The percutaneous vessel closure devices market is likely to be valued at USD 4,570.0 million in 2026, is projected to reach USD 10,622.0 million by 2036, expanding at a CAGR of 8.8% during the forecast period. Demand growth reflects sustained increases in catheter-based cardiovascular and endovascular procedures where reliable access-site management is essential to procedural safety and recovery efficiency. Femoral and large-bore access continues to play a central role in interventional cardiology, peripheral interventions, and structural heart procedures, increasing reliance on closure devices that deliver predictable hemostasis without prolonged manual compression. Hospitals and cath labs prioritize solutions that reduce access-site complications, support early ambulation, and stabilize post-procedure workflows under high procedural throughput.
Adoption momentum is reinforced by healthcare system emphasis on recovery optimization, bed utilization efficiency, and nursing workload reduction following interventional procedures. Closure devices enable standardized post-procedure protocols that shorten monitoring time and facilitate same-day discharge pathways in appropriate cases. Clinical preference is shaped by device familiarity, sheath compatibility, and performance consistency across varied patient anatomies and anticoagulation profiles. Ongoing material innovation, including bioabsorbable components and refined deployment mechanisms, supports broader use while addressing concerns related to vessel integrity and long-term outcomes. Demand expansion reflects procedural intensity, workflow efficiency priorities, and complication risk management rather than expansion of interventional infrastructure.
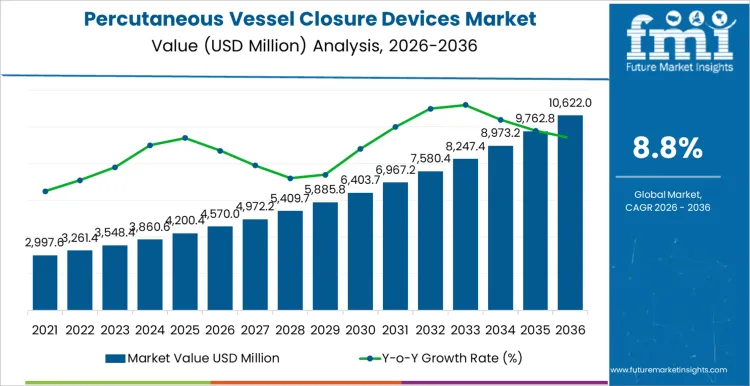
| Metric | Value |
|---|---|
| Market Value (2026) | USD 4,570.0 million |
| Market Forecast Value (2036) | USD 10,622.0 million |
| Forecast CAGR 2026 to 2036 | 8.8% |
Demand for percutaneous vessel closure devices is increasing as interventional cardiology and endovascular procedures expand globally and clinicians seek reliable methods to achieve hemostasis following catheter-based interventions. These devices provide controlled closure of arterial access sites after procedures such as angiography, peripheral interventions, and structural heart repairs, reducing the need for prolonged manual compression and bed rest. Vascular surgeons and interventionalists specify closure systems that deliver consistent sealing, minimize hematoma formation, and support early ambulation, because improved post-procedural recovery contributes to reduced complication rates and shorter hospital stays. Procurement teams in hospitals and ambulatory surgical centers evaluate device profile, ease of deployment, and compatibility with a variety of sheath sizes to ensure seamless integration with existing procedural workflows and team proficiency.
Growth in minimally invasive cardiovascular interventions is reinforcing uptake of percutaneous vessel closure solutions that align with enhanced recovery protocols and value-based care models. As healthcare systems emphasize cost containment without compromising safety, devices that reduce procedure room time, accelerate patient turnover, and lower incidences of access site complications appeal to cath labs and hybrid OR programs. Some hospital groups also evaluate access-site strategy alongside hemorrhage control systems used in emergency and high-acuity cardiovascular care. Training initiatives for cardiology and radiology teams on correct selection and deployment techniques enhance confidence in device performance across diverse patient anatomies. Continued innovation in bioabsorbable materials and sealant technologies supports broader clinical adoption by addressing concerns around long-term vessel integrity and foreign body response. These clinical, operational, and procedural efficiency drivers are contributing to sustained demand growth in the percutaneous vessel closure devices market.
