The pharmaceutical ointments base market is projected to grow from USD 4.7 billion in 2026 to USD 6.9 billion by 2036. Driven by the needs of dermatology and wound care, the demand is poised to rise at a compound annual growth rate (CAGR) of 3.90%. The sector primarily functions as a platform for excipients rather than as a finished dosage form. Procurement teams evaluate suppliers based on consistency of batches, control of impurities, and the thoroughness of documentation. Furthermore, the ease of filling and the required cleaning processes significantly affect production scheduling.
The dynamics of the market are shaped more by product lifecycle management and portfolio considerations than by launch windows. Manufacturing plants organize operations such as melting, blending, and cooling based on temperature and viscosity parameters, adhering to specific homogenization constraints. Product release is contingent upon visual appearance, microbial limits, and rheological assessments.

Formulary stability and manufacturing practice determine purchasing behavior in the pharmaceutical ointments base market more than shifts in finished drug branding. In 2026, at about USD 4.7 billion, demand is anchored in dermatology, wound care, and topical anti-inflammatory products where base selection is fixed during formulation approval. Once a base system is validated for viscosity, release profile, and stability, it stays in use across long product lifecycles. Consumption follows production volumes of established SKUs and line extensions that keep the same excipient framework. Expansion comes from incremental additions to topical portfolios and wider geographic production rather than frequent reformulation. The upward movement reflects steady growth in topical therapies and contract manufacturing output rather than changes in prescribing patterns alone.
Quality control discipline and supply continuity shape the later phase of the pharmaceutical ointments base market. As value approaches roughly USD 6.9 billion by 2036, buyers place weight on batch consistency, impurity profiles, and documentation readiness for inspections. Production planning favors long campaigns of standardized grades that simplify validation and inventory handling. Manufacturers invest in process control and raw material traceability to protect release schedules. The move from the mid four billion into the high six billion reflects accumulated volume growth across many finished products rather than a single therapeutic shift. Suppliers that maintain reliable quality, predictable delivery, and technical support for audits tend to secure long term positions in approved excipient lists.
| Metric | Value |
|---|---|
| Market Value (2026) | USD 4.7 billion |
| Forecast Value (2036) | USD 6.9 billion |
| Forecast CAGR 2026 to 2036 | 3.9% |
Pharmaceutical ointment bases provide the carrier matrix for active ingredients in dermatology, wound care, and ophthalmic products. Formulators choose between hydrocarbon, absorption, water removable, and water soluble systems based on drug solubility, occlusiveness, and release profile. Earlier production lines accepted wide variability in texture and spread, which complicated filling accuracy and dose uniformity. Current programs specify viscosity range, particle tolerance, and microbial limits because base behavior affects mixing, deaeration, and tube crimping. Buyers evaluate oxidative stability, batch to batch consistency, and interaction with actives rather than cosmetic feel alone. Use concentrates in chronic therapies and hospital products where predictable performance matters. Supply planning depends on excipient qualification, packaging compatibility, and documentation that supports regulatory filings and site inspections.
Commercial selection is led by formulation development and quality teams because any base change triggers stability work and potential filing updates. Earlier sourcing allowed multiple grades for similar products, which increased inventory and validation load. Current portfolios aim to narrow base families and hold tight specifications across sites. Suppliers compete on refining control, impurity profiles, and continuity of supply rather than on price per kilogram. Manufacturers track total cost through rework, line speed, and complaint rates, not raw material cost alone. Contract manufacturers value bases that tolerate process shear and temperature variation without phase change. Distributors add value through controlled storage and lot traceability. Over time, demand follows growth in topical therapies and pressure to simplify formulations while keeping release behavior and shelf life within approved limits across markets and production locations.
