Demand for MRSA (Methicillin-resistant Staphylococcus aureus) testing in the USA is expected to grow from USD 1.6 billion in 2025 to USD 3.2 billion by 2035, reflecting a compound annual growth rate (CAGR) of 7%. This growth is driven by the increasing prevalence of MRSA infections, particularly in healthcare settings, and the growing need for rapid, accurate diagnostics to manage and control these infections. MRSA is a major cause of hospital-acquired infections (HAIs), and the demand for testing is expected to rise as healthcare providers focus on improving infection control and patient outcomes.
The market is also supported by advancements in diagnostic technologies, such as molecular and PCR-based tests, which offer faster results with greater accuracy. As the awareness of MRSA’s impact on patient health increases and as regulatory requirements for infection prevention become stricter, the demand for MRSA testing is expected to grow, particularly in hospitals, outpatient centers, and long-term care facilities. Additionally, the increasing focus on antimicrobial resistance (AMR) management will likely further support the expansion of MRSA testing over the next decade.
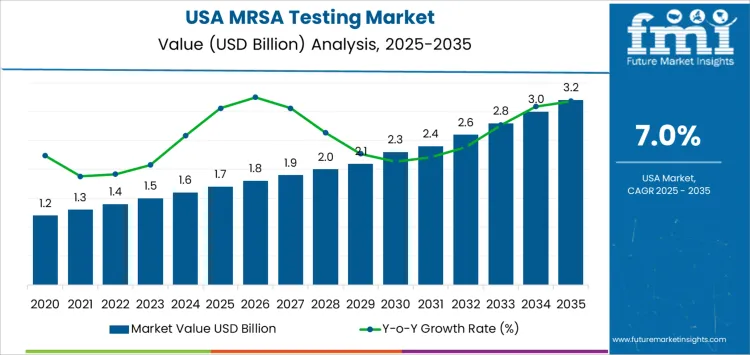
The rolling compound annual growth rate (CAGR) analysis for MRSA testing in the USA shows consistent growth, with the market value rising from USD 1.6 billion in 2025 to USD 1.7 billion in 2026. This initial phase reflects steady demand for MRSA testing driven by ongoing concerns about hospital-acquired infections and the need for effective diagnostics. The CAGR remains stable in the early years, with the market reaching USD 1.8 billion in 2027 and USD 1.9 billion in 2028.
As the market matures, growth begins to accelerate, with the market reaching USD 2.0 billion in 2029, USD 2.1 billion in 2030, and USD 2.3 billion in 2031. This accelerated growth is driven by increasing healthcare awareness and more widespread adoption of rapid MRSA testing technologies, such as PCR and molecular diagnostics, which are contributing to quicker diagnosis and treatment. By 2035, the market is expected to reach USD 3.2 billion, indicating a strong growth trajectory throughout the decade. The rolling CAGR analysis highlights that while growth remains steady over the long term, the adoption of advanced testing technologies and heightened awareness of infection control will continue to fuel the demand for MRSA testing.
| Metric | Value |
|---|---|
| Industry Sales Value (2025) | USD 1.6 billion |
| Industry Forecast Value (2035) | USD 3.2 billion |
| Industry Forecast CAGR (2025-2035) | 7% |
Demand for MRSA (Methicillin Resistant Staphylococcus aureus) testing in the USA continues to grow because MRSA remains a significant public health challenge both in hospitals and in the community. MRSA infections are among the leading causes of hospital acquired infections, contributing to high rates of morbidity, mortality, prolonged hospital stays, and increased healthcare costs. Many individuals carry MRSA asymptomatically, often on the skin or in the nasal passages, without knowing. These carriers can unwittingly transmit the bacterium to vulnerable patients or through community interactions. Because of this dual threat-active infection and silent carriage-routine testing of patients, especially those admitted to hospitals or undergoing surgery, is seen as crucial to prevent outbreaks, guide effective treatment, and protect public health.