Demand for percutaneous vessel closure devices is influenced by growth in minimally invasive procedures, preference for faster hemostasis, and emphasis on early patient mobilization. Clinical adoption reflects rising catheter-based interventions, increasing use of radial and femoral access, and focus on reducing access-site complications. Where radial programs expand, hospitals also stock transradial closure devices to support faster discharge without increasing nursing workload. Purchasing decisions prioritize closure reliability, ease of deployment, and compatibility with varying sheath sizes. Segmentation explains how device design, procedural context, and care setting determine utilization patterns across interventional practices.

Suture-based closure devices hold 34.0%, reflecting broad adoption in procedures requiring secure and predictable femoral access closure. These devices support precise vessel approximation and are widely used across complex interventional workflows. Collagen plug-based devices at 28.0% offer faster deployment and reduced time to hemostasis, supporting efficiency in routine interventions. Clip-based closure systems represent 18.0%, providing mechanical closure without intravascular suturing. Sealant-based closure devices at 12.0% focus on bioresorbable sealing for selected cases. Hybrid closure technologies at 8.0% combine mechanisms to address varied access challenges. Device-type segmentation highlights preference for proven mechanical reliability balanced with procedural efficiency.
Key Points

Interventional cardiology represents 38.0%, driven by high volumes of coronary angiography and percutaneous coronary interventions. Peripheral vascular interventions at 26.0% rely on closure devices to manage access following diagnostic and therapeutic procedures. Case-mix shifts in peripheral vascular devices utilization can change sheath profiles, which directly affects closure inventory and training needs. Structural heart procedures account for 18.0%, reflecting need for secure closure after larger sheath access. Neurovascular procedures at 10.0% emphasize precise closure to minimize bleeding risk. Large-bore access procedures at 8.0% apply specialized closure solutions following complex interventions. Application-based segmentation shows higher utilization where procedural volume and access-site management are critical to outcomes.
Key Points

Hospitals and cath labs account for 48.0%, reflecting concentration of interventional procedures within acute care settings. Specialty cardiac centers at 20.0% focus on high-throughput cardiovascular interventions requiring standardized closure protocols. Ambulatory surgical centers represent 18.0%, emphasizing rapid recovery and same-day discharge models. Standalone cath labs at 10.0% utilize closure devices to support outpatient diagnostic procedures. Military and emergency centers at 4.0% apply devices in trauma and urgent care scenarios. End-user segmentation highlights adoption where procedural volume, recovery efficiency, and clinical oversight converge.
Key Points
Demand for percutaneous vessel closure devices reflects procedural requirements to achieve rapid and reliable hemostasis following catheter-based interventions. Adoption concentrates in hospitals, cardiac catheterization laboratories, and interventional radiology centers. Global scope aligns with growth in minimally invasive cardiovascular and peripheral procedures. Usage centers on collagen-based, suture-mediated, and clip-based devices designed to close femoral and radial access sites after diagnostic or therapeutic interventions.
Catheter-based interventions require prompt vascular closure to reduce bleeding risk and enable early patient mobilization. Many centers align closure-device selection with their broader hemostasis products formulary to standardize bleeding management across service lines. Demand increases where high procedure volumes create pressure to shorten recovery time and improve lab turnover. Closure devices provide faster hemostasis compared with manual compression, supporting same-day discharge pathways. Interventional cardiology relies on predictable closure performance to minimize access-site complications such as hematoma or pseudoaneurysm formation. Device selection depends on access size, anticoagulation status, and vessel anatomy. For higher-risk bleeders, teams frequently stage backup plans that include pre-filled hemostasis agents when closure performance is uncertain. Operator familiarity influences consistent outcomes across cases. Adoption reflects clinical emphasis on workflow efficiency, patient comfort, and reduction of post-procedural complications in high-acuity interventional settings.
