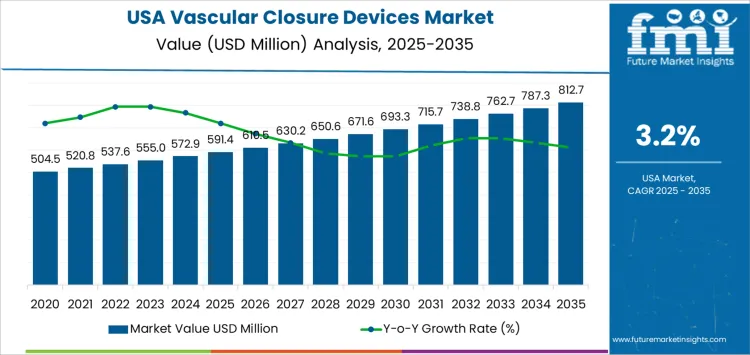
The USA vascular closure devices demand is valued at USD 591.4 million in 2025 and is projected to reach USD 812.7 million by 2035, reflecting a CAGR of 3.2%. Growth is supported by increased catheter-based cardiovascular and peripheral interventions where rapid hemostasis and early ambulation improve patient outcomes. Higher procedural volumes in older populations continue to strengthen demand across interventional cardiology and radiology settings.
The West, South, and Northeast regions show strong adoption driven by large hospital networks and advanced cardiac care infrastructure. Key suppliers include Abbott Laboratories, Terumo Corporation, Medtronic Plc, Teleflex Incorporated, and Johnson & Johnson. Their strategies focus on improved sealant materials, next-generation deployment mechanisms, and product portfolios tailored for both femoral and emerging radial access requirements to support evolving interventional practice standards.
Demand for vascular closure devices in the United States remains in an expansion phase, yet certain indicators show movement toward a gradual saturation point. High adoption across cardiac catheterization labs, interventional radiology units, and peripheral vascular centers has already replaced manual compression in many procedures. Growth continues as procedure volumes rise due to aging demographics and greater use of minimally invasive interventions. Most large hospitals already use closure devices as a standard practice, which limits new site expansion.
Incremental gains now rely on product upgrades such as faster hemostasis, improved biocompatibility, and reduced post-procedure complications. Ambulatory surgical centers add ongoing but slower contribution. Competitive pricing pressure and purchasing consolidation reduce the pace of uptake across mature facilities. Awareness among clinicians is high, meaning expansion is not driven by education gaps. Future gains depend on next-generation technologies that demonstrate clear clinical advantage in complex cases. The sector is advancing but trending toward moderate saturation because core demand from major cardiovascular service providers is already met.
| Metric | Value |
|---|---|
| USA Vascular Closure Devices Sales Value (2025) | USD 591.4 million |
| USA Vascular Closure Devices Forecast Value (2035) | USD 812.7 million |
| USA Vascular Closure Devices Forecast CAGR (2025-2035) | 3.2% |
Demand for vascular closure devices in the USA is increasing because hospitals and surgical centers perform a high volume of interventional cardiology and radiology procedures such as angioplasty, stent placement and endovascular repairs. After using large-bore catheters, clinicians rely on closure devices to seal the access site quickly and minimize bleeding or vascular complications. Use of these devices reduces recovery time and allows faster patient mobilization, which supports same-day discharge protocols and more efficient patient throughput. Expansion of outpatient surgical and catheterization suites drives adoption because closure devices help standardize post-procedure care while reducing demands on nursing staff. Aging demographics and rising cardiovascular disease prevalence contribute to procedural growth and recurring demand for closure solutions.
Manufacturers provide a variety of closure mechanisms, including plug-based, suture-based and collagen-based systems, to suit different clinical scenarios and patient anatomies. Devices with proven safety records gain wider acceptance among clinicians seeking reliable outcomes and lower complication rates. Constraints include device cost and requirement for clinician training to ensure proper deployment. Some vascular access sites remain unsuitable for certain closure systems, which may limit use. Hospitals may evaluate cost per procedure before replacing manual compression when volumes are modest.
