratory analyzers account for 61.0%, reflecting their dominant role in centralized diagnostic laboratories where high-throughput testing is performed. These systems support comprehensive coagulation panels for emergency, pre-operative, and chronic disease monitoring. Large hospitals and reference labs prefer automated analyzers for accuracy, reduced manual error, and greater cost efficiency at scale. Point-of-care systems address bedside needs in critical settings but remain a smaller segment due to higher per-test costs and selective adoption. Consumables support recurring revenue streams for reagent manufacturers, driven by frequent testing requirements and stringent quality control standards across U.S. facilities. Demand alignment reflects a strong preference toward centralized, automated workflows that improve turnaround time and diagnostic precision nationwide.
Key Points:
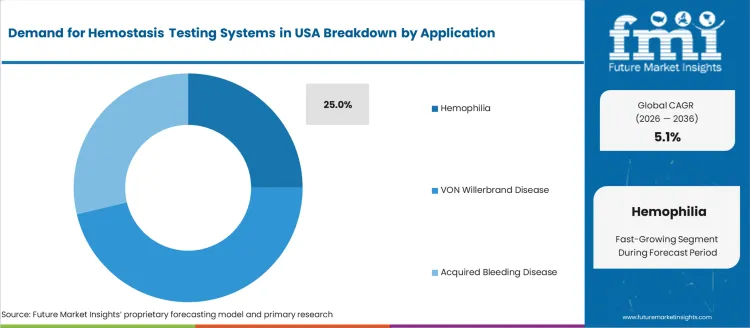
Hemophilia accounts for 25.0% of application-based demand as patients require lifelong coagulation monitoring, especially those receiving factor replacement or gene-based therapies. U.S. treatment guidelines emphasize routine APTT, PT, and inhibitor testing to prevent bleeding complications and optimize dosage. Von Willebrand disease and other acquired bleeding disorders make up the remaining application share, associated with oncology treatments, liver disease, and cardiovascular drug Usage. Testing frequency rises in hospitals and specialty hematology centers where precision monitoring is critical. Broader diagnostic adoption aligns with improving access to specialized care and advancing therapies for clotting factor deficiencies.
Key Points:
Growth of cardiovascular and coagulation disorder screening, increased surgical volume and wider adoption of point of care systems are driving demand.
In the United States, hemostasis testing systems gain steady use as hospitals manage a high prevalence of thrombotic and bleeding disorders linked to aging, obesity and chronic disease. Cardiology and oncology practices regularly monitor clotting status for patients on anticoagulant therapy, creating repeated testing requirements. Operating rooms and emergency departments depend on rapid coagulation testing to guide transfusion decisions during trauma care and complex surgeries. Outpatient clinics and dialysis centers adopt compact analyzers to improve patient management without requiring full laboratory processing. National attention on stroke and venous thromboembolism prevention supports broader physician ordering of PT, aPTT and platelet function tests. These clinical needs maintain continuous procurement across hospital laboratories and decentralized care sites.
High equipment cost, reimbursement variation for point of care tests and staffing shortages in laboratory departments restrain demand.
Advanced analyzers require capital investment and ongoing consumable costs that smaller facilities evaluate carefully before adoption. Reimbursement rules for diagnostic tests differ across private and public payers, which may limit the financial viability of frequent point of care use. Skilled lab personnel are needed to manage calibration and quality control, which can slow procurement in facilities facing workforce gaps. Integration with electronic medical records may require IT support and compliance validation, creating operational barriers for rapid expansion. These financial and logistical factors lead to staged upgrades rather than universal deployment.
Shift toward viscoelastic testing in trauma centers, increased home monitoring interest for anticoagulation therapy and rising automation within hospital labs define key trends.
Trauma and transplant programs are adopting viscoelastic technology to guide real time clotting management and reduce blood product waste. Patients on long term anticoagulants show growing interest in remote monitoring supported by digital reporting systems that link to clinical providers. Hospital laboratories invest in automated analyzers that streamline throughput and reduce manual steps to handle rising test volumes. Data connectivity improves result turnaround times and supports clinical decision protocols during surgery and intensive care. These developments indicate sustained, outcomes focused demand for hemostasis testing systems across the United States healthcare network.
