Pharmaceutical Glass Packaging Market : Global Industry Analysis 2016 - 2025 and Opportunity Assessment 2026 - 2036
Pharmaceutical Glass Packaging Market Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 - 2025 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2026 - 2036
Pharmaceutical Glass Packaging Market Forecast and Outlook 2026 to 2036
The global pharmaceutical glass packaging market is forecasted to reach USD 8.7 billion in 2026 and expand to USD 14.2 billion by 2036, advancing at a CAGR of 5.1%. Government regulations for pharmaceutical glass packaging focus on safety, sterility, and compatibility with drug formulations.
Regulatory authorities require testing and certification of glass containers before they can be manufactured, imported, sold, or used in pharmaceutical applications. These requirements ensure that glass packaging materials used for drug storage and delivery do not interact with pharmaceutical compounds or compromise drug stability.
Compliance with pharmacopeial standards is a key regulatory aspect. Governments mandate alignment with recognized pharmaceutical testing standards that define acceptable methods for measuring chemical resistance, thermal shock resistance, and hydrolytic resistance. Adherence to these standards supports drug stability across formulations and ensures consistent packaging performance for pharmaceutical manufacturers and healthcare providers.
Chemical compatibility and leachable substance regulations also apply to pharmaceutical glass packaging. Containers must meet safety norms that protect drug integrity during storage and transportation. This includes compliance with extractable and leachable limits, surface treatment requirements, and barrier properties to prevent contamination risks.
Quick Stats for Pharmaceutical Glass Packaging Market
- Pharmaceutical Glass Packaging Market Value (2026): USD 8.7 billion
- Pharmaceutical Glass Packaging Market Forecast Value (2036): USD 14.2 billion
- Pharmaceutical Glass Packaging Market Forecast CAGR (2026-2036): 5.1%
- Leading Product Type by Demand Share: Vials
- Fastest-Growing Countries: India, China, USA, Germany, Japan
- Top Players in Global Demand: Schott AG, Gerresheimer AG, West Pharmaceutical Services, Bormioli Pharma, SGD Pharma
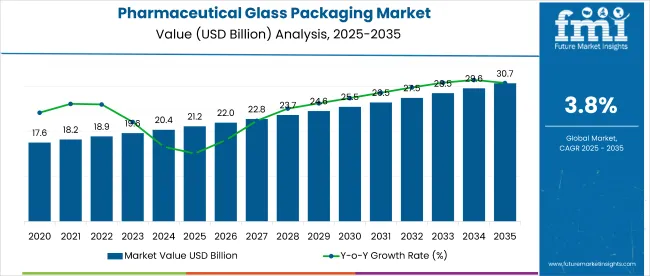
Pharmaceutical Glass Packaging Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 8.7 billion |
| Market Forecast Value (2036) | USD 14.2 billion |
| Forecast CAGR (2026-2036) | 5.1% |
Source: FMI analysis based on primary research and proprietary forecasting model
Who are the Major Pharmaceutical Glass Packaging Exporting and Importing Countries?
Major exporting countries for pharmaceutical glass packaging include manufacturing centers in Europe, North America, and Asia where production of pharmaceutical containers and specialty glass technologies is established. Germany is a leading exporter of pharmaceutical glass containers because of its advanced glass manufacturing ecosystem and specialized production capabilities. The United States and Italy are also significant exporters of pharmaceutical glass packaging to global markets. China and France contribute to exports through advanced glass container manufacturing and technology development. Other exporting countries that participate in the global supply chain include Czech Republic and India.
On the importing side, several countries with expanding pharmaceutical manufacturing and high packaging demand are primary markets for pharmaceutical glass containers, cites FMI. Brazil, Mexico, and India are among the top importers of such packaging to support drug manufacturing and healthcare infrastructure. The United States and China also import specialized pharmaceutical glass containers to complement domestic production and meet regulatory compliance needs. Other notable import markets include Russia and countries focused on pharmaceutical manufacturing expansion.