Demand for vascular closure devices in the United States reflects consistent use of minimally invasive cardiovascular procedures where rapid hemostasis and early ambulation are required. Clinical decision factors include access route, vessel diameter, patient bleeding risk, and interventional workflow efficiency. Increased adoption follows a shift toward catheter-based therapies across cardiology and vascular care networks. Growth benefits from quality-of-care mandates that reduce post-procedure complications and recovery duration.
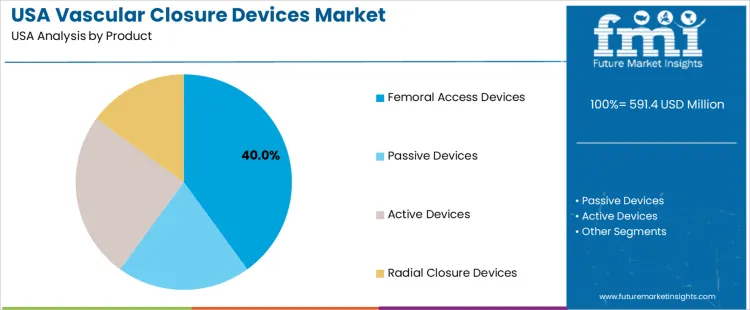
Femoral access closure devices account for 40.0%. Large-bore access remains common in diagnostic angiography and interventional cardiology. These devices support secure vessel sealing that minimizes hospital stay. Active closure devices represent 25.0%. Suture-based and clip-based systems support predictable performance in complex accesses. Passive devices contribute 20.0%. Collagen-based plugs serve lower-risk cases where simplified handling and broad anatomical readiness are prioritized. Radial closure devices hold 15.0%. Use aligns with clinical preference for reduced bleeding and enhanced patient mobility in radial-access interventions. Adoption patterns reflect clinician familiarity, post-procedure discharge goals, and reduction of access-site complications.
Key Points:
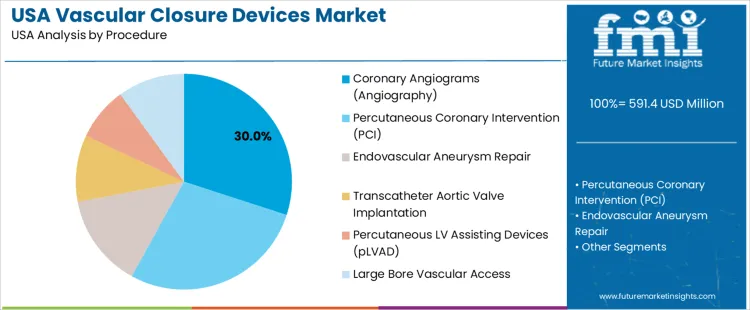
Coronary angiography represents 30.0%. High diagnostic volume drives frequent vascular access closures in hospitals. Percutaneous coronary intervention holds 28.0%. Increased stent placement rates support closure utilization among cardiovascular patients requiring efficient discharge targets. Endovascular aneurysm repair contributes 14.0%, and transcatheter aortic valve implantation accounts for 10.0%. Large bore vascular access procedures collectively represent 10.0%, requiring closure systems engineered for enhanced mechanical integrity. Percutaneous LV assist device placement contributes 8.0%, where hemostasis performance influences device removal outcomes. Procedure mix reflects advanced structural heart therapy expansion paired with strong safety emphasis.
Key Points:
Growth of minimally invasive cardiovascular procedures, rising focus on early ambulation and increased adoption in outpatient settings drive demand.
In the United States, vascular closure devices support hemostasis after catheter-based interventions performed in cardiology and interventional radiology labs. Hospitals and ambulatory surgery centers increase reliance on percutaneous coronary intervention, electrophysiology studies and peripheral angioplasty, which creates a continuous need for rapid and reliable femoral artery closure. Clinicians seek products that reduce manual compression time and shorten bedrest requirements for patients recovering from diagnostic and therapeutic catheter procedures.
