The demand for drug discovery services in Japan is valued at USD 1,191.4 million in 2026 and is projected to reach USD 3,390.6 million by 2036, reflecting a CAGR of 11.0%. Growth from USD 706.3 million in 2020 to USD 1,191.4 million in 2026 is supported by wider use of chemistry and biology services across pharmaceutical and biotechnology companies. Small molecule programs dominate early activity, while oncology, neurology, and infectious disease research anchor service utilization in Japan’s expanding R&D environment.
From 2027 onward, values rise from USD 1,322.8 million to USD 2,009.9 million by 2031, then reach USD 3,390.6 million by 2036 through widening annual gains. Academic institutes and manufacturing partners maintain steady participation as candidate validation, hit to lead work, and optimization processes increase. Biologics programs add measured yet consistent demand. Long term growth reflects deeper outsourcing, complex modality development, and predictable expansion across therapeutic research areas within Japan’s established innovation ecosystem.
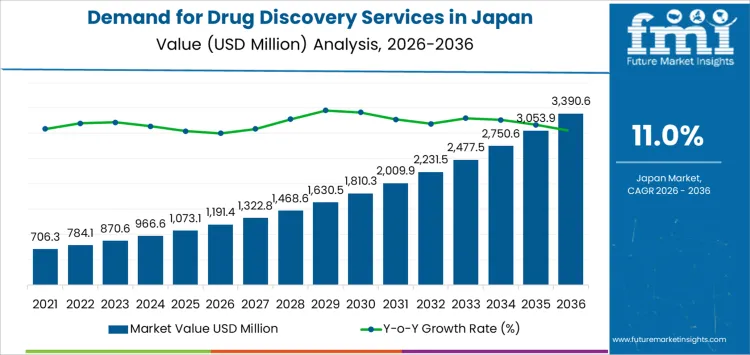
Between 2026 and 2031, demand for drug discovery services in Japan rises from USD 1,191.4 million to USD 2,009.9 million, showing a substantial increase tied to expanding research pipelines and wider outsourcing by pharmaceutical and biotech firms. Growth during this interval reflects heightened emphasis on early-stage candidate identification, increased use of contract screening platforms, and the growing role of specialized CROs supporting complex assays. Earlier expansion was shaped by incremental project-based outsourcing, while the near-term phase captures a marked shift toward multi-year research partnerships and broader reliance on external discovery infrastructure.
From 2031 to 2036, demand advances from USD 2,009.9 million to USD 3,390.6 million, indicating a sharper acceleration as therapeutic discovery efforts diversify. Expanded gene and cell therapy pipelines, rising demand for multi-omics integration, and broader investment in modality-specific discovery frameworks support the steeper trajectory. Earlier growth was driven by capacity limitations within in-house R&D teams, whereas later expansion reflects a structural reconfiguration in Japan’s innovation ecosystem, with discovery outsourcing becoming central to research strategy. The increasing complexity of target validation and higher data-processing intensity further reinforce service demand through 2036.
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 1,191.4 million |
| Forecast Value (2036) | USD 3,390.6 million |
| Forecast CAGR (2026-2036) | 11.0% |
Demand for drug discovery services in Japan grew as pharmaceutical and biotech companies sought external research capacity to support early stage projects. Historical demand reflected limited in-house resources for screening, lead optimisation, and specialized assays. Firms partnered with service providers to access advanced instrumentation, broader compound libraries, and niche expertise in molecular biology and medicinal chemistry. Universities and research institutes collaborated with outsourcing partners to accelerate translational projects. From 2026 to 2036 demand is expected to align with expansion of small and mid-size biotechs, greater emphasis on precision medicine, and wider adoption of computational design.
Companies rely on service providers to support complex target validation, high-content screening, and integrated data interpretation. Demand also reflects the need for flexible capacity as project portfolios shift under changing regulatory and clinical priorities. Providers offering genome editing support, protein engineering expertise, and early pharmacology platforms may see increased engagement. Buyers evaluate scientific capability, reproducibility, data security, and turnaround times when selecting partners. Growth depends on sustained research funding, emerging therapeutic modalities, and competitive pressures to shorten discovery timelines across the sector.
The demand for drug discovery services in Japan from 2026 to 2036 is driven by expansion in biologics research, steady investment in small-molecule programs, and outsourcing by domestic pharma seeking shorter development cycles. Target selection leads by process due to its role in shaping project direction. Chemistry services hold the largest share as they support synthesis, profiling, and structure–activity refinement across a wide range of therapeutic areas.