What are the Various Segments of the Pharmaceutical Glass Packaging Market?
Pharmaceutical glass packaging plays a critical role in delivering drug protection, sterility assurance, and chemical compatibility across injectable drugs, oral medications, and diagnostic applications. Adoption is influenced by formulation requirements, stability specifications, regulatory compliance, and end-use performance criteria. Segmentation by product type, application type, and glass type highlights how pharmaceutical manufacturers select specific packaging solutions to meet drug safety targets, storage capabilities, and quality standards across diverse therapeutic applications.
Which Product Type Holds the Largest Share in Pharmaceutical Glass Packaging?
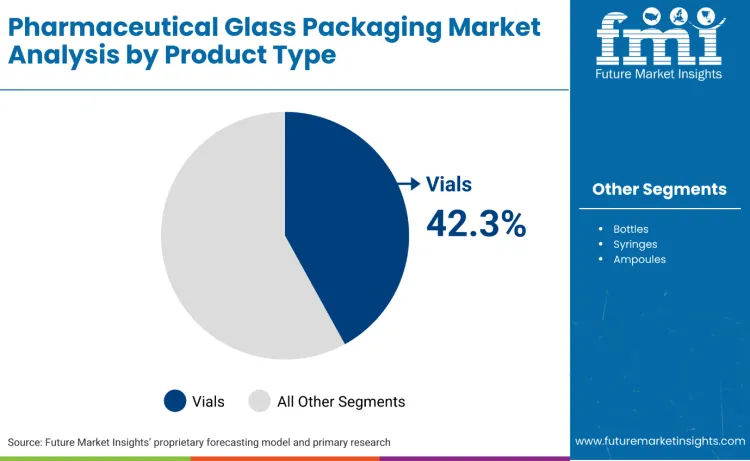
Vials account for 42.3%, driven by extensive use of vial boxes in injectable drug packaging, vaccine storage, and diagnostic sample collection. Bottles hold 28.4%, supporting oral medication packaging, liquid formulation storage, and pharmaceutical distribution applications. Syringes represent 16.7%, favored for pre-filled drug delivery, vaccination programs, and patient convenience applications. Ampoules contribute 8.9%, used in single-dose packaging and sterile drug administration processes. Other products account for 3.7%.
Key Points
- Vials dominate injectable drug and vaccine packaging applications.
- Bottles support oral medication and liquid formulation markets.
- Syringes address pre-filled drug delivery and convenience requirements.
How Do Application Requirements Influence Packaging Selection?
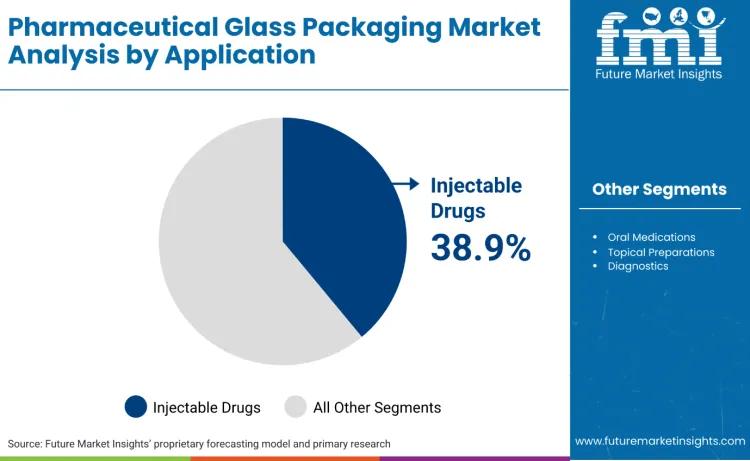
FMI is of the opinion that injectable drugs represent 38.9%, reflecting priority on applications requiring sterile packaging and chemical compatibility capabilities. Oral medications account for 31.2%, suitable for tablet packaging and liquid formulation storage applications requiring moisture protection and stability assurance. Topical preparations hold 18.6%, used for cream packaging and ointment storage requiring barrier properties and contamination prevention. Diagnostics contribute 7.8%, favored for sample collection and reagent storage requirements. Other applications account for 3.5%.