At the same time, evolving antimicrobial resistance and limited treatment options make accurate and timely MRSA diagnostics more important than ever. As antibiotic resistance grows, treatment regimens must be tailored based on susceptibility results to avoid ineffective therapies and reduce selection pressure for further resistance. The growing incidence of both hospital associated MRSA and community associated MRSA means that healthcare providers increasingly rely on MRSA testing-including rapid molecular methods and screening protocols-to identify colonization vs active infection, decide isolation or decolonization strategies, and manage antibiotic use responsibly. Because of disease burden, public health risk, and the critical need for informed treatment decisions, demand for MRSA testing is expected to remain high and grow in coming years across hospitals, clinics, and community health settings.
Overview The MRSA (Methicillin-resistant Staphylococcus aureus) testing market in the USA is segmented by drug class and indication. Glycopeptides are the leading drug class, accounting for 16% of the market share, while skin and soft tissue infections (SSTIs) are the dominant indication, representing 50% of the market. The rising incidence of MRSA infections, coupled with the increasing demand for rapid, accurate diagnostic methods, is driving the growth of the MRSA testing market in the USA.
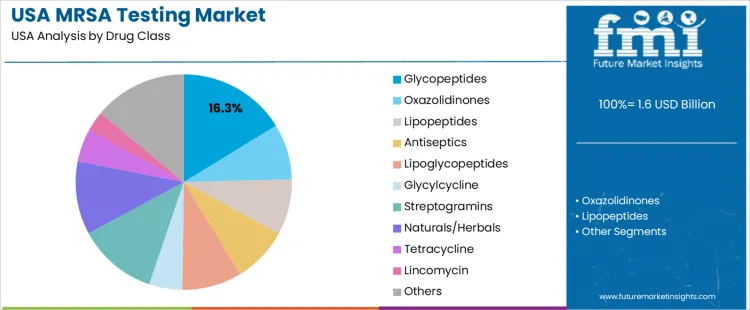
Glycopeptides dominate the drug class segment for MRSA testing in the USA, holding 16% of the market share. This class, which includes antibiotics such as vancomycin, is commonly used in the treatment of MRSA infections. Glycopeptides are particularly effective against Gram-positive bacterial infections like MRSA, making them the first line of defense in treating resistant strains. The growing prevalence of MRSA, especially in healthcare-associated infections, has led to an increased reliance on glycopeptides, driving demand for MRSA testing to guide appropriate treatment. While other drug classes such as oxazolidinones, lipopeptides, and antiseptics are also used, glycopeptides remain the most prescribed and effective treatment for MRSA, contributing significantly to the market.
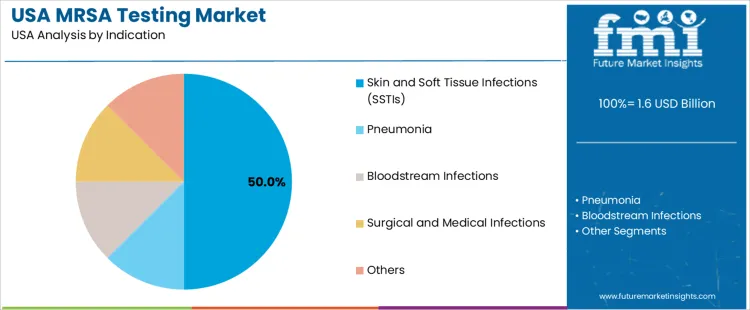
Skin and soft tissue infections (SSTIs) lead the indication segment for MRSA testing, representing 50% of the market share. SSTIs are one of the most common manifestations of MRSA infection, and early detection is crucial for effective treatment. MRSA testing helps clinicians determine whether an infection is caused by resistant strains, enabling them to prescribe the most appropriate antibiotics and reduce the risk of complications. The high incidence of SSTIs, particularly in hospitalized or immunocompromised patients, has driven the demand for MRSA testing. Pneumonia, bloodstream infections, and surgical and medical infections also contribute to the market, as MRSA can cause severe and life-threatening infections in these areas. The demand for MRSA testing in these indications is expected to grow, driven by the rising prevalence of resistant infections and the need for rapid diagnostic solutions in clinical settings.
The demand for MRSA (methicillin-resistant Staphylococcus aureus) testing in the USA is rising as the prevalence of antibiotic-resistant infections continues to be a major public health concern. MRSA is a common cause of healthcare-associated infections, and its resistance to many antibiotics makes it a critical challenge in hospitals, nursing homes, and other healthcare settings. As healthcare providers aim to quickly diagnose and treat MRSA infections, the need for accurate and rapid MRSA testing is increasing. Advances in diagnostic technologies, along with growing awareness of antibiotic resistance, are driving the adoption of MRSA testing in both clinical and outpatient settings.
