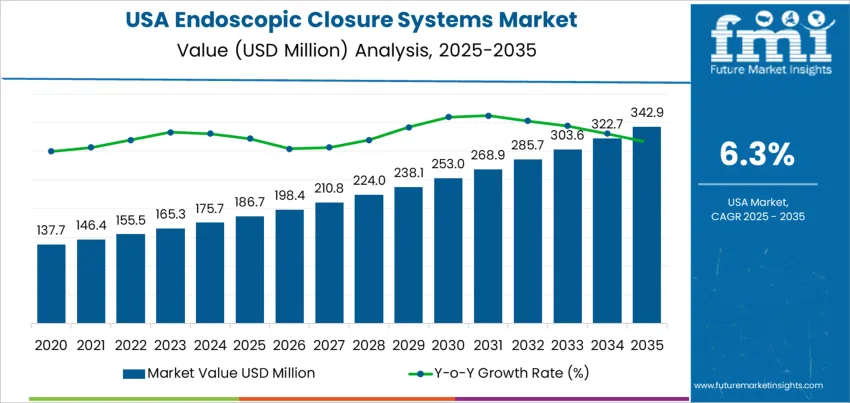
The USA endoscopic closure systems demand is valued at USD 186.7 million in 2025 and is forecast to reach USD 342.9 million by 2035, recording a CAGR of 6.3%. Demand is shaped by expanded utilization of endoscopic interventions for gastrointestinal wall defects, bariatric procedures, and post-operative leakage management. Healthcare providers are adopting closure devices to reduce surgical conversions, minimize hospital stays, and strengthen procedural safety in high-risk patient groups. Increased procedural volumes in ambulatory care settings contribute to wider device deployment across clinical networks.
Endoscopic vacuum-assisted closure systems lead the product landscape. These systems are selected for controlled drainage management, improved tissue granulation, and suitability for complex defect closures where suturing or clipping alone is insufficient. Device improvements include enhanced material biocompatibility, reduced profile for easier device passage, and integrated monitoring of negative-pressure therapy performance.
Demand remains strongest in the West, South, and Northeast due to higher endoscopy procedure throughput, broader adoption of advanced minimally invasive workflows, and the presence of leading tertiary hospitals. Key suppliers include Ethicon (Johnson & Johnson), Boston Scientific Corporation, Medtronic, Apollo Endosurgery, Inc., and Ovesco Endoscopy AG. Their portfolios cover vacuum-assisted systems, over-the-scope clips, and suturing platforms used in gastrointestinal closure and leak-prevention applications.
Growth contribution for demand for endoscopic closure systems in the United States is concentrated within hospital surgical departments and advanced gastrointestinal care centers. These facilities generate the largest share of total revenue because they perform high volumes of minimally invasive procedures requiring reliable closure solutions. Increased adoption of endoscopic resections, bariatric interventions, and gastrointestinal defect repairs supports solid contribution from these clinical environments throughout the forecast period.
Technology enhancements such as improved clip delivery devices and suture-based closure tools add additional contribution by expanding the types of lesions that can be treated endoscopically. Payer support for minimally invasive alternatives to open surgery encourages procedural shifts that increase product use. Ambulatory surgery centers contribute a rising share as their endoscopic infrastructure grows and more procedures shift to outpatient settings.
Later-stage contributions broaden toward trauma and emergency applications where faster recovery and reduced complication risks are priorities. Consistent disease prevalence across digestive disorders ensures ongoing replacement demand for instruments and consumables. The overall contribution index indicates stable gains anchored by core gastrointestinal procedures, with incremental growth from expanded clinical indications and wider outpatient adoption.
| Metric | Value |
|---|---|
| USA Endoscopic Closure Systems Sales Value (2025) | USD 186.7 million |
| USA Endoscopic Closure Systems Forecast Value (2035) | USD 342.9 million |
| USA Endoscopic Closure Systems Forecast CAGR (2025 to 2035) | 6.3% |
Demand for endoscopic closure systems in the USA is increasing because clinicians perform a rising number of minimally invasive gastrointestinal and bariatric procedures that require safe and immediate closure of tissue defects. Hospitals prefer closure devices that reduce procedure time and help avoid surgical conversion, which supports faster recovery for patients and lower overall treatment costs. Expanded use of endoscopic mucosal resection and submucosal dissection for early stage tumors creates consistent demand for reliable closure of post procedural wounds to limit bleeding and leakage risks.
