Pharmaceutical Unit Dose Packaging Market
Pharmaceutical Unit Dose Packaging Market Analysis Size and Share Forecast Outlook 2026 to 2036
Historical Data Covered: 2016 to 2024 | Base Year: 2025 | Estimated Year: 2026 | Forecast Period: 2027 to 2036
Pharmaceutical Unit Dose Packaging Market Forecast and Outlook 2026 to 2036
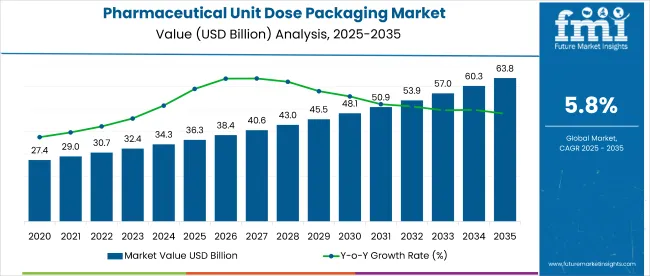
The pharmaceutical unit dose packaging market is expected to be worth about USD 3.5 billion in 2026 and is forecast to grow to USD 9.8 billion by 2036, reflecting a strong CAGR of 10.8%. This growth is closely tied to the healthcare industry’s push for safer, simpler, and more reliable ways to give patients their medications. Unit dose packaging provides each medicine in a ready to use, single serving pack, which helps reduce dosing mistakes, improves hygiene, and makes it easier for nurses, pharmacists, and patients to use medicines correctly.
Demand is rising as hospitals, clinics, and care homes look for better ways to manage large volumes of medications, especially for older patients and those with chronic illnesses. Individually packaged doses make it easier to track what has been given, lower the risk of contamination, and cut down on wasted medicines. The growing use of high value and specialty drugs is also boosting the need for secure, tamper evident packaging that protects product quality.
Producing unit dose packs is more complex and expensive than traditional bottle packaging, and it must meet very strict regulatory standards. To keep up, packaging companies are investing in faster, more automated equipment and smarter packaging features that improve efficiency, support tracking and tracing, and ensure every dose reaches the patient in a safe and reliable way.
Quick Stats for Pharmaceutical Unit Dose Packaging Market
- Pharmaceutical Unit Dose Packaging Market Value 2026: USD 3.5 billion
- Pharmaceutical Unit Dose Packaging Market Forecast Value 2036: USD 9.8 billion
- Pharmaceutical Unit Dose Packaging Market Forecast CAGR 2026 to 2036: 10.8%
- Leading Product Category in Pharmaceutical Unit Dose Packaging Market 2026: Blister packaging with about 58% market share
- Key Growth Regions in Pharmaceutical Unit Dose Packaging Market: North America, Europe, Asia Pacific
- Key Players in Pharmaceutical Unit Dose Packaging Market: Amcor plc, Gerresheimer AG, West Pharmaceutical Services Inc., Schott AG, Berry Global Inc.
Pharmaceutical Unit Dose Packaging Market Key Takeaways
| Metric | Value |
|---|---|
| Market Value (2026) | USD 3.5 billion |
| Market Forecast Value (2036) | USD 9.8 billion |
| Forecast CAGR (2026 to 2036) | 10.8% |
How Is the Pharmaceutical Unit Dose Packaging Market Segmented?
The Pharmaceutical Unit Dose Packaging Market is segmented by packaging type, material type, end use application, and region to support safe, accurate, and efficient medication dispensing. By packaging type, the industry includes blister packaging, strip packaging, pouch packaging, vial and ampoule packaging, pre filled syringes, and other unit dose formats. By material type, solutions use plastic, aluminum foil, paper and paperboard, and glass depending on protection and sterility requirements.
By end use application, demand comes from hospital and institutional pharmacies, retail pharmacies, long term care facilities, clinics, and other healthcare providers. Regionally, the industry covers North America, Europe, Asia Pacific, Latin America, and the Middle East & Africa, reflecting differences in healthcare infrastructure, regulatory standards, and medication distribution systems. This segmentation structure supports precision dosing, patient safety, and regulatory compliance across diverse healthcare environments.
Why Does Blister Packaging Dominate the Pharmaceutical Unit Dose Packaging Market?