What are the Drivers of Demand for MRSA Testing in USA?
Several factors are driving the demand for MRSA testing in the USA. First, the increasing incidence of MRSA infections, especially in healthcare settings where patients are more vulnerable, is creating a need for early detection and prompt treatment. Second, growing concerns about antibiotic resistance and the potential for MRSA to spread in hospitals and other care facilities are encouraging routine MRSA screening as part of infection control protocols. Third, advancements in diagnostic technologies, including rapid molecular testing, are enabling quicker results and making MRSA testing more accessible and efficient. Finally, the rising focus on antimicrobial stewardship programs and infection prevention measures is contributing to the demand for MRSA testing, as these programs rely on accurate data to guide treatment decisions and minimize the spread of resistant infections.
What are the Restraints on Demand for MRSA Testing in USA?
Despite the increasing demand, several challenges may limit the adoption of MRSA testing in the USA. One key restraint is the cost of testing, particularly for newer, rapid diagnostic technologies, which may not be affordable for all healthcare providers, especially smaller clinics or outpatient settings. Another challenge is the lack of widespread awareness or routine screening for MRSA among certain patient populations, which can lead to delayed diagnoses and treatment. Additionally, the complexity of some MRSA testing methods, such as culture-based tests, can result in longer turnaround times, limiting their effectiveness in urgent situations. Finally, resistance to implementing routine screening programs, particularly in low-risk or outpatient settings, may slow the adoption of MRSA testing in some regions.
What are the Key Trends Influencing Demand for MRSA Testing in USA?
Several key trends are shaping the demand for MRSA testing in the USA. One significant trend is the shift toward faster, more accurate molecular testing methods, which allow for quicker detection of MRSA infections, reducing treatment delays and improving patient outcomes. Another trend is the growing integration of MRSA testing into infection control protocols, particularly in hospitals and long-term care facilities, as part of broader efforts to reduce healthcare-associated infections. The increasing focus on antimicrobial resistance and the need for more effective stewardship programs is also driving demand for MRSA testing, as accurate data is crucial for guiding treatment decisions and limiting the spread of resistant infections. Additionally, there is a growing trend toward more widespread screening in outpatient settings, especially among high-risk groups, to prevent the transmission of MRSA in the community.
The MRSA (Methicillin-resistant Staphylococcus aureus) Testing market in the USA is expected to see steady growth, with the West USA leading the way at a projected CAGR of 4.6%. South USA follows closely with a growth rate of 4.1%, while the Northeast USA is expected to grow at 3.7%. The Midwest USA shows more moderate growth with a CAGR of 3.2%. MRSA is a significant healthcare concern due to its resistance to standard antibiotics, and the demand for timely and accurate testing is rising. As hospitals, clinics, and public health organizations continue to combat MRSA infections, the market for MRSA testing solutions is expanding across the country.
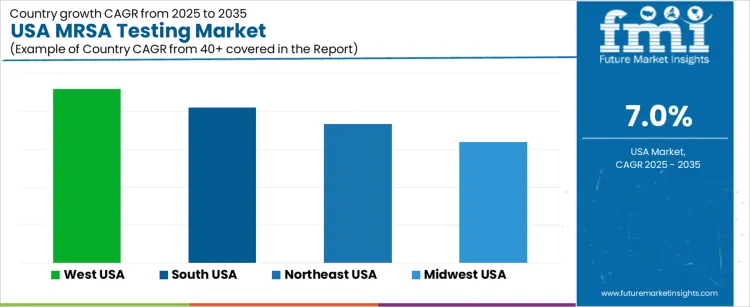
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 4.6% |
| South USA | 4.1% |
| Northeast USA | 3.7% |
| Midwest USA | 3.2% |
The West USA is projected to experience the highest growth in the MRSA Testing market, with a projected CAGR of 4.6%. The region's large and diverse healthcare infrastructure, combined with its focus on innovative medical solutions, is driving the demand for MRSA testing. States like California, Washington, and Oregon are leading the way in adopting advanced diagnostic technologies to detect MRSA infections early. The West USA has a significant number of hospitals and healthcare providers that are increasingly using MRSA testing to manage infection risks and improve patient outcomes. As the prevalence of MRSA continues to rise, the West's healthcare systems are prioritizing rapid diagnostic solutions to reduce the spread of infections. Additionally, public health initiatives aimed at controlling healthcare-associated infections (HAIs) are contributing to the growing demand for MRSA testing in the region.