Ambulatory surgery centers adopt closure systems to strengthen efficiency in outpatient care, especially as payers encourage shifts away from open surgery. Manufacturers provide improved clip mechanisms and suturing technologies that work in complex anatomical locations, supporting adoption by gastroenterologists and advanced endoscopists. Increased screening for colorectal disease leads to a greater volume of polyp removal procedures, reinforcing regular use of closure tools in follow up management. Constraints include high capital and disposable component cost for advanced systems, along with training requirements for proper device handling. Smaller facilities may standardize a limited range of closure tools until procedure volumes justify broader adoption.
Demand for endoscopic closure systems in the United States is supported by the rising adoption of minimally invasive procedures and the need to manage postoperative complications with fewer surgical conversions. Hospitals and ambulatory surgical centers increasingly rely on these devices to reduce recovery time, improve patient safety, and control treatment costs related to gastrointestinal defects. Growth is strengthened by expanding clinical use in complex GI interventions and availability of advanced suturing and clip-based technologies.
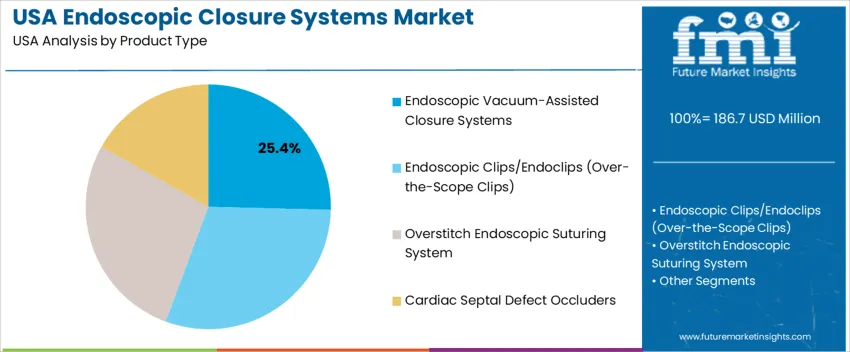
Endoscopic vacuum-assisted closure systems account for 25.4%, driven by their effectiveness in treating anastomotic leaks, fistulas, and perforations that require continuous drainage and tissue granulation. They are widely used in USA tertiary hospitals due to proven clinical outcomes and reduced hospitalization duration. Other product types such as endoscopic clips/endoclips, overstitch suturing systems, and cardiac septal defect occluders also maintain high relevance, particularly for closing mucosal defects, repairing septal openings, and securing stents. Technology adoption depends on patient complexity, anatomical access, and reimbursement support within USA GI endoscopy units. Increasing expertise in advanced gastrointestinal procedures encourages broader utilization of multiple closure modalities, often in hybrid form, ensuring precision sealing and improved procedural success.
