Demand for Vascular Closure Devices in Japan
Demand for Vascular Closure Devices in Japan Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Demand for Vascular Closure Devices in Japan Forecast and Outlook 2025 to 2035
Demand for vascular closure devices in Japan is valued at USD 80.5 million in 2025 and is projected to reach USD 103.6 million by 2035, reflecting a CAGR of 2.6%. The historical path from USD 71.0 million in 2020 to USD 80.5 million in 2025 reflects steady procedural volumes in diagnostic and interventional cardiology. Femoral access devices account for the largest product share due to continued use in coronary angiography and complex catheter-based procedures.
Hospitals represent the primary end users as advanced cardiovascular interventions remain centered in large acute care settings. Passive and active devices maintain a consistent demand linked to post-procedure hemostasis control. Early-phase growth reflects procedural stability and regulated adoption within established clinical protocols rather than expansion into new care settings.
Quick Stats of the Demand for Vascular Closure Devices in Japan
- Demand for Vascular Closure Devices in Japan Value (2025): USD 80.5 million
- Demand for Vascular Closure Devices in Japan Forecast Value (2035): USD 103.6 million
- Demand for Vascular Closure Devices in Japan Forecast CAGR (2025 to 2035): 2.6%
- Demand for Vascular Closure Devices in Japan Leading Product: Femoral Access Devices (40%)
- Demand for Vascular Closure Devices in Japan Key Growth Regions: Kyushu & Okinawa, Kanto, Kansai
- Demand for Vascular Closure Devices in Japan Top Players: Terumo Corporation, Abbott Laboratories, Medtronic Plc, Teleflex Incorporated, Johnson and Johnson
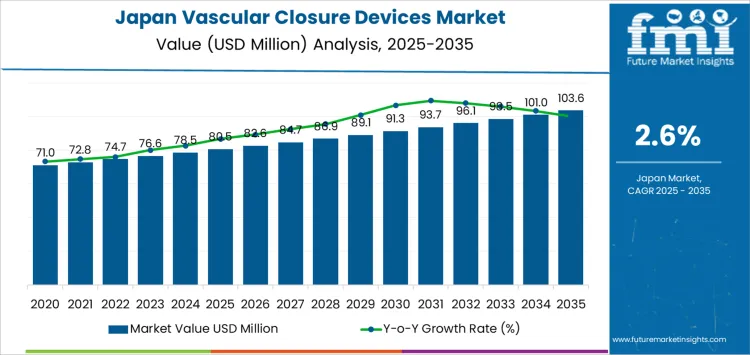
What is the Growth Forecast for Vascular Closure Devices in Japan through 2035?
From 2026, demand increases from USD 82.6 million to USD 93.7 million by 2031, then advances to USD 103.6 million by 2035 through narrow annual gains. Percutaneous coronary intervention remains the largest procedural driver as outpatient catheter volumes remain stable. Endovascular aneurysm repair and transcatheter aortic valve implantation contribute to smaller but consistent device usage. Radial closure devices gain a gradual share of the market as access-site preference continues to shift in selected patient groups.
Independent catheterization laboratories and ambulatory surgical centers show limited but steady participation. After 2030, replacement and procedural volume growth become the primary drivers rather than technology shifts. Annual value movement remains contained across the forecast period.
Between 2025 and 2030, demand for vascular closure devices in Japan increases from USD 80.5 million to USD 91.3 million, reflecting an absolute expansion of USD 10.8 million over five years. Average annual value addition remains slightly above USD 2.0 million during this phase. Growth is shaped by rising catheter-based procedures across cardiology, interventional radiology, and peripheral vascular care. Procedure volume growth in secondary hospitals supports steady device usage. Earlier demand was sustained by manual compression practices. Near term expansion reflects stronger preference for faster hemostasis, lower post procedure immobility time, and consistent adoption in elective diagnostic workflows.
