Pharmaceutical-Grade PCR Blister Packaging Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Pharmaceutical-Grade PCR Blister Packaging Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Pharmaceutical-Grade PCR Blister Packaging Market Forecast and Outlook 2026 to 2036
The pharmaceutical-grade PCR blister packaging market is valued at USD 980 million in 2026 and is projected to reach USD 2,620 million by 2036, reflecting a CAGR of 10.3%. Performance through this period is shaped by qualification timelines, material validation costs, and batch level consistency requirements. Unlike conventional packaging, adoption depends on repeatable barrier performance, machinability on existing blister lines, and stability under regulated storage conditions. Cost structures reflect extended testing cycles, smaller production runs, and higher rejection risk. Suppliers with validated formulations and in house testing retain pricing control. Value concentrates among operators able to amortize compliance costs across long production contracts.
Between 2026 and 2036, adoption is governed by approval inertia rather than unit demand growth. Pharmaceutical buyers prioritize continuity, audit history, and regulatory alignment, reducing tolerance for supplier change. Certification complexity creates durable entry barriers, protecting incumbent margins. Dependency on qualified resin streams limits sourcing flexibility and reinforces long term supply agreements. Strategic tradeoffs emerge between capacity expansion and validation risk, slowing aggressive scaling. Vendors integrating regulatory documentation, traceability, and quality assurance into commercial offerings secure stronger positioning. Profit pools favor firms embedded in customer approval frameworks, where replacement risk is low and contract duration supports predictable margin recovery.
Quick Stats for Pharmaceutical-Grade PCR Blister Packaging Market
- Pharmaceutical-Grade PCR Blister Packaging Market Value (2026): USD 980 million
- Pharmaceutical-Grade PCR Blister Packaging Market Forecast Value (2036): USD 2,620 million
- Pharmaceutical-Grade PCR Blister Packaging Market Forecast CAGR (2026 to 2036): 10.3%
- Leading Type in Pharmaceutical-Grade PCR Blister Packaging Market: Push-through blisters
- Key Growth Regions in Pharmaceutical-Grade PCR Blister Packaging Market: Asia Pacific, Europe, North America
- Top Players in Pharmaceutical-Grade PCR Blister Packaging Market: Klöckner Pentaplast, Südpack Medica, Shanghai Haishun, ACG Packaging, Mitsubishi Chemical Packaging, Tekni-Plex
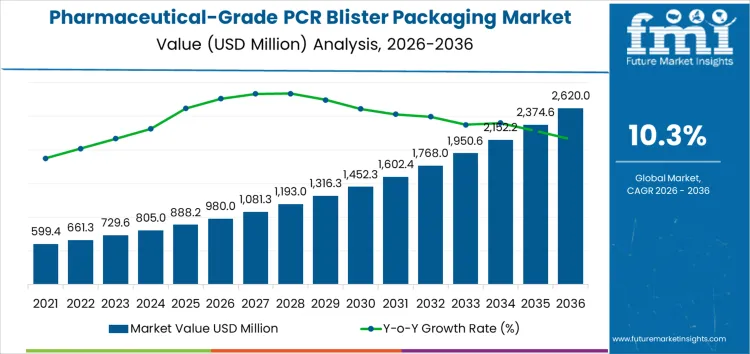
What Is the Growth Forecast for Pharmaceutical-Grade PCR Blister Packaging Market through 2036?
Regulatory validation timelines and material qualification requirements define the early growth path for the Pharmaceutical-Grade PCR Blister Packaging Market between 2026 and 2031. Over this period, the market expands from USD 980 million to USD 1,423 million, adding USD 443 million in value. Annual progression from USD 659 million to USD 980 million reflects cautious adoption, as pharmaceutical companies prioritize stability testing, barrier performance, and compliance with drug safety standards. Historical demand was limited by strict approval processes and conservative procurement practices. Future growth in this phase is supported by successful material trials, increased acceptance in secondary blister formats, and gradual integration within established solid dosage packaging lines.
Supply chain integration and scale efficiency shape the growth trajectory from 2031 to 2036. During this phase, the market increases from USD 1,423 million to USD 2,620 million, adding USD 1,197 million and representing the stronger expansion window. Annual values rise from USD 1,562 million to USD 2,620 million, indicating accelerating year over year momentum. Earlier growth relied on selective adoption, while later expansion reflects broader portfolio level rollouts by pharmaceutical manufacturers. Improved consistency of pharmaceutical grade PCR resins, streamlined regulatory pathways, and higher utilization across prescription and over the counter drug packaging support sustained growth across global healthcare packaging supply chains.
