The global vascular closure devices sector is on track to achieve a valuation of USD 2.2 billion by 2036, accelerating from USD 1.6 billion in 2026 at a CAGR of 3.4%. As per Future Market Insights, expansion is structurally underpinned by the rising volume of catheter-based interventional procedures, the clinical shift from manual compression to active closure in both arterial and venous access sites, and the expansion of structural heart and electrophysiology procedures that require large-bore vascular access management.
The American College of Cardiology (ACC) confirmed in its 2024 Clinical Performance Measures update that percutaneous coronary intervention (PCI) volumes in the United States exceeded 600,000 annually, with same-day discharge protocols increasingly requiring reliable vascular closure for safe patient ambulation. This procedural volume compels interventional cardiologists and electrophysiologists to adopt active closure devices that reduce time to hemostasis and enable earlier ambulation. Simultaneously the competitive landscape is expanding as manufacturers move beyond standard arterial closure to address venous, large-bore, and transradial access sites.
Haemonetics acquired Vivasure Medical for USD 116 million upfront in January 2026 to expand its portfolio into large-bore structural heart and endovascular closure. Christopher Simon, President and CEO of Haemonetics, stated in the acquisition announcement: 'The acquisition of Vivasure is aligned with our vascular closure growth strategy and expands our portfolio into new and attractive market segments, including structural heart and endovascular closure.' FMI opines that Haemonetics' combined VASCADE MVP XL mid-bore venous system and Vivasure large-bore technology will position the company to capture 15 to 20% of the emerging large-bore closure segment by 2030, as structural heart procedure volumes (TAVR, LAAC, mitral repair) continue to accelerate globally.
The competitive landscape in 2024, 2025, and 2026 is defined by venous closure expansion, large-bore portfolio building, and interventional infrastructure acquisition. Haemonetics announced the full USA market release of the VASCADE MVP XL Mid-Bore Venous Vascular Closure System in August 2024 for multi-access venous closures using 10 to 15 French sheaths. Cordis received FDA approval for the MYNX CONTROL Venous VCD in July 2024 and was awarded a national agreement with Premier, Inc. for the device. Teleflex entered a definitive agreement in February 2025 to acquire Biotronik's Vascular Intervention business, complementing its MANTA Large Bore Closure device portfolio.
Terumo Interventional Systems received FDA 510(k) clearance for the OPUSWAVE Dual Sensor Imaging System in October 2025. Zylox-Tonbridge announced plans to acquire equity in a European medical device company in January 2026 to accelerate its global vascular closure expansion. As per FMI, this convergence of venous closure platform launches, large-bore structural heart closure acquisitions, and interventional portfolio integration confirms that the vascular closure devices market is transitioning from a single-modality arterial closure market toward a multi-access, multi-bore, arterio-venous closure ecosystem.
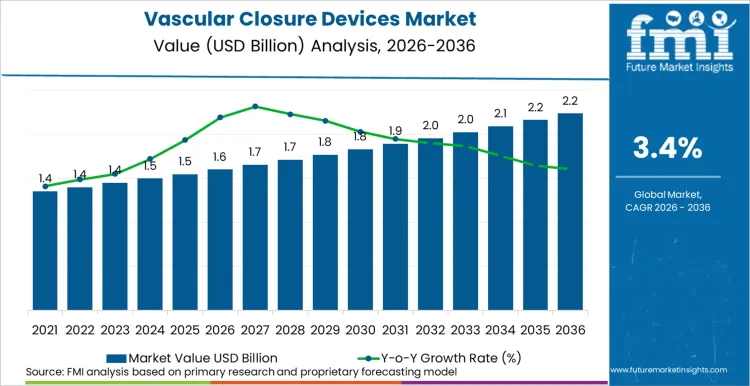
Future Market Insights projects the vascular closure devices industry to expand at a CAGR of 3.4% from 2026 to 2036, increasing from USD 1.6 Billion in 2026 to USD 2.2 Billion by 2036.
FMI Research Approach: FMI proprietary forecasting model based on ACC PCI volume data, structural heart procedure growth analysis, and closure device penetration tracking.
FMI analysts perceive the market evolving toward multi-bore closure ecosystems where venous-specific, large-bore structural heart, and imaging-guided platforms replace standard femoral arterial compression workflows.
FMI Research Approach: Haemonetics VASCADE MVP XL launch and Vivasure large-bore closure acquisition analysis.
The United States holds a significant share of the global vascular closure devices market by value which is supported by 600,000+ annual PCI procedures, same-day discharge adoption, and the concentration of Teleflex, Haemonetics, Abbott, and Cordis operations.