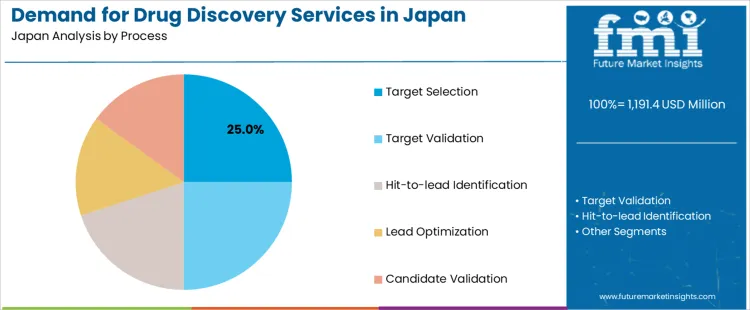
Target selection accounts for 25% of the demand in Japan because research teams increasingly outsource early investigative work to reduce internal screening load and improve project filtration. Consumption increases across therapeutic programs examining receptor families, metabolic pathways, and emerging protein classes that require specialized analytical capabilities. Usage remains steady since target selection influences resource allocation for the remainder of the discovery cycle, prompting teams to rely on external groups with advanced profiling tools. Procurement is guided by firms evaluating data reproducibility, assay quality, and depth of pathway characterization. Price sensitivity stays moderate as companies prioritize accuracy during this phase. Specification control centers on biomarker relevance, mechanistic clarity, and computational modeling support. Demand continues from 2026 to 2036 as Japan’s research ecosystem expands partnerships between pharmaceutical laboratories and contract discovery organizations.
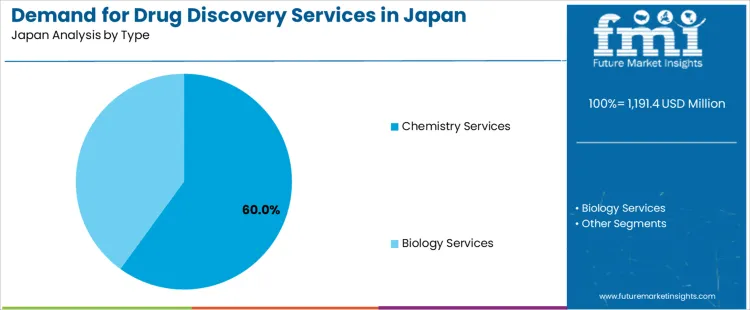
Chemistry services represent 60.0% of the demand in Japan because small-molecule programs require continuous synthesis, optimization, and analytical evaluation throughout the discovery pipeline. Consumption is strong among companies advancing lead series where iterative structural changes shape potency, selectivity, and safety. Usage remains steady since chemistry-based workflows support hit expansion, impurity tracking, and formulation compatibility assessments. Procurement decisions are influenced by partners offering scalable synthetic routes and rapid turnaround on compound libraries. Price sensitivity stays moderate as high-quality chemical output reduces downstream failures. Specification control emphasizes reaction reproducibility, compound purity, and stability documentation. Demand remains consistent from 2026 to 2036 as domestic drug developers maintain active pipelines that rely on external chemistry capacity for sustained project momentum.
Demand grows through Japan’s research hubs where universities, regional laboratories, and mid-sized pharmaceutical firms depend on external support for screening, modeling, and early stage validation. Facilities in Kanagawa request study support for pathway analysis linked to local oncology programs. Research groups in Kyoto rely on outsourced assay development during seasonal capacity peaks. Startups in Osaka use external chemistry services to process compound libraries funded through prefectural grants. Clinical research centers in Tokyo coordinate early validation steps with contracted teams to manage workload variation. These patterns reflect local research practices shaped by Japan’s scientific infrastructure.
Which Local Research Practices Are Driving Demand for Drug Discovery Services in Japan?
Drivers arise from project structures inside Japan’s academic and industrial networks. University laboratories in Kyoto request external screening support when grant cycles create condensed timelines. Pharmaceutical teams in Shizuoka rely on medicinal chemistry services to progress candidates that align with domestic program schedules. Biotech firms in Yokohama use outsourced modeling to compensate for limited in house computational staff. Research hospitals in Tokyo coordinate specialized assay requests during trials linked to local patient cohorts. These activities create practical, locally grounded demand shaped by Japan’s research rhythms and project specific workloads.