Key Points
- Injectable drugs remain the primary packaging segment.
- Oral medications serve solid and liquid formulation applications.
- Application selection depends on drug type and stability requirements.
What are the Certification Requirements in the Pharmaceutical Glass Packaging Market?
Certification requirements in the pharmaceutical glass packaging market focus on product safety, material integrity, and regulatory compliance across global supply chains. Glass containers must comply with pharmacopeial standards such as USP <660> in the United States and corresponding chapters in the European Pharmacopoeia and Japanese Pharmacopoeia, which define hydrolytic resistance, chemical durability, and extractables performance. Manufacturers supplying regulated markets must operate under current good manufacturing practices enforced by authorities such as USA Food and Drug Administration and the European Medicines Agency, covering quality systems, process validation, and traceability.
ISO 15378 certification is widely required, combining ISO 9001 quality management principles with GMP requirements specific to primary packaging for medicinal products. ISO 8362 standards apply to glass vials and cartridges, specifying dimensions, tolerances, and functional performance. Compliance with ISO 10993 risk assessment principles supports evaluation of material interactions. Environmental and occupational certifications such as ISO 14001 and ISO 45001 are increasingly expected by pharmaceutical buyers to ensure responsible manufacturing practices.
How Is the Pharmaceutical Glass Packaging Market Evolving Globally?
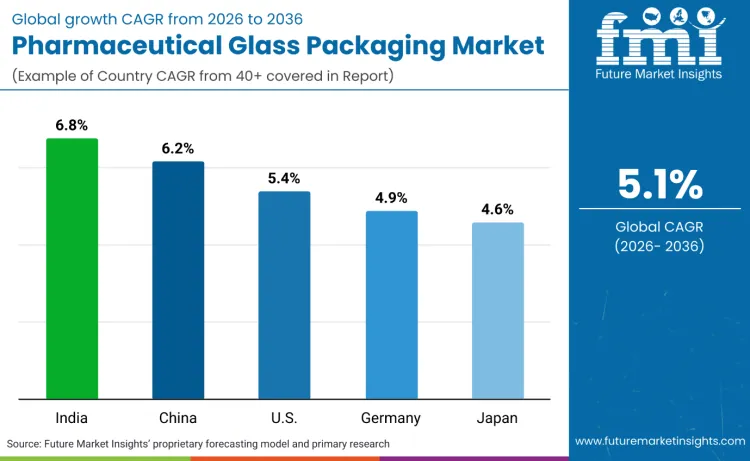
Global demand for pharmaceutical glass packaging is increasing as pharmaceutical manufacturers expand production capacity while addressing drug safety, regulatory compliance, and chemical compatibility application requirements.
Growth reflects rising use of high-quality packaging materials, specialized manufacturing protocols, and precision-controlled sterilization systems across drug production facilities, pharmaceutical companies, and healthcare manufacturing systems. Packaging selection focuses on chemical inertness, thermal stability, and barrier performance under various storage scenarios. India records 6.8% CAGR, China records 6.2% CAGR, USA records 5.4% CAGR, Germany records 4.9% CAGR, and Japan records 4.6% CAGR. Adoption remains driven by drug safety requirements and manufacturing efficiency rather than volume expansion alone.
| Country | CAGR (2026-2036) |
|---|---|
| India | 6.8% |
| China | 6.2% |
| USA | 5.4% |
| Germany | 4.9% |
| Japan | 4.6% |
Source: FMI analysis based on primary research and proprietary forecasting model
What is Driving Growth of Pharmaceutical Glass Packaging Demand in India?
