The PCR packaging for pharma and OTC market is forecasted to be valued at USD 2,360.0 million in 2026 and is set to reach USD 6,946.5 million by 2036, expanding at a 11.4% CAGR. Growth reflects accelerating integration of post-consumer recycled polymers into pharmaceutical and consumer health packaging formats that operate under stringent regulatory control. Adoption concentrates in bottles, blisters, and rigid containers where sustainability targets must be met without compromising moisture protection, light shielding, or chemical compatibility with active ingredients. Demand strength increases as pharma and OTC brands align environmental reporting obligations with packaging redesign programs that prioritize recycled content traceability and material qualification.
Market expansion is shaped by the structural shift from pilot-scale PCR usage toward validated, GMP-aligned packaging systems capable of supporting high-speed filling and serialization. Investment flows toward PCR resins processed through closed-loop, contamination-controlled streams suitable for drug-contact applications. Contract packagers and global pharmaceutical suppliers increasingly standardize PCR packaging specifications across multi-country portfolios to simplify compliance and auditing. Supply reliability, batch consistency, and long-term availability of pharma-grade PCR materials influence procurement decisions more than resin price differentials. Growth momentum remains anchored to regulatory acceptance pathways, lifecycle reporting requirements, and the ability of PCR packaging solutions to replicate virgin-material performance across the full product shelf life.
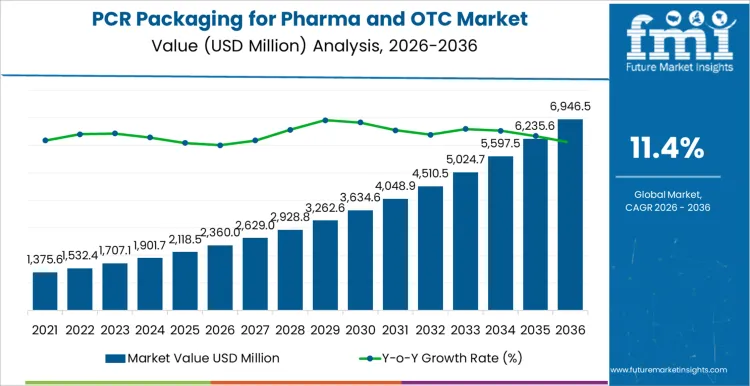
| Metric | Value |
|---|---|
| Market Value (2026) | USD 2,360.0 million |
| Market Forecast Value (2036) | USD 6,946.5 million |
| Forecast CAGR 2026 to 2036 | 11.4% |
The rise in use of post-consumer recycled (PCR) packaging in pharmaceutical and over-the-counter (OTC) products reflects intensified focus on sustainability within highly regulated health sectors. Pharmaceutical brands under pressure to reduce environmental footprint are evaluating PCR bottles, tubes, and blister backing materials that meet strict drug safety and compatibility criteria. PCR content helps companies align with corporate environmental goals while maintaining barrier performance essential for moisture- and light-sensitive APIs (active pharmaceutical ingredients). OTC producers of vitamins, analgesics, and topical treatments require consistent polymer quality to preserve dosage form integrity and avoid extractables or leachables that could interact with formulations.
Packaging engineers specify PCR resins that are traceable, sorted, and purified to meet stringent pharmacopeial acceptance thresholds and maintain tight dimensional tolerance in high-speed filling lines. Regulatory frameworks and buyer expectations in key markets support accelerated adoption of PCR packaging for pharma and OTC products. Regions with mandates on recycled content in consumer packaging extend influence into healthcare sectors where waste reduction is emphasized in procurement policies. Material qualification protocols incorporate accelerated aging and contamination screening to ensure PCR meets stability requirements across product shelf life. Contract manufacturers with multi-product facilities adopt PCR materials to help brand partners achieve sustainability reporting targets while avoiding disruptions in serialization and track-and-trace operations that rely on stable packaging performance.
Demand for PCR packaging in pharmaceutical and OTC applications is shaped by regulatory scrutiny, patient safety requirements, and sustainability commitments from drug manufacturers. Packaging suppliers focus on contamination control, material traceability, and validation alignment with pharma-grade standards. Selection depends on packaging format sensitivity, sterilization compatibility, and audit readiness. Segmentation by packaging type, compliance technology, and end use explains how recycled materials are adopted across regulated pharmaceutical packaging while maintaining quality, safety, and compliance integrity.