What Local Constraints Are Limiting Wider Use of Drug Discovery Services in Japan?
Barriers develop from funding rules, facility routines, and partnership structures. Smaller biotech firms in Fukuoka avoid extensive outsourcing because grant budgets cover only limited external tasks. University labs in Hokkaido maintain established internal protocols that reduce reliance on external service providers. Pharmaceutical plants in Toyama follow procurement routines that prioritize long term partners, which limits diversification. Research centers in rural prefectures face slow sample transport to metropolitan hubs, narrowing collaboration options. These factors constrain broader uptake in regions where budgets, workflows, or logistics shape conservative service use.

| Region | CAGR (%) |
|---|---|
| Kyushu & Okinawa | 13.8% |
| Kanto | 12.7% |
| Kinki | 11.1% |
| Chubu | 9.8% |
| Tohoku | 8.6% |
| Rest of Japan | 8.2% |
The demand for drug discovery services in Japan is accelerating across pharmaceutical, biotechnology, and academic research ecosystems, led by Kyushu and Okinawa at a 13.8% CAGR. Growth in this region is supported by expanding biomedical clusters, increased collaboration between universities and contract research organizations, and stronger investment in early stage research platforms. Kanto follows at 12.7%, driven by major pharma headquarters, advanced R&D infrastructure, and robust outsourcing to specialized discovery service providers. Kinki shows 11.1% growth, reflecting active biotechnology hubs and strong participation from academic research centers. Chubu at 9.8% demonstrates rising adoption linked to pharmaceutical manufacturing networks and regional innovation programs. Tohoku and the Rest of Japan, at 8.6% and 8.2%, show stable but slower growth influenced by smaller research ecosystems and fewer discovery focused institutions.
Demand for drug discovery services in Kyushu and Okinawa is expanding at a CAGR of 13.8% from 2026 to 2036 as universities, biotech firms, and clinical centers outsource screening, assay development, and preclinical studies. Academic laboratories partner with service providers to support early stage research. Emerging biotech firms use external platforms to reduce infrastructure needs. Regional distributors observe active inquiry patterns linked to ongoing projects. Growth aligns with strong research engagement where outsourced capabilities help institutions manage workloads and timelines.What Factors Guide Drug Discovery Services Consumption in Kanto?
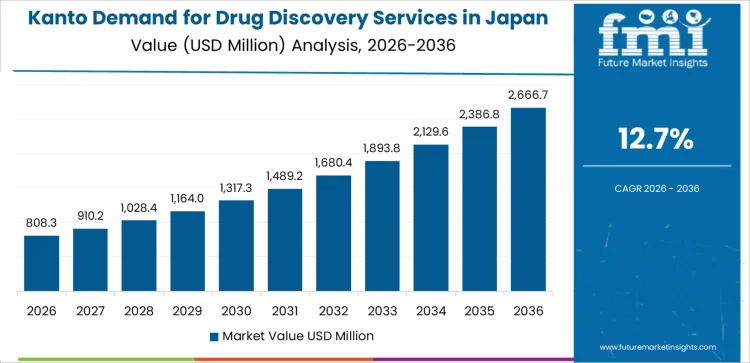
Demand for drug discovery services in Kanto is increasing at a CAGR of 12.7% from 2026 to 2036 as major pharmaceutical firms, advanced hospitals, and research institutions use outsourced capabilities to support discovery workflows. Large firms rely on screening and analytical support for pipeline programs. Hospitals with research divisions collaborate on translational studies. Service providers manage steady engagements driven by metropolitan research density. Growth reflects broad scientific activity across public and private sectors.
Demand for drug discovery services in Kinki is progressing at a CAGR of 11.1% from 2026 to 2036 as research universities, mid scale biotech firms, and hospital laboratories rely on external partners for assays, molecular studies, and in vitro evaluations. Universities use services to advance exploratory programs. Biotech firms outsource complex analytical workflows. Retail scientific suppliers track consistent procurement tied to ongoing research. Growth reflects balanced research participation across academic and commercial settings.
Demand for drug discovery services in Chubu is rising at a CAGR of 9.8% from 2026 to 2036 as hospitals, academic institutions, and contract labs integrate outsourced capabilities into research cycles. Hospitals use services for molecular analysis. Institutions rely on assay development support to manage resource limits. Contract labs coordinate specialized tasks for local clients. Growth reflects methodical research activity in a region where institutions require structured external assistance.