Specification in Pharmaceutical Ointments Base Market is determined by formulation stability requirements, drug release behavior, and regulatory acceptance rather than by raw material substitution. Once a finished product dossier is approved, the base becomes a fixed component of the formulation and is controlled through change management procedures. Any modification requires renewed stability studies and, in many cases, regulatory notification. This links demand to product portfolio decisions, line extensions, and manufacturing capacity planning. Segment structure reflects differences in occlusivity, water handling, and compatibility with active ingredients. Buyers prioritize reproducibility, excipient quality documentation, and long term supply continuity because base performance directly affects therapeutic consistency and shelf life.
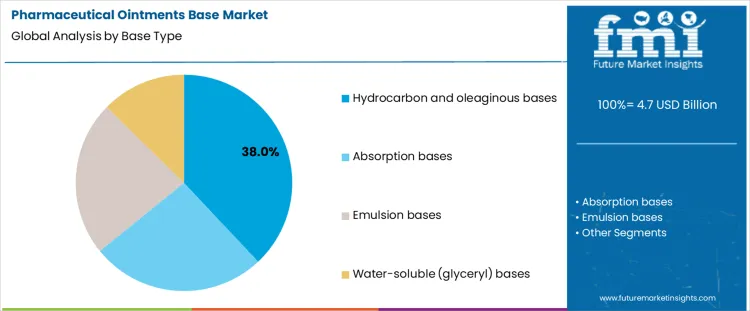
Hydrocarbon or oleaginous bases represent about 38% of demand in the Pharmaceutical Ointments Base Market because they provide strong occlusive properties, chemical stability, and simple processing behavior. These characteristics make them suitable for many chronic dermatological products. Absorption bases are selected where incorporation of aqueous solutions is required, which increases formulation flexibility but also expands control requirements for phase stability. Emulsion bases serve products that require balanced release and improved patient acceptability, though they introduce tighter control over emulsifier systems and processing conditions. Water soluble glyceryl bases are used where non greasy feel and easy wash off are required, which shifts attention toward moisture sensitivity and packaging protection. Each base family establishes distinct compounding, filling, and stability testing regimes.
From a manufacturing perspective, base type choice defines both process complexity and quality risk exposure. Oleaginous systems offer robust stability and simple heating and mixing steps. Absorption and emulsion systems require stricter control of temperature profiles, mixing order, and homogenization. Water soluble bases introduce sensitivity to humidity and microbial control, increasing environmental and packaging constraints. Once a product is validated with a specific base, substitution is avoided because release profile, stability data, and bioequivalence assumptions must be reassessed. Demand by base type therefore follows long product lifecycle decisions rather than short term changes in excipient pricing.
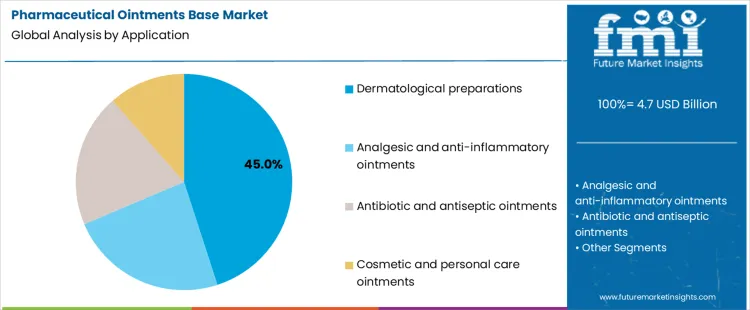
Dermatological preparations account for about 45% of demand in the Pharmaceutical Ointments Base Market because skin disorders require long term topical therapy across large patient populations. These products emphasize consistency, stability, and predictable skin interaction. Analgesic and anti-inflammatory ointments focus on controlled release and local tolerability, which influences base selection and testing depth. Antibiotic and antiseptic ointments require compatibility with sensitive actives and preservative systems, increasing specification strictness. Cosmetic and personal care ointments operate under different regulatory frameworks and place greater weight on sensory properties, though they still rely on pharmaceutical grade base systems for quality and safety.