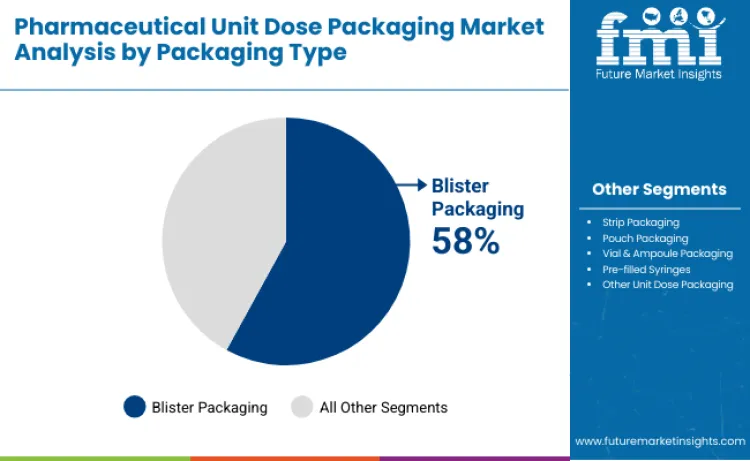
Blister packaging accounts for 58% of total demand because it offers superior protection, tamper resistance, and dose visibility for a wide range of medications. Each tablet or capsule is sealed in its own cavity, protecting it from moisture, oxygen, and contamination while allowing easy identification and tracking. Healthcare providers favor blister packs because they reduce medication errors, simplify inventory management, and support patient adherence through clear labeling. Blister packaging is also highly compatible with automated dispensing and serialization systems, which are increasingly used in hospitals and pharmacies. Manufacturers continue to improve barrier films and sealing technologies, making blister packs suitable for both standard pharmaceuticals and sensitive specialty drugs, reinforcing their leading position in the unit dose packaging market.
Why Do Hospital and Institutional Pharmacies Lead End Use Demand?
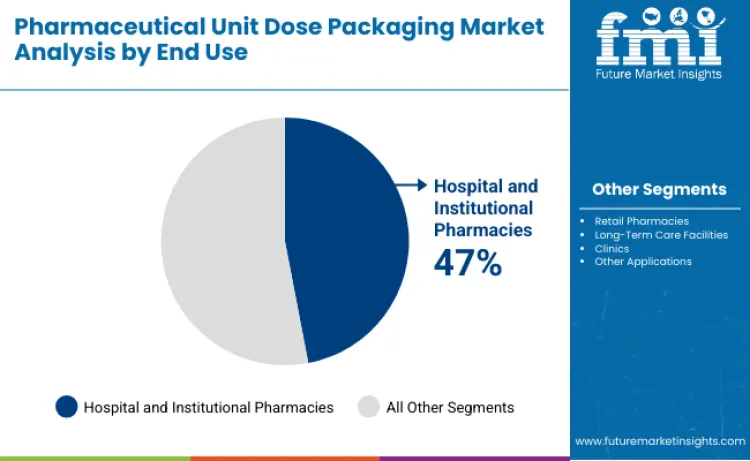
Hospital and institutional pharmacies represent 47% of market growth because they handle large volumes of high risk and time critical medications. These settings require precise dose control, strong traceability, and strict safety standards to protect patients and avoid dispensing errors. Unit dose packaging allows hospitals to prepare and distribute medications more efficiently while maintaining accurate records for compliance and auditing. The rise of automated medication dispensing cabinets and centralized pharmacy systems further increases the use of unit dose formats. As hospitals focus more on patient safety, infection control, and workflow efficiency, they continue to be the primary drivers of demand for pharmaceutical unit dose packaging.
What Are the Key Drivers, Trends, and Restraints in the Pharmaceutical Unit Dose Packaging Market?
The Pharmaceutical Unit Dose Packaging Market is driven by the growing emphasis on medication safety, accuracy, and traceability in modern healthcare systems. Hospitals, long term care facilities, and clinics are under increasing pressure to reduce medication errors, improve patient outcomes, and comply with strict regulatory requirements, all of which favor unit dose formats.
The rising use of high value specialty drugs, biologics, and oncology therapies further increases demand for packaging that ensures precise dosing and product integrity. Key trends include the integration of smart packaging features such as serialization, barcodes, and authentication technologies that support real time tracking, anti counterfeiting, and automated dispensing. There is also strong momentum toward environmentally friendly materials, including recyclable and biodegradable films, as healthcare providers seek to reduce packaging waste.
The industry faces restraints from higher production and material costs compared to bulk packaging, increased waste generation, and complex supply chains that can impact pricing and availability. Compliance with diverse regional regulations also adds operational complexity for manufacturers.
How Is the Pharmaceutical Unit Dose Packaging Market Shaping Safer Drug Delivery Worldwide?