From 2030 to 2035, demand rises from USD 91.3 million to USD 103.6 million, an increase of USD 12.3 million during the later phase of the forecast. Average yearly value addition is moving closer to USD 2.5 million, indicating a slightly faster expansion trajectory than in the earlier period. Growth is supported by higher intervention rates among aging populations, greater use of radial and femoral access management protocols, and wider availability of closure devices across regional hospitals. Earlier growth relied on procedural volume expansion, while later demand reflects standardized post-catheterization care pathways and growing preference for reduced recovery time across institutional treatment settings.
Vascular Closure Devices Industry in Japan Key Takeaways
| Metric | Value |
|---|---|
| Industry Value (2025) | USD 80.5 million |
| Forecast Value (2035) | USD 103.6 million |
| Forecast CAGR (2025 to 2035) | 2.6% |
What Is Driving the Demand for Vascular Closure Devices in Japan with Regard to Medical Trends and Future Needs?
Demand for vascular closure devices (VCDs) in Japan grew as minimally invasive cardiovascular and radiological procedures became more common in hospitals and surgical centers. Increased procedure volumes for catheter-based diagnostics and interventions raised need for reliable arterial puncture closure. VCDs offered faster hemostasis, shorter bed rest, and reduced complication risk compared with manual compression. Hospitals and clinics adopted these devices to streamline workflow, reduce patient discomfort, and improve throughput in interventional cardiology and vascular surgery settings. Growth followed stable patterns of cardiovascular care delivery, periodic equipment upgrades, and preference for disposable or single-use closure devices in high-volume treatment centers.
Future demand is expected to rise as the prevalence of cardiovascular diseases increases alongside aging demographics in Japan. Adoption of newer closure technologies such as advanced plug-based, suture-based, or external hemostatic devices will support broader use across femoral and radial access procedures. Rising preference for outpatient or short-stay interventions makes rapid arterial closure appealing for both providers and patients. Hospitals and ambulatory surgical centers may scale up use of VCDs to manage rising caseloads while controlling stay duration and post-procedure risks. Demand will lean on device safety, ease of use, regulatory compliance, and ability to support varied patient profiles under older-age and comorbidity conditions.
What Is the Demand for Vascular Closure Devices in Japan by Product and Procedure?
Demand for vascular closure devices in Japan is shaped by high catheterization volumes, aging cardiovascular patient base, and strong preference for rapid hemostasis in post procedure care. Femoral access devices lead by product due to continued reliance on femoral routes for complex interventions. Coronary angiograms lead by procedure as diagnostic catheterization remains a routine step across cardiac care pathways. Procurement is driven by tertiary hospitals, cardiovascular centers, and government supported acute care institutions. Import reliance remains high for proprietary closure mechanisms and bioabsorbable materials. Substitution pressure exists from manual compression in low risk cases. Demand stability is supported by rising interventional cardiology workload and structured post procedure care protocols.
Why do femoral access devices account for 40% of the demand for vascular closure devices in Japan by product?
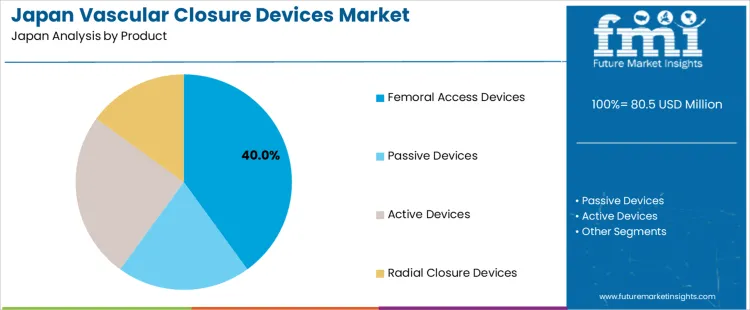
Femoral access devices account for 40% of the demand for vascular closure devices in Japan by product, reflecting their continued use in high complexity and large bore procedures. Consumption intensity is driven by structural heart interventions, peripheral angiography, and emergency catheterization where femoral routes provide stable access. Usage remains stable because many interventional procedures require sheath sizes that exceed radial compatibility.