Application mix also shapes procurement and qualification practices. Prescription dermatology and antibiotic products follow formal regulatory change control and supplier approval procedures. Pain relief and antiseptic products balance regulatory oversight with high volume manufacturing requirements. Cosmetic and personal care segments adopt faster development cycles but still require documented excipient quality and stability support. The resulting demand structure concentrates volume in therapeutic dermatology while technical scrutiny varies by therapeutic category. Segment shares therefore move with product portfolio expansion, treatment guideline changes, and lifecycle management strategies rather than with short term shifts in consumer purchasing behavior.
The Pharmaceutical Ointments Base Market is shaped by how drug developers balance formulation performance with regulatory expectations and manufacturing repeatability. Ointment bases directly influence drug release, shelf life, skin feel, and patient acceptance, which makes base selection a core formulation decision rather than a secondary excipient choice. Buyers focus on purity, consistency, compatibility with active ingredients, and documented quality. Selection decisions involve formulation scientists, quality teams, and regulatory affairs. This positions ointment bases as part of the product approval strategy and long-term manufacturing plan rather than as interchangeable raw materials.
Many pharmaceutical companies aim to reuse the same base systems across multiple topical products to simplify development, validation, and manufacturing. This is changing expectations in the Pharmaceutical Ointments Base Market toward base materials that perform reliably across different active ingredients and processing conditions. Development teams prefer bases with predictable rheology, stable behavior during heating and cooling, and consistent batch-to-batch performance. This reduces the need for repeated reformulation work. As a result, suppliers are evaluated on long-term specification stability, technical support, and the ability to supply identical material across multiple production sites.
Steady volume demand is being built in chronic dermatology treatments, over-the-counter products, and hospital formulations that are produced continuously over long periods. Once a topical product is established, its base system usually remains unchanged for many years, creating stable and predictable raw material demand. The Pharmaceutical Ointments Base Market also benefits when companies extend existing product lines using the same base platform. There is opportunity for suppliers that become embedded in approved formulations and supply agreements, securing recurring business tied to product lifecycles rather than to short-term development projects.
Any change in an ointment base can affect drug release behavior, stability data, and patient experience, which often triggers additional testing and regulatory review. This makes pharmaceutical companies cautious about changing base suppliers or base compositions. The Pharmaceutical Ointments Base Market also operates under strict quality and documentation requirements, which increases the cost and effort of supplier qualification. Even when alternative materials are available, the risk of disrupting an approved product often outweighs potential cost savings. These factors create a conservative sourcing environment where continuity and proven performance are prioritized over frequent material changes.
| Country | CAGR |
|---|---|
| USA | 3.5% |
| UK | 3.2% |
| China | 4.2% |
| India | 4.5% |
| Brazil | 3.8% |
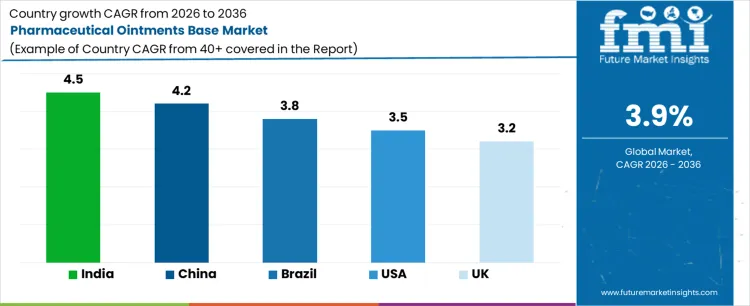
Demand for pharmaceutical ointments base is growing steadily as topical formulations remain widely used in dermatology, wound care, and pain management therapies. India leads with a 4.5% CAGR, supported by expanding domestic drug manufacturing, rising access to basic healthcare, and higher volumes of generic topical medicines. China follows at 4.2%, driven by large scale production of dermatological and traditional medicine based topical products. Brazil records 3.8%, reflecting growth in public healthcare supply and over the counter skin treatments. The USA grows at 3.5%, shaped by stable prescription and OTC usage and reformulation activity. The UK, at 3.2%, reflects a mature market with replacement and incremental volume growth.