The pharmaceutical unit dose packaging market is playing a growing role in improving how medicines are distributed and administered across healthcare systems. From 2026 to 2036, rising hospital admissions, growth in chronic disease treatment, and expansion of outpatient care are increasing demand for precise and reliable packaging. Unit dose packs reduce the risk of medication errors, improve hygiene, and simplify tracking for both hospitals and pharmacies.
Pharmaceutical companies also benefit because individually packed doses protect product integrity and support accurate labeling. Digital health systems are further increasing the value of unit dose packaging by linking each dose to patient records. These combined trends are turning unit dose packaging into a core part of modern pharmaceutical supply chains across developed and emerging markets.
| Country | CAGR |
|---|---|
| China | 12.9% |
| United States | 11.1% |
| Germany | 10.6% |
| United Kingdom | 10.3% |
| Japan | 10.0% |
How Is China Becoming the Largest Growth Engine for Unit Dose Packaging?
China’s pharmaceutical unit dose packaging market is projected to grow at a CAGR of 12.9% from 2026 to 2036, driven by large scale healthcare expansion and rapid pharmaceutical modernization. Hospitals across major cities are shifting toward standardized medication dispensing systems that require unit dose formats. Growing use of biologics, injectables, and specialty drugs further increases the need for individual packaging. Pharmaceutical manufacturers are investing in advanced blister and sachet technologies to serve hospital pharmacies and outpatient clinics. Government efforts to improve medication safety and traceability are accelerating adoption. As China continues to upgrade its healthcare infrastructure, unit dose packaging is becoming essential for efficient drug delivery and patient protection.
How Is the United States Reinforcing Unit Dose Packaging Demand?
The United States pharmaceutical unit dose packaging market is expected to grow at a CAGR of 11.1% from 2026 to 2036, supported by strict safety standards and a highly developed hospital network. Hospitals and long term care facilities depend on unit dose systems to minimize dispensing errors and improve workflow. Pharmaceutical companies increasingly offer ready to administer doses to meet clinical needs. Growth in home healthcare and specialty pharmacy services also supports wider use of individual packaging. Digital prescription and barcoding systems further strengthen the role of unit dose formats. Together, these factors keep the United States at the forefront of unit dose packaging adoption.
How Is Germany Expanding Its Use of Pharmaceutical Unit Dose Packaging?
Germany’s pharmaceutical unit dose packaging market is forecast to grow at a CAGR of 10.6% from 2026 to 2036, reflecting its strong focus on medication accuracy and healthcare quality. Hospitals and pharmacies are adopting unit dose systems to improve dispensing control and reduce drug waste. Pharmaceutical manufacturers are aligning packaging with European safety and labeling standards, making unit dose formats more common. Centralized pharmacy services and hospital automation also favor individually packed medications. As Germany continues to upgrade its healthcare delivery systems, unit dose packaging is gaining importance as a reliable way to improve efficiency and patient safety.
How Is the United Kingdom Integrating Unit Dose Packaging into Healthcare Systems?
The United Kingdom pharmaceutical unit dose packaging market is projected to grow at a CAGR of 10.3% from 2026 to 2036 as hospitals and clinics emphasize safer medication practices. NHS facilities are expanding centralized dispensing and ward level distribution systems that rely on unit dose packs. Pharmaceutical suppliers are providing more drugs in ready to use formats, helping to streamline administration. The growing burden of chronic illness and an aging population further support demand. With continued focus on safety, traceability, and efficiency, unit dose packaging is becoming a standard part of the UK healthcare supply chain.
How Is Japan Supporting Steady Growth in Unit Dose Packaging?
Japan’s pharmaceutical unit dose packaging market is expected to grow at a CAGR of 10.0% from 2026 to 2036, driven by an aging population and strong emphasis on healthcare quality. Hospitals and community pharmacies use unit dose formats to ensure correct dosing and prevent contamination. Pharmaceutical companies design packaging that supports convenience and precision, especially for outpatient and home care. As Japan’s healthcare system adapts to rising demand for long term treatment, individually packaged medicines help maintain safety and efficiency. These trends allow unit dose packaging to remain an important part of Japan’s pharmaceutical infrastructure.
What Is the Competitive Landscape of the Pharmaceutical Unit Dose Packaging Market?
The pharmaceutical unit dose packaging market is defined by competition among a concentrated group of global healthcare packaging suppliers and medication delivery specialists that serve hospital, pharmacy, and clinical distribution channels. Unlike conventional packaging segments, this market is driven by strict regulatory oversight, patient safety requirements, and the need for accurate, tamper evident, and contamination free dosing. As a result, purchasing decisions are influenced more by compliance, reliability, and technical performance than by packaging cost alone.