Key Points:
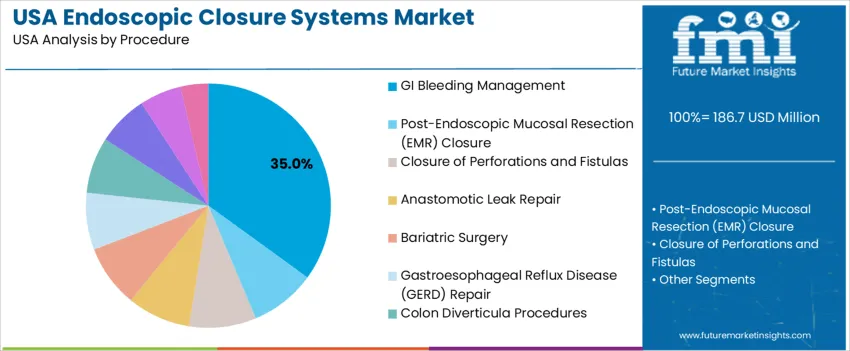
GI bleeding management represents 35.0%, reflecting high procedure frequency and the need for reliable hemostatic closure during endoscopy. These devices support ulcer stabilization, vessel sealing, and post-intervention bleeding prevention. Other interventions such as post-EMR closure, treatment of perforations and fistulas, anastomotic leak repair, bariatric and GERD procedures, diverticula repair, stent fixation, and pediatric applications continue to use closure systems depending on severity and risk of leakage. USA clinical protocols increasingly emphasize early, device-based closure to reduce emergency surgery rates. Growth remains tied to aging demographics and higher prevalence of GI conditions requiring therapeutic endoscopy, solidifying closure systems as standard equipment in advanced GI centers nationwide.
Key Points:
Growth of minimally invasive surgery, increased focus on faster postoperative recovery and wider adoption of advanced endoscopic procedures are driving demand.
In the United States, endoscopic closure systems are increasingly required in hospitals and ambulatory surgery centers performing gastrointestinal and bariatric interventions where reducing post-procedural leaks is essential. Clinicians use mechanical closure tools and tissue approximation devices to avoid conversion to open surgery, supporting shorter hospital stays under value-based reimbursement models. The prevalence of colorectal cancer screening and therapeutic endoscopy contributes to routine use of closure devices for polypectomy and mucosal resection. Bariatric surgery programs expand closure adoption to reduce complications during procedures for obesity management. Training by gastroenterology societies and manufacturer support strengthen surgeon familiarity with closure options, supporting steady procurement across high-volume centers.
Higher device cost, variability in insurance reimbursement and limited adoption in low-complexity cases restrain demand.
Advanced closure tools may carry higher per-case cost than basic clips or manual suturing, leading administrators to evaluate cost-benefit for routine indications. Reimbursement for closure devices depends on procedural coding and outcomes justification, which may discourage extensive use in minor resections. Some community facilities with lower procedure complexity continue using traditional hemostatic clips when complication risk is minimal. Device learning curves require procedural training that smaller centers may delay until case volumes increase. These financial and operational considerations contribute to selective deployment aligned with clinical risk.
Shift toward full-thickness defect management, increased use in outpatient GI suites and rising integration with digital visualization systems define key trends.
Hospitals adopt closure systems capable of managing full-thickness perforations and large defects during advanced endoscopic resection, reducing the need for emergency surgery. Ambulatory GI centers expand closure device USAge to support safe same-day discharge for polyp removal and therapeutic procedures. Device manufacturers integrate improved articulation and visualization compatibility to support precise placement within confined anatomy. Real-time performance data collected through connected imaging platforms assists physicians in quality tracking and device selection. These developments indicate sustained, safety-driven demand for endoscopic closure systems across the United States digestive health and minimally invasive surgery sectors.
Demand for endoscopic closure systems in the United States is rising as minimally invasive gastrointestinal, bariatric, and colorectal procedures expand in both ambulatory and hospital settings. Procurement priorities include suture strength, device ergonomics, deployment precision, and compatibility with varied endoscopic platforms used during defect management and perforation closure. Increased reimbursement support and surgeon familiarity are accelerating uptake across digestive-care networks. West USA leads with 7.2% CAGR, followed by South USA at 6.5%, Northeast USA at 5.8%, and Midwest USA at 5.0%. Growth aligns with clinical volume concentration, training access, and endoscopy-suite modernization across regional healthcare systems.