Formulation portfolio management keeps the pharmaceutical ointments base market in the United States on a 3.5% CAGR path. Generic manufacturers, contract development groups, and brand owners represent the main users. Selection of bases focuses on consistency, drug compatibility, and stability under storage. Procurement follows product lifecycle planning rather than short term purchasing. Quality teams require certificates, impurity limits, and traceability documentation. Production schedules link base consumption to batch release plans. Replacement of approved bases occurs rarely due to validation cost. Inventory planning reflects continuous manufacturing operations. Technical service support influences supplier continuity. Import and domestic supply share volumes across grades. Change control governs any modification to composition or source. Documentation supports inspections and customer audits. Commercial position depends on approved material lists and consistent performance rather than competition for one time supply orders.
Manufacturing governance places the pharmaceutical ointments base market in the United Kingdom on a 3.2% CAGR trajectory. Contract manufacturers and branded product owners form the main customer base. Base selection follows compatibility studies and stability programs. Central technical teams manage approved excipient lists across multiple sites. Procurement aligns volumes with annual production plans. Storage and handling conditions receive attention due to temperature sensitivity. Production teams track batch consistency and yield. Change management procedures limit frequent supplier switching. Distributor networks support documentation and delivery scheduling. Import reliance influences logistics planning. Audit records remain part of regulatory files. Replacement demand links to reformulation or product transfer projects. Commercial access depends on inclusion in approved excipient schedules and reliable technical support rather than competition for small volume spot material orders.
High volume topical production places the pharmaceutical ointments base market in China on a 4.2% growth path. Dermatology, anti-infective, and pain relief products account for most usage. Corporate formulation groups define base compositions and performance criteria. Local excipient production supports volume availability and cost control. Procurement flows through group purchasing structures. Plant trials confirm texture, spreadability, and stability performance. Quality teams monitor viscosity, homogeneity, and microbial limits. Inventory planning follows continuous filling schedules. Replacement of qualified bases occurs infrequently due to process risk. Export oriented production adds documentation requirements. Payment schedules follow long term supply agreements. Commercial success depends on placement within large manufacturing groups and approved formulation platforms rather than fragmented sales to small workshops.
Rising topical medicine output fixes the pharmaceutical ointments base market in India at a 4.5% CAGR. Generic producers, export focused plants, and contract manufacturers represent core users. Procurement decisions focus on consistency, regulatory acceptance, and service support. Many facilities follow group formulation standards across products. Incoming material testing remains mandatory before release. Storage and handling controls receive attention due to climatic conditions. Production teams track batch uniformity and filling behavior. Import and domestic supply compete under qualification rules. Inventory planning aligns with production schedules. Replacement demand follows formulation updates or product transfers. Distributor technical teams support trials and audits. Commercial results depend on inclusion in approved excipient lists and maintaining technical service coverage rather than competition for one-time material supply to individual plants.
Pharmaceutical production development keeps the pharmaceutical ointments base market in Brazil near a 3.8% CAGR. Local manufacturers and regional exporters form the main customer base. Base choice reflects regulatory acceptance, stability performance, and handling characteristics. Procurement often occurs through long term supply agreements. Import procedures influence lead times and stock policies. Quality teams perform routine checks on viscosity, appearance, and microbial limits. Change approval processes limit frequent base switching. Inventory management follows steady batch production runs. Technical service support influences plant confidence. Replacement cycles link to formulation changes or site transfers. Distributor networks manage local delivery and audits. Commercial position depends on approved supplier status and consistent performance rather than competition for short term material sales to individual production lines.
