Demand for Hemostasis Testing Systems in Japan
A comprehensive overview of the demand for hemostasis testing systems in japan market is delivered in this report, featuring market size insights, revenue projections, competitive assessment, demand evaluation, growth propellers, restraining factors, Product Type trends, Application trends, supply chain analysis, and strategic opportunities ahead.
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Demand for Hemostasis Testing Systems in Japan Size, Market Forecast and Outlook By FMI
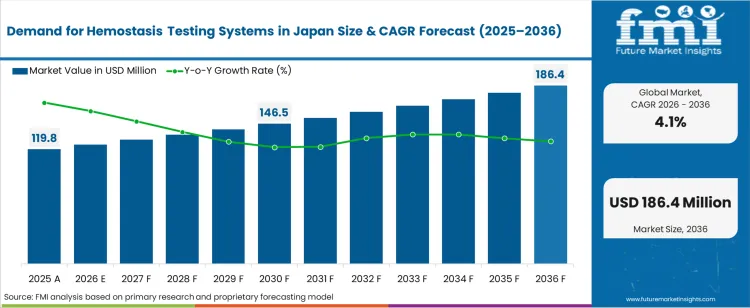
The demand for hemostasis testing systems in japan was valued at USD 119.80 million in 2025, projected to reach USD 124.71 million in 2026, and is forecast to expand to USD 186.39 million by 2036 at a 4.1% CAGR. Japan's aging population and the associated rise in anticoagulant therapy prescriptions are increasing the volume of coagulation testing performed across hospital laboratories and point-of-care settings. National health insurance coverage for routine hemostasis panels and the growing complexity of perioperative bleeding risk assessment protocols are sustaining institutional procurement of automated coagulation analysers and testing consumables.
Summary of Demand for Hemostasis Testing Systems in Japan
- Market Snapshot
- The demand for hemostasis testing systems in japan is valued at USD 119.80 million in 2025 and is projected to reach USD 186.39 million by 2036.
- The industry is expected to grow at a 4.1% CAGR from 2026 to 2036, creating an incremental opportunity of USD 61.68 million.
- The market is anchored in Japan's universal health insurance system, where coagulation testing reimbursement codes and the expanding anticoagulant therapy patient base define procurement volumes.
- Demand and Growth Drivers
- Japan's aging population and the associated rise in anticoagulant therapy prescriptions are increasing coagulation testing volumes across hospital laboratories.
- National health insurance coverage for expanded hemostasis test panels sustains institutional procurement of automated analyser platforms and consumables.
- Growing use of direct oral anticoagulants is generating demand for drug-specific coagulation assays requiring new reagent kits.
- Among tracked regions, Kyushu and Okinawa lead at 5.1% CAGR, followed by Kanto at 4.7%, Kansai at 4.1%, Chubu at 3.6%, Tohoku at 3.2%, and the Rest of Japan at 3.0%.
- Product and Segment View
- The market covers laboratory analysers, point-of-care testing systems, and consumables (reagent kits, calibrators, controls) used for coagulation and bleeding disorder diagnostics.
- Core applications include hemophilia testing, Von Willebrand disease diagnostics, and acquired bleeding disease assessment across hospital and outpatient laboratory settings.
- Laboratory Analyzers lead by product type with 61.0% share in 2026.
- Hemophilia leads by application with 25.0% share in 2026.
- The scope includes hemostasis testing instruments and consumables and excludes general clinical chemistry analysers, hematology cell counters, and blood banking equipment.
- Geography and Competitive Outlook
- Kyushu, Okinawa, and Kanto are the fastest-growing sub-national markets, anchored by elderly population density and hospital laboratory modernisation programmes.
- Kansai and Chubu reflect steady institutional procurement tied to tertiary care hospital coagulation testing infrastructure.