FMI Research Approach: FMI country-level revenue modeling by ACC procedural volume data and hospital GPO contract tracking.
The global vascular closure devices market is projected to reach USD 2.2 Billion by 2036.
FMI Research Approach: FMI long-term revenue forecast derived from structural heart procedure growth and large-bore closure adoption curves.
The vascular closure devices market includes suture-mediated, collagen-based, sealant-based (PEG), clip-based, and compression-assisted closure systems used to achieve hemostasis at arterial and venous catheter access sites in hospitals and catheterisation laboratories.
FMI Research Approach: FMI market taxonomy aligned with ACC/AHA vascular access management guidelines and FDA device classification codes.
Globally unique trends include large-bore structural heart closure expansion (Haemonetics/Vivasure USD 116M, Teleflex MANTA/Biotronik), venous-specific PEG sealant closure (Cordis MYNX CONTROL Venous/Premier agreement), and imaging-guided closure integration (Terumo OPUSWAVE FDA clearance).
FMI Research Approach: Haemonetics/Vivasure acquisition and Cordis MYNX CONTROL clinical data tracking.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 1.6 Billion |
| Industry Value (2036) | USD 2.2 Billion |
| CAGR (2026 to 2036) | 3.4% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The North American vascular closure devices market is being developed with the help of major medical device manufacturers, hospitals, and healthcare providers who are initiating the adoption of catheter-based interventions and at the same time are addressing the growing peripheral vascular diseases.
The USAA. was in the leading position traditionally, as it is well-equipped with healthcare facilities that are always in need of fast and effective vascular access closure complications and support with the use and FDA approvals for new bioabsorbable designs. On the contrary, factors like high procedural costs, rigor reimbursement policies, and device-related problems present obstacles to the market's growth.
Medical institutions and technology firms are at the forefront of innovation with the integration of AI-powered vascular imaging, the advancement of same-day discharge protocols for catheterization procedures, and the reinforcement of collaborations between hospitals and med-tech companies.
These improvements in efficiency and patient care will allow the North American vascular closure devices market to progress and consequently become a pioneer in interventional cardiology and vascular access management
Fostering investments in interventional cardiology and progressing towards the adoption of less invasive procedures, vascular closure devices market in Europe is being propelled by healthcare providers, regulatory bodies and medical device manufacturers. The introduction of advanced closure technologies and strong regulatory frameworks has support medical device innovation.
Germany, France, and the UK are the front runners in this sector, Cardiovascular treatment settings are actively using suture-based and bioabsorbable passive devices to have femoral access. Hospitals settings in collaboration with the governments offering patient safety initiatives in catheterization procedures.
The market growth is also hindered by complicated MDR regulations, expensive next-generation closure devices and the slow embracing of bioresorbable technologies in public healthcare institutions.
The key players and the research institutes spur the growth by integrating hybrid closure systems and cardiovascular management systems. The demand for disposable vascular closure devices and low-profile closure techniques that are modern in the industry to avoid complications in the patient's healing process have made Europe a top spot for the vascular closure innovation drive.
The Asia-Pacific region is currently enjoying a flourishing market for vascular closure devices due to investments in healthcare infrastructure, increased incidences of cardiovascular diseases, and mandated rise in demand for economical closure products.
Catheter-based interventions that are now widely available, government support with regard to cardiac care that is on the rise, along with increased interest in and the demand for less invasive closure products are the key aspects of the potential growth markets in countries like China, Japan, and India.
However, barriers like the inability to afford the costs, lack of formal vascular closure training, and delayed regulatory approval of new closure technologies can slow down the market entry. Moreover, merging technology such as self-expanding closure device and exploration of hybrid vascular heat-sealing technologies are augmenting procedural efficacy and patient comfort in the region.
Challenges
The vascular closure devices market encounters difficulties that include steep expenses for the latest closure technologies, the probability of vascular complications like hematomas and pseudo-aneurysms, and the legal barriers hindering the introduction of new materials for the closure.
The necessity for the better training of vascular access management, the complications of the closure devices being compatible with different sheath sizes, and the unequal adoption of technology between high-tech and low-infrastructure hospitals that are additional barriers to the market growth.
Furthermore, the problems in obtaining the proper closure for large access vessels, the high costs to incorporate the closure system in the outpatient procedure and the reluctance to shift from manual compression to device-based closure, hinder the growth of the market.