Payers and providers align around value-based care models that prioritize shorter recovery periods, lower complication rates and faster discharge. Ambulatory facilities add closure devices to enable same-day procedures with predictable throughput and efficient room turnover. These clinical and operational priorities maintain steady procurement across USA institutions performing high volumes of vascular access procedures.
Pricing pressure, anatomical access shifts and standardized purchasing controls restrain growth.
Cost evaluation for single-use closure devices influences selection in facilities where budgets remain tight. Adoption may slow when clinicians transition coronary access to the radial artery, reducing femoral punctures in certain procedural categories. Purchasing committees enforce product standardization policies that limit brand variety and extend vendor review cycles across multi-campus systems. Training requirements influence usage in centers with nursing turnover or limited device education time. Low-acuity diagnostic cases with minimal bleeding risk sometimes continue using manual compression, reducing device demand in selected populations. These considerations limit accelerated expansion across all care environments.
Shift toward extravascular technologies, increased use in structural heart interventions and rising integration with radial-to-femoral hybrid workflows define key trends.
Manufacturers advance closure products that avoid intravascular components to support simplified deployment and reduce imaging needs. Structural heart programs adopt reliable closure devices during large-bore femoral access used for transcatheter valve therapies and endovascular aortic procedures. Workflow planning aligns closure decisions with recovery pathways that target walking readiness within hours of the procedure.
Device design emphasizes smaller profiles and tactile feedback to improve handling consistency among diverse operator skill levels. Postmarket analytics track bleeding events and recovery metrics to refine purchasing strategies in high-volume centers. These developments indicate durable, procedure-driven demand for vascular closure devices across the United States supported by efficiency goals, outpatient care growth and ongoing cardiovascular innovation.
Demand for vascular closure devices in the United States grows as hospitals and outpatient cardiovascular centers seek efficient hemostasis solutions after catheter-based diagnostic and interventional procedures. Healthcare systems prioritize workflow efficiency, mobility recovery, and reduced post-procedure complications. Manufacturers supply plug-, suture-, and clip-based devices used in angiography, percutaneous coronary interventions, and electrophysiology cases.
Procurement planning considers patient-volume forecasts and operator learning requirements. West USA retains the fastest expansion at 3.7% CAGR, followed by South USA at 3.3%, Northeast USA at 3.0%, and Midwest USA at 2.6%. Facility administrators examine product-performance consistency, vascular-access compatibility, and supply-chain resilience supporting uninterrupted procedural schedules.
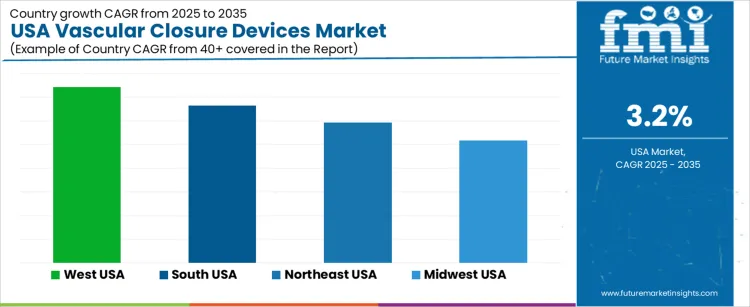
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 3.7% |
| South USA | 3.3% |
| Northeast USA | 3.0% |
| Midwest USA | 2.6% |
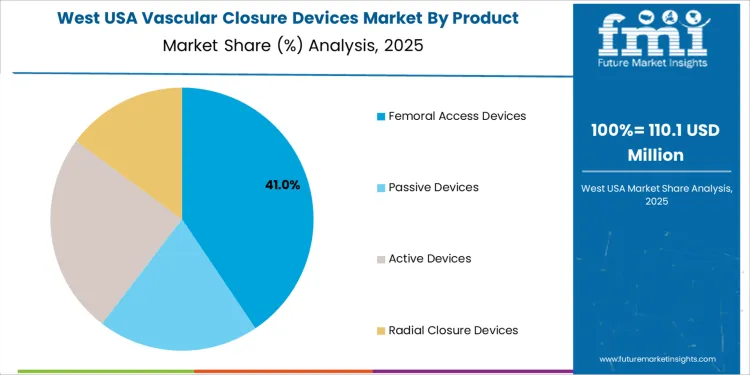
West USA posts 3.7% CAGR, supported by high interventional cardiology activity in California and surrounding states. Hospital networks emphasize devices that support rapid patient mobilization to increase room turnover and reduce extended post-procedure monitoring. Physicians evaluate closure reliability in large-bore procedures associated with structural-heart and electrophysiology interventions. Tender evaluations include seal-strength stability in diverse vessel conditions. Inventory coverage extends to ambulatory surgical centers that maintain short-stay recovery pathways. Procurement teams assess compatibility with existing guide-sheath profiles and staff familiarity to reduce variability.