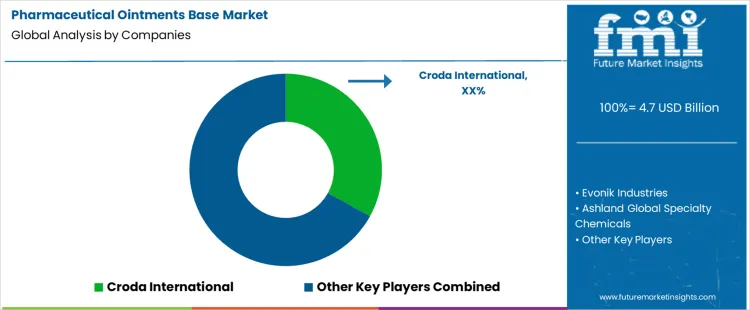
Pharmaceutical ointments base selection happens during preformulation and stability planning rather than at purchasing stage. Development teams define drug solubility, occlusion needs, water activity, sensory targets, and manufacturing method before approving materials. Croda, Evonik, Ashland, BASF, Lubrizol compete inside those formulation programs. Once a base system enters the dossier, change requires new stability studies and bioavailability checks. Croda positions structured lipid systems and functional esters for controlled release profiles. Evonik supplies polymeric and lipid excipients with defined rheology windows. Ashland focuses on cellulose derivatives and consistency modifiers used across topical ranges. BASF supports petrolatum, polyethylene glycols, and specialty wax systems. Lubrizol provides carbomer and acrylic technologies that control viscosity and yield behavior across shear ranges.
Competitive outcomes reflect documentation quality, lot consistency, and supply assurance rather than list pricing. Formulators review impurity profiles, microbial limits, peroxide values, and batch to batch variation during qualification. Scale up team’s favor suppliers that support transfer from pilot to commercial lines without rework of mixing or filling steps. Regulatory groups value DMF coverage and change notification discipline. Croda benefits from application labs that help optimize skin feel and release behavior. Evonik secures positions where polymer performance governs stability across temperature cycles. Ashland holds share in programs that rely on cellulose based structuring systems. BASF competes through breadth of base materials and global availability. Lubrizol wins where precise viscosity control and suspension stability determine acceptance. Market positions vary by dosage form, therapeutic class, and internal development standards.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD billion |
| Base Type | Hydrocarbon and oleaginous bases, Absorption bases, Emulsion bases, Water-soluble (glyceryl) bases |
| Application | Dermatological preparations, Analgesic and anti-inflammatory ointments, Antibiotic and antiseptic ointments, Cosmetic and personal care ointments |
| Region | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | China, Japan, South Korea, India, Australia & New Zealand, ASEAN, Germany, United Kingdom, France, Italy, Spain, Nordic, BENELUX, United States, Canada, Mexico, Brazil, Chile, Saudi Arabia, Turkey, South Africa, and other regional markets |
| Key Companies Profiled | Croda International, Evonik Industries, Ashland Global Specialty Chemicals, BASF SE, Lubrizol Corporation |
| Additional Attributes | Dollar by sales by base type and application; demand linked to dermatology and wound care portfolios; selection based on stability, release behavior, and compatibility; procurement driven by formulation lock-in and change control; focus on batch consistency, impurity limits, documentation readiness, long production campaigns, and audit support. |
How big is the pharmaceutical ointments base market in 2026?
The global pharmaceutical ointments base market is estimated to be valued at USD 4.7 billion in 2026.
What will be the size of pharmaceutical ointments base market in 2036?
The market size for the pharmaceutical ointments base market is projected to reach USD 6.9 billion by 2036.
How much will be the pharmaceutical ointments base market growth between 2026 and 2036?
The pharmaceutical ointments base market is expected to grow at a 3.9% CAGR between 2026 and 2036.
What are the key product types in the pharmaceutical ointments base market?
The key product types in pharmaceutical ointments base market are hydrocarbon and oleaginous bases , absorption bases, emulsion bases and water‑soluble (glyceryl) bases.
Which application segment to contribute significant share in the pharmaceutical ointments base market in 2026?
In terms of application, dermatological preparations segment to command 45.0% share in the pharmaceutical ointments base market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.