Opportunities
The deployment of hybrid vascular closure systems serves as a breakthrough marketing strategy for the industry as it directly deals with the drawbacks of the presently available closure devices. Hybrid systems composed of suture-based components, collagen plugs, and bioresorbable materials have several advantages compared to the standalone suture-based or standalone collagen plug polymer.
They are more versatile, faster to stop the bleeding, and have fewer complications. While hospitals and ambulatory surgical centers are looking for cost-efficient and effective closure devices, hybrid apparatuses have the advantage due to the fact that they can fit different arterial access sizes and also can treat high-risk patient groups.
The producers of medical devices as well as the governing bodies in health care are coming to the realization of this multi-component system's advantages, which in turn encourages their use in interventional cardiology and peripheral vascular procedures.
Moreover, the continuous clinical trials and technological progress are the key factors of an ongoing development in hybrid closure technology. By becoming a universal solution, hybrid closure technologies can not only reduce the complications after the procedures but also lead to better vascular access management.
Between 2020 and 2024, the vascular closure devices (VCDs) market showed an upward trend as a result of the increased popularity of catheter-based operations and the need for effective hemostasis solutions. Preferred by hospitals and ambulatory surgical centers VCDs instead of manual compression, thereby reducing both patient recovery time and procedure duration.
The introduction of technological innovations such as biodegradable and active closure devices, which rendered surgical procedures safer and more effective, was a breakthrough. The regulatory challenges and high cost have caused the adoption of biodegradable less in the emerging markets.
North America was the major player thanks to the amount of surgical procedures, while Asia-Pacific registered the most dynamic growth due to the increased interest of the public and the healthcare improvements. Although the pandemic slowed down the process at first, the market began to flourish as more patients sought interventional treatment.
Between 2026 and 2036, The market will be heading towards artificial intelligence-based vascular access management, bioresorbable materials and hybrid closure systems using suture, collagen and polymer technologies. Drug-eluting and nanotechnology-based VCDs will reduce infection risks and vascular inflammation.
As same-day discharge protocols increase, there will be a need for simple, cheap, and effective closure devices. Manufacturers will go to the smart, data-driven solutions by making stricter regulatory oversight and real-time monitoring.
Ethical sourcing, sustainability, and supply chain efficiency will gain importance, thus will lead to a more affordable and fairer product. Aging population and the prevalence rate of cardiovascular cases will render the VCD market to be in a constant boost, with countless breakthroughs.
Market Shifts: A Comparative Analysis (2021 to 2025 vs. 2026 to 2036)
| Market Shift | 2021 to 2025 |
|---|---|
| Regulatory Landscape | Adoption of regulations that will ensure the efficacy and safety of vascular closure devices, resulting in standard operating protocols and usage directives |
| Technological Advancements | Introduction of new-generation VCDs with better efficacy and safety profiles, improving patient outcomes and decreasing recovery times |
| Consumer Demand | Greater familiarity with and acceptance of VCDs, resulting in greater demand in many healthcare environments, especially for minimally invasive medical procedures |
| Market Growth Drivers | Increase in cardiovascular disease prevalence, technology innovations in VCD technologies, and a trend towards cost-effective and efficient vascular closure solutions |
| Sustainability | First attempts at eco-friendly production processes and the evolution of VCDs with lower environmental footprints. |
| Supply Chain Dynamics | Reliance on expert suppliers of device components, with attempts at localizing manufacturing to avoid supply chain interruptions witnessed in international events. |
| Market Shift | 2026 to 2036 |
|---|---|
| Regulatory Landscape | Ongoing oversight and potential modifications of guidelines to balance the patient safety advantage against technological advance, coupled with enhanced quality evaluation measures to maximize product performance |
| Technological Advancements | Creation of next-generation VCDs with better portability, easy-to-use interfaces, and compatibility with artificial intelligence for customized vascular closure solutions |
| Consumer Demand | Increase in the demand for integrated VCDs and patient-focused care, fuelled by advances in device technologies and concerns for patient safety and cost efficiency in operations |
| Market Growth Drivers | Development of healthcare infrastructure in emerging markets, growth in healthcare spending, and ongoing technological advancements that boost procedural efficiency and patient outcomes |
| Sustainability | Embracing green practices in manufacturing and distribution, such as the utilization of recyclable materials and energy-saving processes, conforming to international environmental standards |
| Supply Chain Dynamics | Local manufacturing capabilities are being improved through technology development and collaborative partnerships, and as a result, the dependence on imports has decreased and the supply chain has become more robust. |
The vascular closure devices market is expected to grow over the forecast period, fuelled by advancements in vascular technology, increasing use of these devices in catheterization labs and outpatient settings, and a move towards patient-centric care. Manufacturers need to prioritize innovation, adherence to changing regulations, and measures to make it more affordable and accessible to take advantage of future opportunities in this dynamic market.