Demand for hemostasis testing systems in the United States is growing due to the rising need for coagulation monitoring in surgery, trauma care, and chronic disease management. Hospitals and diagnostic labs are expanding platforms for rapid PT/INR, aPTT, platelet function, and D-dimer testing to support clinical response times. Cardiovascular disease prevalence, aging demographics, and anticoagulant therapy use strengthen baseline testing volumes. Automated analyzers and point-of-care devices improve sample-to-result efficiency in emergency settings. Vendor evaluation focuses on reagent continuity, digital reporting compatibility, and clinical data accuracy. Growth differs across regions: West USA leads at 5.9% CAGR, South USA follows with 5.3%, Northeast USA at 4.7%, and Midwest USA at 4.1%, reflecting infrastructure density and patient-care access levels.
.webp)
| Region | CAGR (2026-2036) |
|---|---|
| West USA | 5.9% |
| South USA | 5.3% |
| Northeast USA | 4.7% |
| Midwest USA | 4.1% |

West USA records a 5.9% compound annual growth rate, reflecting strong adoption of coagulation analyzers across California, Washington, and Arizona’s acute-care hospitals. Trauma centers and high-volume surgical facilities require accurate clotting assessment to manage perioperative bleeding and anticoagulant reversal. Point-of-care models are deployed inside emergency departments to reduce diagnostic turnaround times for stroke and trauma treatment. Research networks assess innovative markers such as thromboelastography to evaluate clot quality rather than time-based indicators alone. Diagnostic labs emphasize full automation, reagent-stability assurance, and digital integration with hospital information systems to support continuous reporting workflows. Population diversity increases screening needs related to genetic clotting disorders and chronic disease comorbidities.
South USA exhibits approximately 5.3% CAGR, driven by cardiovascular disease prevalence and extensive surgical-care networks in Texas, Florida, and Georgia. Hospitals manage anticoagulation therapy for aging populations, which increases follow-up monitoring frequency. Healthcare systems invest in analyzers capable of handling fluctuation in inpatient and outpatient sample volumes. Emergency facilities emphasize fast INR confirmation to accelerate thrombolysis eligibility in stroke cases. Training programs support consistent reagent USAge and sample handling to maintain accuracy in community-based labs. Forecasting teams monitor supply chain continuity for consumables, given humid climate conditions that can affect storage.
Northeast USA grows at around 4.7% CAGR, supported by advanced tertiary hospitals throughout New York, Pennsylvania, and Massachusetts that prioritize complex coagulation diagnostics. Teaching institutions validate new hemostasis markers and ensure clinical staff remain updated on testing protocols. Dense metropolitan patient populations lead to significant sample throughput managed by automated systems with strong uptime reliability. Specialized oncology and transplant centers require precise coagulation tracking under high-risk therapy conditions. Laboratories emphasize LIS integration, calibration quality, and reagent traceability for regulatory compliance. Seasonal respiratory illness waves increase hospitalization rates and indirectly elevate clot-risk monitoring demand.
Midwest USA advances at a 4.1% growth rate, reflecting steady expansion across regional hospitals and community laboratories in Illinois, Ohio, Missouri, and Michigan. Healthcare networks focus on reliable analyzer performance supporting emergency stabilization and chronic therapy management. Investment decisions emphasize durability, cost-efficient reagents, and simplified maintenance cycles that suit dispersed facility layouts. Point-of-care units enhance testing availability in smaller hospitals, decreasing reliance on centralized lab transport times. Education initiatives improve anticoagulation oversight to prevent adverse bleeding episodes in primary-care environments. Procurement strategies evaluate consistent vendor support to maintain uptime during severe weather events affecting logistics.
