Pharmaceutical Plastic Packaging Market
The Pharmaceutical Plastic Packaging Market is segmented by Material (Polyethylene, Polypropylene, PET, PVC), End Use (Pharmaceuticals, Medical Devices, Biotechnology, Home Healthcare), and Region. Forecast for 2026 to 2036.
Pharmaceutical Plastic Packaging Market Size, Market Forecast and Outlook By FMI
In 2025, the pharmaceutical plastic packaging market exceeded a valuation of USD 27.4 billion. Based on Future Market Insights’ analysis, demand is estimated to grow to USD 28.7 billion in 2026 and USD 44.9 billion by 2036. FMI projects a CAGR of 4.6% during the forecast period.
The fundamental procurement logic in this sector is shifting from "cost-per-unit" to "cost-of-compliance." Pharmaceutical manufacturers are no longer just buying bottles; they are purchasing risk mitigation against regulatory failure. With the rise of sensitive large-molecule biologics, the cost of a packaging failure, be it through leaching, moisture ingress, or lack of serialization, far outweighs the savings from commoditized plastic sourcing.
Summary of Pharmaceutical Plastic Packaging Market
- Pharmaceutical Plastic Packaging Market Definition
- The industry covers specialized polymer containment solutions engineered for drug stability, patient safety, and regulatory compliance.
- Demand Drivers in the Market
- Rapid expansion of the biologics and GLP-1 sector requires high-performance barrier films.
- Rising generic exports from Asia Pacific fuel volume demand for cost-effective bottles and blisters.
- Sustainability mandates force a switch from PVC to recyclable polyolefins and bio-based alternatives.
- Key Segments Analyzed in the FMI Report
- Material: Polyethylene leads with 35% share due to its versatility in bottle applications.
- End Use: Pharmaceuticals segment dominates with 40% of demand driven by oral solid dosages.
- Geography: High-growth Asia Pacific contrasts with the regulatory-heavy European market.
- Analyst Opinion at FMI
- Ismail Sutaria, Principal Consultant for Packaging at Future Market Insights, opines, "In the updated version of the Pharmaceutical Plastic Packaging Market Report for 2026 to 2036, CXOs will find insights into the critical 'Compliance Cliff' facing the industry. Besides this, my findings point at a strategic pivot where Western markets will prioritize bio-based barriers to bypass fossil-plastic taxes, while Asian hubs will double down on automation to secure global export contracts."
- Strategic Implications/Executive Takeaways
- Prioritize investment in sustainable pharmaceutical packaging alternatives to mitigate future plastic tax liabilities.
- Shift focus from capacity volume to value-added barrier technologies for biologics.
- Integrate digital serialization features to meet global track-and-trace regulations.
- Methodology
- Validated through first-hand corporate production and capacity data.
- Zero reliance on speculative third-party market research reports.
- Based on verifiable industrial benchmarks and verified sources.
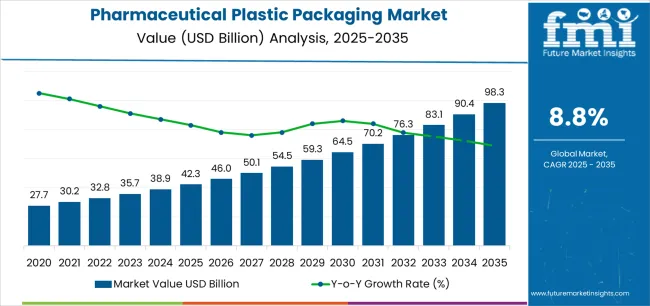
This "compliance gate" is forcing buyers to decouple their procurement strategies: while generics still chase the lowest bidder for standard polyethylene bottles, the high-margin biologics sector is locking in long-term contracts for engineered barrier films and pre-filled syringe components. Consequently, suppliers unable to provide validated stability data and anti-counterfeit pharmaceutical packaging features are being effectively gated out of the premium tier of the market.
As Dietmar Siemssen, CEO of Gerresheimer AG, noted regarding the industry's trajectory, “Our long-term growth prospects remain positive. We are growing strongly in systems and solutions for large-molecule biologics for which we are systematically expanding our production capacities.” [1] This statement underscores a critical strategic divergence: capital is moving away from generic capacity and toward specialized assets. For buyers, this implies that securing supply for biologic-compatible packaging will become tighter, necessitating earlier vendor engagement. Suppliers, in turn, must pivot their capex toward "system" solutions rather than standalone components to capture the value migrating toward complex therapies.