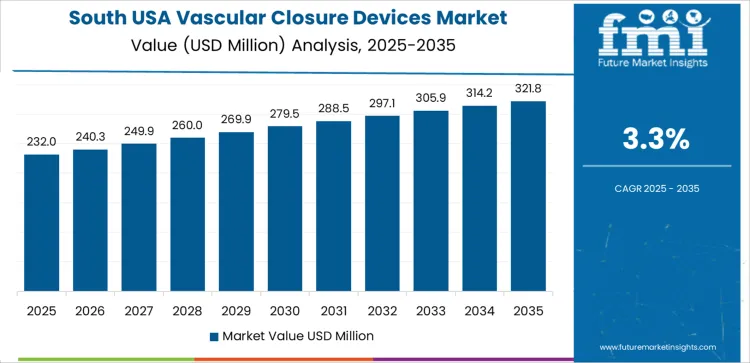
South USA expands at 3.3% CAGR, influenced by extensive cardiovascular disease incidence across Texas and Gulf Coast states. Providers integrate closure devices to support hemostasis during routine catheter-based care in high-volume facilities. Buyers focus on predictable deployment mechanisms that reduce dependence on manual compression during peak caseloads. Hospital administrators include closure-related efficiency metrics when evaluating treatment pathways that support faster discharge readiness. Training access strengthens user confidence among new interventional teams.
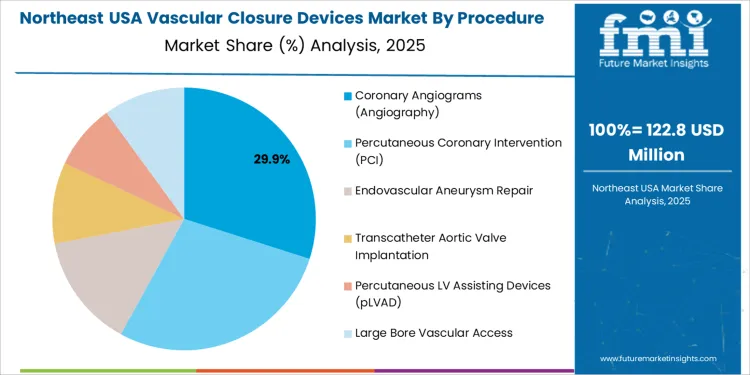
Northeast USA shows 3.0% CAGR, shaped by procedure concentration in academic medical centers and research-driven hospitals in New York, New Jersey, and Massachusetts. Clinicians review closure behavior in complex access sites during high-acuity interventions. Decision-makers prioritize clinically validated deployment systems delivering reliable outcomes with variable patient physiology. Predictable recovery supports bed-management planning in dense urban locations. Pharmacy and procurement teams monitor post-market evidence to confirm performance benchmarks.
Midwest USA maintains 2.6% CAGR, with adoption driven by structured catheter-based care across Illinois, Michigan, and Ohio. Clinicians seek closure devices that maintain workflow predictability in facilities serving mixed-acuity patient populations. Product pricing and supply stability influence purchase decisions where budget planning remains conservative. Adoption expands steadily as staff certification and familiarity improve. Vendors support training linked to procedural-volume growth forecasts across regional cardiovascular centers.
