The demand for drug induced immune hemolytic anemia treatment in Japan is valued at USD 450.6 million in 2026 and is projected to reach USD 699.7 million by 2036, reflecting a CAGR of 4.5%. The rise from USD 346.0 million in 2020 to USD 450.6 million in 2026 is shaped by hospital centered management of hemolytic reactions linked to cephalosporins, penicillin derivatives, levodopa, methyldopa, and quinidine. Hospitals retain the largest share as diagnostic confirmation and acute care oversight remain essential.
From 2027 onward, values increase from USD 470.9 million to USD 561.5 million by 2031, then advance to USD 699.7 million by 2036 through measured annual gains. Clinical laboratories support consistent diagnostic demand as testing volumes widen with improved detection practices. Treatment patterns remain stable, reflecting established therapeutic responses and monitoring requirements. Growth across the decade is influenced by prescription patterns, ageing patient populations, and incremental increases in confirmatory testing within Japan’s healthcare system.
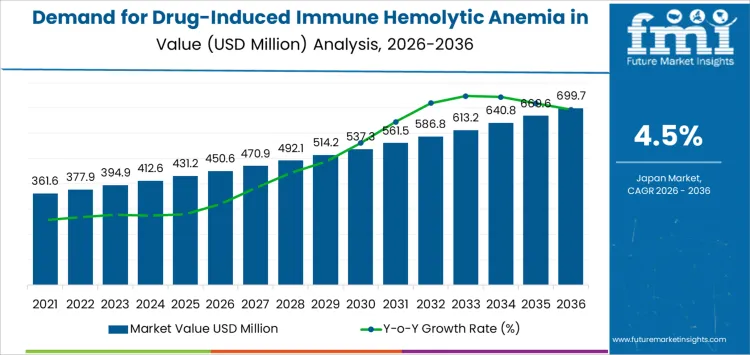
Between 2026 and 2031, demand for drug-induced immune hemolytic anemia management in Japan rises from USD 450.6 million to USD 537.3 million. The pattern across these years shows little evidence of short-term seasonality, since incidence typically results from medication exposure rather than environmental or infectious drivers. Fluctuations instead reflect cyclical prescription trends for high-risk drugs, periodic pharmacovigilance adjustments, and evolving diagnostic awareness. Earlier increases tracked medication utilization patterns, while the near-term phase reflects broader screening and more consistent reporting by hematology services.
From 2031 to 2036, demand increases from USD 537.3 million to USD 699.7 million, revealing a smoother upward trajectory rather than distinct cycles. Growth aligns with long-term prescribing behaviors, shifts in therapeutic classes, and broader adoption of risk-mitigation protocols. Cyclicality appears more structural than seasonal, influenced by population aging, chronic disease management, and periodic replacement of treatment regimens. Earlier variation stemmed from evolving drug portfolios, while later expansion is shaped by stable referral pathways, higher diagnostic precision, and structured monitoring frameworks across Japan’s hematology networks.
| Metric | Value |
|---|---|
| Industry Value (2026) | USD 450.6 million |
| Forecast Value (2036) | USD 699.7 million |
| Forecast CAGR (2026-2036) | 4.5% |
Demand for care related to drug-induced immune hemolytic anemia in Japan increased as clinicians improved detection of hemolysis linked to medication exposure. Historical growth reflected closer monitoring of antibiotics, cardiovascular drugs, and oncology treatments known to trigger immune reactions in rare cases. Hospitals expanded laboratory protocols for direct antiglobulin testing when unexplained anemia appeared during therapy. Providers focused on early identification to prevent severe outcomes, and pharmacovigilance teams tracked adverse reactions more systematically across major treatment centres.
Demand from 2026 to 2036 is likely to rise through wider use of complex drug regimens in aging populations. Broader access to biologics, combination therapies, and high-dose antimicrobials increases the need for diagnostic precision and follow-up. Hospitals emphasise coordinated care between hematologists, pharmacists, and primary physicians when managing suspected cases. Research into drug-immune interactions supports updated diagnostic criteria and refined risk profiles. Demand aligns with improved reporting practices, expanded testing capacity, and consistent evaluation of hemolytic responses across diverse clinical settings.
The demand for drug-induced immune hemolytic anemia treatment in Japan from 2026 to 2036 is shaped by broad antimicrobial usage, increased screening for adverse drug reactions, and higher hospitalization rates among older adults. Cephalosporins hold the leading position by drug type due to frequent inpatient prescribing. Hospitals dominate by end user because DIHA cases require rapid evaluation, laboratory confirmation, and intensive supportive care managed within acute medical environments.