Growth across key economies reflects this divergence between mature replacement markets and emerging volume hubs. China (6.5%) and India (6.0%) lead expansion through massive generic export volume, while the USA (4.5%), Germany (4.0%), and the UK (3.5%) drive value through innovation in biologic delivery systems and sustainability compliance.
Pharmaceutical Plastic Packaging Market Definition
The pharmaceutical plastic packaging market comprises the global production and trade of rigid and flexible polymer-based containment solutions designed specifically for the storage, protection, and dispensing of drug products. This includes primary packaging such as bottles, vials, ampoules, and blister packs, as well as closures and medical device components. Unlike standard consumer packaging, these materials are engineered to meet strict pharmacopeial standards (USP, EP, JP) for stability, moisture barriers, and extractables/leachables profiles.
Pharmaceutical Plastic Packaging Market Inclusions
The report includes a comprehensive analysis of market dynamics, featuring Global and Regional Market Sizes (Volume and Value) and a 10-year Forecast (2026-2036). It covers segmental breakdowns by polymer type (PE, PP, PET, PVC), application (Oral, Parenteral, Topical), and end-use sectors including contract packaging and retail pharmacy. Analysis extends to regulatory frameworks governing child resistance, serialization for track-and-trace, and the integration of pharmaceutical packaging equipment compatibility.
Pharmaceutical Plastic Packaging Market Exclusions
The scope excludes non-polymer packaging materials such as glass tubing, aluminum foil (unless part of a blister lidding structure), and rubber stoppers, except where they form integral parts of a plastic hybrid system. It also omits secondary and tertiary packaging materials like paperboard cartons, corrugated boxes, and shipping pallets that do not come into direct contact with the pharmaceutical product. Equipment used for filling and sealing is also outside the resin-focused scope.
Pharmaceutical Plastic Packaging Market Research Methodology
- Primary Research: Interviews were conducted with packaging engineers, procurement heads at pharma companies, resin suppliers, and regulatory consultants to gauge material shifts and compliance pain points.
- Desk Research: Production data from plastic industry associations, pharmaceutical export councils, and company financial disclosures supported volume benchmarking and revenue estimation.
- Market-Sizing and Forecasting: A hybrid top-down and bottom-up model was developed.
- Data Validation and Update Cycle: Outputs undergo anomaly screening, variance checks against regional pharma output, and structured peer review prior to release.
Segmental Analysis
Pharmaceutical Plastic Packaging Market Analysis by Material
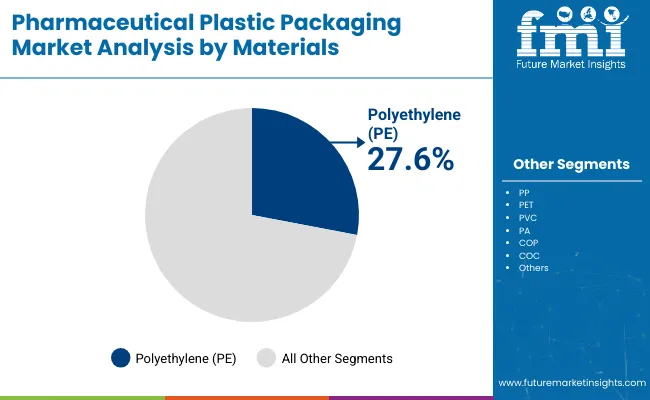
Polyethylene remains the foundational resin for the industry, securing a dominant position due to its chemical inertness and cost-efficiency. According to FMI's estimates, Polyethylene holds a 35% market share, driven by its ubiquity in rigid bottles for oral solid dosages and liquid formulations. However, the material landscape is fracturing; while commodity PE sustains the generic market, high-value segments are aggressively substituting traditional PVC. The industry is witnessing a structural migration toward advanced barrier PET and PP systems that offer recyclability without compromising stability. This shift is exemplified by recent patent activity, such as Amcor's European patent for its "AmFiber" platform, which uses paper-based technologies to replicate the barrier properties of plastics, signaling a future where material definitions blur to meet sustainability goals [2].