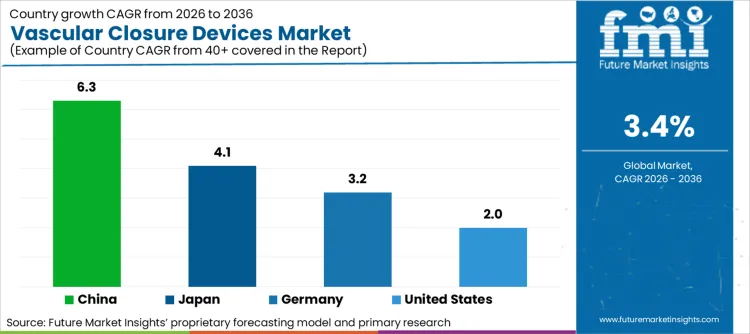
Market Outlook
The United States holds the largest share in the vascular closure devices (VCDs) market due to high procedure volumes, robust hospital infrastructure, and quick adoption of sophisticated closure technologies. VCDs are favoured by healthcare providers over manual compression due to quicker recovery and fewer complications. The availability of top medical device firms and FDA approvals for newer-generation devices are fuelling the market growth
Market Growth Factors
Market Forecast
| Country | CAGR (2026 to 2036) |
|---|---|
| United States | 2.0% |
Market Outlook
The European vascular closure devices market is primarily ruled by Germany because of its strict regulatory compliance, developed hospital infrastructures, and the extensive use of minimally invasive procedures.
Cardiovascular interventions are being performed with the help of hospitals now using bioresorbable closure devices as well as the continuation of product innovation driven by government-sponsored research programs. The use of hybrid closure systems is on the rise in the high-tech medical sector of Germany, especially at the high-tech medical facilities.
Market Growth Factors
Market Forecast
| Country | CAGR (2026 to 2036) |
|---|---|
| Germany | 3.2% |
Market Outlook
Japan's vascular closure device market is growing fast due to technological advancement, the high rate of minimally invasive procedures, and healthcare practices centered on precision. Medical facilities employ AI-powered closure devices for enhancing vascular access management. Japan's growing geriatric population and increasing burden of cardiovascular diseases are speeding up the demand for more efficient and safe VCDs.
Market Growth Factors
Market Forecast
| Country | CAGR (2026 to 2036) |
|---|---|
| Japan | 4.1% |
Market Outlook
China's market for vascular closure devices is on the rise due to rising health care access, growing interventional cardiology, and significant healthcare investments by the government. Key local medical device companies are working on low-cost, high-performance VCDs, as urbanization and increasing cases of heart disease keep vascular interventions and closing solutions in the demand limelight.
Market Growth Factors
Market Forecast
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 6.3% |
Market Outlook
The Indian market for vascular closure devices is having a really strong development rate because of the increase in the number of cardiovascular disease cases, the rise of Catherization lab procedures, and the better quality offered by the health facilities.
Government-funded health programs alongside the growing private sector investments are facilitating the supply of vascular closure devices that are affordable to the mass population, while Tier 1 medical centers are specializing in advanced minimally invasive procedures.
Market Growth Factors
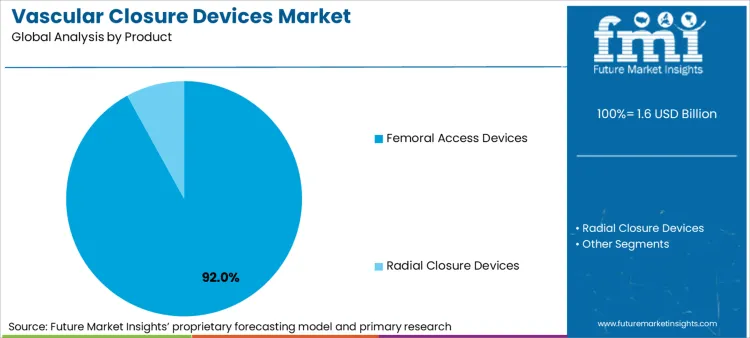
Increasing demand for Suture-Based and Minimally Invasive Hemostasis Systems driving Active Vascular Closure Devices
Active vascular closure devices are designed to offer instant hemostasis post catheterization procedures via either mechanical, suture-based, or collagen-based technologies. VCDs result in reduced recovery times, fewer complications, and better mobility for patients after the procedure.