Demand for drug discovery services in Tohoku is growing at a CAGR of 8.6% from 2026 to 2036 as universities, clinical laboratories, and public research centers collaborate with service providers for screening and molecular evaluations. Universities use external partners to manage complex studies. Clinical labs incorporate outsourced testing for research oriented projects. Regional suppliers observe stable engagement from dispersed institutions. Growth reflects practical research dependencies across smaller networks.
Demand for drug discovery services in Rest of Japan is moving at a CAGR of 8.2% from 2026 to 2036 as hospitals, local universities, and small research groups outsource analytical and screening tasks. Hospitals engage providers for targeted studies. Universities incorporate external support for selected projects. Distributors manage measured demand linked to project cycles. Growth reflects practical outsourcing across smaller institutions requiring flexible research capabilities.
The demand for drug discovery services in Japan is shaped by steady investment in oncology, regenerative medicine, rare diseases, and immunology research. Domestic pharmaceutical firms outsource early discovery steps to shorten project timelines and expand access to specialized platforms. University hospitals and national research institutes increase use of outsourced assay development, target validation, and preclinical evaluation to supplement limited in house capacity. Charles River Laboratories International Inc participates through in vivo and in vitro models widely referenced in Japanese preclinical workflows. Laboratory Corporation of America Holdings supports demand through bioanalytical, ADME, and early safety testing accessed by research teams across pharmaceutical clusters. Thermo Fisher Scientific Inc influences discovery through screening platforms, reagent supply, and specialized laboratory services used in translational programs. These organizations complement domestic capabilities by providing scale, speed, and diversified technology access during early drug development.
Evotec SE participates through integrated discovery programs involving chemistry, biology, and computational design used by Japanese firms building pipeline diversification strategies. Pharmaron Beijing Co Ltd supports project based assignments in medicinal chemistry, pharmacology, and early toxicology. Selection in Japan is governed by data integrity, model reproducibility, compliance with national research standards, and close communication between sponsor and external teams. Buyers emphasize predictable timelines and transparent documentation that supports later regulatory work. Demand visibility tracks growth in venture backed biotech, partnerships between global and Japanese drug developers, and sustained investment in precision medicine research.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Process | Target Selection; Target Validation; Hit to Lead Identification; Lead Optimization; Candidate Validation |
| Service Type | Chemistry Services; Biology Services |
| Drug Type | Small Molecule Drugs; Biologics Drugs |
| Therapeutic Area | Oncology; Neurology; Infectious and Immune Systems Diseases; Digestive System Diseases; Other Therapeutic Areas |
| End User | Pharmaceutical & Biotechnology Companies; Academic Institutes; Manufacturing; Other End Users |
| Regions Covered | Kyushu & Okinawa; Kanto; Kansai; Chubu; Tohoku; Rest of Japan |
| Countries Covered | Japan |
| Key Companies Profiled | Charles River Laboratories International Inc; Laboratory Corporation of America Holdings; Thermo Fisher Scientific Inc; Evotec SE; Pharmaron Beijing Co Ltd |
| Additional Attributes | Dollar by sales across processes, service types, and therapeutic areas; screening platform utilization; early stage validation workflows; assay development demand; computational design and modeling requirements; data integrity and reproducibility standards; outsourcing cycle timing; project scalability needs; collaboration structures between academic, biotech, and pharmaceutical groups. |
How big is the demand for drug discovery services in Japan in 2026?
The demand for drug discovery services in Japan is estimated to be valued at USD 1,191.4 million in 2026.
What will be the size of drug discovery services in Japan in 2036?
The market size for the drug discovery services in Japan is projected to reach USD 3,390.6 million by 2036.
How much will be the demand for drug discovery services in Japan growth between 2026 and 2036?
The demand for drug discovery services in Japan is expected to grow at a 11.0% CAGR between 2026 and 2036.
What are the key product types in the drug discovery services in Japan?
The key product types in drug discovery services in Japan are target selection, target validation, hit-to-lead identification, lead optimization and candidate validation.
Which type segment is expected to contribute significant share in the drug discovery services in Japan in 2026?
In terms of type, chemistry services segment is expected to command 60.0% share in the drug discovery services in Japan in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.