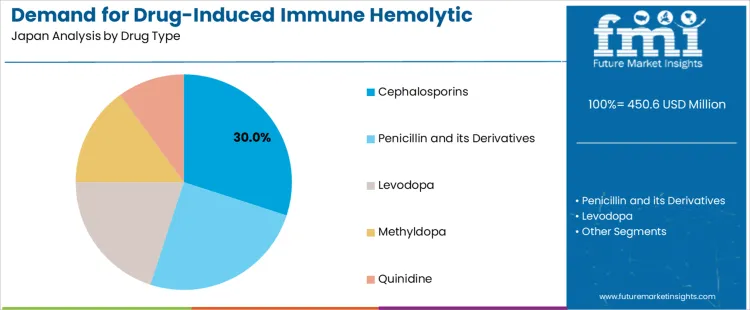
Cephalosporins account for 30% of DIHA-related treatment demand in Japan because they remain core agents in inpatient antimicrobial protocols, especially for respiratory infections, abdominal conditions, and perioperative prophylaxis. Their broad coverage ensures steady prescribing volumes across major hospitals, increasing the likelihood of immune-mediated reactions in susceptible patients. Clinical teams monitor hemolysis indicators closely in older adults and multimorbid individuals who often receive prolonged antibiotic courses. Procurement is streamlined through hospital formularies that maintain multiple cephalosporin generations, supporting rapid therapy adjustment when immune reactions emerge. Demand remains stable through 2026 to 2036, sustained by conservative antimicrobial practices, periodic infection surges, and the need for rapid substitution within established treatment pathways.
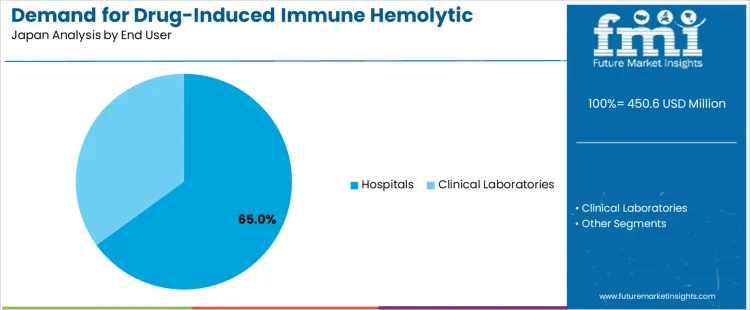
Hospitals represent 65.0% of the demand for DIHA-related treatment in Japan because most cases require urgent assessment, controlled monitoring, and access to extensive diagnostic panels. Patients often present with abrupt hemoglobin decline, dark urine, or acute fatigue, prompting rapid admission to emergency or hematology units. Hospital laboratories perform direct antiglobulin testing and biochemical assessments needed to confirm DIHA quickly. Treatment requires close supervision, especially when transfusion support, high-dose corticosteroids, or immunoglobulin therapy is initiated. Japanese hospitals maintain structured adverse reaction pathways that ensure timely reporting and coordinated adjustment of the offending medication. Demand remains steady from 2026 to 2036 as inpatient antimicrobial exposure, aging demographics, and multidisciplinary management models continue to shape diagnostic and therapeutic workloads.
Interest in diagnostic and management services grows through routine hospital workflows in Japan that monitor adverse drug reactions. Hematology units in major cities track cases linked to specific medications that require rapid confirmation tests. Community clinics refer patients with unexplained anemia for specialized analysis, which increases local use of testing panels. Municipal health programs support awareness across elder care settings where polypharmacy is common. These factors create steady pull for clinical evaluation without relying on broad global narratives.
Which Local Clinical Factors Are Driving Demand for Drug Induced Immune Hemolytic Anemia Services in Japan?
Drivers originate within departments that manage medication related complications. Hospitals in Tokyo, Nagoya, and Kobe review anemia cases that appear after antibiotic or antiplatelet use, prompting requests for direct antiglobulin testing. Elder care facilities in Chiba and Fukuoka address polypharmacy among residents, which leads to more referrals when sudden hemoglobin drops occur. Clinics participating in regional safety programs instruct physicians to report suspected reactions for confirmatory analysis. These settings increase diagnostic activity because staff follow structured local protocols that focus on medication monitoring in routine care.
What Local Constraints Are Limiting Broader Utilization of Drug Induced Immune Hemolytic Anemia Diagnostics in Japan?
Barriers reflect workflow limits inside smaller facilities that lack in house hematology testing. Rural clinics send samples to distant laboratories, which slows confirmation and reduces testing frequency. Some hospitals assign limited budgets to rare adverse event panels, so departments reserve requests for severe cases. Elder care homes with small medical teams may overlook early signs related to medication reactions because of tight schedules. Pharmacy units working within fixed formularies rarely flag lower risk drugs, which narrows detection opportunities. These patterns constrain broader uptake across regions with limited resources or slower referral routes.