Successful deployment depends on precise technique and familiarity with device-specific steps. Demand sensitivity rises where inadequate training increases failure or complication rates. Device cost exceeds manual compression, affecting use in cost-controlled environments. Anatomical variability and calcified vessels limit suitability in some patient populations. Failure scenarios require fallback strategies, increasing procedural complexity. Inventory management challenges arise due to multiple device types and sizes. Reimbursement structures influence routine use across procedure categories. Sites that treat high-anticoagulation populations often expand hemostasis testing systems to support safer closure decisions and post-procedure monitoring. Scalability remains constrained by operator training requirements, patient selection limits, and economic evaluation relative to procedural risk reduction benefits.
Demand for percutaneous vessel closure devices is expanding globally as minimally invasive cardiovascular and endovascular procedures increase. Faster hemostasis, early ambulation, and reduced complication risk support broader adoption across cath labs. Growth of transcatheter interventions and large-bore access procedures elevates device utilization. Large-bore pathways increasingly reference arterial occlusion devices as contingency tools when access-site bleeding control requires escalation. Reimbursement clarity, operator training, and protocol standardization influence adoption pace. Growth rates in India at 11.3%, Brazil at 10.8%, China at 10.4%, the USA at 7.2%, and Germany at 7.1% indicate sustained expansion driven by procedural volumes, recovery optimization, and clinical efficiency rather than expansion of hospital infrastructure alone.
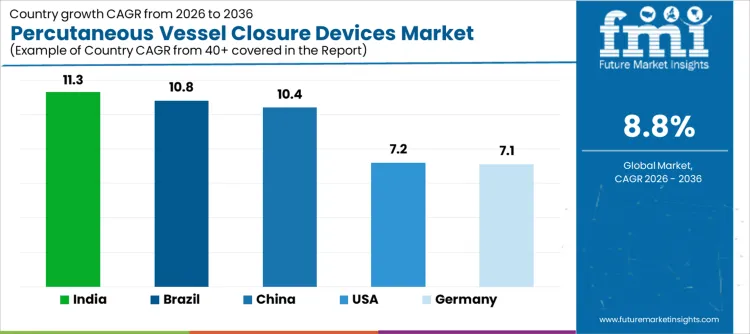
| Country | CAGR (%) |
|---|---|
| India | 11.3% |
| Brazil | 10.8% |
| China | 10.4% |
| USA | 7.2% |
| Germany | 7.1% |
Demand for percutaneous vessel closure devices in India is growing at a CAGR of 11.3%, supported by rising interventional cardiology and peripheral procedures. High femoral access volumes increase need for reliable closure to reduce bed rest and nursing burden. Private cardiac centers prioritize early ambulation to improve throughput and patient turnover. Growing use of complex interventions increases reliance on predictable hemostasis. Expanded operator training improves confidence in device deployment. Growth reflects procedural intensity, workflow efficiency priorities, and private sector investment rather than shifts in access site preference.
Percutaneous vessel closure device demand in Brazil is expanding at a CAGR of 10.8%, driven by modernization of catheterization laboratories. Public and private hospitals seek to reduce post-procedure complications and length of stay. Growth in structural heart and peripheral interventions increases device relevance. Procurement programs support adoption of standardized closure solutions. Regional centers expand interventional capacity beyond major metros. Growth reflects institutional efficiency goals and complication reduction strategies rather than expansion of cath lab counts.
Demand for percutaneous vessel closure devices in China is growing at a CAGR of 10.4%, aligned with high-volume interventional care delivery. Large tertiary hospitals perform extensive femoral access procedures requiring consistent closure outcomes. Adoption supports faster patient mobilization and bed availability. Domestic manufacturers increase affordability and supply continuity. National clinical guidance encourages standardized post-procedure care. Growth reflects scale-driven utilization, standardized protocols, and domestic device availability rather than isolated technology trials.