The interventional cardiology and peripheral vascular interventions boom, the change of trend towards close to no invasive hemostasis for patients and usage of suture-based closure systems are the main drivers for growth in the market. North America and Europe dominate active VCD adoption due to the presence of cardiovascular care centers, while Asia-Pacific is expanding due to the increase in catheterization lab investments.
Passive Vascular Closure Devices are Affordable Haemostasis Solutions for Outpatient and Radial Access Procedures
Passive VCDs utilize external compression, gel-type sealants, or manual compression devices to ensure hemostasis after arterial access procedures. Passive VCDs are used as the first option in low-risk patients and scenarios where cost-saving is a focus.
The expanding demand for cost-effective alternatives for active closure devices, expanding adoption in outpatient sites, and enhancing trend towards radial artery access procedures are driving the growth of the market.
North America and Europe lead in passive VCD use as a result of robust uptake of radial closure methods, whereas Asia-Pacific is observing increased demand as interventional cardiology grows. Future developments are headed towards more sophisticated hemostatic agents for greater closure, intelligent compression devices with real-time pressure feedback, and AI-driven vascular access site management systems
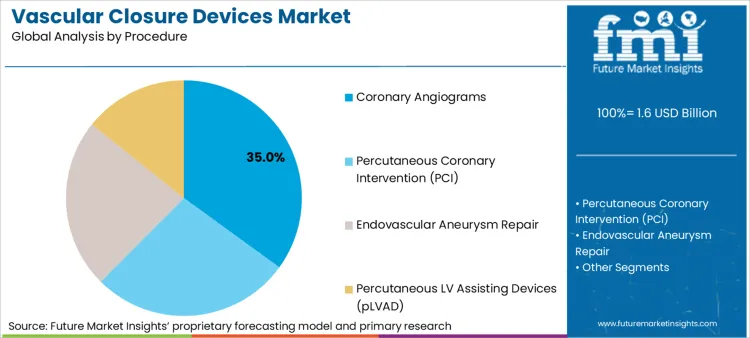
Rising Cardiovascular Cases and Technological Innovations Fuelling Growth in Hospitals and Catheterization Labs
Hospitals and Catherization labs are the major customers of vascular closure devices, as they perform high volumes of percutaneous coronary intervention (PCI), angioplasty, and diagnostic catheterization procedures. These centres reap the benefits of sophisticated closure technologies that minimize hospital stays, improve patient outcomes, and decrease post-procedure bleeding risks.
The growing incidence of cardiovascular diseases, increasing volume of catheter-based procedures, and expanding need for effective haemostasis solutions are fueling market demand.
North America and Europe are the leaders in hospital-based adoption because of robust reimbursement policies, whereas Asia-Pacific is witnessing growth because of increasing healthcare infrastructure. Future developments involve robot-assisted vascular closure and integrated closure devices with automated sealing systems.
Ambulatory Surgical Centers are Expanding Outpatient Cardiovascular Procedures and Demand for Fast, Cost-Effective Closure Solutions
ASCs are turning out to be the primary adopters of vascular closure devices, especially for same-day peripheral vascular and angiographic interventions. They appreciate portable and simple-to-use closure systems for quicker patient discharge and enhanced efficiency in workflow.
The increasing tendency towards outpatient vascular procedures, improving availability of miniature closure devices, and heightened concern for cost-saving healthcare solutions are driving market growth. North America and Europe lead in ASC-based vascular closure uptake because of good regulatory support, whereas Asia-Pacific is witnessing growing demand with an increase in outpatient cardiovascular services.
Future directions include miniaturized vascular closure systems for ASC procedures, patient recovery tracking, and bioresorbable closure technology for improved patient comfort.
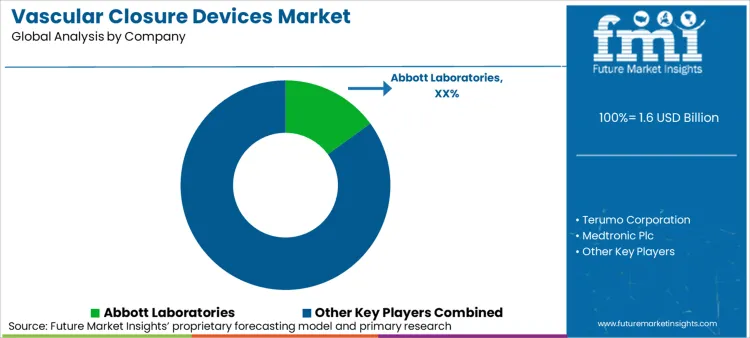
The market for vascular closure devices is competitive, with the rising number of minimally invasive cardiovascular procedures, the evolution of hemostasis technologies, and the increased use of femoral artery closure solutions. Bioabsorbable closure systems, suture-based devices, and fast hemostasis solutions are being invested in by companies to stay competitive.