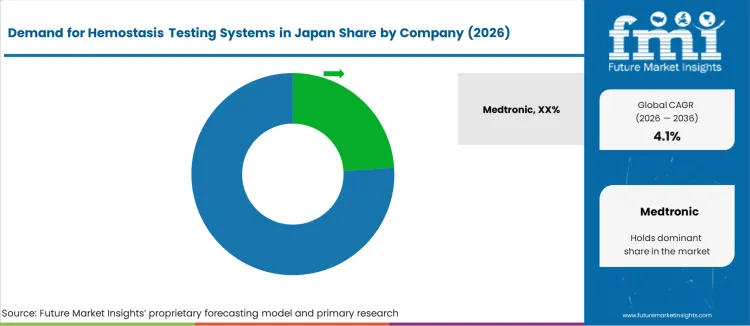
- Competition is concentrated among global IVD manufacturers with established PMDA-approved product portfolios, including Medtronic, Siemens Healthcare GmbH, Abbott, Roche Diagnostics Limited, Nihon Kohden Corporation.
Demand for Hemostasis Testing Systems in Japan Key Takeaways
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 124.71 million |
| Industry Value (2036) | USD 186.39 million |
| CAGR (2026 to 2036) | 4.1% |
Source: Future Market Insights, 2026
The incremental opportunity between 2026 and 2036 amounts to USD 61.68 million. FMI analysts note that the installed base of automated coagulation analysers in Japanese hospitals is entering a replacement cycle, with facilities upgrading from single-channel to multi-channel platforms capable of running expanded test menus. The growth of direct oral anticoagulant prescriptions is generating incremental demand for drug-specific coagulation assays that require new reagent kits and calibration consumables.
Sub-national growth patterns reflect hospital infrastructure concentration and elderly population density. Kyushu & Okinawa at 5.1%, Kanto at 4.7%, Kansai at 4.1%, Chubu at 3.6%, Tohoku at 3.2%, Rest of Japan at 3.0% CAGR characterise the regional landscape. Kyushu and Okinawa lead due to a high proportion of elderly patients on anticoagulant therapy and active prefectural hospital laboratory modernisation programmes. Kanto follows, anchored by Tokyo's concentration of tertiary care hospitals with high-throughput coagulation testing laboratories. Kansai benefits from Osaka's medical device procurement networks, while Chubu and Tohoku reflect incremental gains from rural hospital laboratory automation investments.
Demand for Hemostasis Testing Systems in Japan Definition
Hemostasis testing systems are diagnostic instruments and consumables used to evaluate blood coagulation function and bleeding disorder severity. The Japan market covers laboratory analysers, point-of-care testing systems, and associated consumables (reagent kits, calibrators, quality controls) used for hemophilia assessment, Von Willebrand disease diagnostics, and acquired bleeding disease evaluation. These systems are deployed across hospital central laboratories, outpatient clinics, and perioperative testing environments.
Demand for Hemostasis Testing Systems in Japan Inclusions
Market scope includes automated coagulation analysers, semi-automated coagulation instruments, point-of-care coagulation testing devices, and associated consumables including reagent kits, calibrators, and quality control materials. Replacement parts and service contracts for installed analyser platforms are included.
Demand for Hemostasis Testing Systems in Japan Exclusions
General clinical chemistry analysers, hematology cell counters, blood banking equipment, and blood gas analysers are excluded. Surgical hemostatic agents, topical sealants, and transfusion medicine products are outside the analytical scope.
Demand for Hemostasis Testing Systems in Japan Research Methodology
- Primary Research: Analysts engaged with procurement managers, laboratory directors, and supply chain specialists to map decision triggers, specification requirements, and adoption timelines across institutional and commercial channels.
- Desk Research: Data collection aggregated regulatory filings, public procurement records, industry association publications, and manufacturer financial disclosures to establish baseline market parameters.
- Market-Sizing and Forecasting: Baseline values derive from a bottom-up aggregation of unit shipment data and average selling prices, applying region-specific adoption curves to project forward demand trajectories.
- Data Validation and Update Cycle: Projections are cross-validated against publicly reported financial disclosures from leading manufacturers and verified against institutional procurement records.
Why is the Demand for Hemostasis Testing Systems in Japan Growing?
The demand for hemostasis testing systems in Japan is growing due to the increasing need for accurate and reliable diagnostic tools for managing bleeding disorders, cardiovascular diseases, and other medical conditions related to blood clotting. Hemostasis testing, which measures the body’s ability to form and dissolve blood clots, is crucial in diagnosing and monitoring conditions such as hemophilia, deep vein thrombosis (DVT), and pulmonary embolism (PE). With the growing focus on early detection and personalized healthcare, the demand for advanced hemostasis testing systems is on the rise.