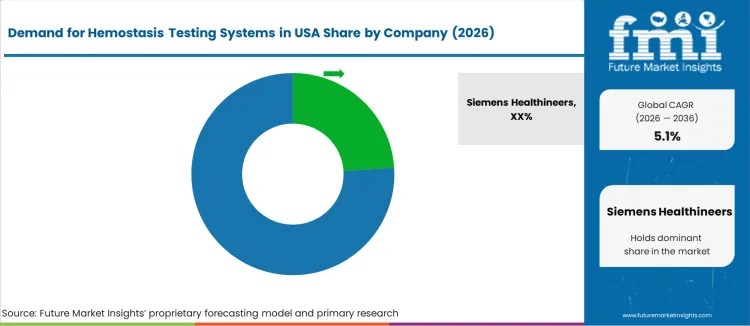
Demand for hemostasis testing systems in the United States is driven by hospitals, diagnostic laboratories, transfusion centers, and emergency-care facilities that require reliable coagulation panels, rapid clotting-time analysis, and stable thrombosis/bleeding-risk assessment. Requirements focus on test accuracy, throughput capacity, regulatory compliance, and integration with lab-information systems. Procurement decisions favour suppliers offering validated reagent kits, software support for result interpretation, and strong after-sales service networks to maintain 24/7 lab operations. Siemens Healthineers holds an estimated 24.2% share. Its coagulation analyzers and reagent systems are widely deployed in U.S. hospital laboratories and large reference labs, valued for consistent clotting-time measurement, validated quality control, and integration with laboratory-information workflows.
Roche Diagnostics contributes significantly with immuno-coagulation and automated analyzer solutions used in hospitals and blood-bank screening labs to manage hemostatic disorders and pre-surgical workups. Abbott Laboratories supports demand through compact coagulation systems suited for both central and point-of-care lab environments, offering reliability in throughput and ease of use for smaller clinical settings. Becton Dickinson (BD) adds presence via sample-management and cartridge-based systems used in transfusion and outpatient clinics, where quick turnaround and ease of operation are priorities. Instrumentation Laboratory (Werfen) serves specialized coagulation testing needs, such as thromboelastography and advanced clot-dynamics monitoring, supporting trauma centers and critical-care units managing complex bleeding or anticoagulation therapies. Competition in the USA emphasizes analytical precision, system automation, reagent stability, wide test panels, and accreditation support. Demand remains stable as healthcare providers rely on hemostasis diagnostics for safe surgical planning, thrombosis risk assessment, and patient-specific anticoagulant management.
| Metric | Value |
|---|---|
| Quantitative Units | USD 0.84 to USD 1.38 billion, at a CAGR of 5.1% |
| Market Definition | Hemostasis testing systems are diagnostic instruments and consumables used to evaluate blood coagulation function, detect bleeding disorders, and monitor anticoagulant therapy. The USA market covers laboratory analysers, point-of-care testing systems, and associated consumables (reagent kits, calibrators, quality controls) used for hemophilia assessment, Von Willebrand disease diagnostics, and acquired bleeding disease evaluation. |
| Segmentation | Product Type (Laboratory Analyzers, Point-of-Care Testing Systems, Consumables), Application (Hemophilia, Von Willebrand Disease, Acquired Bleeding Disease) |
| Regions Covered | West, South, Northeast, Midwest |
| Key Companies Profiled | Siemens Healthineers, Roche Diagnostics, Abbott Laboratories, Becton Dickinson (BD), Instrumentation Laboratory (Werfen) |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with unit shipment and average selling price data, cross-validated against manufacturer financial disclosures and institutional procurement records. |
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
How large is the demand for hemostasis testing systems in usa in 2026?
The demand for hemostasis testing systems in usa is estimated to be valued at USD 0.84 billion in 2026.
What will be the market size of demand for hemostasis testing systems in usa by 2036?
Market size for demand for hemostasis testing systems in usa is projected to reach USD 1.38 billion by 2036.
What is the expected growth rate for demand for hemostasis testing systems in usa between 2026 and 2036?
The demand for hemostasis testing systems in usa is expected to grow at a CAGR of 5.1% between 2026 and 2036.
Which product type is poised to lead by 2026?
Laboratory Analyzers accounts for 61.0% share in 2026, driven by established procurement patterns and institutional adoption.
How is hemophilia positioned in driving adoption in 2026?
Hemophilia holds 25.0% share of the application segment in 2026, reflecting concentrated institutional demand.
What is the growth outlook for West?
West is projected to grow at a CAGR of 5.9% during 2026 to 2036.
What is included in the scope of this report?
Market scope includes automated coagulation analysers, semi-automated coagulation instruments, point-of-care coagulation testing devices, and associated consumables including reagent kits, calibrators, and quality control materials. Replacement parts, software upgrades, and service contracts for installed analyser platforms are included.
How does FMI build and validate the demand for hemostasis testing systems in usa forecast?
Forecasting models apply a bottom-up methodology starting with unit shipment and average selling price data, cross-validated against manufacturer financial disclosures and institutional procurement records.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.