- Polyethylene: Demand is sustained by its extensive use in pharmaceutical plastic bottles for generics, where cost-per-unit is the primary decision metric for bulk procurement.
- Polypropylene: Preferred for closures, caps, and syringes due to its high heat resistance and autoclavability, essential for sterile environments, with companies like Berry Global refining closure systems to enhance sealing integrity [3].
- PET: Gaining traction in liquid oral packaging for its clarity and superior oxygen barrier properties compared to standard polyolefins, offering glass-like visibility without the breakage risk.
Pharmaceutical Plastic Packaging Market Analysis by End Use
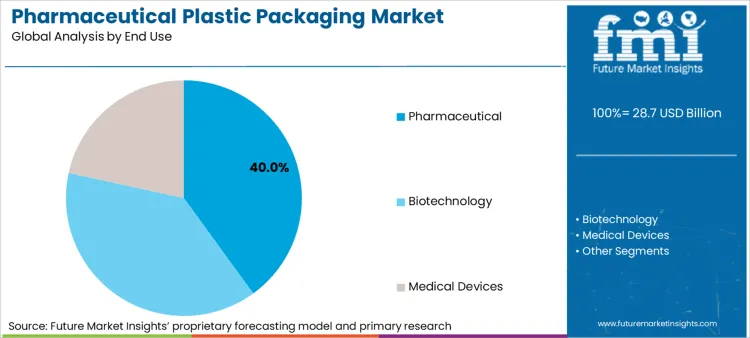
The Pharmaceuticals segment commands the largest share of demand, accounting for 40.0% of the total market value. This dominance is underpinned by the chronic disease burden which necessitates consistent volumes of daily medication packaging. However, the "Pharmaceuticals" category is bifurcating: standard oral solids are becoming commoditized, while high-value growth is concentrating in the Biotechnology and specialized delivery sectors. FMI analysts opine that the Biotechnology segment is the fastest-growing vertical, fueled by the surge in GLP-1 agonists and large-molecule drugs. AptarGroup’s recent performance validates this trend, with their Pharma segment delivering 8% sales growth specifically driven by proprietary drug delivery systems [4]. This indicates that value is no longer in the "bottle" but in the integrated device that delivers the drug.
- Pharmaceuticals: Volume growth is anchored by the generic drug boom in emerging markets, driving demand for standard blister packaging formats.
- Biotechnology: High-value growth tracks the expansion of injectable therapies; West Pharmaceutical Services reported their High-Value Product (HVP) components business grew 15.1% organically, driven by robust demand for GLP-1 related packaging components [5].
- Medical Devices: Packaging requirements are shifting toward sterile barrier systems that maintain device integrity through rigorous distribution chains, often utilizing Tyvek-plastic combinations.
Pharmaceutical Plastic Packaging Market Drivers, Restraints, and Opportunities
The surge in biologics and complex therapies is a primary engine for market value expansion. This reflects a broader industry movement where packaging is no longer a passive container but an active component of the drug delivery mechanism, requiring tighter integration between the drug formulation and the plastic device. This trend is creating opportunities for suppliers who can co-develop integrated dispensing solutions.
Conversely, stringent environmental regulations pose a significant restraint, forcing a "redesign or retire" approach for legacy materials. The European Union's Packaging and Packaging Waste Regulation (PPWR) has set a countdown for recyclability, although it offers a temporary exemption for contact-sensitive pharma packaging until 2035 [6]. This regulatory grace period creates a critical window of opportunity for innovation. Startups like Kelpi are seizing this chance, securing £4.35 million to develop seaweed-based barrier coatings that can replace fossil-fuel plastics without compromising product safety, potentially offering a route to compliance before the exemption expires [7].
- Regulatory Serialization: Mandates for track-and-trace are driving the integration of anti-counterfeit pharmaceutical packaging features directly into plastic molds, turning packaging into a data carrier.
- Export Compliance: Alignment with FDA and EMA standards is essential for Asian manufacturers targeting Western markets, necessitating higher grade resins.
- Bio-material Innovation: Development of scalable, plant-based polymers offers a pathway to bypass future plastic taxes and appeal to eco-conscious healthcare procurement.
Regional Analysis
Based on the regional analysis, the pharmaceutical plastic packaging market is segmented into North America, Latin America, Europe, East Asia, South Asia, Oceania, and Middle East & Africa across 40+ countries. The full report also offers market attractiveness analysis based on regional trends.