Procurement is led by cardiac catheterization laboratories and interventional radiology departments. Price sensitivity remains moderate because access site complications increase length of stay and total treatment cost. Specification control emphasizes closure strength, hemostasis time consistency, bioabsorbable material behavior, and ease of deployment under fluoroscopic guidance.
Femoral access devices also generate consistent repeat demand through high daily catheterization throughput in large volume hospitals. Repeat utilization remains predictable as most devices are single use per procedure. Buyers favor platforms that support a wide range of sheath sizes to standardize inventory. Margin structure remains controlled under competitive tendering and bundled procurement with access kits. Regulatory exposure remains elevated due to implant safety monitoring and adverse event reporting. Import reliance persists for collagen plugs, suture-mediated systems, and polymer anchors. Substitution pressure from radial techniques exists in diagnostic cases but does not eliminate femoral demand in advanced interventions.
Why do coronary angiograms represent 30.0% of the demand for vascular closure devices in Japan by procedure?
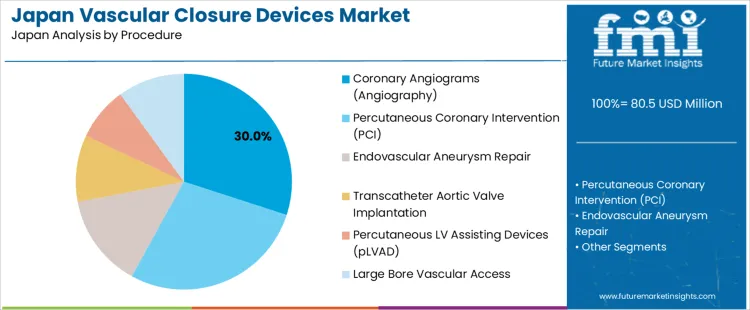
Coronary angiograms represent 30.0% of the demand for vascular closure devices in Japan by procedure, reflecting high diagnostic catheterization volume across cardiac care networks. Consumption intensity is driven by ischemic heart disease screening, pre surgical evaluation, and post intervention surveillance. Usage remains stable because angiography remains the primary diagnostic standard for coronary assessment. Procurement is dominated by cardiovascular centers, public hospitals, and emergency care units. Price sensitivity remains moderate because rapid access site management supports faster patient turnover. Specification control emphasizes immediate sealing performance, reduced rebleeding risk, and compatibility with diagnostic sheath profiles.
Coronary angiogram related closure use also generates steady repeat demand through daily outpatient and inpatient diagnostic schedules. Repeat utilization remains predictable due to routine screening and follow up protocols. Buyers favor closure systems that minimize bed rest time to support same day discharge. Margin structure remains controlled under hospital purchasing frameworks. Regulatory exposure remains elevated due to post-procedure bleeding surveillance and device tracking requirements. Import reliance persists for most proprietary closure platforms. Substitution pressure from compression bands remains present in low-risk radial diagnostics but does not displace femoral closure use in selected angiographic workflows.
What Is Driving the Demand for Vascular Closure Devices in Japan Catheter Laboratory Workflow and Patient Mobility Needs?
Demand for vascular closure devices in Japan is shaped by high catheter procedure volume, rapid sheath removal protocols, and pressure to shorten post procedure bed rest. Cardiology, peripheral intervention, and neuro angiography units use closure devices to control access site bleeding and restore mobility sooner. Elderly patient prevalence increases bleeding management sensitivity after femoral access. Hospitals prioritize early ambulation to free beds and lower nursing workload. National insurance recognizes closure use within defined indications, which stabilizes utilization. Demand grows through cath lab workflow optimization, complication avoidance, and turnover efficiency rather than through elective expansion of interventional case volume alone across
How Is the Shift Toward Complex Structural and Peripheral Interventions Influencing Device Use?