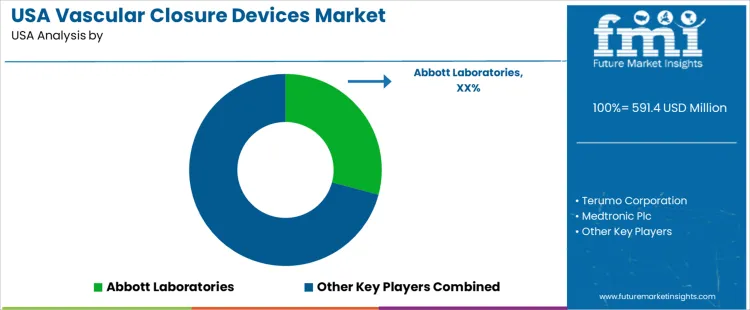
Demand for vascular closure devices in the United States reflects strong adoption in interventional cardiology and peripheral vascular procedures. Hospitals and catheterization labs select products that support quick hemostasis, consistent access site integrity, and shorter patient ambulation times. Procurement teams evaluate device compatibility with common sheath diameters, operator learning requirements, and long-term safety evidence. Reimbursement stability strengthens vendor relationships with large health systems.
Brand familiarity and widely published clinical data support repeat purchasing. Terumo Corporation supplies polymer-based and angiographic solutions used in radial and femoral pathways. USA deployment from established distribution networks maintains visibility among cardiologists and vascular surgeons.
Medtronic plc provides implantable and extravascular closure options designed to reduce reliance on manual compression. Healthcare providers note standardized workflows during high-volume procedures. Teleflex Incorporated offers access site management systems that support predictable seal formation with ease of use for new operators. Johnson & Johnson participates through surgical and interventional subsidiaries where closure capability aligns with broader cardiovascular portfolios.
Competitive positioning depends on clinical performance across varied anatomies, inventory availability for 24-hour intervention units, and training programs for staff proficiency. Institutions track outcomes through internal quality metrics that influence continuing product selection.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Product | Femoral Access Devices, Passive Devices, Active Devices, Radial Closure Devices |
| Procedure | Coronary Angiograms (Angiography), Percutaneous Coronary Intervention (PCI), Endovascular Aneurysm Repair, Transcatheter Aortic Valve Implantation, Percutaneous LV Assisting Devices (pLVAD), Large Bore Vascular Access |
| End User | Hospitals, Independent Catheterization Labs, Ambulatory Surgical Centers |
| Regions Covered | West USA, South USA, Northeast USA, Midwest USA |
| Key Companies Profiled | Abbott Laboratories, Terumo Corporation, Medtronic Plc, Teleflex Incorporated, Johnson & Johnson |
| Additional Attributes | Dollar sales by product category, procedure type, and end-user segments; regional use influenced by PCI volumes, catheterization lab capacity, and adoption of radial access; competitive landscape with active and passive sealing technologies; growth driven by minimally invasive cardiovascular interventions and reduced time-to-ambulation post-procedure; procurement via hospitals and outpatient cardiac centers. |
How big is the demand for vascular closure devices in USA in 2025?
The demand for vascular closure devices in USA is estimated to be valued at USD 591.4 million in 2025.
What will be the size of vascular closure devices in USA in 2035?
The market size for the vascular closure devices in USA is projected to reach USD 812.7 million by 2035.
How much will be the demand for vascular closure devices in USA growth between 2025 and 2035?
The demand for vascular closure devices in USA is expected to grow at a 3.2% CAGR between 2025 and 2035.
What are the key product types in the vascular closure devices in USA?
The key product types in vascular closure devices in USA are femoral access devices, passive devices, active devices and radial closure devices.
Which procedure segment is expected to contribute significant share in the vascular closure devices in USA in 2025?
In terms of procedure, coronary angiograms (angiography) segment is expected to command 30.0% share in the vascular closure devices in USA in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.