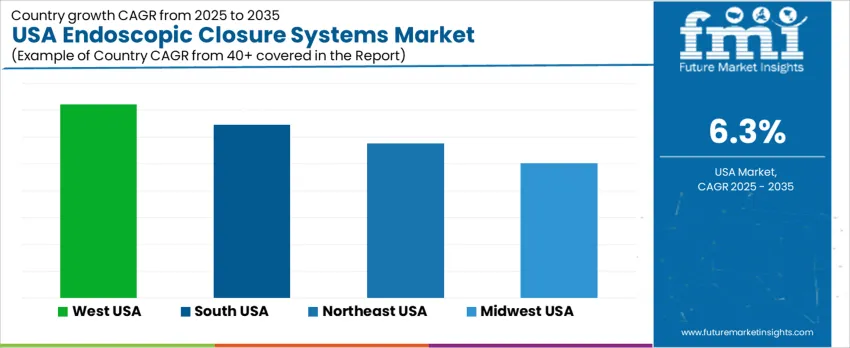
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 7.2% |
| South USA | 6.5% |
| Northeast USA | 5.8% |
| Midwest USA | 5.0% |
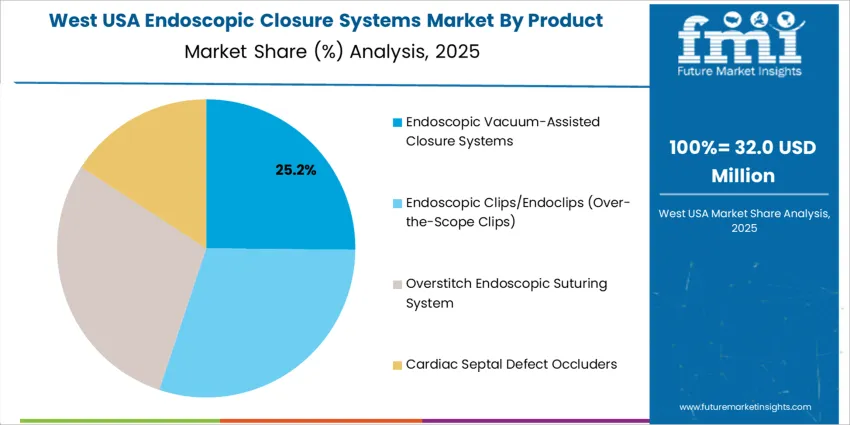
West USA demonstrates strong expansion at 7.2% CAGR, supported by high procedural intensity across California and adjacent states where gastroenterology practices adopt advanced closure tools to reduce postoperative complications. Bariatric-surgery volumes stimulate procurement for leak prevention and defect reinforcement. Teaching hospitals utilize closure systems for training on complex endoscopic interventions, while outpatient centers adopt solutions that shorten recovery and same-day discharge timelines. Clinical evaluations emphasize deployment accuracy in narrow anatomical areas and time-saving mechanics improving procedural efficiency. Vendor access through established medical-device distribution networks strengthens product availability. Purchasing decisions incorporate compatibility with robotic and digital visualization platforms increasingly used in advanced GI suites. Hospitals track durability and repeatable performance under varying tissue conditions.
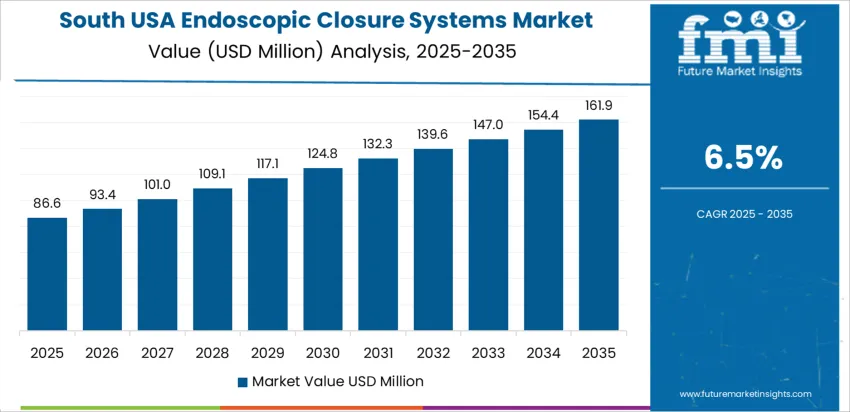
South USA shows 6.5% CAGR, tied to broader endoscopy access and higher obesity-related GI procedure rates across Texas, Florida, and Georgia. Healthcare networks integrate closure systems to manage bleeding and perforation in therapeutic interventions, reducing escalation to invasive surgery. Ambulatory surgical centers expand use as reimbursement policies favor minimally invasive defect repair. Procurement departments prioritize devices that reduce operator learning curves while ensuring consistent tissue-approximation strength. Growing populations in suburban regions expand procedural throughput, prompting inventory scaling among distributors. Evaluation criteria include ergonomic handle design, flexible system sizing for varied anatomy, and validated performance in inflamed or friable tissues.
