The global medical-grade PCR packaging market is likely to total USD 1,920 million in 2026 and is expected to rise to USD 5,840 million by 2036, advancing at an 11.8% CAGR. FMI analysis indicates that growth is being shaped by a structural shift in procurement logic. PCR is increasingly treated as a controlled input into regulated packaging systems, where the acceptance threshold is determined by validation rigor, contamination risk tolerance, and supplier documentation maturity rather than circularity claims.
Regulatory and quality frameworks are tightening the definition of what counts as acceptable recycled content in medical packaging contexts. For medical devices, the EU MDR intensifies accountability across technical documentation and conformity assessment, making packaging material decisions more exposed to scrutiny when sterility and integrity are at stake. For pharmaceuticals, compendial expectations for plastic packaging systems and material characterization are reinforcing a compliance environment where PCR use must be justified through structured suitability and safety evidence.
FMI also observes a parallel evolution in qualification pathways. Firms are investing in validation-grade PCR supply programs that can demonstrate consistent resin provenance, controlled processing, and traceable conversion into regulated packaging formats. This is accelerating as quality teams look to prevent revalidation churn and protect filing stability across product lifecycles, especially for high volume therapies and widely distributed medical device portfolios.
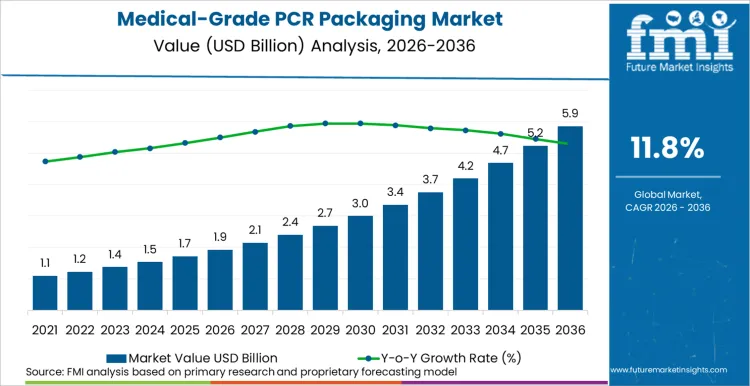
| Metric | Value |
|---|---|
| Expected Value 2026E | USD 1,920 million |
| Forecast Value 2036F | USD 5,840 million |
| CAGR 2026 to 2036 | 11.8% |
Source: Future Market Insights analysis based on proprietary forecasting model and primary research
The primary growth engine is the formalization of quality evidence requirements for packaging materials in regulated environments. In medical devices, sterile barrier expectations and validation disciplines supported by standards such as ISO 11607 increase the burden of proof for any material change, which includes introducing PCR content into packaging systems that must maintain sterility until point of use. This shifts adoption toward PCR programs that can be qualified without increasing validation volatility. (ISO)
A second driver is the rising strategic value of documentation readiness. Compendial and regulatory expectations for plastic packaging systems in pharma raise the importance of chemical characterization, suitability assessment, and controlled change management, which collectively favor PCR supply approaches that can be represented clearly in technical files and quality dossiers.
Growth is also supported by policy and system level pressure to strengthen plastics resource circulation, which expands the incentive for regulated sectors to develop compliant recycled content pathways without compromising patient safety or product integrity. This macro direction is visible across major markets that are strengthening plastics circularity policies and compliance mechanisms.
The segment structure reflects where medical-grade PCR adoption is operationally defensible under regulated packaging regimes. FMI retains only the dominant segment in each category and expands the analysis to reflect qualification depth, governance logic, and execution realism.