Pharmaceutical-Grade PCR Blister Packaging Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 980 million |
| Forecast Value (2036) | USD 2,620 million |
| Forecast CAGR (2026 to 2036) | 10.3% |
What Is Driving Growth in the Pharmaceutical-Grade PCR Blister Packaging Market?
Growth in the pharmaceutical-grade post-consumer recycled (PCR) blister packaging market arises from intersecting dynamics that extend beyond conventional sustainability claims. Pharmaceutical packaging must meet stringent safety, traceability, and leachability criteria because it directly contacts sensitive dosage forms.
Historically, PCR content in blister film was limited by inconsistent feedstock quality and concerns about residual contaminants that could interact with active pharmaceutical ingredients or compromise barrier performance. Pharmaceutical firms therefore prioritised virgin polymers to ensure regulatory compliance and product integrity, confining early recycled material use to ancillary packaging and non-critical components rather than primary blister structures.
Future demand in this market is being shaped by formalised regulatory pathways, documented material performance standards, and pharmaceutical supply chain commitments to circular material use. Regulatory bodies in key regions are beginning to recognise controlled recycled content when accompanied by validated purification and traceability processes that ensure equivalent safety outcomes to virgin resins. Initiatives that define acceptable extractables and leachables profiles for recycled polymer compounds are creating pathways for PCR blister films to meet pharmacopeia requirements.
Advances in PCR feedstock segregation, decontamination techniques, and polymer reconditioning improve consistency in barrier properties, tensile strength, and optical clarity, aligning recycled polymers with strict quality attributes required for blister packaging. Growth is further influenced by pharmaceutical manufacturers integrating environmental impact metrics into procurement, linking recycled content adoption to corporate sustainability strategies and lifecycle assessments without compromising safety. Demand will expand as validated material specifications and documented performance
What Factors Are Affecting the Demand for Pharmaceutical-Grade PCR Blister Packaging Market in Terms of Blister Type and Material?
The Pharmaceutical-Grade PCR Blister Packaging Market is driven by sustainability targets, regulatory scrutiny on medical packaging waste, and the need to maintain drug safety and integrity. Demand concentrates on blister formats and materials that allow recycled content while meeting stringent pharmaceutical compliance standards. Adoption is shaped by compatibility with existing blistering lines, barrier performance, and validation requirements. Market leadership reflects feasibility of PCR integration without compromising patient safety, dosing accuracy, or regulatory approval pathways.
What Is Driving Leadership by Blister Type in the Pharmaceutical-Grade PCR Blister Packaging Market?

Push-through blisters account for about 50% of demand because they are the most widely used format for solid oral dosage forms. Their simple structure, ease of use, and compatibility with high-speed packaging lines make them the default choice across branded and generic pharmaceuticals. Push-through designs tolerate limited material modification, allowing gradual incorporation of PCR content without altering patient interaction or dispensing performance. Pharmaceutical companies favor this format because validation protocols are well established and regulatory acceptance is clearer. High prescription volumes for tablets and capsules further reinforce scale advantages. As sustainability targets expand, manufacturers prioritize adapting existing push-through formats rather than introducing new blister architectures, supporting continued dominance.
Child-resistant, unit-dose, and alu–plastic blisters serve important but narrower roles. Child-resistant formats are used selectively for safety-sensitive drugs and involve more complex structures, complicating PCR adoption. Unit-dose blisters support compliance in hospital and elderly care settings but represent lower volumes. Alu–plastic blisters offer superior barrier protection but face recycling and material separation challenges. These formats grow incrementally but do not displace push-through blisters, reinforcing leadership driven by volume, simplicity, and regulatory familiarity rather than functional specialization.
What Is Driving Leadership by Material in the Pharmaceutical-Grade PCR Blister Packaging Market?
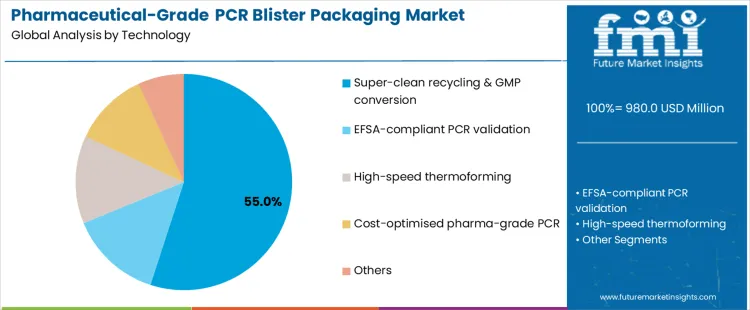
PCR-PET and PCR-PVC account for about 55% of material demand because they align most closely with existing blister specifications and regulatory frameworks. These materials offer predictable thermoforming behavior, acceptable clarity, and compatibility with common sealing layers. Pharmaceutical converters adopt PCR-PET and PCR-PVC where recycled content can be introduced without affecting moisture or oxygen barrier requirements. Established recycling streams and improving decontamination technologies support consistent resin quality. Regulatory approvals and customer audits further concentrate demand on these materials, especially for non-sensitive formulations packaged at scale.
PCR-PET alone is increasingly favored where PVC reduction strategies are pursued, but performance tradeoffs remain for certain drugs. PCR-PVC continues to dominate legacy systems due to installed equipment and proven performance. Other materials remain limited due to barrier or validation constraints. Material leadership therefore reflects feasibility, regulatory confidence, and supply consistency rather than innovation breadth.
How Is the Pharmaceutical-Grade PCR Blister Packaging Market Being Applied Across Drug Manufacturing and Distribution?
Use appears in hospitals, pharmacies, and pharmaceutical manufacturing where high-purity post-consumer recycled (PCR) plastics are used for blister packs, strips, and unit-dose packaging. Manufacturers adopt PCR-based packaging to meet sustainability targets while ensuring compliance with stringent regulatory standards for drug safety and stability. Pharmaceutical companies integrate PCR blister packs for tablets, capsules, and nutraceuticals without compromising barrier properties, moisture resistance, or chemical compatibility. These applications reflect operational and compliance priorities rather than decorative purposes, with adoption driven by recyclate utilization, regulatory adherence, and product safety.
What Operational and Material Conditions Support Adoption of Pharmaceutical-Grade PCR Blister Packaging?
Selection aligns with facilities capable of processing high-quality PCR feedstock with low contamination, controlled intrinsic viscosity, and consistent transparency. Thermoforming, cold forming, or composite lamination processes are optimized to maintain dimensional stability, barrier performance, and mechanical strength. Quality control monitors polymer purity, microbial safety, and chemical compatibility to ensure compliance with FDA, EMA, or ISO standards. These conditions emerge from operational efficiency, material integrity, and regulatory compliance priorities in structured pharmaceutical packaging workflows.
Which Practical Considerations Limit Wider Use of Pharmaceutical-Grade PCR Blister Packaging?
Feedstock variability, contamination, and mechanical inconsistencies can compromise barrier performance or clarity. Higher production costs compared with virgin plastics may affect adoption in cost-sensitive drugs or over-the-counter products. Compatibility with active pharmaceutical ingredients, adhesives, and labeling must be verified. Regulatory approval, food-contact certification, and stability testing are required for each application. These factors lead to selective deployment where recyclate utilization, compliance, and packaging performance justify incremental cost and operational oversight.
What is the Demand for Pharmaceutical Grade PCR Blister Packaging by Country?
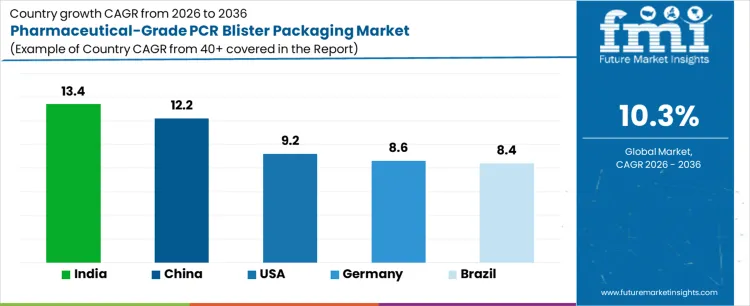
| Country | CAGR (%) |
|---|---|
| USA | 9.2% |
| Germany | 8.6% |
| China | 12.2% |
| India | 13.4% |
| Brazil | 8.4% |
The demand for pharmaceutical grade PCR blister packaging varies across countries, driven by pharmaceutical production, sustainable packaging adoption, and regulatory requirements for medical products. India leads with a 13.4% CAGR, supported by expanding pharmaceutical manufacturing, increasing exports, and growing adoption of recycled plastic packaging in drug delivery systems. China follows at 12.2%, driven by large scale pharmaceutical production, modernization of packaging lines, and regulatory compliance. The USA grows at 9.2%, reflecting steady adoption in domestic pharmaceutical manufacturing and distribution. Brazil records 8.4%, shaped by growing pharmaceutical sector and sustainable packaging initiatives. Germany posts 8.6%, supported by mature pharmaceutical markets and strict regulatory standards.
How Is the Pharmaceutical-Grade PCR Blister Packaging Market Expanding in the United States?
In the United States, the Pharmaceutical-Grade PCR Blister Packaging Market is growing at a CAGR of 9.2% through 2036, driven by increasing adoption of recycled-content blister packs in pharmaceutical manufacturing and packaging operations. Companies are integrating PCR blister solutions to meet sustainability goals, reduce virgin plastic usage, and comply with FDA and state-level packaging regulations. Demand is concentrated in pharmaceutical production facilities, contract packaging units, and hospital supply chains. Compared to Germany, where regulatory compliance is the primary driver, the U.S. market balances sustainability with operational efficiency. Recurring production cycles and high-volume packaging operations sustain consistent adoption nationwide.
- Pharmaceutical production facilities, contract packaging units, and hospital supply chains drive adoption
- PCR blister packs reduce virgin plastic usage and support sustainability goals
- Domestic suppliers provide high-performance, regulatory-compliant blister solutions
- Recurring production cycles sustain consistent adoption
How Are Regulatory Standards Driving Growth in Germany?
In Germany, revenue is growing at a CAGR of 8.6% through 2036, supported by EU mandates on recycled packaging content and strict pharmaceutical packaging regulations. Pharmaceutical manufacturers, contract packaging units, and hospital supply chains are adopting PCR blister packs to comply with environmental and quality standards. Demand is concentrated in industrial pharmaceutical hubs, contract manufacturing facilities, and urban packaging zones. Compared to the U.S., adoption is more compliance-driven than efficiency-focused. Domestic suppliers provide high-quality, EU-compliant PCR blister solutions compatible with automated production lines. Recurring packaging operations and regulatory inspections sustain steady adoption nationwide.
- Pharmaceutical hubs, contract manufacturing facilities, and urban packaging zones drive adoption
- PCR blister packs ensure regulatory compliance and environmental sustainability
- Domestic suppliers provide EU-compliant, high-performance blister solutions
- Recurring packaging operations sustain steady adoption
How Is Industrial Scale and Healthcare Growth Driving Adoption in China?
In China, the market is expanding at a CAGR of 12.2% through 2036, fueled by rapid growth in pharmaceutical production, healthcare infrastructure, and urban packaging demand. Facilities are adopting PCR blister packaging to reduce virgin plastic use, comply with local environmental standards, and enhance sustainability. Demand is concentrated in industrial pharmaceutical hubs, contract manufacturing centers, and hospital supply chains. Domestic suppliers are scaling production of high-performance PCR blister solutions compatible with automated packaging lines. Compared to Brazil, adoption focuses on large-scale industrial integration and urban manufacturing efficiency. Recurring production cycles sustain rapid adoption nationwide.
- Industrial pharmaceutical hubs, contract manufacturing centers, and hospital supply chains drive adoption
- PCR blister packs reduce virgin plastic use and improve sustainability
- Domestic suppliers scale high-performance, automated-compatible solutions
- Recurring production cycles sustain rapid adoption
How Are Emerging Pharmaceutical and Packaging Zones Driving Market Growth in India?
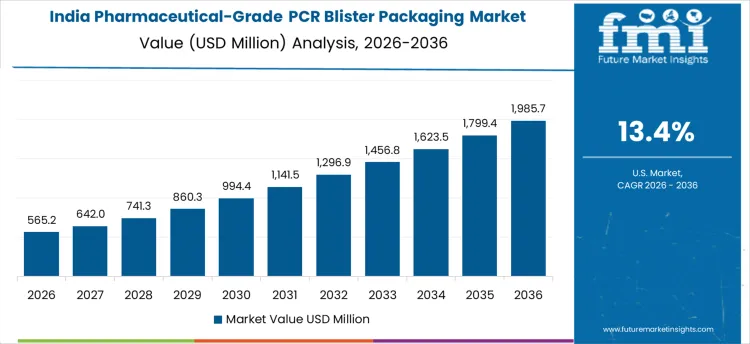
In India, revenue is growing at a CAGR of 13.4% through 2036, supported by expansion of pharmaceutical manufacturing units, contract packaging facilities, and healthcare infrastructure. Companies are adopting PCR blister packaging to meet sustainability targets, reduce environmental impact, and comply with emerging packaging regulations. Demand is concentrated in urban industrial clusters, pharmaceutical hubs, and hospital supply chains. Domestic suppliers provide cost-effective, high-performance PCR blister solutions suitable for automated packaging operations. Compared to China, adoption in India is influenced more by emerging zones and government incentives promoting recycled-content packaging. Recurring production cycles sustain robust adoption nationwide.
- Pharmaceutical hubs, contract packaging facilities, and hospital supply chains drive adoption
- PCR blister packs reduce environmental impact and support sustainability
- Domestic suppliers provide cost-effective, high-performance solutions
- Recurring production cycles sustain robust adoption
How Are Industrial and Packaging Hubs Supporting Growth in Brazil?
In Brazil, the market is growing at a CAGR of 8.4% through 2036, driven by adoption of PCR blister packaging in established pharmaceutical manufacturing centers, contract packaging units, and hospital supply chains. Companies are integrating recycled-content blister packs to reduce virgin plastic consumption, comply with environmental standards, and meet corporate sustainability initiatives. Demand is concentrated in industrial pharmaceutical hubs, urban packaging zones, and distribution facilities. Domestic suppliers provide high-performance PCR blister solutions compatible with existing packaging lines. Compared to India and China, adoption focuses on established hubs rather than rapid industrial expansion. Recurring production cycles sustain steady market growth nationwide.
- Pharmaceutical hubs, contract packaging units, and distribution facilities drive adoption
- PCR blister packs reduce virgin plastic use and support sustainability
- Domestic suppliers provide high-performance, production-compatible solutions
- Recurring production cycles sustain steady adoption
Which Strategic Approaches Define Competition in the Pharmaceutical-Grade PCR Blister Packaging Market through Regulatory Confidence and Process Discipline?
Competition in the Pharmaceutical-Grade PCR Blister Packaging Market is governed by regulatory acceptance and the ability to control contamination risk across the full value chain. Suppliers are evaluated on how PCR content is introduced without altering barrier performance, sealing behavior, or extractables profiles. Klöckner Pentaplast and Tekni Plex compete by embedding PCR layers within multilayer structures that preserve proven pharma facing surfaces, allowing continuity with existing drug approvals.
Südpack Medica emphasizes material traceability and process segregation, positioning PCR blister films as extensions of validated platforms rather than new materials. Pharmaceutical customers prioritize audit readiness, documentation depth, and reproducibility across batches, making conservative integration strategies more competitive than aggressive recycled content targets.
Regional execution and customer alignment shape secondary competitive dynamics. ACG Packaging influences adoption through close relationships with solid dose manufacturers, aligning PCR blister materials with filling line behavior and long qualification cycles.
Mitsubishi Chemical Packaging applies polymer science depth to manage optical clarity, stiffness, and forming consistency in PCR containing films used for regulated markets. Shanghai Haishun competes through localized supply and cost discipline, targeting regional pharmaceutical producers where regulatory pathways differ and approval timelines are shorter. Across the market, competitive advantage depends on segregation of waste streams, validation support, and long term supply reliability. Success is defined by trust, consistency, and regulatory continuity rather than speed of innovation or volume expansion.
Key Players in the Pharmaceutical-Grade PCR Blister Packaging Market
- Klöckner Pentaplast
- Südpack Medica
- Shanghai Haishun
- ACG Packaging
- Mitsubishi Chemical Packaging
- Tekni-Plex
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Type | Push-Through Blisters, Child-Resistant Blisters, Unit-Dose Blisters, Alu-Plastic Blisters, Other Types |
| Application | Solid Oral Dosages, Prescription Medicines, Generic Pharmaceuticals, Export-Oriented Pharma, High-Compliance Packaging |
| End User | Pharmaceutical Manufacturers, Contract Packaging Organizations, Hospital Supply Chains, Generic Drug Producers |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | USA, Germany, China, India, Japan, South Korea, Brazil, United Kingdom, France, Italy, Canada, Mexico, ASEAN countries, GCC countries, and other global regions |
| Key Companies Profiled | Klöckner Pentaplast, Südpack Medica, Tekni-Plex, ACG Packaging, Mitsubishi Chemical Packaging, Shanghai Haishun |
| Additional Attributes | Dollar sales by blister type and material categories, regional adoption trends across Asia Pacific, Europe, and North America, competitive landscape across global and regional pharmaceutical blister packaging suppliers, regulatory validation and audit requirements under FDA, EMA, and GMP frameworks, material traceability and quality assurance requirements for pharmaceutical-grade PCR integration |
Pharmaceutical-Grade PCR Blister Packaging Market Segmentation
By End-use:
- Solid oral dosages
- Prescription medicines
- Generic pharmaceuticals
- Export-oriented pharma
- Domestic pharma
- High-compliance packaging
By Blister Type:
- Push-through blisters
- Child-resistant blisters
- Unit-dose blisters
- Alu–plastic blisters
- Others
By Material:
- PCR-PET / PCR-PVC
- PCR-PET
- PCR-PVC
- Others
By Technology:
- Super-clean recycling & GMP conversion
- EFSA-compliant PCR validation
- High-speed thermoforming
- Cost-optimised pharma-grade PCR
- Others
By Region
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- United States
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Frequently Asked Questions
How big is the pharmaceutical-grade PCR blister packaging market in 2026?
The global pharmaceutical-grade PCR blister packaging market is estimated to be valued at USD 980.0 million in 2026.
What will be the size of pharmaceutical-grade PCR blister packaging market in 2036?
The market size for the pharmaceutical-grade PCR blister packaging market is projected to reach USD 2,620.0 million by 2036.
How much will be the pharmaceutical-grade PCR blister packaging market growth between 2026 and 2036?
The pharmaceutical-grade PCR blister packaging market is expected to grow at a 10.3% CAGR between 2026 and 2036.
What are the key product types in the pharmaceutical-grade PCR blister packaging market?
The key product types in pharmaceutical-grade PCR blister packaging market are solid oral dosages, prescription medicines, generic pharmaceuticals, export-oriented pharma, domestic pharma and high-compliance packaging.
Which blister type segment to contribute significant share in the pharmaceutical-grade PCR blister packaging market in 2026?
In terms of blister type, push-through blisters segment to command 50.0% share in the pharmaceutical-grade PCR blister packaging market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Million) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Million) Projections, 2026 to 2036
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By End-use
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End-use , 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End-use , 2026 to 2036
- Solid oral dosages
- Prescription medicines
- Generic pharmaceuticals
- Export-oriented pharma
- Domestic pharma
- High-compliance packaging
- Solid oral dosages
- Y to o to Y Growth Trend Analysis By End-use , 2021 to 2025
- Absolute $ Opportunity Analysis By End-use , 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Blister Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Blister Type, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Blister Type, 2026 to 2036
- Push-through blisters
- Child-resistant blisters
- Value (USD Million)-dose blisters
- Alu–plastic blisters
- Push-through blisters
- Y to o to Y Growth Trend Analysis By Blister Type, 2021 to 2025
- Absolute $ Opportunity Analysis By Blister Type, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Material
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Material, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Material, 2026 to 2036
- PCR-PET / PCR-PVC
- PCR-PET
- PCR-PVC
- PCR-PET
- Others
- PCR-PET / PCR-PVC
- Y to o to Y Growth Trend Analysis By Material, 2021 to 2025
- Absolute $ Opportunity Analysis By Material, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Technology
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Technology, 2021 to 2025
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Technology, 2026 to 2036
- Super-clean recycling & GMP conversion
- EFSA-compliant PCR validation
- High-speed thermoforming
- Cost-optimised pharma-grade PCR
- Others
- Super-clean recycling & GMP conversion
- Y to o to Y Growth Trend Analysis By Technology, 2021 to 2025
- Absolute $ Opportunity Analysis By Technology, 2026 to 2036
- Global Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2021 to 2025
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2026 to 2036
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- USA
- Canada
- Mexico
- By End-use
- By Blister Type
- By Material
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Blister Type
- By Material
- By Technology
- Key Takeaways
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Brazil
- Chile
- Rest of Latin America
- By End-use
- By Blister Type
- By Material
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Blister Type
- By Material
- By Technology
- Key Takeaways
- Western Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By End-use
- By Blister Type
- By Material
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Blister Type
- By Material
- By Technology
- Key Takeaways
- Eastern Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By End-use
- By Blister Type
- By Material
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Blister Type
- By Material
- By Technology
- Key Takeaways
- East Asia Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- China
- Japan
- South Korea
- By End-use
- By Blister Type
- By Material
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Blister Type
- By Material
- By Technology
- Key Takeaways
- South Asia and Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By End-use
- By Blister Type
- By Material
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Blister Type
- By Material
- By Technology
- Key Takeaways
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2021 to 2025
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2026 to 2036
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By End-use
- By Blister Type
- By Material
- By Technology
- By Country
- Market Attractiveness Analysis
- By Country
- By End-use
- By Blister Type
- By Material
- By Technology
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- Canada
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- Mexico
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- Brazil
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- Chile
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- Germany
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- UK
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- Italy
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- Spain
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- France
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- India
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- China
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- Japan
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- South Korea
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- Russia
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- Poland
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- Hungary
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- South Africa
- Pricing Analysis
- Market Share Analysis, 2025
- By End-use
- By Blister Type
- By Material
- By Technology
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By End-use
- By Blister Type
- By Material
- By Technology
- Competition Analysis
- Competition Deep Dive
- Klöckner Pentaplast
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Südpack Medica
- Shanghai Haishun
- ACG Packaging
- Mitsubishi Chemical Packaging
- Tekni-Plex
- Klöckner Pentaplast
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2021 to 2036
- Table 2: Global Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 3: Global Market Value (USD Million) Forecast by Blister Type, 2021 to 2036
- Table 4: Global Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 5: Global Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 6: North America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 7: North America Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 8: North America Market Value (USD Million) Forecast by Blister Type, 2021 to 2036
- Table 9: North America Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 10: North America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 11: Latin America Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 12: Latin America Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 13: Latin America Market Value (USD Million) Forecast by Blister Type, 2021 to 2036
- Table 14: Latin America Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 15: Latin America Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 16: Western Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 17: Western Europe Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 18: Western Europe Market Value (USD Million) Forecast by Blister Type, 2021 to 2036
- Table 19: Western Europe Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 20: Western Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 21: Eastern Europe Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 22: Eastern Europe Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 23: Eastern Europe Market Value (USD Million) Forecast by Blister Type, 2021 to 2036
- Table 24: Eastern Europe Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 25: Eastern Europe Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 26: East Asia Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 27: East Asia Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 28: East Asia Market Value (USD Million) Forecast by Blister Type, 2021 to 2036
- Table 29: East Asia Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 30: East Asia Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 31: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 32: South Asia and Pacific Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 33: South Asia and Pacific Market Value (USD Million) Forecast by Blister Type, 2021 to 2036
- Table 34: South Asia and Pacific Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 35: South Asia and Pacific Market Value (USD Million) Forecast by Technology, 2021 to 2036
- Table 36: Middle East & Africa Market Value (USD Million) Forecast by Country, 2021 to 2036
- Table 37: Middle East & Africa Market Value (USD Million) Forecast by End-use , 2021 to 2036
- Table 38: Middle East & Africa Market Value (USD Million) Forecast by Blister Type, 2021 to 2036
- Table 39: Middle East & Africa Market Value (USD Million) Forecast by Material, 2021 to 2036
- Table 40: Middle East & Africa Market Value (USD Million) Forecast by Technology, 2021 to 2036
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2021-2036
- Figure 3: Global Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 4: Global Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 5: Global Market Attractiveness Analysis by End-use
- Figure 6: Global Market Value Share and BPS Analysis by Blister Type, 2026 and 2036
- Figure 7: Global Market Y-o-Y Growth Comparison by Blister Type, 2026-2036
- Figure 8: Global Market Attractiveness Analysis by Blister Type
- Figure 9: Global Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 10: Global Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 11: Global Market Attractiveness Analysis by Material
- Figure 12: Global Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 13: Global Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 14: Global Market Attractiveness Analysis by Technology
- Figure 15: Global Market Value (USD Million) Share and BPS Analysis by Region, 2026 and 2036
- Figure 16: Global Market Y-o-Y Growth Comparison by Region, 2026-2036
- Figure 17: Global Market Attractiveness Analysis by Region
- Figure 18: North America Market Incremental Dollar Opportunity, 2026-2036
- Figure 19: Latin America Market Incremental Dollar Opportunity, 2026-2036
- Figure 20: Western Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 21: Eastern Europe Market Incremental Dollar Opportunity, 2026-2036
- Figure 22: East Asia Market Incremental Dollar Opportunity, 2026-2036
- Figure 23: South Asia and Pacific Market Incremental Dollar Opportunity, 2026-2036
- Figure 24: Middle East & Africa Market Incremental Dollar Opportunity, 2026-2036
- Figure 25: North America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 26: North America Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 27: North America Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 28: North America Market Attractiveness Analysis by End-use
- Figure 29: North America Market Value Share and BPS Analysis by Blister Type, 2026 and 2036
- Figure 30: North America Market Y-o-Y Growth Comparison by Blister Type, 2026-2036
- Figure 31: North America Market Attractiveness Analysis by Blister Type
- Figure 32: North America Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 33: North America Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 34: North America Market Attractiveness Analysis by Material
- Figure 35: North America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 36: North America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 37: North America Market Attractiveness Analysis by Technology
- Figure 38: Latin America Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 39: Latin America Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 40: Latin America Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 41: Latin America Market Attractiveness Analysis by End-use
- Figure 42: Latin America Market Value Share and BPS Analysis by Blister Type, 2026 and 2036
- Figure 43: Latin America Market Y-o-Y Growth Comparison by Blister Type, 2026-2036
- Figure 44: Latin America Market Attractiveness Analysis by Blister Type
- Figure 45: Latin America Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 46: Latin America Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 47: Latin America Market Attractiveness Analysis by Material
- Figure 48: Latin America Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 49: Latin America Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 50: Latin America Market Attractiveness Analysis by Technology
- Figure 51: Western Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 52: Western Europe Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 53: Western Europe Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 54: Western Europe Market Attractiveness Analysis by End-use
- Figure 55: Western Europe Market Value Share and BPS Analysis by Blister Type, 2026 and 2036
- Figure 56: Western Europe Market Y-o-Y Growth Comparison by Blister Type, 2026-2036
- Figure 57: Western Europe Market Attractiveness Analysis by Blister Type
- Figure 58: Western Europe Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 59: Western Europe Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 60: Western Europe Market Attractiveness Analysis by Material
- Figure 61: Western Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 62: Western Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 63: Western Europe Market Attractiveness Analysis by Technology
- Figure 64: Eastern Europe Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 65: Eastern Europe Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 66: Eastern Europe Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 67: Eastern Europe Market Attractiveness Analysis by End-use
- Figure 68: Eastern Europe Market Value Share and BPS Analysis by Blister Type, 2026 and 2036
- Figure 69: Eastern Europe Market Y-o-Y Growth Comparison by Blister Type, 2026-2036
- Figure 70: Eastern Europe Market Attractiveness Analysis by Blister Type
- Figure 71: Eastern Europe Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 72: Eastern Europe Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 73: Eastern Europe Market Attractiveness Analysis by Material
- Figure 74: Eastern Europe Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 75: Eastern Europe Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 76: Eastern Europe Market Attractiveness Analysis by Technology
- Figure 77: East Asia Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 78: East Asia Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 79: East Asia Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 80: East Asia Market Attractiveness Analysis by End-use
- Figure 81: East Asia Market Value Share and BPS Analysis by Blister Type, 2026 and 2036
- Figure 82: East Asia Market Y-o-Y Growth Comparison by Blister Type, 2026-2036
- Figure 83: East Asia Market Attractiveness Analysis by Blister Type
- Figure 84: East Asia Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 85: East Asia Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 86: East Asia Market Attractiveness Analysis by Material
- Figure 87: East Asia Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 88: East Asia Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 89: East Asia Market Attractiveness Analysis by Technology
- Figure 90: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 91: South Asia and Pacific Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 92: South Asia and Pacific Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 93: South Asia and Pacific Market Attractiveness Analysis by End-use
- Figure 94: South Asia and Pacific Market Value Share and BPS Analysis by Blister Type, 2026 and 2036
- Figure 95: South Asia and Pacific Market Y-o-Y Growth Comparison by Blister Type, 2026-2036
- Figure 96: South Asia and Pacific Market Attractiveness Analysis by Blister Type
- Figure 97: South Asia and Pacific Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 98: South Asia and Pacific Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 99: South Asia and Pacific Market Attractiveness Analysis by Material
- Figure 100: South Asia and Pacific Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 101: South Asia and Pacific Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 102: South Asia and Pacific Market Attractiveness Analysis by Technology
- Figure 103: Middle East & Africa Market Value Share and BPS Analysis by Country, 2026 and 2036
- Figure 104: Middle East & Africa Market Value Share and BPS Analysis by End-use , 2026 and 2036
- Figure 105: Middle East & Africa Market Y-o-Y Growth Comparison by End-use , 2026-2036
- Figure 106: Middle East & Africa Market Attractiveness Analysis by End-use
- Figure 107: Middle East & Africa Market Value Share and BPS Analysis by Blister Type, 2026 and 2036
- Figure 108: Middle East & Africa Market Y-o-Y Growth Comparison by Blister Type, 2026-2036
- Figure 109: Middle East & Africa Market Attractiveness Analysis by Blister Type
- Figure 110: Middle East & Africa Market Value Share and BPS Analysis by Material, 2026 and 2036
- Figure 111: Middle East & Africa Market Y-o-Y Growth Comparison by Material, 2026-2036
- Figure 112: Middle East & Africa Market Attractiveness Analysis by Material
- Figure 113: Middle East & Africa Market Value Share and BPS Analysis by Technology, 2026 and 2036
- Figure 114: Middle East & Africa Market Y-o-Y Growth Comparison by Technology, 2026-2036
- Figure 115: Middle East & Africa Market Attractiveness Analysis by Technology
- Figure 116: Global Market - Tier Structure Analysis
- Figure 117: Global Market - Company Share Analysis