The market is influenced by established medical device companies, cardiovascular solution providers, and new interventional technology companies, all of which contribute to the changing landscape of vascular closure systems.
Recent Developments:
In February 2025, Teleflex entered a definitive agreement to acquire Biotronik's Vascular Intervention business to complement its MANTA Large Bore Closure device.
Market Share Analysis by Company
| Company Name | Estimated Market Share (%) |
|---|---|
| Abbott Laboratories | 15-20% |
| Terumo Corporation | 12-15% |
| Medtronic Plc | 8-12% |
| Teleflex Incorporated | 8-10% |
| Johnson & Johnson | 5-9% |
| Other Companies (combined) | 52-34% |
| Company Name | Key Offerings/Activities |
|---|---|
| Abbott Laboratories | Market leader offering Perclose ProGlide and StarClose SE suture-based and clip-mediated closure systems. |
| Terumo Corporation | Develops Angio-Seal vascular closure devices designed for rapid hemostasis and patient comfort. |
| Medtronic Plc | Specializes in VenaSeal™ Closure System with vascular closure technology. |
| Teleflex Incorporated | Provides MANTA large-bore vascular closure systems optimized for structural heart and endovascular procedures. |
| Johnson & Johnson | Focuses on EXOSEAL™ Vascular Closure Device and aims to maintain a diverse and innovative product portfolio to meet the changing needs of cardiovascular healthcare professionals and patients |
Key Company Insights
Beyond the leading companies, several other manufacturers contribute significantly to the market, enhancing product diversity and technological advancements. These include:
These companies focus on expanding the reach of vascular closure solutions, offering competitive pricing and cutting-edge innovations to meet diverse interventional cardiology and radiology needs.
The vascular closure devices market represents revenue generated from the manufacture and sale of devices used to achieve hemostasis at percutaneous vascular access sites following catheter-based diagnostic and interventional procedures. The market measures the value of suture-mediated, collagen-based, sealant-based (PEG), clip-based, and compression-assisted closure systems sold to hospitals, catheterisation laboratories, ambulatory surgery centers, and electrophysiology laboratories.
Inclusions cover active arterial closure devices (Perclose ProGlide, Angio-Seal), active venous closure devices (VASCADE MVP XL, MYNX CONTROL Venous), large-bore closure systems (MANTA, Vivasure), transradial closure bands, and compression-assisted closure devices used at femoral, radial, and large-bore access sites.
Exclusions include manual compression bandages without active closure mechanism, topical hemostatic agents, catheter introducer sheaths, vascular stents and grafts, and surgical vascular repair sutures used in open surgical access.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 1.6 Billion |
| Product Type | Suture-Mediated, Collagen-Based, Sealant (PEG), Clip-Based, Compression-Assisted |
| Access Site | Femoral Arterial, Femoral Venous, Radial, Large-Bore |
| End User | Hospitals, Catheterisation Labs, Ambulatory Surgery Centers |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Key Companies Profiled | Teleflex, Haemonetics, Abbott, Cordis, Terumo, Zylox-Tonbridge |
What is the current global market size for Vascular Closure Devices?
The global market is valued at USD 1.6 Billion in 2026, driven by rising interventional procedure volumes and expansion from arterial to venous and large-bore closure.
What is the projected CAGR for the market over the next 10 years?
The market is projected to grow at a CAGR of 3.4% from 2026 to 2036.
Which regions are experiencing the fastest expansion?
China leads with 8.2% CAGR driven by catheterisation lab expansion, while the USA retains the largest value share through 600,000+ annual PCI procedures and structural heart program growth.
What are the primary market drivers?
Rising interventional procedure volumes, venous and large-bore closure modality expansion, structural heart program growth (TAVR, LAAC), and same-day discharge protocol adoption are the primary catalysts.
Who are the leading suppliers in the industry?
Teleflex (MANTA), Haemonetics (VASCADE, Vivasure), Abbott (Perclose ProGlide), and Cordis (MYNX CONTROL) are key players, differentiating through large-bore and venous closure technology.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.