Japan interventional teams increasingly favor radial access for coronary cases, which reshapes closure device mix at femoral sites used for structural heart and complex peripheral work. Large bore access for valve replacement and mechanical support raises reliance on advanced closure systems with suture mediated designs. Procedure planning includes pre close steps that integrate devices before sheath removal. Nursing teams observe lower groin compression time and faster transfer to step down beds. This workflow raises utilization per complex case even as simple diagnostic procedures shift away from femoral routes. Demand aligns with case complexity growth rather than with raw procedure counts
Why Is the Aging Anticoagulated Patient Profile Increasing Dependence on Closure Devices?
Japan aging inpatient profile includes high rates of atrial fibrillation, chronic kidney disease, and dual antiplatelet therapy. These conditions raise access site bleeding risk after catheter interventions. Vascular closure devices support predictable hemostasis under anticoagulated states where manual compression becomes labor intensive. Physicians select device type based on artery size, calcification burden, and coagulation status. Early mobilization lowers back pain and delirium risk among frail patients. Nursing staffing benefit appears through shorter manual hold times and reduced sheath related monitoring intensity. Demand rises through patient safety management and ward workload balance in high acuity cardiovascular units nationwide across regional hospitals
What Institutional and Technical Factors Limit Faster Standardization of Closure Device Use?
Vascular closure device use in Japan faces limits tied to operator training depth, device cost, and anatomical constraints. Complex femoral punctures with heavy calcification narrow device suitability. Learning curves affect outcomes across low volume centers that lack constant repetition. Budget oversight committees review per case consumable spend under fixed reimbursement schedules. Some clinicians retain preference for manual compression based on tactile control and zero inventory reliance. Access site complications still require surgical backup in a small share of cases. These factors moderate universal standardization across all catheter laboratories and keep adoption weighted toward tertiary cardiac and hybrid operating suite environments
What is the Demand for Vascular Closure Devices in Japan by Region?
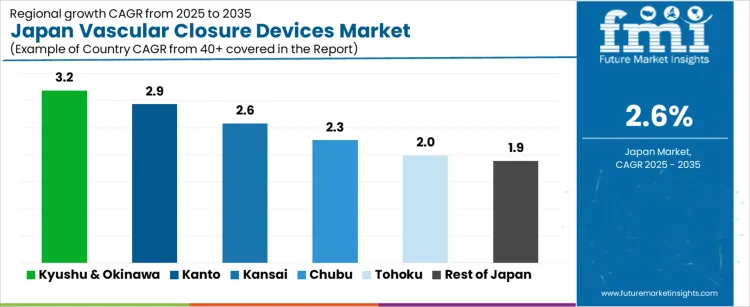
| Region | CAGR (%) |
|---|---|
| Kyushu & Okinawa | 3.2% |
| Kanto | 2.9% |
| Kansai | 2.6% |
| Chubu | 2.3% |
| Tohoku | 2.0% |
| Rest of Japan | 1.9% |
The demand for vascular closure devices in Japan is increasing at a measured pace across interventional cardiology and radiology centers, led by Kyushu and Okinawa at a 3.2% CAGR. Growth in this region is supported by rising catheter based procedures, expanding peripheral intervention programs, and steady modernization of cath lab infrastructure in regional hospitals. Kanto follows at 2.9%, driven by dense concentrations of tertiary hospitals, high procedure volumes in coronary and structural heart care, and consistent replacement demand.
Kansai records 2.6% growth, reflecting stable use across university hospitals and private cardiovascular centers. Chubu at 2.3% shows moderate uptake linked to regional interventional programs. Tohoku and the Rest of Japan, at 2.0% and 1.9%, reflect slower growth shaped by lower procedure density and longer device replacement cycles.
How Is Catheter Based Procedure Volume Supporting Vascular Closure Device Demand in Kyushu and Okinawa?