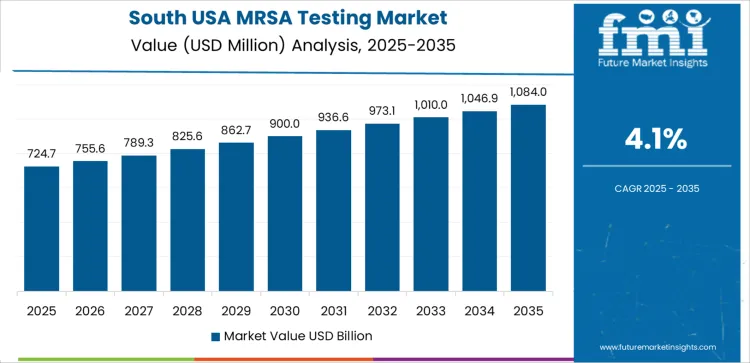
In South USA, the demand for MRSA testing is projected to grow at a CAGR of 4.1%. The region's increasing focus on improving healthcare outcomes and preventing the spread of drug-resistant infections is contributing to the adoption of MRSA testing. States like Texas, Florida, and Georgia have large populations and a high number of hospital admissions, making the early detection of MRSA infections a priority. The growing awareness of the risks associated with MRSA, particularly in healthcare settings, is driving healthcare providers in the South to adopt better diagnostic tools. With increasing healthcare costs and the need for more efficient infection control practices, MRSA testing is seen as a valuable tool in reducing healthcare-associated infections and improving patient care. The growing support for infection control programs and antimicrobial stewardship in the South USA is also supporting the market's growth.
In Northeast USA, the MRSA Testing market is projected to grow at a CAGR of 3.7%. The region's dense population and well-established healthcare systems are driving the demand for timely and accurate MRSA testing. Cities like New York, Boston, and Philadelphia are at the forefront of combating healthcare-associated infections, with many healthcare facilities adopting MRSA testing as part of their infection control programs. As MRSA continues to be a significant concern in hospitals and long-term care facilities, healthcare providers in the Northeast are increasingly turning to advanced diagnostic tools for early detection and treatment. The growing awareness of antibiotic resistance and the need for effective infection control strategies are further contributing to the rise in MRSA testing. The region's emphasis on patient safety and healthcare quality is expected to continue driving the demand for MRSA testing solutions.
In Midwest USA, the MRSA Testing market is expected to grow at a CAGR of 3.2%. While the growth rate in the Midwest is more moderate compared to other regions, the increasing awareness of MRSA infections and the need for better diagnostic solutions are driving the market forward. States like Illinois, Michigan, and Ohio are seeing rising healthcare demands, and hospitals and healthcare providers are adopting MRSA testing to prevent the spread of infections. The Midwest's healthcare systems are placing greater emphasis on infection prevention and control, which is contributing to the demand for MRSA testing. As the region’s hospitals and clinics continue to invest in advanced diagnostic technologies, the adoption of MRSA testing is expected to rise gradually. Public health initiatives focused on reducing healthcare-associated infections are also playing a role in driving market growth in the Midwest.