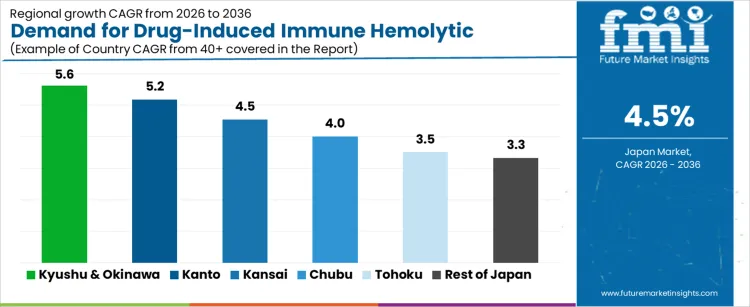
| Region | CAGR (%) |
|---|---|
| Kyushu & Okinawa | 5.6% |
| Kanto | 5.2% |
| Kansai | 4.5% |
| Chubu | 4.0% |
| Tohoku | 3.5% |
| Rest of Japan | 3.3% |
The demand for drug induced immune hemolytic anemia treatment in Japan is growing steadily as diagnostic awareness improves and hematology care pathways expand. Kyushu and Okinawa lead at a 5.6% CAGR, supported by increasing referrals to regional hematology units, broader use of diagnostic panels, and improved monitoring of high risk medications. Kanto follows at 5.2%, driven by large tertiary hospitals, higher testing volumes, and access to advanced therapeutic protocols. Kansai records 4.5% growth, reflecting consistent patient evaluations within university hospitals and specialty clinics. Chubu at 4.0% shows moderate uptake shaped by growing hematology departments and structured treatment planning. Tohoku and the Rest of Japan, at 3.5% and 3.3%, reflect slower progression influenced by fewer specialists, lower case identification, and more conservative treatment adoption.
Demand for drug induced immune hemolytic anemia treatment in Kyushu and Okinawa is rising at a CAGR of 5.6% from 2026 to 2036 as hospitals and clinics manage cases linked to medication related blood cell destruction. Providers rely on diagnostic testing, supportive care, and medication adjustments that require steady clinical resources. Facilities maintain consistent procurement of treatment related supplies aligned with patient volume. Retail medical distributors supply essential items needed for monitoring and follow up. Growth reflects ongoing clinical attention to rare but significant drug related hematologic conditions across regional care environments.
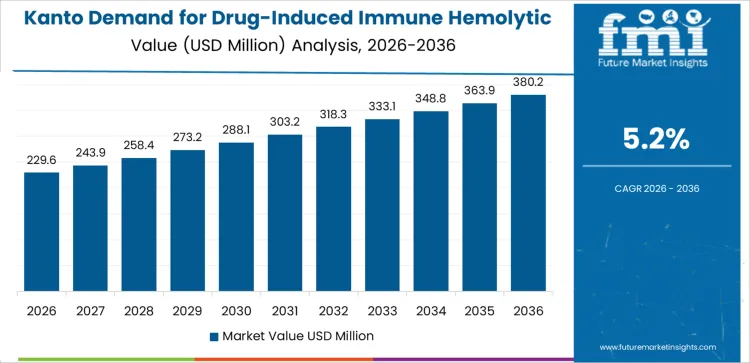
Demand for drug induced immune hemolytic anemia treatment in Kanto is progressing at a CAGR of 5.2% from 2026 to 2036 as large medical centers treat varied patient populations requiring consistent diagnostic and therapeutic support. Providers use specialized testing to confirm hemolytic responses linked to medication exposure. Clinics modify drug regimens and offer ongoing monitoring. Retail medical suppliers handle steady turnover of consumables tied to patient management. Growth follows dense regional healthcare activity where early detection and structured follow up remain central to treatment.
Demand for drug induced immune hemolytic anemia treatment in Kansai is increasing at a CAGR of 4.5% from 2026 to 2036 as healthcare facilities evaluate medication related reactions and manage supportive therapies. Providers rely on laboratory testing and periodic monitoring to track blood parameters. Clinics offer follow up visits to review medication impact and adjust therapies. Retail distributors maintain steady supply of related medical items. Regional demand reflects consistent clinical oversight across suburban and urban care settings where rare hematologic events require precise management.
Demand for drug induced immune hemolytic anemia treatment in Chubu is trending at a CAGR of 4.0% from 2026 to 2036 as hospitals and clinics address medication linked hemolytic responses through structured testing and clinical management. Providers use laboratory tools to evaluate red cell destruction patterns. Clinics adjust drug plans and monitor recovery through scheduled visits. Retail medical outlets manage measured inventory aligned with regional usage. Growth reflects predictable treatment needs across mid sized healthcare systems.