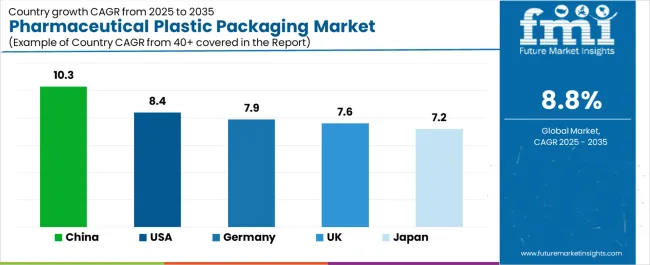
| Country | CAGR (2026 to 2036) |
|---|---|
| China | 6.5% |
| India | 6.0% |
| United States | 4.5% |
| Germany | 4.0% |
| United Kingdom | 3.5% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
Asia Pacific Pharmaceutical Plastic Packaging Market Analysis
Asia Pacific is rapidly consolidating its status as the global pharmacy of the world, driving massive volume demand for compliant packaging. As per FMI's projection, the region's growth is structurally supported by the dual engines of domestic healthcare modernization and export-oriented generic manufacturing. Governments are tightening quality controls to ensure their outputs meet global standards, effectively forcing an upgrade cycle from basic packaging to export-grade materials. This is not just about volume; it is about "compliant volume." Manufacturers are investing in clean-room capabilities and ISO-certified packaging lines to ensure their products are accepted in Western markets, turning the region into a battleground for global resin suppliers.
- China: A CAGR of 6.5% is underpinned by the National Medical Products Administration's (NMPA) rigorous digitization drive, which enforced full electronic submission for registrations starting September 2024; this mandate compels manufacturers to adopt standardized, traceable packaging formats to ensure data compliance and market access [8].
- India: Growth tracks a CAGR of 6.0% as the country reinforces its position as a top exporter; this expansion is linked to the pharmaceutical sector's robust export performance, which reached USD 30.47 billion in FY 2024-25, necessitating high-quality, international-standard packaging solutions to satisfy the regulatory requirements of key markets like the US and Europe [9].
FMI’s report includes a detailed analysis of growth in the Asia Pacific region, along with a country-wise assessment that includes China and India. Readers can also find regional trends, regulations, and market growth based on different segments and countries in Asia Pacific, including key markets like Japan and South Korea which serve as high-tech hubs for machinery and advanced materials.
North America Pharmaceutical Plastic Packaging Market Analysis
North America remains the value leader, characterized by a preference for advanced drug delivery systems and strict adherence to safety protocols. The market is defined by a "quality over quantity" dynamic, where premium pricing for plastic vials and ampoules is justified by superior barrier properties and patient adherence features. The region is seeing a distinct shift in capital allocation; companies are moving away from broad capacity expansion and focusing on efficiency and specialized capabilities to protect margins in a high-inflation environment.
- United States: At 4.5% CAGR, the country outlook is shaped by capital efficiency strategies among major players; for instance, West Pharmaceutical Services optimized its CapEx to USD 286 million in 2024 to focus on high-margin HVP componentry rather than broad capacity, aligning with the shift toward specialized biologic packaging [5].
FMI’s report includes a detailed analysis of growth in the North America region, along with a country-wise assessment that includes the United States. Readers can also find regional trends, regulations, and market growth based on different segments and countries in North America, such as Canada where government healthcare spending continues to support steady demand for medical packaging.
Europe Pharmaceutical Plastic Packaging Market Analysis
Europe serves as the global regulatory laboratory for sustainable packaging, driving a hard shift away from non-recyclable composites. The market is navigating a complex transition where circular economy goals must be balanced against immutable patient safety requirements. This region is characterized by intense M&A activity as traditional players seek to acquire the technological capabilities needed to survive the "green transition." The focus here is less on adding new volume and more on replacing existing legacy formats with sustainable pharmaceutical packaging solutions that meet upcoming EU mandates.
- Germany: A CAGR of 4.0% reflects the strategic consolidation in the market, highlighted by Gerresheimer’s €800 million acquisition of Bormioli Pharma (completed Dec 2024) to create a comprehensive pharmaceutical glass packaging and plastic solutions powerhouse capable of serving pan-European demand [10].