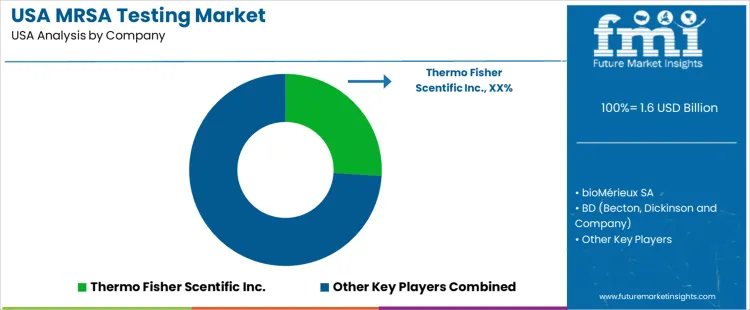
Demand for testing for Methicillin Resistant Staphylococcus aureus (MRSA) remains high in the USA due to the persistent prevalence of MRSA infections in hospital and community settings, rising concern over antibiotic resistant bacteria, and growing emphasis on infection control and antimicrobial stewardship. The frequency of invasive MRSA infections, combined with awareness among clinicians that timely identification is essential for effective treatment and prevention of outbreaks, fuels demand for accurate, rapid diagnostics. The increasing use of molecular and point of care testing methods has made MRSA detection more accessible and practical for hospitals, clinics, long term care facilities, and public health labs. This shift supports broader adoption of MRSA testing as part of standard screening protocols for at risk patients and infection control programs.
On the supply side, several major diagnostics and life science companies dominate the MRSA testing market. Leading providers include Thermo Fisher Scientific, bioMérieux SA, Becton, Dickinson and Company (BD), Abbott Laboratories, and Cepheid. Thermo Fisher is identified as a market leader with around 26% share among the listed firms. These companies supply comprehensive MRSA testing solutions, including molecular based PCR assays, immunodiagnostic kits, instruments, reagents, and workflow software, that support both routine screening and outbreak containment in medical and laboratory settings.
Competition among these firms is shaped by factors such as speed and accuracy of test results, breadth of pathogen and antibiotic resistance detection, ease of automation, regulatory clearance, and integration with hospital lab workflows. Suppliers that deliver rapid, reliable, and easy to use MRSA tests, especially molecular or point of care formats, combined with strong customer support and supply chain stability, are best positioned to capture growth as MRSA concerns remain a major public health priority in the USA.
| Items | Details |
|---|---|
| Quantitative Units | USD Billion |
| Regions Covered | USA |
| Drug Class | Glycopeptides, Oxazolidinones, Lipopeptides, Antiseptics, Lipoglycopeptides, Glycylcycline, Streptogramins, Naturals/Herbals, Tetracycline, Lincomycin, Others |
| Indication | Skin and Soft Tissue Infections (SSTIs), Pneumonia, Bloodstream Infections, Surgical and Medical Infections, Others |
| Route of Administration | Intravenous, Oral, Topical, Nasal, Cutaneous, Intramuscular |
| Type of MRSA | Healthcare Acquired MRSA, Community Acquired MRSA, Hospital Acquired MRSA |
| Distribution Channel | Hospital Pharmacies, Retail Pharmacies, Drug Stores, Online Pharmacies |
| Key Companies Profiled | Thermo Fisher Scientific Inc., bioMérieux SA, BD (Becton, Dickinson and Company), Abbott Laboratories, Cepheid (a Danaher Company) |
| Additional Attributes | Dollar sales by indication and distribution channel show significant growth in MRSA testing, especially for skin and soft tissue infections and bloodstream infections. The shift towards molecular diagnostics (including PCR) is expected to drive demand, with hospitals being the primary end-users. Growing antibiotic resistance concerns contribute to increased testing in both hospital and outpatient settings. |
How big is the demand for MRSA testing in USA in 2025?
The demand for MRSA testing in USA is estimated to be valued at USD 1.6 billion in 2025.
What will be the size of MRSA testing in USA in 2035?
The market size for the MRSA testing in USA is projected to reach USD 3.2 billion by 2035.
How much will be the demand for MRSA testing in USA growth between 2025 and 2035?
The demand for MRSA testing in USA is expected to grow at a 7.0% CAGR between 2025 and 2035.
What are the key product types in the MRSA testing in USA?
The key product types in MRSA testing in USA are glycopeptides, oxazolidinones, lipopeptides, antiseptics, lipoglycopeptides, glycylcycline, streptogramins, naturals/herbals, tetracycline, lincomycin and others.
Which indication segment is expected to contribute significant share in the MRSA testing in USA in 2025?
In terms of indication, skin and soft tissue infections (sstis) segment is expected to command 50.0% share in the MRSA testing in USA in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.