Demand for vascular closure devices in Kyushu and Okinawa is advancing at a CAGR of 3.2% through 2035, supported by steady growth in catheter guided cardiac procedures and rising use of minimally invasive diagnostic interventions. Regional hospitals rely on closure devices to reduce post procedure immobility and improve patient turnover efficiency. Emergency departments also apply these devices for trauma related vascular access management. Growth reflects increasing use of angiography, expanding cardiology service capacity, and gradual replacement of manual compression practices with device based hemostasis solutions.
- Cardiac catheter labs sustain procedural demand
- Emergency access management supports usage breadth
- Faster patient mobilization improves care workflow
- Manual compression shifts toward device based closure
Why Is Large Hospital Density Sustaining Vascular Closure Device Growth in Kanto?
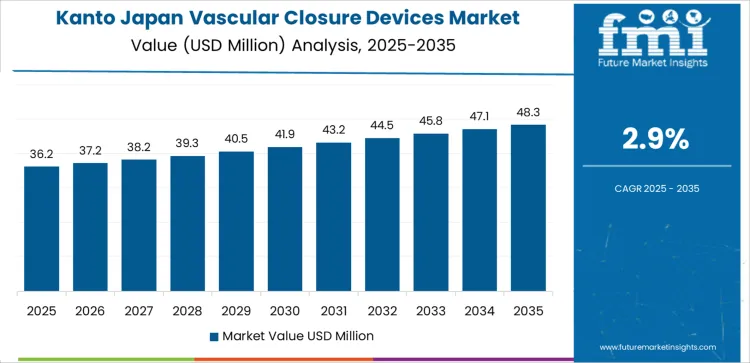
Demand for vascular closure devices in Kanto is rising at a CAGR of 2.9% through 2035, driven by high concentration of tertiary hospitals, large scale imaging centers, and steady growth of interventional cardiology procedures. Kanto records strong adoption across femoral access coronary interventions and peripheral angioplasty workflows. Hospitals prioritize rapid hemostasis to reduce bed occupancy pressure. Growth reflects continuous patient inflow, strong reimbursement coverage for interventional procedures, and rising preference for early ambulation protocols in high volume clinical settings.
- Tertiary centers anchor high procedural throughput
- Early ambulation protocols support clinical adoption
- Peripheral angioplasty drives femoral access usage
- Bed occupancy management shapes procurement priorities
What Is Supporting Measured Vascular Closure Device Adoption Across Kansai?
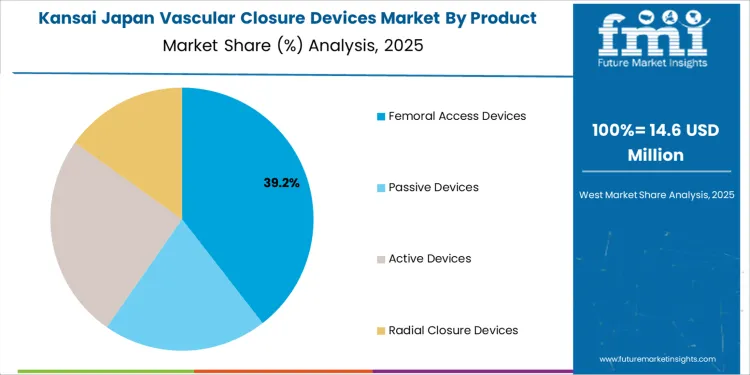
Demand for vascular closure devices in Kansai is progressing at a CAGR of 2.6% through 2035, supported by balanced growth in diagnostic catheterization labs and controlled expansion of structural heart programs. Kansai hospitals apply closure devices mainly for routine angiography and selected intervention cases. Adoption remains structured with clear protocol alignment. Growth reflects moderate cardiology case volume growth, stable hospital budgets, and gradual increase in physician familiarity with device assisted arterial closure techniques.
- Diagnostic angiography anchors baseline device usage
- Structured procedural protocols guide utilization rates
- Physician training improves technical usage confidence
- Moderate case volume shapes controlled demand growth
How Is Regional Hospital Modernization Shaping Vascular Closure Device Demand in Chubu?