- United Kingdom: Expansion at 3.5% CAGR is linked to NHS England's clinical waste strategy, which targets the management of 156,000 tonnes of waste annually; this initiative incentivizes packaging designs that minimize incineration volumes and support a greener supply chain, effectively creating a market preference for low-carbon materials [11].
FMI’s report includes a detailed analysis of growth in the Europe region, along with a country-wise assessment that includes Germany and the United Kingdom. Readers can also find regional trends, regulations, and market growth based on different segments and countries in Europe, including France and Italy which remain critical hubs for luxury cosmetic and pharmaceutical packaging design.
Competitive Aligners for Market Players
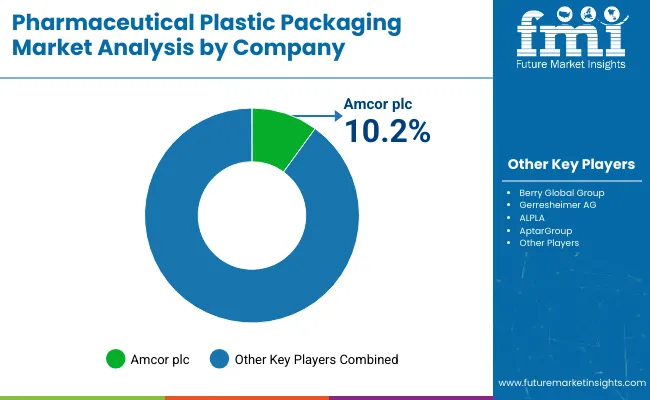
The competitive landscape is bifurcated between large-scale integrators and specialized innovators. Major entities are aggressively pursuing vertical integration to control the value chain from resin sourcing to final delivery system assembly. This scale allows them to absorb compliance costs that smaller players cannot, effectively creating a barrier to entry based on regulatory capital rather than just manufacturing capability. For example, Berry Global’s spin-off of its Health & Hygiene division signals a strategic move toward pure-play specialization, allowing for more focused capital allocation in high-growth segments [12].
Mid-tier competitors are surviving by occupying niche segments such as plastic tube packaging or ophthalmic dispensing, where precision molding offers protection against commoditization. These players often partner with regional generic manufacturers to secure long-term volume contracts, insulating themselves from spot market volatility. The ability to offer "ready-to-file" packaging documentation is becoming a critical service differentiator, as pharma companies seek to reduce their own regulatory burden.
Innovation-led startups are disrupting the lower end of the barrier market with bio-based materials. While currently low-volume, these entities are forcing incumbents to accelerate their own sustainability roadmaps. The acquisition of such technologies by global leaders is a likely consolidation endgame, ensuring that the giants remain relevant in a post-fossil-plastic regulatory environment.
Recent Developments
The report includes full coverage of key trends from competitive benchmarking. Some of the recent developments covered in the reports:
- In May 2024, Amcor plc opened its fourth global Innovation Center in Ghent, Belgium, to facilitate collaborative plastic healthcare packaging design and material science advancements with European partners [13].
- In December 2024, Gerresheimer AG completed the acquisition of Bormioli Pharma for approximately €800 million, significantly expanding its portfolio of glass and plastic solutions to offer a "one-stop-shop" for pharmaceutical customers [10].
- In July 2025, Constantia Flexibles invested €6.5 million in advanced 5-layer blown film with MDO technology, enhancing film strength and uniformity. The development enables recyclable mono-PE packaging solutions, supporting sustainability and high-performance requirements in pharmaceutical packaging.
Key Players in Pharmaceutical Plastic Packaging Market
- Berry Global
- Amcor plc
- Gerresheimer AG
- AptarGroup, Inc.