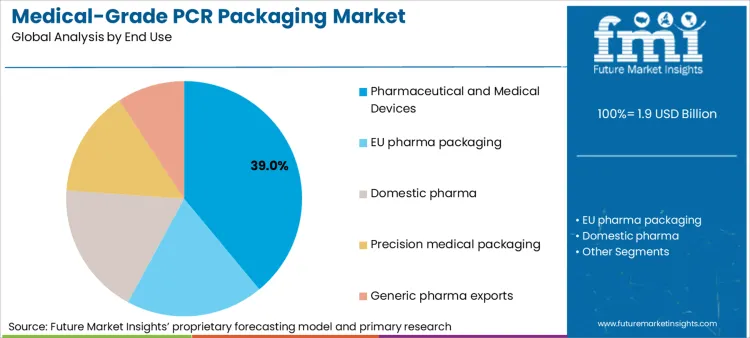
Pharmaceutical and medical devices account for 39% of demand, making this the structural anchor for medical-grade PCR adoption because it operates under the most explicit material accountability frameworks. Packaging decisions in this end use are directly linked to patient safety, sterility assurance, and regulatory filing stability, which elevates the importance of controlled material provenance and documented processing history. PCR adoption progresses only where recycled polymers can be integrated without increasing validation volatility or triggering additional regulatory scrutiny during audits and inspections.
A second reinforcing factor is lifecycle duration. Pharmaceutical and medical device packaging formats often remain unchanged for extended periods once validated, which means any material transition carries long-term compliance implications. This creates a strong preference for PCR solutions that can demonstrate long-run consistency in resin quality, traceability, and conversion conditions. FMI observes that adoption is most successful where PCR is introduced as part of a structured change control strategy rather than as a sustainability-driven substitution, ensuring continuity across markets, dossiers, and post-approval change management processes.
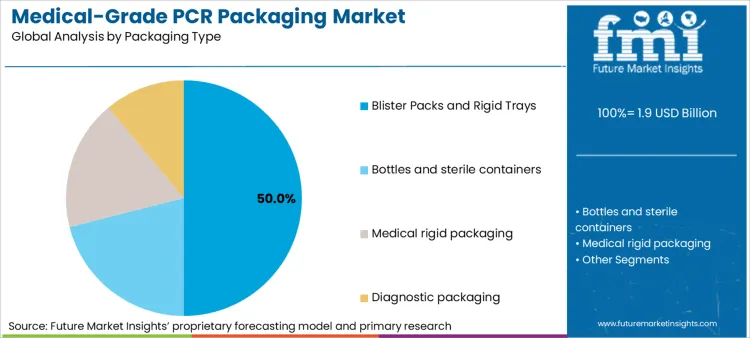
Blister packs and rigid trays represent 50% of packaging type demand because they offer the most standardized qualification and validation pathways within regulated packaging ecosystems. These formats are deeply embedded in pharmaceutical and device packaging supply chains, with well-established performance criteria for barrier integrity, dimensional stability, and sterilization compatibility. This maturity lowers the incremental risk of introducing PCR, provided that resin processing and forming conditions remain tightly controlled and documented.
From an operational perspective, these formats also benefit from clearer accountability across the value chain. Material suppliers, converters, and brand owners typically operate under defined roles with established audit expectations, which simplifies traceability and change management when PCR content is introduced. FMI analysis shows that converters favor these formats when scaling PCR because they allow quality teams to ring-fence risk, isolate variables, and demonstrate compliance without cascading revalidation across unrelated packaging systems.
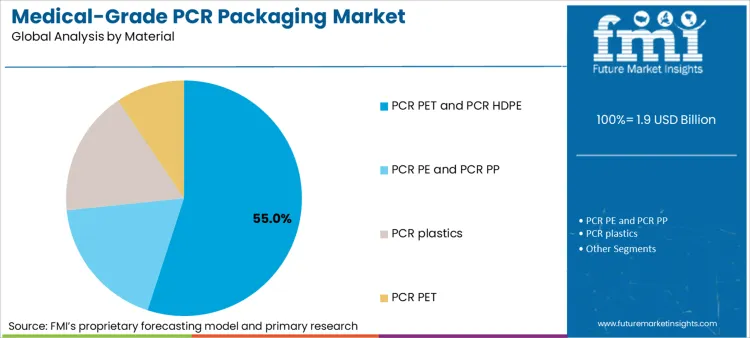
PCR PET and PCR HDPE account for 55% of material demand because they represent the most industrialized and technically understood recycled polymer pathways available at scale. Their dominance is not driven by sustainability preference but by qualification pragmatism. These polymers benefit from established analytical methods, historical use in regulated packaging contexts, and clearer expectations around material characterization and impurity risk management.