Leading companies such as Amcor plc, Gerresheimer AG, West Pharmaceutical Services Inc., Schott AG, and Berry Global Inc. occupy dominant positions through their advanced materials, sterile manufacturing environments, and strong regulatory expertise. These firms supply a wide range of unit dose formats including blister packs, vials, ampoules, prefilled syringes, and sachets that protect drug stability while enabling precise and consistent dosing across hospital and retail pharmacy settings. Their global production networks and quality systems allow them to support both large pharmaceutical companies and emerging biotech manufacturers.
AptarGroup Inc. and Becton, Dickinson and Company strengthen competition by integrating packaging with drug delivery technologies such as injectors, inhalers, and dispensing systems. This combination improves patient adherence, dosing accuracy, and healthcare workflow efficiency, making these suppliers highly attractive for complex therapies and self administered medications.
Catalent Inc. and CCL Industries Inc. expand competitive intensity through contract packaging, labeling, and clinical trial services that help pharmaceutical companies manage complex regulatory and serialization requirements. WestRock Company also plays a role through secondary unit dose cartons and pharmacy automation compatible packaging that supports inventory management and automated dispensing.
As healthcare systems increasingly focus on medication safety, traceability, and individualized dosing, competitive advantage in this market will continue to favor companies that can combine material performance, regulatory readiness, and seamless integration into clinical and pharmacy workflows.
Key Players in the Pharmaceutical Unit Dose Packaging Market
- • Amcor plc
- • Gerresheimer AG
- • West Pharmaceutical Services Inc.
- • Schott AG
- • Berry Global Inc.
- • AptarGroup Inc.
- • Becton, Dickinson and Company
- • Catalent Inc.
- • CCL Industries Inc.
- • WestRock Company
Scope of Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Market Size Reference | Market size in value terms |
| Market Parameter | Revenue in USD billion |
| Packaging Type | Blister packaging, strip packaging, pouch packaging, vial and ampoule packaging, pre filled syringes, other unit dose packaging |
| Material Type | Plastic, aluminum foil, paper and paperboard, glass |
| End Use Application | Hospital and institutional pharmacies, retail pharmacies, long term care facilities, clinics, other applications |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa, other regions |
| Countries Covered | United States, Germany, United Kingdom, China, Japan, and 40+ countries |
| Key Companies Profiled | Amcor plc, Gerresheimer AG, West Pharmaceutical Services Inc., Schott AG, Berry Global Inc., and other leading pharmaceutical packaging companies |
| Additional Attributes | Dollar sales by packaging type, material type, end use application, and region, regional demand trends, competitive landscape, technological advancements in materials engineering, safety design integration initiatives, quality enhancement programs, and premium product development strategies |
Pharmaceutical Unit Dose Packaging Market by Segments
By Packaging Type:
- Blister Packaging
- Strip Packaging
- Pouch Packaging
- Vial & Ampoule Packaging
- Pre-filled Syringes
- Other Unit Dose Packaging
By Material Type:
- Plastic
- Aluminum Foil
- Paper & Paperboard
- Glass
By End-Use Application:
- Hospital & Institutional Pharmacies
- Retail Pharmacies
- Long-Term Care Facilities
- Clinics
- Other Applications
By Region:
- North America
- United States
- Canada
- Mexico
- Europe
- Germany
- United Kingdom
- France
- Italy
- Spain
- Nordic Countries
- BENELUX
- Rest of Europe
- Asia Pacific
- China
- Japan
- South Korea
- India
- Australia
- Rest of Asia Pacific
- Latin America
- Brazil
- Argentina
- Rest of Latin America
- Middle East & Africa
- Kingdom of Saudi Arabia
- United Arab Emirates
- South Africa
- Rest of Middle East & Africa
- Other Regions
- Oceania
- Central Asia
- Other Markets
Bibliography
- World Health Organization. (2024). Medication safety in high-risk situations. World Health Organization.
- U.S. Food and Drug Administration. (2024). Drug Supply Chain Security Act (DSCSA): Requirements for packaging, labeling, and product tracing. U.S. Food and Drug Administration.
- European Commission. (2024). Safety features and serialization requirements for medicinal products under EU Falsified Medicines Directive. European Union.
- International Society for Pharmaceutical Engineering. (2024). Pharmaceutical packaging and labeling: Good practice guide. International Society for Pharmaceutical Engineering.
- International Organization for Standardization. (2024). ISO 15378: Primary packaging materials for medicinal products – Quality management systems with GMP integration. International Organization for Standardization.