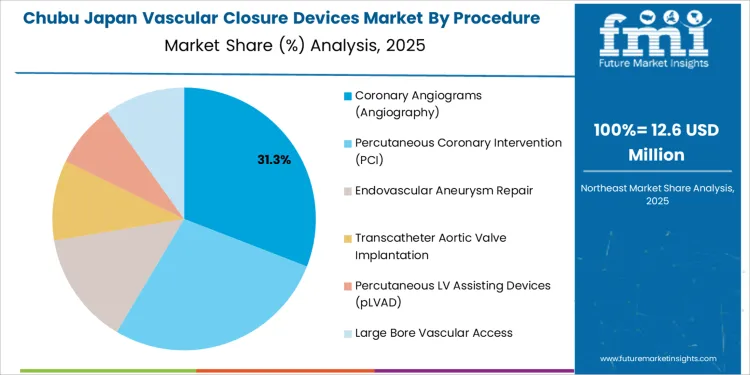
Demand for vascular closure devices in Chubu is advancing at a CAGR of 2.3% through 2035, supported by regional hospital upgrades, steady interventional radiology procedures, and controlled adoption of minimally invasive vascular access management. Chubu hospitals apply these devices mainly in elective procedures and post diagnostic imaging interventions. Growth reflects phased modernization of cath labs, gradual shift toward shorter recovery pathways, and consistent demand from orthopedic and vascular surgery access management requirements.
- Elective procedures guide predictable device demand
- Cath lab upgrades support long term usage expansion
- Recovery time reduction supports adoption decisions
- Vascular surgery access adds secondary application base
Why Is Healthcare Infrastructure Scale Influencing Vascular Closure Device Growth in Tohoku?
Demand for vascular closure devices in Tohoku is advancing at a CAGR of 2.0% through 2035, supported by public hospital interventions, limited but stable catheter procedure volume, and gradual modernization of cardiovascular care services. Tohoku hospitals rely on selective use for high risk patients where manual compression presents mobility limitations. Growth reflects public healthcare funding control, steady outpatient interventional procedures, and cautious adoption of premium closure systems across regional medical centers.
- Public hospitals guide baseline procedural demand
- High risk patient management supports selective usage
- Outpatient intervention growth supports device application
- Public funding control shapes procurement pacing
How Is Small City Interventional Care Shaping Vascular Closure Device Demand in Rest of Japan?
Demand for vascular closure devices in Rest of Japan is advancing at a CAGR of 1.9% through 2035, supported by community hospital catheter access procedures, steady trauma related vascular interventions, and limited but stable angiographic imaging services. These areas rely primarily on basic closure systems rather than premium automated platforms. Growth remains stable and guided by small facility budgets, consistent emergency procedure volume, and gradual awareness of post procedure mobility benefits among regional clinical teams.
- Community hospitals guide essential usage volume
- Trauma access procedures support routine demand
- Basic closure systems dominate device selection
- Post procedure mobility benefits drive gradual adoption
What Is Driving the Demand for Vascular Closure Devices in Japan and Which Companies Shape Hospital Adoption
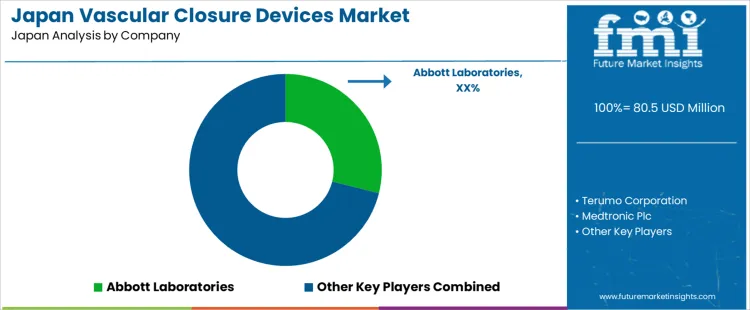
The demand for vascular closure devices in Japan is shaped by high catheterization procedure volumes, growth in transcatheter cardiac interventions, and strong preference for early patient mobilization after angiographic procedures. Terumo Corporation holds a central domestic position through femoral and radial access closure systems used widely across cardiovascular centers and regional hospitals.