A significant driver of this growth is Japan's aging population, which is more prone to developing clotting disorders and cardiovascular diseases. As the number of elderly individuals increases, there is a higher demand for regular monitoring and testing to manage these conditions effectively. Hemostasis testing systems are essential tools for clinicians to assess patients’ risk of bleeding or clotting, particularly in those undergoing surgery or being treated for heart disease.
The growing awareness of the importance of preventive healthcare is encouraging the adoption of hemostasis testing in clinical and hospital settings. Technological advancements, such as automated and point-of-care testing devices, are also driving demand by improving the speed and accuracy of test results. As Japan continues to prioritize high-quality healthcare and the management of chronic diseases, the demand for hemostasis testing systems is expected to grow steadily through 2035.
What is the Segment-Wise Analysis of Demand for Hemostasis Testing Systems in Japan?
Demand for hemostasis testing systems in Japan is segmented by product type and application. By product type, demand is divided into laboratory analyzers, point-of-care testing systems, and consumables, with laboratory analyzers leading at 61%. The demand is also segmented by application, including hemophilia, von Willebrand disease, and acquired bleeding disease, with hemophilia accounting for 25% of the demand. Regionally, demand is divided into Kanto, Kinki, Chubu, Kyushu & Okinawa, Tohoku, and the Rest of Japan.
Why Do Laboratory Analyzers Lead the Demand for Hemostasis Testing Systems in Japan?
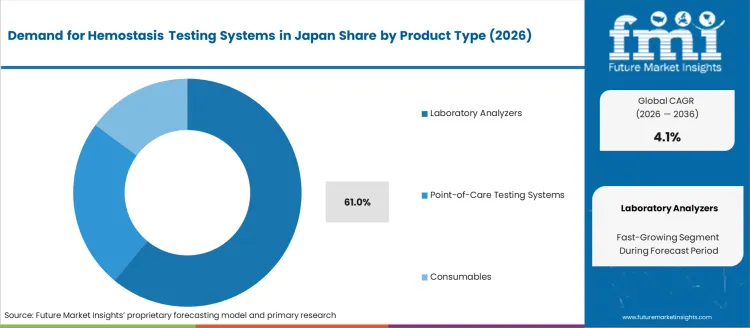
Laboratory analyzers account for 61% of the demand for hemostasis testing systems in Japan due to their high accuracy, reliability, and capacity for handling large volumes of tests. These systems are widely used in hospitals and diagnostic laboratories for comprehensive testing of blood clotting disorders, such as hemophilia and von Willebrand disease. Laboratory analyzers provide detailed and precise results that are crucial for diagnosing bleeding disorders and determining appropriate treatments. The advanced technology and ability to perform multiple tests in a single system make laboratory analyzers the preferred choice for healthcare providers. With Japan’s focus on advanced healthcare and precision medicine, the demand for laboratory analyzers is expected to remain strong, particularly in settings requiring routine or complex hemostasis testing.
Why Does Hemophilia Lead the Demand for Hemostasis Testing Systems in Japan?
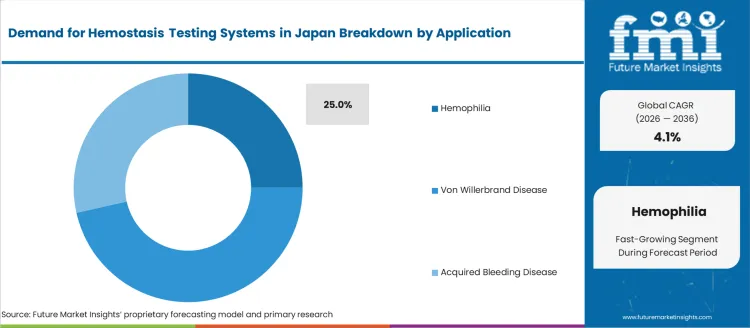
Hemophilia accounts for 25% of the demand for hemostasis testing systems in Japan, driven by the need for accurate diagnosis and ongoing management of this inherited bleeding disorder. Hemophilia is characterized by the lack of clotting factors in the blood, leading to difficulty in controlling bleeding. Regular hemostasis testing is crucial for monitoring clotting factor levels, assessing bleeding risks, and guiding treatment decisions for individuals with hemophilia. The demand for hemostasis testing systems in Japan is also influenced by the country’s advanced healthcare system, which prioritizes early detection and personalized care for patients with hemophilia. As Japan’s population with hemophilia continues to require regular and precise blood clotting tests, the demand for hemostasis testing systems will remain robust, particularly in specialized healthcare centers and hospitals focused on managing bleeding disorders.