Equally important is supply continuity. Medical-grade packaging programs require predictable resin availability over multi-year horizons. PCR PET and PCR HDPE are supported by broader recycling infrastructures and more mature processing technologies, which improves the feasibility of maintaining consistent resin specifications. FMI observes that quality teams are more willing to approve PCR when material variability can be demonstrably constrained and when resin suppliers can provide repeatable documentation aligned with compendial and regulatory expectations.
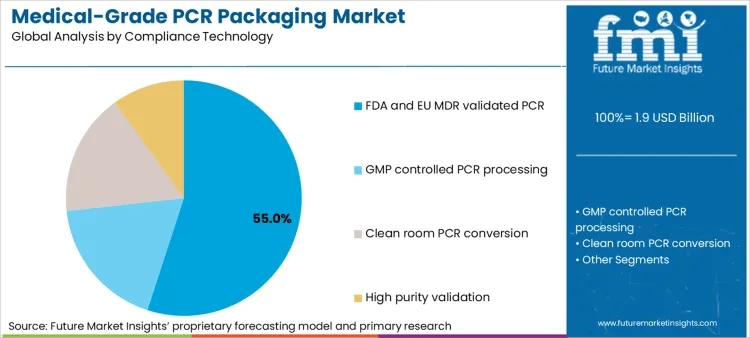
FDA and EU MDR validated PCR accounts for 55% of compliance technology demand because it directly addresses the central barrier to adoption, which is audit defensibility. In regulated packaging, the acceptability of PCR hinges less on recycled content percentage and more on whether the material pathway can be justified through structured evidence, including process controls, contamination risk mitigation, and documented suitability assessments.
These validation-led approaches also reduce organizational risk. By aligning PCR adoption with recognized regulatory frameworks, packaging teams can integrate recycled materials without creating ambiguity during inspections or technical file reviews. FMI finds that companies increasingly prioritize PCR programs that fit cleanly into existing quality systems, rather than experimental approaches that require bespoke justifications for each product or market. This concentrates demand around compliance pathways that emphasize repeatability, traceability, and formal validation discipline.
The key driver is the rise of compliance led procurement, where PCR packaging is adopted only when it can be validated with the same rigor as virgin materials. MDR accountability for medical devices and compendial expectations for pharmaceutical packaging systems are increasing the emphasis on documentation completeness and suitability evidence.
Market expansion is restrained by validation cost and change control friction. Even when PCR resin supply exists, organizations must prevent instability in filings, audits, and supplier qualification. This slows scaling in highly risk averse product categories and in portfolios where packaging changes cascade across multiple markets.
The largest opportunity lies in building validation grade PCR supply and conversion programs that reduce requalification frequency and stabilize compliance documentation across regions. Companies that can provide traceable, controlled pathways will capture premium positioning as regulated buyers seek to scale PCR without increasing audit exposure.
The dominant trend is the shift toward controlled, documented PCR processing and conversion environments, including tighter segregation, traceability, and qualification protocols that align with sterile barrier and compendial expectations, rather than broad sustainability led adoption narratives.