- West Pharmaceutical Services
- Kelpi
- PulpaTronics
- Aluflexpack
- Parcel Health
Scope of the Report
| Metric | Value |
|---|---|
| Quantitative Units | USD 28.7 billion (2026) to USD 44.9 billion (2036), at a CAGR of 4.6% |
| Market Definition | The pharmaceutical plastic packaging market comprises the global production and trade of rigid and flexible polymer-based containment solutions designed specifically for the storage, protection, and dispensing of drug products. |
| Material Segmentation | Polyethylene, Polypropylene, PET, PVC |
| End Use Segmentation | Pharmaceuticals, Medical Devices, Biotechnology, Home Healthcare |
| Application Coverage | Oral, Parenteral, Topical, Transdermal, Nasal, Ophthalmic |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East & Africa |
| Countries Covered | United States, China, India, Germany, United Kingdom, and 40+ others |
| Key Companies Profiled | Berry Global, Amcor plc, Gerresheimer AG, AptarGroup, Inc., West Pharmaceutical Services, Kelpi, PulpaTronics, Aluflexpack, Parcel Health |
| Forecast Period | 2026 to 2036 |
| Approach | Hybrid top down and bottom up market modeling validated through primary interviews with packaging engineers and pharma procurement heads, supported by trade data benchmarking and plant level capacity verification |
Pharmaceutical Plastic Packaging Market Analysis by Segments
Material:
- Polyethylene
- Polypropylene
- PET
- PVC
End Use:
- Pharmaceuticals
- Medical Devices
- Biotechnology
- Home Healthcare
Region:
- Asia Pacific
- India
- China
- Japan
- South Korea
- Indonesia
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- Italy
- France
- United Kingdom
- Spain
- Benelux
- Nordics
- Central & Eastern Europe
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Argentina
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Turkey
- Rest of Middle East & Africa
Bibliography
- [1] Gerresheimer AG. (2025). Gerresheimer delivers solid results in the financial year 2024 despite market headwinds (Press release, 26 February 2025). Gerresheimer (Company News).
- [2] Amcor plc. (2024). Amcor AmFiber™ paper-based packaging receives European patent (Press release, 11 November 2024). Amcor (Newsroom).
- [3] United States Patent and Trademark Office (USPTO). (2022). USA Patent No. 11,433,591 (Berry Global, Inc.) (Patent grant, 6 September 2022). USPTO.
- [4] AptarGroup, Inc. (2024). Aptar Reports Third Quarter 2024 Results (Earnings press release, 24 October 2024). Aptar (Investor Relations).
- [5] USA Securities and Exchange Commission. (2026). West Pharmaceutical Services, Inc.: Current Report (Form 8-K) dated 12 February 2026, including Exhibit 99.1 "West Reports Fourth-Quarter and Full-Year 2025 Results". SEC EDGAR.
- [6] European Union. (2024). Regulation (EU) 2025/40 of the European Parliament and of the Council of 19 December 2024 on packaging and packaging waste. EUR-Lex.
- [7] University of Bath. (2024). Seaweed packaging innovator Kelpi secures £4.35 million in new funding. University of Bath (News).
- [8] National Medical Products Administration (NMPA). (2024). Announcement on the full implementation of electronic submission for cosmetics and new cosmetic ingredients registration/notification (Announcement No. 91 of 2024) (Issued 8 July 2024; effective from 1 September 2024). Government of China (NMPA).
- [9] India Brand Equity Foundation (IBEF). (2026). India’s pharma sector bullish on export growth (News update, 6 January 2026). IBEF.
- [10] Gerresheimer AG. (2024). Gerresheimer to acquire Blitz LuxCo Sarl, the holding company of the Bormioli Pharma Group (Press release, 23 May 2024). Gerresheimer (Company News).
- [11] NHS England. (2023). NHS Clinical Waste Strategy. NHS England.
- [12] USA Securities and Exchange Commission. (2025). Berry Global Group, Inc.: Current Report (Form 8-K) referencing completion of the spin-off and merger of its Health, Hygiene & Specialties Global Nonwovens and Films business (completed 4 November 2024). SEC EDGAR.
- [13] Amcor plc. (2024). Annual Report 2024 (12 months ended 30 June 2024); and Amcor opens its European Innovation Center in Belgium (Press release, 30 May 2024). Amcor (Investor Relations / Newsroom).
This Report Addresses
- Market intelligence to enable structured strategic decision making across mature and emerging packaging economies
- Market size estimation and 10 year revenue forecasts from 2026 to 2036, supported by validated capacity and trade benchmarks
- Growth opportunity mapping across polyethylene, polypropylene, and PET forms with emphasis on sustainability transitions
- Segment and regional revenue forecasts covering pharmaceuticals, biotechnology, and medical device applications
- Competition strategy assessment including integration models, cost survivability positioning, and compliance capability benchmarking
- Product reformulation and sustainability compliance tracking including EU PPWR and bio-material adoption
- Regulatory impact analysis covering serialization mandates, child-resistance standards, and export driven specification upgrades
- Market report delivery in PDF, Excel, PPT, and interactive dashboard formats for executive and operational usea
Frequently Asked Questions
How large is the demand for Pharmaceutical Plastic Packaging in the global market in 2026?