What are the Key Trends, Drivers, and Restraints in Demand for Hemostasis Testing Systems in Japan?
Demand for hemostasis testing systems in Japan is rising as the healthcare sector faces an increasing need for accurate coagulation and bleeding‑risk diagnostics. Factors such as an aging population, rising prevalence of cardiovascular and thrombotic disorders (e.g., stroke, atrial fibrillation), growing number of surgeries and interventional procedures, and heightened awareness of anticoagulant management fuel this demand. Hospitals and diagnostic labs need reliable coagulation assays, point‑of‑care tests, and advanced analyzers to monitor patient clotting status pre‑ and post‑treatment. High cost of advanced testing equipment, maintenance requirements, and limited adoption in smaller clinics or rural hospitals may slow widespread use.
Why is Demand for Hemostasis Testing Systems Growing in Japan?
Demand is growing because more patients require coagulation monitoring due to increased incidence of thrombotic diseases, aging‑related health risks, and use of anticoagulant and antithrombotic therapies. Accurate hemostasis testing becomes essential to prevent bleeding or clotting complications, improving patient safety and treatment outcomes. Also, as medical protocols become more stringent, perioperative and chronic disease management increasingly call for frequent coagulation checks. Growing awareness among physicians and patients about the importance of individualized coagulation monitoring further supports adoption of hemostasis testing systems.
How are Technological & Industry Innovations Driving Hemostasis Testing System Demand in Japan?
Advancements in diagnostic technologies including point‑of‑care coagulation devices, automated analyzers, rapid assay kits, and integration with hospital information systems are boosting demand. New systems offer faster turnaround times, miniaturization, improved sensitivity and specificity, and lower sample volume requirements. Portable or bed‑side testing enables better perioperative, emergency, and outpatient management. Innovations also include assays tailored to newer anticoagulants, enabling reliable monitoring even for patients on modern therapies. These developments make hemostasis testing more convenient, reliable, and scalable across different care settings encouraging broader uptake across hospitals and clinics.
What are the Key Challenges and Risks That Could Limit Hemostasis Testing System Demand in Japan?
Despite growing need, several challenges may limit adoption. The high cost of purchasing and maintaining advanced testing equipment could be a barrier for smaller hospitals or clinics. Training staff and ensuring quality control for complex assays requires resources and expertise. Variability in reimbursement policies or lack of coverage for certain tests may discourage frequent testing. Also, in rural or smaller healthcare settings, limited patient volume may not justify investment in advanced systems. Finally, adoption may be slowed by regulatory hurdles, standardization issues across assay types, or the emergence of alternative less‑invasive monitoring approaches.
What is the Regional Demand Outlook for Hemostasis Testing Systems in Japan?
.webp)
| Region | CAGR (%) |
|---|---|
| Kyushu & Okinawa | 5.1% |
| Kanto | 4.7% |
| Kansai | 4.1% |
| Chubu | 3.6% |
| Tohoku | 3.2% |
| Rest of Japan | 3.0% |

Demand for hemostasis testing systems in Japan is steadily growing, with Kyushu & Okinawa leading at a 5.1% CAGR, driven by an aging population and increasing healthcare access. Kanto follows with a 4.7% CAGR, supported by its advanced healthcare infrastructure and high demand for diagnostic tools. Kansai shows a 4.1% CAGR, fueled by its focus on healthcare improvements and preventive care. Chubu experiences a 3.6% CAGR, driven by an aging population and rising healthcare investments. Tohoku and the Rest of Japan show moderate growth at 3.2% and 3.0%, respectively, as access to healthcare services and diagnostic technologies increases in rural areas. As healthcare awareness and the need for early diagnosis continue to grow, demand for hemostasis testing systems is expected to rise steadily across Japan.