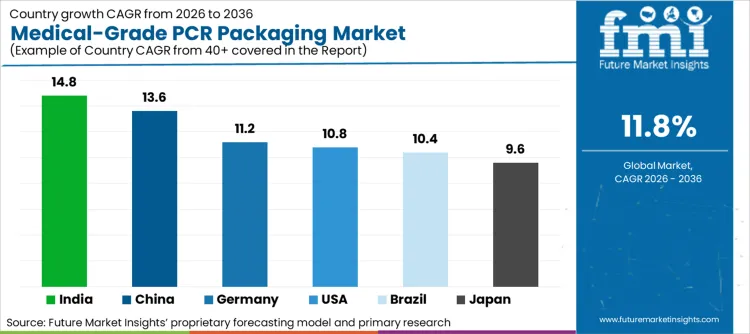
The country-level analysis reflects differences in regulatory enforcement intensity, healthcare manufacturing scale, and packaging governance maturity. FMI expands each country profile to capture both demand drivers and structural constraints.
USA is projected to grow at a 10.8% CAGR, expanding steadily as pharmaceutical and medical device manufacturers seek to reconcile recycled content goals with conservative risk management practices. PCR adoption is anchored in evidence-based qualification, where material acceptability is evaluated through documented processing controls and suitability assessments rather than sustainability positioning alone. This approach aligns with broader expectations around material change management in regulated packaging systems.
A second growth factor is enterprise-scale portfolio governance. Large U.S.-based pharma and device companies operate extensive SKU portfolios across multiple therapeutic areas and device classes. This scale amplifies the economic value of standardizing PCR pathways that can be reused across product lines without repeated validation. FMI observes that U.S. adoption accelerates where PCR is embedded into long-term packaging strategies designed to minimize cumulative regulatory and quality overhead.
Germany is forecast to grow at an 11.2% CAGR, reflecting its role as a compliance benchmark within the European regulated packaging ecosystem. MDR has increased the visibility of packaging decisions within technical documentation and conformity assessment processes, making material selection more exposed to review. This environment favors PCR adoption only when supported by comprehensive documentation and tightly controlled processing conditions.
German manufacturers also benefit from strong engineering cultures and formalized quality systems, which support structured scaling of medical-grade PCR. FMI analysis indicates that adoption is most pronounced where packaging suppliers and brand owners collaborate closely on validation protocols, enabling PCR integration without disrupting established approval pathways. This disciplined approach allows Germany to scale PCR adoption while maintaining alignment with stringent EU regulatory expectations.
China is expected to grow at a 13.6% CAGR, driven by the scale of domestic pharmaceutical production and the operational value of supply resilience. High-volume manufacturing environments create strong incentives to optimize material sourcing and reduce dependency on virgin polymers, provided quality risks can be contained. FMI finds that PCR adoption in China is strongest where conversion takes place within tightly controlled manufacturing environments that limit contamination exposure.
Policy direction toward plastics pollution control and circular economy development further supports long-term momentum. While regulatory expectations for medical packaging remain conservative, the broader system-level push for plastics management is encouraging investment in cleaner, more traceable PCR processing infrastructure. Over time, this infrastructure development is expected to lower barriers for regulated sectors seeking compliant recycled content pathways.
Japan is projected to grow at a 9.6% CAGR, reflecting high expectations around material purity, process stability, and quality assurance. Medical packaging decisions are tightly governed, and any material change must demonstrate negligible impact on performance and safety. This results in slower but more durable adoption, where PCR is introduced incrementally under controlled pilot programs rather than broad portfolio conversions.
The national emphasis on resource circulation reinforces the strategic rationale for PCR, but execution remains gated by validation rigor. FMI observes that Japanese adoption is strongest in precision packaging segments where suppliers can demonstrate exceptional control over processing variables and documentation quality. This approach prioritizes long-term reliability over rapid scale-up, aligning with Japan’s broader manufacturing philosophy.
India is projected to grow at a 14.8% CAGR, driven by its position as a global hub for generic pharmaceutical manufacturing and exports. As Indian manufacturers serve regulated markets worldwide, packaging decisions increasingly need to satisfy international quality and compliance expectations. This creates strong incentives to develop PCR pathways that can withstand scrutiny from multiple regulatory authorities without compromising export continuity.