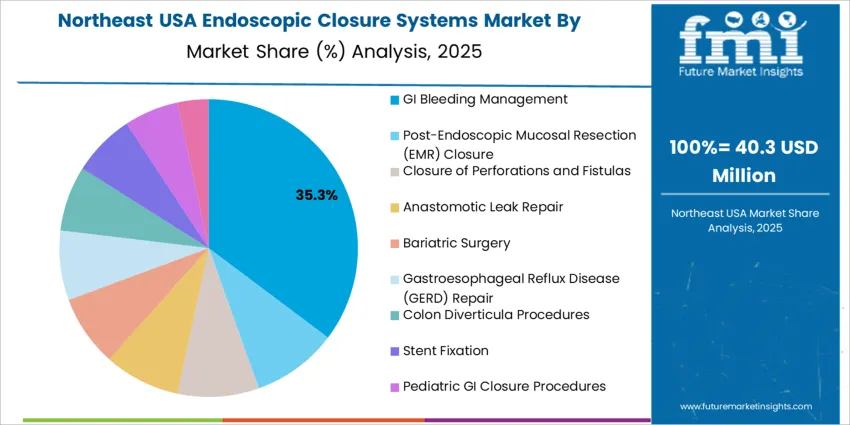
Northeast USA increases at 5.8% CAGR, driven by dense academic hospital networks in New York, Pennsylvania, and Massachusetts performing high-complexity therapeutic endoscopy. Clinicians implement closure systems to reduce adverse event rates in polypectomy, EMR, and ESD cases. Evidence-based procurement emphasizes controlled-deployment mechanisms supported by clinical-trial validation. Strong regulatory compliance expectations guide supplier qualification and traceable material sourcing. Urban medical systems coordinate cross-facility standardization to simplify training and inventory. Hospitals analyze product lifecycle economics, balancing single-use advantages with sustainability considerations in device-usage planning.
Midwest USA posts 5.0% CAGR, reflecting steady uptake in integrated hospital networks across Illinois, Ohio, and Michigan. Closure systems support perforation prevention during routine polypectomy in aging populations with elevated GI disease prevalence. Facilities value durability and reliable deployment performance for efficient procedure completion. Procurement strategies focus on cost-effectiveness due to budget-managed purchasing environments. Distributors maintain predictable supply aligned with stable procedure counts rather than surge-driven variability. Clinician education programs coordinate with regional conferences to expand familiarity with newer clip-and-suture technologies.