Abbott Laboratories supports demand through arterial closure platforms adopted in interventional cardiology and peripheral vascular procedures. Medtronic Plc participates through integrated access management solutions used in structural heart and electrophysiology labs. These suppliers anchor routine clinical use through deep relationships with teaching hospitals, cath lab operators, and purchasing groups tied to prefectural medical networks.
Teleflex Incorporated supports demand through vascular access and closure systems used in diagnostic and interventional radiology settings. Johnson and Johnson participates through selected access management technologies supplied into tertiary care hospitals performing complex endovascular procedures. System selection in Japan is governed by hemostasis reliability, reduction of post procedure complications, ease of deployment in elderly patients, and compatibility with anticoagulation protocols.
Buyer preference favors suppliers with stable domestic distribution, Japanese language training support, and clear procedural documentation. Demand visibility tracks expansion of structural heart programs, growth in catheter based stroke intervention, and steady increase in minimally invasive vascular procedures across aging urban populations and regional medical hubs.
Key Players in the Vascular Closure Devices Industry in Japan
- Emergex Vaccines
- Indian Immunologicals Ltd.
- GlaxosmithKline
- Merck
- Codagenix
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2025) | USD million |
| Type | Femoral Access Devices, Passive Devices, Active Devices, Radial Closure Devices |
| Application | Coronary Angiograms (Angiography), Percutaneous Coronary Intervention (PCI), Endovascular Aneurysm Repair, Transcatheter Aortic Valve Implantation, Percutaneous LV Assisting Devices (pLVAD), Large Bore Vascular Access |
| End User | Hospitals, Independent Catheterization Labs, Ambulatory Surgical Centers |
| Regions Covered | Kyushu and Okinawa, Kanto, Kansai, Chubu, Tohoku, Rest of Japan |
| Countries Covered | Japan |
| Key Companies Profiled | Terumo Corporation, Abbott Laboratories, Medtronic Plc, Teleflex Incorporated, Johnson and Johnson |
| Additional Attributes | Dollar sales by product and procedure categories, procedure driven replacement demand under high catheterization volumes, adoption linked to early ambulation and reduced post procedure immobility, reimbursement aligned utilization in femoral and radial access management, import reliance for bioabsorbable plugs and suture mediated systems, regulatory tracking for adverse events and implant safety under national cardiovascular care standards |
Vascular Closure Devices Industry in Japan Segmentation
Product:
- Femoral Access Devices
- Passive Devices
- Active Devices
- Radial Closure Devices
Procedure:
- Coronary Angiograms (Angiography)
- Percutaneous Coronary Intervention (PCI)
- Endovascular Aneurysm Repair
- Transcatheter Aortic Valve Implantation
- Percutaneous LV Assisting Devices (pLVAD)
- Large Bore Vascular Access
End User:
- Hospitals
- Independent Catheterization Labs
- Ambulatory Surgical Centers
Region
- Kyushu & Okinawa
- Kanto
- Kansai
- Chubu
- Tohoku
- Rest of Japan
Frequently Asked Questions
How big is the demand for vascular closure devices in Japan in 2025?
The demand for vascular closure devices in Japan is estimated to be valued at USD 80.5 million in 2025.
What will be the size of vascular closure devices in Japan in 2035?
The market size for the vascular closure devices in Japan is projected to reach USD 103.6 million by 2035.
How much will be the demand for vascular closure devices in Japan growth between 2025 and 2035?
The demand for vascular closure devices in Japan is expected to grow at a 2.6% CAGR between 2025 and 2035.
What are the key product types in the vascular closure devices in Japan?