How is Demand for Hemostasis Testing Systems Growing in Kyushu & Okinawa?
Kyushu & Okinawa shows the highest demand for hemostasis testing systems, growing at a 5.1% CAGR. The region’s increasing focus on healthcare improvements, particularly in diagnostics and blood-related disorders, is driving the adoption of hemostasis testing systems. As the aging population grows, the prevalence of conditions like cardiovascular diseases and diabetes, which often require hemostasis testing, is also rising. Furthermore, with advancements in medical technology, healthcare facilities in Kyushu & Okinawa are increasingly adopting high-performance testing systems. The region’s strong healthcare infrastructure, coupled with a growing awareness of the importance of early diagnosis, contributes to the steady demand. As more hospitals and clinics in the region prioritize patient care and efficient diagnostics, demand for hemostasis testing systems is expected to continue its steady growth.
Why is Demand for Hemostasis Testing Systems Rising in Kanto?
Kanto is experiencing steady demand for hemostasis testing systems, with a 4.7% CAGR. The region's advanced healthcare infrastructure, particularly in Tokyo, plays a major role in driving the demand for diagnostic technologies, including hemostasis testing systems. With a high prevalence of cardiovascular diseases and diabetes in the urban population, there is an increasing need for accurate and timely blood coagulation tests. Kanto’s healthcare facilities, which are equipped with state-of-the-art medical technologies, are increasingly adopting hemostasis testing systems to improve diagnosis and patient outcomes. The region’s focus on research and innovation in the healthcare sector, particularly in clinical diagnostics, is further contributing to this demand. As healthcare awareness and the need for efficient diagnostic tools continue to grow, Kanto’s demand for hemostasis testing systems will likely increase steadily.
How is Demand for Hemostasis Testing Systems Expanding in Kansai?
Kansai is seeing steady demand for hemostasis testing systems, with a 4.1% CAGR. The region’s growing focus on healthcare improvements, particularly in diagnostics, is driving the demand for advanced testing systems. With cities like Osaka and Kyoto at the forefront of healthcare innovation, there is a growing need for reliable diagnostic tools to address blood coagulation issues, such as those associated with heart disease and diabetes. Kansai’s strong medical infrastructure, including research centers and hospitals, is increasingly adopting hemostasis testing systems to provide accurate and efficient testing for patients. The region’s increasing focus on preventive healthcare and early diagnosis contributes to the growing demand for these testing systems. As the population becomes more health-conscious and diagnostic technologies continue to evolve, Kansai’s demand for hemostasis testing systems is expected to grow steadily.
Why is Demand for Hemostasis Testing Systems Growing in Chubu?
Chubu is experiencing moderate demand for hemostasis testing systems, growing at a 3.6% CAGR. The region’s focus on improving healthcare infrastructure and services, particularly in urban areas like Nagoya, is contributing to the growing adoption of diagnostic technologies, including hemostasis testing systems. As the population ages and the incidence of diseases requiring blood coagulation tests rises, Chubu’s healthcare providers are increasingly investing in advanced diagnostic solutions. The region’s growing emphasis on preventive healthcare and early diagnosis further drives the demand for hemostasis testing systems. As healthcare facilities expand and the need for accurate and timely testing increases, Chubu’s demand for these systems is expected to grow steadily. The increasing awareness of blood-related health risks among the population is also supporting this demand.
How is Demand for Hemostasis Testing Systems Expanding in Tohoku?
Tohoku is seeing moderate demand for hemostasis testing systems, with a 3.2% CAGR. While the industry is smaller compared to more urbanized regions, there is steady growth in interest as healthcare facilities in the region adopt advanced diagnostic technologies. Tohoku’s aging population is a key driver, as older adults are more prone to conditions requiring blood coagulation testing. As the region continues to modernize its healthcare infrastructure, there is increasing access to diagnostic tools, including hemostasis testing systems. The focus on preventive healthcare and early detection of diseases is contributing to the growing demand for these systems. Although growth is slower compared to urban areas, Tohoku’s demand for hemostasis testing systems is expected to rise gradually, driven by both healthcare improvements and greater awareness of blood-related health conditions.