Demand for drug induced immune hemolytic anemia treatment in Tohoku is moving at a CAGR of 3.5% from 2026 to 2036 as providers incorporate diagnostic and therapeutic steps into routine hematology care. Clinics use structured follow up to track patient response. Hospitals conduct testing to confirm drug related hemolysis. Retail suppliers follow steady procurement cycles that align with patient management needs. Growth reflects consistent oversight across diverse clinical environments where careful monitoring supports effective treatment.
Demand for drug induced immune hemolytic anemia treatment in Rest of Japan is advancing at a CAGR of 3.3% from 2026 to 2036 as clinics and regional hospitals manage medication related hemolytic events through staged evaluation and therapeutic adjustments. Providers use laboratory assessments and patient review to guide treatment. Retail distributors maintain modest yet consistent stock levels for required supplies. Regional demand aligns with predictable clinical needs across smaller communities where stable diagnostic and support services remain central to patient care.
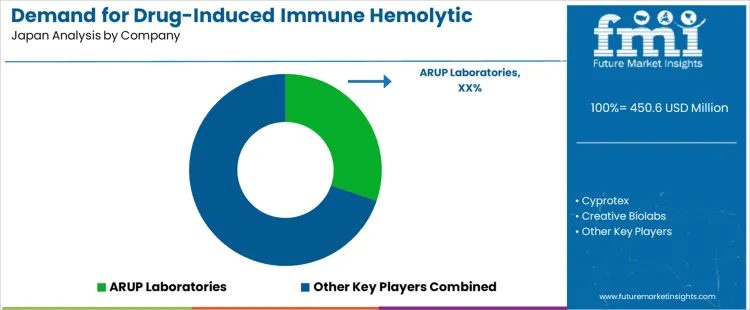
The demand for drug induced immune hemolytic anemia assessment in Japan is shaped by increased use of complex drug regimens, wider hematology screening in hospitals, and rising attention to rare adverse reactions in aging patients. University laboratories and large hospital centers handle most direct testing through antibody screening, Coombs testing, and detailed medication history reviews. Machaon Diagnostics participates through specialized testing referenced in complex case consultations and research collaboration. Creative Biolabs contributes at the research level through assay development that informs long term diagnostic capability rather than routine clinical use.
ARUP Laboratories influences clinical practice through published testing protocols and reference methods accessed by Japanese specialists. F Hoffmann La Roche Ltd shapes upstream awareness through pharmacovigilance studies linked to drug safety monitoring across therapeutic areas. Diagnostic selection in Japan is governed by test sensitivity, reagent stability, clear interpretation guidelines, and strong alignment with national hematology practice. Hospitals emphasize rapid differentiation between immune mediated hemolysis and other causes of anemia to guide medication withdrawal and supportive therapy. Clinicians value access to confirmatory methods that reduce diagnostic uncertainty in complex polypharmacy cases. Buyer preference centers on validated assays, reliable reagent supply, and detailed documentation that supports case review in multidisciplinary teams.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD million |
| Drug Type | Cephalosporins; Penicillin and its Derivatives; Levodopa; Methyldopa; Quinidine |
| End Users | Hospitals; Clinical Laboratories |
| Regions Covered | Kyushu & Okinawa; Kanto; Kansai; Chubu; Tohoku; Rest of Japan |
| Countries Covered | Japan |
| Key Companies Profiled | ARUP Laboratories; Cyprotex; Creative Biolabs; Machaon Diagnostics; F. Hoffmann-La Roche Ltd. |
| Additional Attributes | Dollar by sales across drug types and end users; diagnostic workflow structure; direct antiglobulin and antibody screening practices; regulated testing protocols; hospital centred evaluation pathways; reagent and assay supply requirements; pharmacovigilance linked detection patterns; integration with hematology and clinical laboratory operations. |
How big is the demand for drug-induced immune hemolytic anemia treatment in Japan in 2026?
The demand for drug-induced immune hemolytic anemia treatment in Japan is estimated to be valued at USD 450.6 million in 2026.
What will be the size of drug-induced immune hemolytic anemia treatment in Japan in 2036?
The market size for the drug-induced immune hemolytic anemia treatment in Japan is projected to reach USD 699.7 million by 2036.
How much will be the demand for drug-induced immune hemolytic anemia treatment in Japan growth between 2026 and 2036?
The demand for drug-induced immune hemolytic anemia treatment in Japan is expected to grow at a 4.5% CAGR between 2026 and 2036.
What are the key product types in the drug-induced immune hemolytic anemia treatment in Japan?
The key product types in drug-induced immune hemolytic anemia treatment in Japan are cephalosporins, penicillin and its derivatives, levodopa, methyldopa and quinidine.
Which end user segment is expected to contribute significant share in the drug-induced immune hemolytic anemia treatment in Japan in 2026?
In terms of end user, hospitals segment is expected to command 65.0% share in the drug-induced immune hemolytic anemia treatment in Japan in 2026.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.