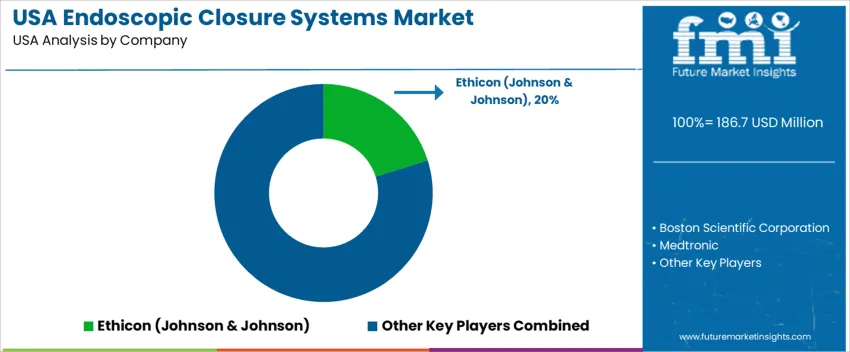
Endoscopic-closure systems in the United States are used in gastrointestinal procedures including polyp removal, perforation repair, bariatric surgery, and controlled tissue approximation. Adoption is guided by the shift toward minimally invasive intervention across ambulatory surgery centers and hospital GI suites. Systems must support secure closure under varied anatomical and access conditions, while enabling clinicians to reduce complication risk and avoid conversion to open surgery. Device dependability, ergonomic control, and compatibility with widely installed flexible endoscopes influence procurement decisions. Ethicon (Johnson & Johnson) holds an estimated 20.2% share. Its endoscopic stapling and surgical-closure portfolio maintains strong penetration in U.S. hospitals due to procedure familiarity and broad surgeon training support.
Boston Scientific Corporation contributes through endoluminal closure clips and suturing tools used in complex GI interventions. Medtronic supports demand with advanced mechanical closure solutions that integrate into bariatric and colorectal workflows. Apollo Endosurgery participates with endoscopic suturing systems adopted in obesity treatment and defect repair across specialized GI programs. Ovesco Endoscopy AG provides over-the-scope clip devices valued in challenging defect closures and emergency GI response scenarios. Competitive positioning in the United States depends on closure reliability, learning-curve efficiency, proven clinical outcomes, and access to field-based technical support that assists surgical teams adopting advanced endoluminal techniques for improved patient recovery and reduced hospital length of stay.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Product Type | Endoscopic Vacuum-Assisted Closure Systems, Endoscopic Clips/Endoclips (Over-the-Scope Clips), OverStitch Endoscopic Suturing System, Cardiac Septal Defect Occluders |
| Procedure | GI Bleeding Management, Post-Endoscopic Mucosal Resection (EMR) Closure, Closure of Perforations and Fistulas, Anastomotic Leak Repair, Bariatric Surgery, Gastroesophageal Reflux Disease (GERD) Repair, Colon Diverticula Procedures, Stent Fixation, Pediatric GI Closure Procedures, Other Procedures |
| Regions Covered | West USA, South USA, Northeast USA, Midwest USA |
| Key Companies Profiled | Ethicon (Johnson & Johnson), Boston Scientific Corporation, Medtronic, Apollo Endosurgery, Inc., Ovesco Endoscopy AG |
| Additional Attributes | Dollar sales by product type and procedure; regional demand differences driven by GI disease prevalence and advanced endoscopy adoption; growth in minimally-invasive closure techniques for perforations, leaks, and bariatric complications; technology innovation in clip-based and suturing systems; positioning of U.S. hospitals and ambulatory centers as major adopters for complex GI interventions. |
How big is the demand for endoscopic closure systems in USA in 2025?
The demand for endoscopic closure systems in USA is estimated to be valued at USD 186.7 million in 2025.
What will be the size of endoscopic closure systems in USA in 2035?
The market size for the endoscopic closure systems in USA is projected to reach USD 342.9 million by 2035.
How much will be the demand for endoscopic closure systems in USA growth between 2025 and 2035?
The demand for endoscopic closure systems in USA is expected to grow at a 6.3% CAGR between 2025 and 2035.
What are the key product types in the endoscopic closure systems in USA?
The key product types in endoscopic closure systems in USA are endoscopic vacuum-assisted closure systems, endoscopic clips/endoclips (over-the-scope clips), overstitch endoscopic suturing system and cardiac septal defect occluders.
Which procedure segment is expected to contribute significant share in the endoscopic closure systems in USA in 2025?
In terms of procedure, gi bleeding management segment is expected to command 35.0% share in the endoscopic closure systems in USA in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.