Why is Demand for Hemostasis Testing Systems Steady in the Rest of Japan?
The Rest of Japan is experiencing steady demand for hemostasis testing systems, growing at a 3.0% CAGR. While demand is lower in rural regions compared to major urban centers, there is consistent growth as local healthcare facilities increasingly adopt diagnostic technologies to address blood coagulation disorders. As the region’s population ages, the need for hemostasis testing to diagnose and monitor conditions like cardiovascular diseases and diabetes is rising. The growing focus on improving healthcare services in rural areas, coupled with increasing access to advanced medical technologies, supports steady demand for hemostasis testing systems. As awareness of the importance of early diagnosis and preventive care continues to grow, demand for these systems in the Rest of Japan is expected to rise slowly but steadily.
What is the Competitive Landscape of Hemostasis Testing Systems Demand in Japan?
Demand for hemostasis testing systems in Japan is rising steadily as hospitals, clinics, and diagnostic labs face growing needs for coagulation and blood-clotting assessments. Aging demographics, rising incidence of cardiovascular and thrombotic disorders, and increasing volumes of surgeries and invasive procedures drive the demand for reliable hemostasis diagnostics. Advanced coagulation analyzers and point-of-care testing devices are becoming part of regular diagnostic workflows from pre-operative screening and anticoagulant therapy monitoring to emergency care and chronic disease management.
Major suppliers in the Japanese industry include Medtronic (24.0%), Siemens Healthcare GmbH, Abbott, Roche Diagnostics Limited, and Nihon Kohden Corporation. These firms offer a range of products, from fully automated laboratory analyzers to portable point-of-care systems and consumables. Medtronic leads with an estimated share of 24.0%, offering integrated testing solutions used across hospitals and specialized clinics. Competitors such as Siemens, Abbott, Roche, and Nihon Kohden complement this with high-throughput analyzers, reagent kits, and support for routine coagulation tests (e.g. PT/INR, aPTT, D-dimer), as well as advanced hemostasis assays.
Growth is driven by a combination of demographic pressures (aging population), rising burden of thrombotic and bleeding disorders, and increasing demand for rapid, accurate diagnostics. Technological advances such as automation, microfluidics, and integration with lab information systems are enabling faster throughput, improved accuracy, and reduced manual workload, which are critical for busy Japanese hospitals. Point-of-care hemostasis devices are gaining traction due to convenience and speed, especially in emergency and outpatient settings. Vendors compete on system reliability, throughput, ease of use, reagent availability, and after-sales support. Those that offer comprehensive solutions hardware, reagents, and services are best positioned to win hospital contracts and long-term lab partnerships.
Key Players in Japan Hemostasis Testing Systems Demand
- Medtronic
- Siemens Healthcare GmbH
- Abbott
- Roche Diagnostics Limited
- Nihon Kohden Corporation
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 124.71 to USD 186.39 million, at a CAGR of 4.1% |
| Market Definition | Hemostasis testing systems are diagnostic instruments and consumables used to evaluate blood coagulation function and bleeding disorder severity. The Japan market covers laboratory analysers, point-of-care testing systems, and associated consumables (reagent kits, calibrators, quality controls) used for hemophilia assessment, Von Willebrand disease diagnostics, and acquired bleeding disease evaluation. |
| Segmentation | Product Type (Laboratory Analyzers, Point-of-Care Testing Systems, Consumables), Application (Hemophilia, Von Willebrand Disease, Acquired Bleeding Disease) |
| Regions Covered | Kyushu & Okinawa, Kanto, Kansai, Chubu, Tohoku, Rest of Japan |
| Key Companies Profiled | Medtronic, Siemens Healthcare GmbH, Abbott, Roche Diagnostics Limited, Nihon Kohden Corporation |
| Forecast Period | 2026 to 2036 |
| Approach | Forecasting models apply a bottom-up methodology starting with unit shipment and average selling price data, cross-validated against manufacturer financial disclosures and institutional procurement records. |
Japan Hemostasis Testing Systems Demand by Key Segments
Product Type
- Laboratory Analyzers
- Point-of-Care Testing Systems
- Consumables
Application
- Hemophilia
- Von Willerbrand Disease
- Acquired Bleeding Disease
Region
- Kyushu & Okinawa
- Kanto
- Kansai
- Chubu
- Tohoku
- Rest of Japan
Bibliography
- 1. Ministry of Health, Labour and Welfare, Japan. (2024). National Health Insurance Reimbursement Schedule for Laboratory Testing. MHLW.