Frequently Asked Questions
How big is the pharmaceutical unit dose packaging market in 2026?
The global pharmaceutical unit dose packaging market is valued at USD 3.5 billion in 2026.
What will be the size of the pharmaceutical unit dose packaging market in 2036?
The pharmaceutical unit dose packaging market is projected to reach USD 9.8 billion by 2036.
How much will the pharmaceutical unit dose packaging market grow between 2026 and 2036?
The pharmaceutical unit dose packaging market is expected to grow at a CAGR of 10.8% between 2026 and 2036.
What are the key product types in the pharmaceutical unit dose packaging market?
The key product types include blister packaging, strip packaging, pouch packaging, vial and ampoule packaging, pre filled syringes, and other unit dose formats.
Which end use application segment contributes the largest share in the pharmaceutical unit dose packaging market in 2026?
Hospital and institutional pharmacies lead with 47% share in 2026 due to high patient volumes and strict medication safety requirements.
Table of Content
- Executive Summary
- Market Overview
- Key Findings
- Growth Projections
- Market Overview
- Definition and Scope
- Market Segmentation
- Market Drivers and Restraints
- Trends and Opportunities
- Market Dynamics
- Growth Drivers
- Patient Safety and Medication Adherence
- Regulatory Standards and Compliance
- Technological Advancements in Packaging
- Challenges
- High Production and Material Costs
- Complex Supply Chain Management
- Opportunities
- Smart Packaging and Serialization
- Sustainability in Packaging
- Growth Drivers
- Market Segmentation
- By Packaging Type
- Blister Packaging
- Strip Packaging
- Pouch Packaging
- Vial and Ampoule Packaging
- Pre-filled Syringes
- Other Unit Dose Formats
- By Material Type
- Plastic
- Aluminum Foil
- Paper and Paperboard
- Glass
- By End-Use Application
- Hospital and Institutional Pharmacies
- Retail Pharmacies
- Long-Term Care Facilities
- Clinics
- Other Applications
- By Region
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa
- By Packaging Type
- Regional Market Analysis
- North America
- Europe
- Asia Pacific
- Latin America
- Middle East and Africa
- Competitive Landscape
- Key Players and Market Share Analysis
- Strategic Initiatives
- Market Positioning and Competitive Strategies
- Market Forecast
- Market Size and Growth Projections (2026-2036)
- Future Trends and Opportunities
- Forecast by Region, Packaging Type, and Material
- Key Players in the Pharmaceutical Unit Dose Packaging Market
- Amcor plc
- Gerresheimer AG
- West Pharmaceutical Services Inc.
- Schott AG
- Berry Global Inc.
- AptarGroup Inc.
- Becton, Dickinson and Company
- Catalent Inc.
- CCL Industries Inc.
- WestRock Company
- Conclusion and Strategic Recommendations
- Appendix
- Research Methodology
- Data Sources
- Glossary of Terms
List of Tables
- Table 1 Global Pharmaceutical Unit Dose Packaging Market Value and Volume Forecast 2026 to 2036
- Table 2 Pharmaceutical Unit Dose Packaging Market Value and Volume by Packaging Type 2026 to 2036
- Table 3 Pharmaceutical Unit Dose Packaging Market Value and Volume by Material Type 2026 to 2036
- Table 4 Pharmaceutical Unit Dose Packaging Market Value and Volume by End-Use Application 2026 to 2036
- Table 5 Pharmaceutical Unit Dose Packaging Market Value and Volume by Region 2026 to 2036
- Table 6 North America Pharmaceutical Unit Dose Packaging Market Value by End-Use Application 2026 to 2036
- Table 7 Europe Pharmaceutical Unit Dose Packaging Market Value by Packaging Type 2026 to 2036
List of Figures
- Figure 1 Global Pharmaceutical Unit Dose Packaging Market Growth by Packaging Type 2026 to 2036
- Figure 2 Global Pharmaceutical Unit Dose Packaging Market Growth by Material Type 2026 to 2036
- Figure 3 Global Pharmaceutical Unit Dose Packaging Market Growth by End-Use Application 2026 to 2036
- Figure 4 Global Pharmaceutical Unit Dose Packaging Market Growth by Region 2026 to 2036
- Figure 5 North America Pharmaceutical Unit Dose Packaging Market Share by Packaging Type 2026 to 2036
- Figure 6 Europe Pharmaceutical Unit Dose Packaging Market Share by Material Type 2026 to 2036
- Figure 7 Asia Pacific Pharmaceutical Unit Dose Packaging Market Growth Trends 2026 to 2036