Demand for pharmaceutical glass packaging in India is expanding as pharmaceutical facilities develop manufacturing capabilities and quality improvement programs. Growth at 6.8% CAGR reflects rising use of high-quality packaging materials, pharmaceutical manufacturing applications, and drug production systems addressing healthcare and export market requirements. Packaging quality improvement under manufacturing conditions remains critical for drug safety and regulatory performance acceptance. Cost considerations encourage glass packaging delivering chemical inertness and thermal stability at competitive price points. Pharmaceutical engineers prioritize containers compatible with existing manufacturing infrastructure and sterilization capabilities. Demand concentrates within pharmaceutical manufacturing clusters, drug production zones, and healthcare facilities serving domestic and international markets.
- Expansion of pharmaceutical manufacturing capabilities
- Need for drug production and quality assurance solutions
- Cost-effective high-quality packaging materials
- Growth of pharmaceutical export capacity
Why is China Seeing Expansion in Pharmaceutical Glass Packaging Adoption?
Pharmaceutical glass packaging demand in China is rising as medical packaging manufacturers expand quality enhancement initiatives. Growth at 6.2% CAGR reflects strong activity in pharmaceutical manufacturing, biopharmaceutical production, and specialty drug packaging applications. Large-scale healthcare development is driving demand for containers delivering chemical inertness, thermal performance, and manufacturing efficiency. Pharmaceutical facilities increase utilization of precision glass packaging due to quality requirements and international regulatory specifications. Domestic healthcare policies support advanced drug manufacturing and packaging quality standards. Demand remains centered on major pharmaceutical regions and integrated manufacturing complexes serving national and global markets.
- Expansion of pharmaceutical manufacturing programs
- Healthcare system support for advanced drug production
- Growth of biopharmaceutical production and specialty drug packaging services
- Development of precision manufacturing capabilities
What Factors are Shaping Pharmaceutical Glass Packaging Sales in USA?
Sales of pharmaceutical glass packaging in USA are increasing as manufacturers expand drug production facilities and quality improvement capabilities aligned with pharmaceutical services and healthcare processing needs. Growth at 5.4% CAGR reflects rising use in drug manufacturing, biopharmaceutical packaging, and specialty packaging applications. Container performance under manufacturing conditions and supply chain reliability drives technology selection. Pharmaceutical financing mechanisms increase access to high-quality packaging for qualifying manufacturing facilities. Drug production programs focus on chemical inertness and thermal performance to demonstrate product value. Demand remains strongest within pharmaceutical manufacturing centers and healthcare facilities serving regional markets.
- Expansion of drug production facilities
- Manufacturer access to precision packaging
- Focus on chemical inertness and thermal performance
- Growth of pharmaceutical manufacturing and specialty packaging services
How is Germany Supporting Growth of Pharmaceutical Glass Packaging Demand?
FMI states that pharmaceutical glass packaging market growth in Germany is advancing as manufacturing systems expand drug production capabilities across pharmaceutical manufacturing, specialty packaging, and healthcare applications. Growth at 4.9% CAGR reflects strong use in drug manufacturing, regulatory compliance, and quality assurance applications. Advanced pharmaceutical operations introduce complex packaging scenarios requiring consistent container performance and chemical reliability. Manufacturing strategies prioritize containers with proven quality characteristics and sterilization stability. Major pharmaceutical facilities emphasize packaging specifications and quality control protocols to optimize manufacturing efficiency. Demand is driven by performance evidence and cost effectiveness rather than raw packaging availability trends.
- Expansion of pharmaceutical manufacturing and specialty packaging programs
- Advanced pharmaceutical facility management
- Focus on quality characteristics and sterilization reliability
- Growth of precision manufacturing centers
What is Driving Expansion of Pharmaceutical Glass Packaging Demand in Japan?
Demand for pharmaceutical glass packaging in Japan is rising as drug manufacturing programs expand quality capabilities across pharmaceutical production, healthcare manufacturing, and precision packaging applications. Growth at 4.6% CAGR reflects utilization driven by quality standards and performance-based packaging protocols. Container consistency under long-term storage scenarios influences technology selection and manufacturing management approaches. Pharmaceutical operators prioritize systems supporting chemical inertness, thermal stability, and sterilization performance. Quality acceptance standards elevate focus on chemical compatibility, container reliability, and supply consistency. Demand remains tied to pharmaceutical manufacturing program development and healthcare capacity rather than general packaging expansion.
- Quality standard-driven packaging utilization
- Expansion of pharmaceutical manufacturing programs
- Focus on chemical compatibility and thermal stability
- Performance-based packaging supply protocols
Who are the Major Players Active in the Pharmaceutical Glass Packaging Market?
Performance requirements in drug manufacturing, pharmaceutical packaging, and precision applications are shaping demand for pharmaceutical glass packaging. Pharmaceutical engineers assess chemical inertness, thermal stability, sterilization compatibility, and supply reliability during container selection processes. Quality assessment includes chemical resistance, barrier properties, manufacturing compatibility, and integration with existing pharmaceutical production protocols.
Procurement behavior reflects extensive performance validation, sterilization reliability requirements, and reliance on suppliers offering technical support and consistent packaging specifications. Trends in the pharmaceutical glass packaging market reflect emphasis on drug safety and manufacturing efficiency in pharmaceutical production, healthcare manufacturing, and specialty packaging applications.
Schott AG accounts for a leading position through integrated pharmaceutical glass operations designed to deliver chemical precision, container reliability, and supply consistency capabilities. Gerresheimer AG competes by combining high-quality glass containers with comprehensive sterilization systems supporting manufacturing performance across diverse pharmaceutical applications. West Pharmaceutical Services supports demand through specialized packaging platforms aligned with pharmaceutical production and regulatory compliance requirements.
Bormioli Pharma maintains relevance by supplying precision glass containers enhancing drug protection and packaging performance potential. SGD Pharma participates with packaging emphasizing chemical inertness and manufacturing integration. Competitive differentiation centers on container quality, sterilization reliability, technical specifications, and depth of pharmaceutical engineering collaboration.
Key Players in the Pharmaceutical Glass Packaging Market
- Schott AG
- Gerresheimer AG
- West Pharmaceutical Services
- Bormioli Pharma
- SGD Pharma
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Product Type | Vials; Bottles; Syringes; Ampoules; Others |
| Application | Injectable Drugs; Oral Medications; Topical Preparations; Diagnostics; Others |
| Glass Type | Borosilicate Glass; Soda-lime Glass; Others |
| End-Use Industry | Pharmaceutical Companies; Biotechnology; Contract Manufacturing; Research Institutions; Others |
| Regions Covered | Asia Pacific, Europe, North America, Latin America, Middle East & Africa |
| Countries Covered | India, China, USA, Germany, Japan, and 40+ countries |
| Key Companies Profiled | Schott AG; Gerresheimer AG; West Pharmaceutical Services; Bormioli Pharma; SGD Pharma; Others |
| Additional Attributes | Dollar sales by product type, application, and glass type; performance in chemical inertness and thermal stability across pharmaceutical manufacturing, drug production, and healthcare matrices; chemical compatibility improvement, manufacturing efficiency enhancement, and quality benefit under pharmaceutical operations; impact on drug protection, sterilization control, and barrier reliability during manufacturing processes; compatibility with pharmaceutical systems and quality control targets; procurement dynamics driven by pharmaceutical expansion, quality improvement programs, and long-term packaging supply partnerships. |
Source: FMI analysis based on primary research and proprietary forecasting model
Pharmaceutical Glass Packaging Market by Segments
Product Type:
- Vials
- Bottles
- Syringes
- Ampoules
- Others
Application:
- Injectable Drugs
- Oral Medications
- Topical Preparations
- Diagnostics
- Others
Glass Type:
- Borosilicate Glass
- Soda-lime Glass
- Others
End-Use Industry:
- Pharmaceutical Companies
- Biotechnology
- Contract Manufacturing
- Research Institutions
- Others
Region:
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- North America
- USA
- Canada
- Mexico
- Latin America
- Brazil
- Chile
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
Bibliography
- Pharmaceutical Glass Manufacturing Association. (2023). Container Standards for Drug Packaging: Technical Guidelines. PGMA.
- International Pharmaceutical Glass Institute. (2023). Package Specifications for Glass Containers: Quality and Sterilization Requirements. IPGI.
- Society of Pharmaceutical Engineering Technology. (2024). SPET Guidelines for Container Selection in Pharmaceutical Manufacturing (updated quality specifications for chemical compatibility and thermal applications). SPET.
- Martinez, C. L., et al. (2023). Chemical compatibility effects of precision pharmaceutical glass packaging on drug manufacturing efficiency: A comprehensive analysis of pharmaceutical and biotechnology applications. Journal of Pharmaceutical Manufacturing Technology, 31, 78-95.
- Thompson, R. K., et al. (2023). Sixth annual pharmaceutical glass packaging report: Special focus on sterilization reliability and chemical compatibility in drug manufacturing. International Pharmaceutical Review, 28, 1-22.
Frequently Asked Questions
How big is the pharmaceutical glass packaging market in 2026?
The global pharmaceutical glass packaging market is estimated to be valued at USD 8.7 billion in 2026.
What will be the size of the pharmaceutical glass packaging market in 2036?
The market size for the pharmaceutical glass packaging market is projected to reach USD 14.2 billion by 2036.
How much will the pharmaceutical glass packaging market grow between 2026 and 2036?
The pharmaceutical glass packaging market is expected to grow at a 5.1% CAGR between 2026 and 2036.
What are the key product types in the pharmaceutical glass packaging market?
The key product types in the pharmaceutical glass packaging market include vials, bottles, syringes, ampoules, and other specialized pharmaceutical containers.
Which product segment will contribute a significant share in the pharmaceutical glass packaging market in 2026?
In terms of product type, the vials segment is set to command a 42.3% share in the pharmaceutical glass packaging market in 2026.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand Side Trends
- Supply Side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Research Methodology
- Chapter Orientation
- Analytical Lens and Working Hypotheses
- Market Structure, Signals, and Trend Drivers
- Benchmarking and Cross-market Comparability
- Market Sizing, Forecasting, and Opportunity Mapping
- Research Design and Evidence Framework
- Desk Research Programme (Secondary Evidence)
- Company Annual and Sustainability Reports
- Peer-reviewed Journals and Academic Literature
- Corporate Websites, Product Literature, and Technical Notes
- Earnings Decks and Investor Briefings
- Statutory Filings and Regulatory Disclosures
- Technical White Papers and Standards Notes
- Trade Journals, Industry Magazines, and Analyst Briefs
- Conference Proceedings, Webinars, and Seminar Materials
- Government Statistics Portals and Public Data Releases
- Press Releases and Reputable Media Coverage
- Specialist Newsletters and Curated Briefings
- Sector Databases and Reference Repositories
- Roots Internal Knowledge Base and Historical Datasets
- Subscription Datasets and Paid Sources
- Social Channels, Communities, and Digital Listening Inputs
- Additional Desk Sources
- Expert Input and Fieldwork (Primary Evidence)
- Qualitative Interviews and Expert Elicitation
- Quantitative Surveys and Structured Data Capture
- Blended Approach
- Governance, Ethics, and Data Stewardship
- Research Ethics
- Data Integrity and Handling
- Desk Research Programme (Secondary Evidence)
- Tooling, Models, and Reference Databases
- Data Engineering and Model Build
- Data Acquisition and Ingestion
- Cleaning, Normalisation, and Verification
- Synthesis, Triangulation, and Analysis
- Quality Assurance and Audit Trail
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter's Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Pharmaceutical Glass Packaging Market Analysis 2021 to 2025 and Forecast, 2026 to 2036
- Historical Market Size Value (USD Billion) Analysis, 2021 to 2025
- Current and Future Market Size Value (USD Billion) Projections, 2026 to 2036
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Pharmaceutical Glass Packaging Market Pricing Analysis 2021 to 2025 and Forecast 2026 to 2036
- Y-o-Y Growth Trend Analysis 2021 to 2025
- Absolute $ Opportunity Analysis 2026 to 2036
- Global Pharmaceutical Glass Packaging Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Product Type
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Product Type, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Product Type, 2026 to 2036
- Vials
- Bottles
- Syringes
- Ampoules
- Others
- Global Pharmaceutical Glass Packaging Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Billion) Analysis By Application, 2021 to 2025
- Market Size Value (USD Billion) Forecast By Application, 2026 to 2036
- Injectable Drugs
- Oral Medications
- Topical Preparations
- Diagnostics
- Others
- Global Pharmaceutical Glass Packaging Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Glass Type
- Borosilicate Glass
- Soda-lime Glass
- Others
- Global Pharmaceutical Glass Packaging Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Region
- North America
- Latin America
- Europe
- Asia Pacific
- Middle East & Africa
- North America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- USA
- Canada
- Mexico
- Latin America Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Brazil
- Chile
- Rest of Latin America
- Europe Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic
- BENELUX
- Rest of Europe
- Asia Pacific Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- China
- Japan
- South Korea
- India
- Australia & New Zealand
- ASEAN
- Rest of Asia Pacific
- Middle East & Africa Market Analysis 2021 to 2025 and Forecast 2026 to 2036, By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkey
- South Africa
- Other African Union
- Rest of Middle East & Africa
- Key Countries Market Analysis
- Pricing Analysis
- Market Share Analysis By Product Type
- Market Share Analysis By Application
- Market Share Analysis By Glass Type
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Region
- By Product Type
- By Application
- Competition Analysis
- Competition Deep Dive
- Schott AG
- Gerresheimer AG
- West Pharmaceutical Services
- Bormioli Pharma
- SGD Pharma
- Competition Deep Dive
- Assumptions & Acronyms Used
List of Tables
- Pharmaceutical Glass Packaging Market Key Takeaways
- Global Pharmaceutical Glass Packaging Market Size (USD Billion), 2021-2036
- Global Pharmaceutical Glass Packaging Market Y-o-Y Growth (%), 2021-2036
- Global Pharmaceutical Glass Packaging Market Absolute $ Opportunity Analysis (USD Billion), 2026-2036
- Global Pharmaceutical Glass Packaging Market Pricing Analysis, 2021-2036
- Global Market Size By Product Type, 2021-2036
- Global Market Size By Application, 2021-2036
- Global Market Size By Glass Type, 2021-2036
- Global Market Size By Region, 2021-2036
- Market Share Analysis By Product Type, 2025
- Market Share Analysis By Application, 2025
- Market Share Analysis By Glass Type, 2025
- Company-wise Market Share Analysis, 2025
List of Figures
- Pharmaceutical Glass Packaging Market Size (USD Billion), 2021-2036
- Pharmaceutical Glass Packaging Market Y-o-Y Growth (%), 2021-2036
- Pharmaceutical Glass Packaging Market Absolute $ Opportunity Analysis
- Pharmaceutical Glass Packaging Market Value Chain Analysis
- Pharmaceutical Glass Packaging Market Supply Chain Analysis
- Pharmaceutical Glass Packaging Market Investment Feasibility Matrix
- Pharmaceutical Glass Packaging Market PESTLE Analysis
- Pharmaceutical Glass Packaging Market Porter’s Five Forces Analysis
- Pharmaceutical Glass Packaging Market Product Life Cycle Analysis
- Pharmaceutical Glass Packaging Market Opportunity Map Analysis
- Global Market Share By Product Type, 2025 and 2036
- Global Market Share By Application, 2025 and 2036
- Global Market Share By Glass Type, 2025 and 2036
- Global Market Share By Region, 2025 and 2036
- Competitive Landscape Dashboard