- 2. World Federation of Hemophilia. (2024). Annual Global Survey: Bleeding Disorder Prevalence and Treatment. WFH.
- 3. Pharmaceuticals and Medical Devices Agency, Japan. (2024). In Vitro Diagnostic Device Approval Database. PMDA.
- 4. Siemens Healthcare GmbH. (2024). Annual Report 2024: Diagnostics Division. Siemens Healthineers.
- 5. Japanese Society on Thrombosis and Hemostasis. (2024). Clinical Practice Guidelines for Coagulation Testing. JSTH.
- 6. World Health Organization. (2024). Global Status Report on Blood Safety and Availability. WHO.
This bibliography is provided for reader reference. The full Future Market Insights report contains the complete reference list with primary research documentation.
Frequently Asked Questions
How large is the demand for hemostasis testing systems in japan in 2026?
The demand for hemostasis testing systems in japan is estimated to be valued at USD 124.71 million in 2026.
What will be the market size of demand for hemostasis testing systems in japan by 2036?
Market size for demand for hemostasis testing systems in japan is projected to reach USD 186.39 million by 2036.
What is the expected growth rate for demand for hemostasis testing systems in japan between 2026 and 2036?
The demand for hemostasis testing systems in japan is expected to grow at a CAGR of 4.1% between 2026 and 2036.
Which product type is poised to lead by 2026?
Laboratory Analyzers accounts for 61.0% share in 2026, driven by established procurement patterns and broad institutional adoption.
How is the role of hemophilia in driving adoption in 2026?
Hemophilia holds 25.0% share of the application segment in 2026, reflecting concentrated demand from primary end-use sectors.
What is the growth outlook for Kyushu & Okinawa?
Kyushu & Okinawa is projected to grow at a CAGR of 5.1% during 2026 to 2036.
What is included in the scope of this report?
Market scope includes automated coagulation analysers, semi-automated coagulation instruments, point-of-care coagulation testing devices, and associated consumables including reagent kits, calibrators, and quality control materials. Replacement parts and service contracts for installed analyser platforms are included.
How does FMI build and validate the demand for hemostasis testing systems in japan forecast?
Forecasting models apply a bottom-up methodology starting with unit shipment and average selling price data, cross-validated against manufacturer financial disclosures and institutional procurement records.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product Type , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product Type , 2026 to 2036
- Laboratory Analyzers
- Point-of-Care Testing Systems
- Consumables
- Laboratory Analyzers
- Y-o-Y Growth Trend Analysis By Product Type , 2021 to 2025
- Absolute $ Opportunity Analysis By Product Type , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application, 2026 to 2036
- Hemophilia
- Von Willerbrand Disease
- Acquired Bleeding Disease
- Hemophilia
- Y-o-Y Growth Trend Analysis By Application, 2021 to 2025
- Absolute $ Opportunity Analysis By Application, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Product Type
- By Application
- By Country
- Market Attractiveness Analysis
- By Country
- By Product Type
- By Application
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Product Type
- By Application
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Medtronic
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Siemens Healthcare GmbH
- Abbott
- Roche Diagnostics Limited
- Nihon Kohden Corporation
- Medtronic
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by Application, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Product Type , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by Application, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Product Type
- Figure 6: Global Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Application
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Product Type
- Figure 23: North America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by Application
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Product Type
- Figure 30: Latin America Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by Application
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Product Type
- Figure 37: Western Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by Application
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Product Type
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by Application
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Product Type
- Figure 51: East Asia Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by Application
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Product Type
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by Application
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Product Type , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Product Type , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Product Type
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by Application, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by Application, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by Application
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis