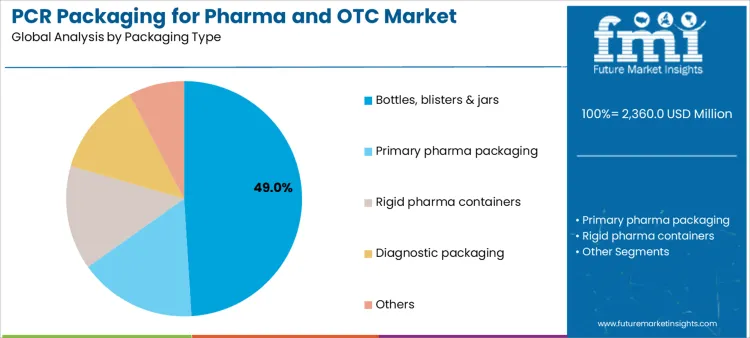
Bottles, blisters, and jars account for 49.0%, reflecting their widespread use in solid oral dosages, liquid medicines, and OTC products. These formats benefit from established validation pathways and predictable material performance. Adoption is strongest where packaging geometry, barrier performance, and patient handling requirements are well defined. Other packaging types, including primary pharma packaging, rigid containers, diagnostic packaging, and specialty formats, support additional use cases without disclosed share values. Packaging-type segmentation highlights concentration in high-volume, standardized formats suited to validated PCR integration.
Key Points:
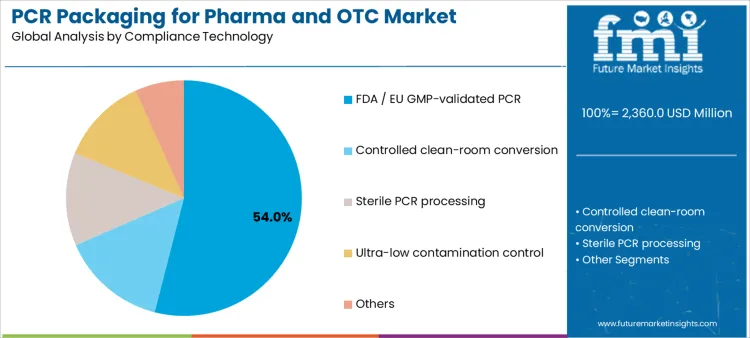
FDA and EU GMP-validated PCR technologies represent 54.0%, underscoring the importance of regulatory acceptance and documented safety controls. These systems emphasize traceability, audit readiness, and consistency across production batches. Controlled clean-room conversion, sterile PCR processing, ultra-low contamination control, and other technologies address varying risk profiles without quantified shares. Compliance-technology segmentation reflects how investment in validation infrastructure enables broader PCR use in sensitive pharmaceutical packaging environments.
Key Points:
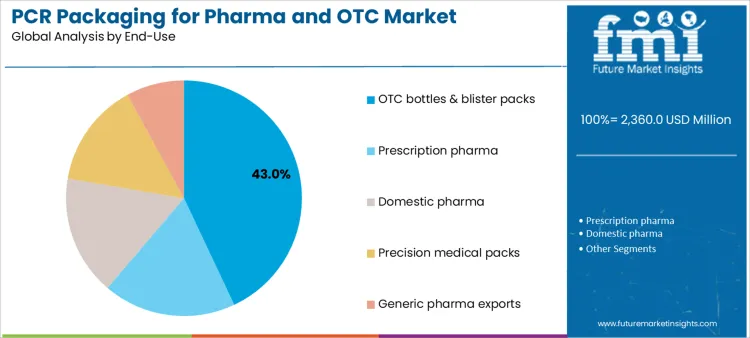
OTC bottles and blister packs represent 43.0%, driven by high unit volumes, retail visibility, and sustainability initiatives from consumer health brands. These products balance regulatory oversight with faster innovation cycles compared to prescription medicines. Other end uses, including prescription pharma, domestic pharmaceutical supply, precision medical packs, and generic pharma exports, contribute to demand without disclosed share values. End-use segmentation shows early PCR adoption concentration in OTC formats where scale, branding, and sustainability messaging align with compliance feasibility.
Key Points:
The demand for PCR packaging in the pharmaceutical and over-the-counter (OTC) market is driven by the need to balance high regulatory standards with growing sustainability goals. Pharmaceutical manufacturers and consumer health companies require PCR materials that meet strict regulatory requirements regarding safety, quality, and traceability. The selection of PCR packaging materials depends on their ability to maintain consistent barrier properties, stability during sterilization, and compatibility with packaging formats used for pills, liquids, and topical products.
Regulatory and safety standards are a key factor in material selection for pharma and OTC packaging. Recycled materials that do not meet the necessary purity or safety standards, such as control of additives or residual chemicals, are often not suitable for use. Packaging for sterile drugs and oral medicines, for instance, must meet strict pharmacopeial guidelines for leachables and extractables. PCR materials must pass rigorous testing to ensure their performance mirrors that of virgin plastics, particularly for sensitive products like blister packs and bottles, where integrity, safety, and barrier properties are critical.