Percutaneous vessel closure device demand in the United States is expanding at a CAGR of 7.2%, supported by mature interventional practice and reimbursement stability. Country-level pricing pressure and protocol governance can be cross-checked in the United States vascular closure devices outlook to calibrate adoption assumptions. Closure devices are routinely used to improve patient comfort and reduce nursing workload. Increasing large-bore procedures sustain demand across cardiac centers. Clinical evidence supports complication reduction and workflow efficiency. Replacement and technology upgrade cycles contribute to steady purchasing. Growth reflects utilization optimization and evidence-based practice rather than expansion of procedure indications.
Percutaneous vessel closure device demand in Germany is growing at a CAGR of 7.1%, shaped by evidence-based interventional cardiology and procedural governance. Certified centers apply closure devices within defined indications. Emphasis on patient safety and outcome monitoring guides adoption. Statutory reimbursement supports appropriate use without volume inflation. Operator training and protocol adherence ensure consistent deployment. Growth remains steady, reflecting structured utilization and clinical oversight rather than rapid procedural expansion.
Demand for percutaneous vessel closure devices is driven by rising interventional cardiology procedures, shorter recovery targets, and bleeding risk reduction priorities. Interventionalists evaluate closure reliability, time to hemostasis, compatibility with sheath sizes, ease of deployment, and performance across femoral access profiles. Buyer assessment includes learning curve, complication rates, imaging compatibility, and suitability for same day discharge protocols. Procurement behavior reflects cath lab standardization, clinician preference alignment, and outcome driven value analysis. Trend in the percutaneous vessel closure devices market shows increasing use following complex and large bore procedures supporting faster patient mobilization.
Abbott leads competitive positioning through widely adopted closure platforms supported by extensive clinical evidence and broad cath lab penetration. Terumo competes with device designs emphasizing procedural simplicity and consistent hemostasis across access sizes. Cardinal Health supports demand through distribution strength and closure solutions aligned with procedural supply chains. Haemonetics maintains relevance by focusing on vascular management technologies supporting bleeding control and workflow efficiency. Merit Medical participates with closure devices positioned for reliability and cost effectiveness in interventional settings. Competitive differentiation centers on hemostasis speed, safety outcomes, physician familiarity, and integration within cath lab workflows.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Device Types | Suture-based closure devices; Collagen plug-based devices; Clip-based closure systems; Sealant-based closure devices; Hybrid closure technologies |
| Clinical Applications | Interventional cardiology; Peripheral vascular interventions; Structural heart procedures; Neurovascular procedures; Large-bore access procedures |
| End Users | Hospitals & cath labs; Ambulatory surgical centers; Specialty cardiac centers; Standalone cath labs; Military & emergency centers |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, Brazil, China, USA, Germany, and 40+ countries |
| Key Companies Profiled | Abbott; Terumo; Cardinal Health; Haemonetics; Merit Medical; Others |
| Additional Attributes | Dollar sales by device type, clinical application, and end user; closure efficacy, hemostasis time, and complication rates across suture, plug, clip, and sealant technologies; suitability for small and large-bore arterial access; learning curve and workflow integration in cath labs; post-procedure mobility and discharge implications; procurement dynamics influenced by procedure volumes, physician preference, and bundled cath lab purchasing agreements. |
How big is the percutaneous vessel closure devices market in 2026?
The global percutaneous vessel closure devices market is estimated to be valued at USD 4,570.0 million in 2026.
What will be the size of percutaneous vessel closure devices market in 2036?
The market size for the percutaneous vessel closure devices market is projected to reach USD 10,622.0 million by 2036.
How much will be the percutaneous vessel closure devices market growth between 2026 and 2036?
The percutaneous vessel closure devices market is expected to grow at a 8.8% CAGR between 2026 and 2036.
What are the key product types in the percutaneous vessel closure devices market?
The key product types in percutaneous vessel closure devices market are suture-based closure devices, collagen plug-based devices, clip-based closure systems, sealant-based closure devices and hybrid closure technologies.
Which clinical applications segment to contribute significant share in the percutaneous vessel closure devices market in 2026?
In terms of clinical applications, interventional cardiology segment to command 38.0% share in the percutaneous vessel closure devices market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.