Demand for Pharmaceutical Plastic Packaging in the global market is estimated to be valued at USD 28.7 billion in 2026.
What will be the market size of Pharmaceutical Plastic Packaging in the global market by 2036?
Market size for Pharmaceutical Plastic Packaging is projected to reach USD 44.9 billion by 2036.
What is the expected demand growth for Pharmaceutical Plastic Packaging in the global market between 2026 and 2036?
Demand for Pharmaceutical Plastic Packaging in the global market is expected to grow at a CAGR of 5.0% between 2026 and 2036.
Which Material is poised to lead global sales by 2026?
Polyethylene is expected to be the dominant material, capturing approximately 35% of global market share in 2026 due to its cost-efficiency and versatility.
How significant is the role of Pharmaceuticals in driving Pharmaceutical Plastic Packaging adoption in 2026?
Pharmaceuticals represents a critical segment, projected to hold a substantial 40.0% share of the total market in 2026 as chronic disease treatment drives volume.
What is driving demand in China?
Growth is driven by the NMPA's digitization mandates which compel manufacturers to adopt traceable, high-quality packaging standards.
What compliance standards or regulations are referenced for China?
The National Medical Products Administration (NMPA) electronic submission requirements are referenced as a key regulatory benchmark.
What is the China growth outlook in this report?
China is projected to grow at a CAGR of 6.5% during 2026 to 2036.
Why is Europe described as a priority region in this report?
Europe is described as a priority region because it acts as a regulatory laboratory for sustainable packaging and circular economy initiatives.
What type of demand dominates in Europe?
Demand is dominated by replacement needs for high-value, sustainable, and compliant packaging solutions rather than volume expansion.
What is India growth outlook in this report?
India is projected to expand at a CAGR of 6.0% during 2026 to 2036.
Does the report cover United States in its regional analysis?
Yes, the United States is included within North America under the regional scope of analysis.
What are the sources referred to for analyzing the United States?
Corporate financial disclosures from major players like West Pharmaceutical Services and AptarGroup are cited as primary reference sources.
What is the main demand theme linked to United States in its region coverage?
North America demand is associated with a shift toward high-value biologic delivery systems and capital efficiency.
Does the report cover Germany in its regional analysis?
Yes, Germany is included within Europe under the regional coverage framework.
What is the main Germany related demand theme in its region coverage?
Consolidation and strategic M&A activities to build comprehensive glass and plastic solution portfolios are emphasized.
Which product formats or configurations are strategically important for Asia Pacific supply chains?
Standard rigid bottles and blister packs are prioritized to support the high-volume generic export market.
What is Pharmaceutical Plastic Packaging and what is it mainly used for?
Pharmaceutical plastic packaging refers to polymer-based containers used for protecting, storing, and dispensing drug products like tablets and liquids.
What does Pharmaceutical Plastic Packaging Market mean in this report?
Pharmaceutical Plastic Packaging Market refers to the global production, trade, and industrial consumption of polymer packaging for healthcare applications.
What is included in the scope of this Pharmaceutical Plastic Packaging report?
Scope covers primary and secondary polymer packaging by form and material, including applications in pharmaceuticals, biotechnology, and medical devices.
What is excluded from the scope of this report?
Non-polymer materials like glass and aluminum foil, secondary packaging like paperboard, and filling equipment are excluded.
What does market forecast mean on this page?
Market forecast represents a model based projection built on defined assumptions for strategic planning purposes.
How does FMI build and validate the Pharmaceutical Plastic Packaging forecast?
Forecast is developed using hybrid top down and bottom up modeling validated through primary interviews and trade data benchmarking.
What does zero reliance on speculative third party market research mean here?