The uptake of PCR packaging in the pharmaceutical and OTC market is impacted by several challenges. The availability of high-quality recycled plastics that meet the stringent medical-grade criteria is often limited, especially when the feedstock comes from non-industrial sources. Sorting and decontamination processes for PCR materials suitable for pharmaceutical use add complexity and cost. The performance of PCR plastics must be consistent to meet the exacting demands of injection and blow molding processes, which are crucial for producing precise closures and sterile vials. The varying regional regulations governing packaging materials also affect the global adoption of PCR packaging, as differing safety standards may require requalification or adjustments in material formulations for specific markets.
Global demand for PCR (Post-Consumer Recycled) packaging for pharmaceutical and over-the-counter (OTC) products is rising as industries focus on sustainability and reducing plastic waste. Growth reflects increasing regulatory pressure, consumer demand for eco-friendly products, and the shift toward circular economy practices. PCR packaging provides an environmentally friendly alternative while maintaining the safety and integrity of pharmaceutical and OTC products. India records 14.8% CAGR, China records 13.6% CAGR, USA records 10.6% CAGR, Germany records 11.0% CAGR, and Japan records 9.8% CAGR. Adoption is driven by sustainability goals, regulatory frameworks, and the increasing use of recycled materials in packaging.
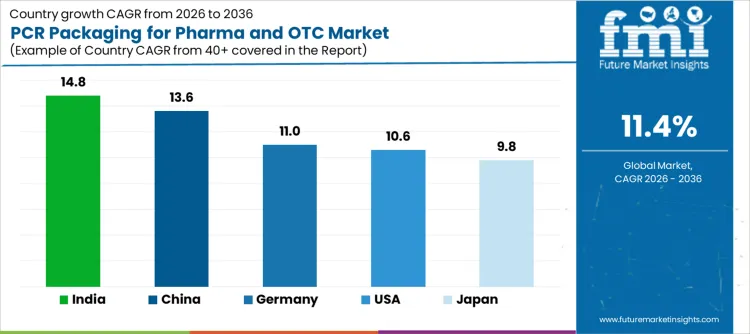
| Country | CAGR (%) |
|---|---|
| India | 14.8% |
| China | 13.6% |
| USA | 10.6% |
| Germany | 11.0% |
| Japan | 9.8% |
Demand for PCR packaging for pharma and OTC products in India is expanding as the pharmaceutical and healthcare sectors embrace sustainability initiatives to meet regulatory requirements and consumer demand for eco-friendly packaging. Growth at 14.8% CAGR reflects increasing adoption of PCR materials in packaging for OTC products, medicines, and supplements. As India focuses on reducing plastic waste and promoting the use of recycled materials, the demand for PCR packaging solutions in the pharma sector continues to rise. Local manufacturers are investing in technology to improve the quality of PCR materials, making them suitable for high-sensitivity pharmaceutical products.
PCR packaging demand for pharma and OTC products in China is rising as the country strengthens its recycling infrastructure and focuses on reducing plastic waste. Growth at 13.6% CAGR reflects China’s push for the adoption of recycled content in packaging, including pharmaceutical products and OTC items. Stringent regulations and government policies on plastic reduction are driving investments in PCR packaging solutions. As the Chinese pharmaceutical and consumer goods sectors embrace sustainability, the use of PCR materials in packaging continues to increase, particularly in the pharmaceutical and healthcare sectors where product integrity and safety are paramount.
Sales of PCR packaging for pharma and OTC products in the USA are increasing as pharmaceutical manufacturers respond to sustainability goals and regulatory pressures. Growth at 10.6% CAGR reflects the increasing use of PCR packaging in OTC products, medicines, and supplements as the USA. market prioritizes eco-friendly packaging solutions. The shift towards sustainable materials is driven by consumer preference for products with reduced environmental impact. Manufacturers in the pharmaceutical and healthcare industries are exploring cost-effective ways to incorporate PCR content into packaging while maintaining product safety and regulatory compliance.
PCR packaging for pharma and OTC products is gaining traction in Germany as the country leads in sustainability and circular economy practices. Growth at 11.0% CAGR reflects Germany’s strong commitment to reducing plastic waste and improving recycling systems. As the pharmaceutical and healthcare sectors embrace green packaging alternatives, PCR materials are increasingly used to meet both regulatory standards and consumer demand for eco-friendly products. Germany's robust recycling infrastructure and regulations supporting the use of recycled content in packaging further boost demand for PCR packaging in the pharma sector.
Demand for PCR packaging for pharma and OTC products in Japan is increasing as the country focuses on improving its recycling rates and reducing plastic waste. Growth at 9.8% CAGR reflects Japan’s commitment to sustainable packaging solutions, particularly in the pharmaceutical and OTC sectors. The government’s push for greater use of recycled materials, along with consumer demand for eco-friendly products, is encouraging the adoption of PCR packaging in pharma products. Manufacturers are exploring ways to incorporate recycled content into packaging without compromising product quality or safety, driving demand for high-quality PCR packaging materials.