The key product types in vascular closure devices in Japan are femoral access devices, passive devices, active devices and radial closure devices.
Which procedure segment is expected to contribute significant share in the vascular closure devices in Japan in 2025?
In terms of procedure, coronary angiograms (angiography) segment is expected to command 30.0% share in the vascular closure devices in Japan in 2025.
Table of Content
- Executive Summary
- Japan Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Japan Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Japan Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Japan Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Product
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Product , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Product , 2025 to 2035
- Femoral Access Devices
- Passive Devices
- Active Devices
- Radial Closure Devices
- Y to o to Y Growth Trend Analysis By Product , 2020 to 2024
- Absolute $ Opportunity Analysis By Product , 2025 to 2035
- Japan Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Procedure
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Procedure, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Procedure, 2025 to 2035
- Coronary Angiograms (Angiography)
- Percutaneous Coronary Intervention (PCI)
- Endovascular Aneurysm Repair
- Transcatheter Aortic Valve Implantation
- Percutaneous LV Assisting Devices (pLVAD)
- Large Bore Vascular Access
- Y to o to Y Growth Trend Analysis By Procedure, 2020 to 2024
- Absolute $ Opportunity Analysis By Procedure, 2025 to 2035
- Japan Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2025 to 2035
- Hospitals
- Independent Catheterization Labs
- Ambulatory Surgical Centers
- Y to o to Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Product
- By Procedure
- By End User
- Competition Analysis
- Competition Deep Dive
- Abbott Laboratories
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Terumo Corporation
- Medtronic Plc
- Teleflex Incorporated
- Johnson & Johnson
- Abbott Laboratories
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Japan Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: Japan Market Value (USD Million) Forecast by Product , 2020 to 2035
- Table 3: Japan Market Value (USD Million) Forecast by Procedure, 2020 to 2035
- Table 4: Japan Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 5: Japan Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 6: Japan Market Value (USD Million) Forecast by Product , 2020 to 2035
- Table 7: Japan Market Value (USD Million) Forecast by Procedure, 2020 to 2035
- Table 8: Japan Market Value (USD Million) Forecast by End User, 2020 to 2035
List of Figures
- Figure 1: Japan Market Pricing Analysis
- Figure 2: Japan Market Value (USD Million) Forecast 2020-2035
- Figure 3: Japan Market Value Share and BPS Analysis by Product , 2025 and 2035
- Figure 4: Japan Market Y-o-Y Growth Comparison by Product , 2025-2035
- Figure 5: Japan Market Attractiveness Analysis by Product
- Figure 6: Japan Market Value Share and BPS Analysis by Procedure, 2025 and 2035
- Figure 7: Japan Market Y-o-Y Growth Comparison by Procedure, 2025-2035
- Figure 8: Japan Market Attractiveness Analysis by Procedure
- Figure 9: Japan Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 10: Japan Market Y-o-Y Growth Comparison by End User, 2025-2035
- Figure 11: Japan Market Attractiveness Analysis by End User
- Figure 12: Japan Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 13: Japan Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 14: Japan Market Attractiveness Analysis by Region
- Figure 15: Japan Market Incremental Dollar Opportunity, 2025-2035
- Figure 16: Japan Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: Japan Market Value Share and BPS Analysis by Product , 2025 and 2035
- Figure 18: Japan Market Y-o-Y Growth Comparison by Product , 2025-2035
- Figure 19: Japan Market Attractiveness Analysis by Product
- Figure 20: Japan Market Value Share and BPS Analysis by Procedure, 2025 and 2035
- Figure 21: Japan Market Y-o-Y Growth Comparison by Procedure, 2025-2035
- Figure 22: Japan Market Attractiveness Analysis by Procedure
- Figure 23: Japan Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 24: Japan Market Y-o-Y Growth Comparison by End User, 2025-2035
- Figure 25: Japan Market Attractiveness Analysis by End User
- Figure 26: Japan Market - Tier Structure Analysis
- Figure 27: Japan Market - Company Share Analysis