Primary interviews and verifiable public datasets are used instead of unverified syndicated market estimates.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- FMI Internal Proprietary Databases and Historical Market Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Primary Modes
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Why Primary Evidence is Used
- Field Techniques
- Interviews
- Surveys
- Focus Groups
- Observational and In-context Research
- Social and Community Interactions
- Stakeholder Universe Engaged
- C-suite Leaders
- Board Members
- Presidents and Vice Presidents
- R&D and Innovation Heads
- Technical Specialists
- Domain Subject-matter Experts
- Scientists
- Physicians and Other Healthcare Professionals
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Primary Modes
- Tooling, Models, and Reference Databases
- Desk Research Programme (Secondary Evidence)
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material , 2026 to 2036
- Polyethylene
- Polypropylene
- PET
- Polyethylene
- Y to o to Y Growth Trend Analysis By Material , 2021 to 2025
- Absolute $ Opportunity Analysis By Material , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End Use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End Use, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End Use, 2026 to 2036
- Pharmaceutical
- Biotechnology
- Medical Devices
- Pharmaceutical
- Y to o to Y Growth Trend Analysis By End Use, 2021 to 2025
- Absolute $ Opportunity Analysis By End Use, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By Material
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Material
- By End Use
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Material
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Material
- By End Use
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Material
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Material
- By End Use
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Material
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Material
- By End Use
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By Material
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Material
- By End Use
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Material
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Material
- By End Use
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Material
- By End Use
- By Country
- Market Attractiveness Analysis
- By Country
- By Material
- By End Use
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By Material
- By End Use
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Material
- By End Use
- Competition Analysis
- Competition Deep Dive
- Berry Global
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Amcor plc
- Gerresheimer AG
- AptarGroup, Inc.
- West Pharmaceutical Services
- Kelpi
- PulpaTronics
- Berry Global
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 4: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 5: North America Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 7: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 8: Latin America Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 9: Latin America Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 10: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 11: Western Europe Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 12: Western Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 13: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 14: Eastern Europe Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 15: Eastern Europe Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 16: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: East Asia Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 18: East Asia Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 19: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 20: South Asia and Pacific Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 21: South Asia and Pacific Market Value (USD Million) Forecast by End Use, 2021 to 2036
- Table 22: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 23: Middle East & Africa Market Value (USD Million) Forecast by Material , 2021 to 2036
- Table 24: Middle East & Africa Market Value (USD Million) Forecast by End Use, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by Material
- Figure 6: Global Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by End Use
- Figure 9: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Region
- Figure 12: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 13: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 14: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 15: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 16: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 17: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 18: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 20: North America Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 21: North America Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 22: North America Market Attractiveness Analysis by Material
- Figure 23: North America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 24: North America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 25: North America Market Attractiveness Analysis by End Use
- Figure 26: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 27: Latin America Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 28: Latin America Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 29: Latin America Market Attractiveness Analysis by Material
- Figure 30: Latin America Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 31: Latin America Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 32: Latin America Market Attractiveness Analysis by End Use
- Figure 33: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 34: Western Europe Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 35: Western Europe Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 36: Western Europe Market Attractiveness Analysis by Material
- Figure 37: Western Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 38: Western Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 39: Western Europe Market Attractiveness Analysis by End Use
- Figure 40: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 41: Eastern Europe Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 42: Eastern Europe Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 43: Eastern Europe Market Attractiveness Analysis by Material
- Figure 44: Eastern Europe Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 45: Eastern Europe Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 46: Eastern Europe Market Attractiveness Analysis by End Use
- Figure 47: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 48: East Asia Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 49: East Asia Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 50: East Asia Market Attractiveness Analysis by Material
- Figure 51: East Asia Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 52: East Asia Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 53: East Asia Market Attractiveness Analysis by End Use
- Figure 54: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 55: South Asia and Pacific Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 56: South Asia and Pacific Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 57: South Asia and Pacific Market Attractiveness Analysis by Material
- Figure 58: South Asia and Pacific Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 59: South Asia and Pacific Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 60: South Asia and Pacific Market Attractiveness Analysis by End Use
- Figure 61: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 62: Middle East & Africa Market Value Share and BPS Analysis by Material , 2026 and 2036
- Figure 63: Middle East & Africa Market Y-o-Y Growth Comparison by Material , 2026-2036
- Figure 64: Middle East & Africa Market Attractiveness Analysis by Material
- Figure 65: Middle East & Africa Market Value Share and BPS Analysis by End Use, 2026 and 2036
- Figure 66: Middle East & Africa Market Y-o-Y Growth Comparison by End Use, 2026-2036
- Figure 67: Middle East & Africa Market Attractiveness Analysis by End Use
- Figure 68: Global Market - Tier Structure Analysis
- Figure 69: Global Market - Company Share Analysis
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