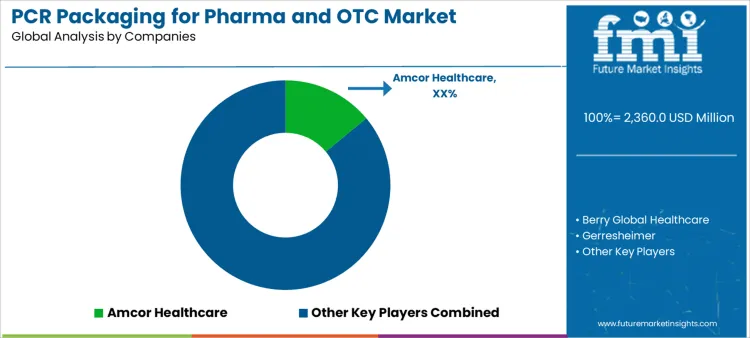
The PCR packaging for pharma and OTC market is driven by increasing demand for sustainable packaging solutions in the pharmaceutical and over-the-counter (OTC) sectors, as well as rising regulatory pressures for recycled content usage. Manufacturers assess packaging strength, moisture and oxygen barrier properties, and compliance with stringent pharmaceutical regulations. Buyer evaluation includes the ability to maintain product integrity, compatibility with sterilization methods, and recyclability without compromising safety or efficacy. Procurement behavior reflects preference for suppliers offering high-quality PCR packaging solutions that meet regulatory requirements while delivering the same performance as virgin materials. The market trend is shifting towards adopting PCR in packaging materials to align with circular economy goals and sustainability initiatives.
Amcor Healthcare leads competitive positioning with its broad range of sustainable packaging solutions, including high-quality PCR content integration while meeting regulatory standards for pharmaceuticals. Berry Global Healthcare competes by offering a wide variety of PCR packaging options designed for both pharma and OTC products, with a focus on robust performance and compliance. Gerresheimer stands out through its specialized glass and plastic packaging solutions that incorporate PCR content while ensuring high-quality standards for the pharmaceutical sector. SCHOTT supports demand with its high-quality pharmaceutical glass packaging, now including sustainable options with PCR materials, and a strong presence in regulated markets. Shandong Pharmaceutical Glass and Nipro provide strong regional competition, focusing on the integration of PCR content into pharmaceutical packaging pharmaceutical packaging while maintaining cost-effectiveness. Toyo Seikan participates with sustainable packaging innovations catering to the OTC market, especially in Asia. Competitive differentiation is driven by packaging performance, regulatory adherence, environmental impact, and ability to scale production while maintaining quality standards.
| Items | Values |
|---|---|
| Quantitative Units | USD million |
| Packaging Type | Bottles, blisters & jars; Primary pharma packaging; Rigid pharma containers; Diagnostic packaging; Others |
| Compliance Technology | FDA / EU GMP-validated PCR; Controlled clean-room conversion; Sterile PCR processing; Ultra-low contamination control; Others |
| End-Use | OTC bottles & blister packs; Prescription pharma; Domestic pharma; Precision medical packs; Generic pharma exports |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, USA, Germany, Japan, and 40+ countries |
| Key Companies Profiled | Amcor Healthcare; Berry Global Healthcare; Gerresheimer; SCHOTT; Shandong Pharmaceutical Glass; Nipro; Toyo Seikan; Others |
| Additional Attributes | Dollar sales by packaging type, compliance technology, and end-use; role of FDA and EU GMP-validated PCR in ensuring safety and quality across pharma packaging; performance of clean-room and sterile PCR processing in maintaining packaging integrity; impact on domestic and export pharmaceutical packaging needs; procurement dynamics shaped by global pharmaceutical packaging regulations and long-term supplier partnerships in the healthcare sector. |
How big is the pcr packaging for pharma and otc market in 2026?
The global pcr packaging for pharma and otc market is estimated to be valued at USD 2,360.0 million in 2026.
What will be the size of pcr packaging for pharma and otc market in 2036?
The market size for the pcr packaging for pharma and otc market is projected to reach USD 6,946.5 million by 2036.
How much will be the pcr packaging for pharma and otc market growth between 2026 and 2036?
The pcr packaging for pharma and otc market is expected to grow at a 11.4% CAGR between 2026 and 2036.
What are the key product types in the pcr packaging for pharma and otc market?
The key product types in pcr packaging for pharma and otc market are bottles, blisters & jars, primary pharma packaging, rigid pharma containers, diagnostic packaging and others.
Which compliance technology segment to contribute significant share in the pcr packaging for pharma and otc market in 2026?
In terms of compliance technology, fda / eu gmp-validated pcr segment to command 54.0% share in the pcr packaging for pharma and otc market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.