At the same time, domestic plastic waste governance is strengthening the compliance environment for packaging materials. FMI analysis shows that adoption is concentrated among export-oriented manufacturers and converters that are investing in controlled processing, traceability systems, and documentation capabilities to align sustainability objectives with regulated market access.
Brazil is forecast to expand at a 10.4% CAGR, reflecting the gradual strengthening of reverse logistics and packaging accountability. While medical-grade PCR adoption remains cautious, increasing regulatory clarity around waste management and recycling credit mechanisms is encouraging longer-term investment in structured PCR supply chains.
In regulated medical packaging, this translates into incremental adoption where suppliers can demonstrate controlled sourcing and processing aligned with quality expectations. FMI expects Brazil’s market to develop through phased implementation, with PCR adoption expanding as documentation practices, traceability infrastructure, and regulatory confidence mature over the forecast period.
The competitive landscape is characterized by high intensity as packaging leaders position medical grade PCR as a compliance ready offering rather than a sustainability add on. Success is increasingly determined by the ability to provide validated material pathways with traceability, controlled processing, and documentation packages that reduce audit risk for pharma and device customers.
Competitive differentiation is shifting toward governance strength. Suppliers that can standardize qualification protocols, support customer change control processes, and demonstrate consistent conversion controls are better positioned to scale across global regulated portfolios where material change volatility is heavily penalized.
The medical grade PCR packaging market comprises revenues generated from packaging materials and rigid packaging formats that incorporate post consumer recycled polymers and are intended for pharmaceutical and medical device packaging applications where documented compliance and quality validation are required.
The scope includes blister packs and rigid trays, bottles and sterile containers, and medical rigid packaging manufactured using PCR polymer families including PCR PET, PCR HDPE, PCR PE, and PCR PP, where processing and conversion are controlled to support regulated use.
The scope excludes active drug delivery devices, primary drug formulation components, sterilization equipment, and general purpose PCR packaging not positioned for regulated medical applications with quality evidence requirements.
| Items | Values |
|---|---|
| Quantitative Units | USD 1,920 million |
| End Use | Pharmaceutical and medical devices, EU pharma packaging, Domestic pharma, Precision medical packaging, Generic pharma exports, Others |
| Packaging Type | Blister packs and rigid trays, Bottles and sterile containers, Medical rigid packaging, Diagnostic packaging, Others |
| Material | PCR PET and PCR HDPE, PCR PE and PCR PP, PCR plastics, PCR PET, Others |
| Compliance Technology | FDA and EU MDR validated PCR, GMP controlled PCR processing, Clean room PCR conversion, High purity validation, Others |
| Regions Covered | North America, Western Europe, East Asia, South Asia and Pacific, Latin America, Middle East and Africa |
| Countries Covered | USA, Germany, China, Japan, India, Brazil and 40 plus countries |
| Key Companies | Amcor Healthcare, Berry Global Healthcare, Gerresheimer, SCHOTT, Shandong Pharmaceutical Glass, Nipro, Toyo Seikan |
Source: Future Market Insights analysis based on proprietary forecasting model and primary research
How big is the medical-grade pcr packaging market in 2026?
The global medical-grade pcr packaging market is estimated to be valued at USD 1.9 billion in 2026.
What will be the size of medical-grade pcr packaging market in 2036?
The market size for the medical-grade pcr packaging market is projected to reach USD 5.9 billion by 2036.
How much will be the medical-grade pcr packaging market growth between 2026 and 2036?
The medical-grade pcr packaging market is expected to grow at a 11.8% CAGR between 2026 and 2036.
What are the key product types in the medical-grade pcr packaging market?
The key product types in medical-grade pcr packaging market are pharmaceutical and medical devices , eu pharma packaging, domestic pharma, precision medical packaging and generic pharma exports.
Which packaging type segment to contribute significant share in the medical-grade pcr packaging market in 2026?
In terms of packaging type, blister packs and rigid trays segment to command 50.0% share in the medical-grade pcr packaging market in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.