Demand for Blood Cancer Treatment in Japan
Demand for Blood Cancer Treatment in Japan Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Demand for Blood Cancer Treatment in Japan Forecast and Outlook 2025 to 2035
Blood cancer treatment demand in Japan is valued at USD 356.0 million in 2025 and is projected to reach USD 689.0 million by 2035 at a CAGR of 6.8%. Early growth is driven by rising diagnosis rates of leukemia, lymphoma, and multiple myeloma linked to population aging and improved detection through routine hematological screening. Hospital-based chemotherapy and targeted drug regimens account for the largest share of current spending, supported by national reimbursement coverage for oncology medications. University hospitals and designated cancer care centers remain the primary treatment hubs due to their access to specialist hematology units, transplant facilities, and integrated diagnostic infrastructure. Drug utilization patterns reflect structured treatment protocols under Japan’s clinical guideline framework.
After 2030, demand growth reflects therapy intensification rather than patient volume expansion alone. Market value rises from about USD 495.3 million in 2030 toward USD 689.0 million by 2035 as greater use of targeted therapies, immune-based treatments, and combination drug regimens increases per-patient treatment cost. Stem cell transplantation volumes remain limited but continue to support high-value procedural demand. Outpatient infusion services expand as hospital capacity is optimized for complex care. Domestic pharmaceutical manufacturers and global oncology firms shape supply through oncology-focused portfolios aligned to Japan regulatory and pricing systems. Long-term spending growth is guided by clinical pathway advancement, treatment duration, and post-remission maintenance approaches rather than cycle-to-cycle drug substitution.
Quick Stats of the Demand for Blood Cancer Treatment in Japan
- Demand for Blood Cancer Treatment in Japan Value (2025): USD 356.0 million
- Demand for Blood Cancer Treatment in Japan Forecast Value (2035): USD 689.0 million
- Demand for Blood Cancer Treatment in Japan Forecast CAGR (2025–2035): 6.8%
- Demand for Blood Cancer Treatment in Japan Leading Application: Leukemia (46%)
- Demand for Blood Cancer Treatment in Japan Key Growth Regions: Kyushu & Okinawa, Kanto, Kansai, Chubu, Tohoku, Rest of Japan
- Demand for Blood Cancer Treatment in Japan Top Players: Roche Holding AG, Novartis AG, Bristol-Myers Squibb, Johnson & Johnson, Gilead Sciences, Inc.
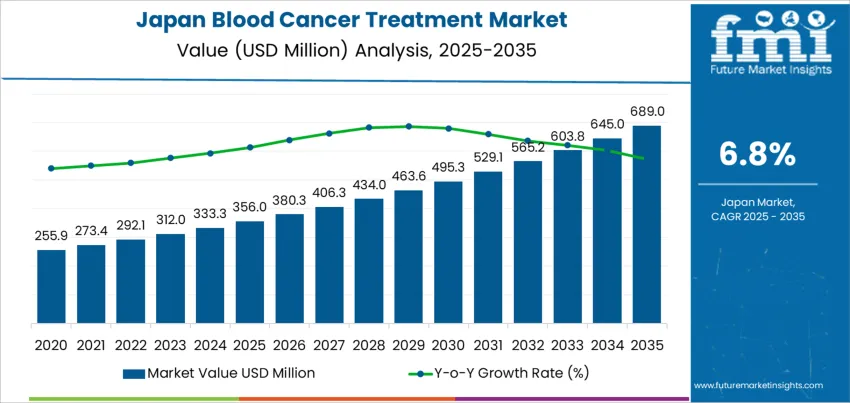
What is the Demand Forecast for Blood Cancer Treatment in Japan through 2035?
Blood cancer treatment in Japan is being reshaped by the convergence of demographic pressure, molecular diagnostics, and long-duration therapy protocols rather than by short-cycle drug adoption alone. Demand increases from USD 356.0 million in 2025 to USD 406.3 million by 2028 and reaches USD 495.3 million by 2030, adding USD 139.3 million from the 2025 base. This phase reflects rising detection rates of leukemia, lymphoma, and multiple myeloma through improved screening, along with longer treatment durations enabled by targeted therapies. Hospital oncology departments expand infusion capacity and outpatient treatment programs, while national reimbursement listings continue to favor precision oncology over broad cytotoxic regimens.
From 2030 to 2035, the market expands from USD 495.3 million to USD 689.0 million, adding USD 193.7 million in the second half of the decade. This back weighted acceleration reflects wider deployment of CAR-T therapies, bispecific antibodies, and next-generation immunotherapies that significantly raise per-patient treatment value. Demand also strengthens as relapse management becomes increasingly drug-intensive and survival periods extend. As Japan continues to prioritize advanced oncology care within its national health system, blood cancer treatment shifts from episodic intervention toward long-term disease management, structurally elevating demand growth through 2035.
Blood Cancer Treatment Industry in Japan Key Takeaways
| Metric | Value |
|---|---|
| Industry Value (2025) | USD 356.0 million |
| Forecast Value (2035) | USD 689.0 million |
| Forecast CAGR (2025–2035) | 6.8% |
What Is Driving the Demand for Blood Cancer Treatment in Japan?
The demand for blood cancer treatment in Japan has grown alongside structural changes in population age, diagnostic capability, and disease awareness. Historically, many hematologic cancers were detected at later stages due to limited screening and symptom overlap with common age related conditions. As clinical diagnostics improved and routine blood testing became widespread, earlier detection increased across hospitals and outpatient settings. Japan rapid demographic aging expanded the patient base for leukemias, lymphomas, and plasma cell disorders, which occur more frequently in older adults. Treatment demand also rose as hospitals adopted standardized care pathways, centralized oncology services, and supportive care infrastructure. Longer survival following initial treatment created ongoing demand for follow up therapy, relapse management, and complication control across the care continuum.
Future demand for blood cancer treatment in Japan will be shaped less by diagnosis expansion and more by treatment complexity, duration of care, and therapy sequencing. New targeted therapies and immune based treatments are extending survival but also increasing the need for long term disease management rather than short treatment cycles. The healthcare system will face rising pressure to deliver continuous outpatient therapy, monitoring, and supportive services for aging patients living with chronic hematologic cancer. Barriers will include high therapy costs, uneven access to specialist centers outside major cities, and workforce strain in oncology nursing and hematology. Demand growth will depend on how effectively Japan balances innovation adoption with cost control, regional access equity, and long term survivorship care capacity.
How Are Application and Treatment Type Structuring the Demand for Blood Cancer Treatment in Japan?
The demand for blood cancer treatment in Japan is structured by disease application and treatment type. Leukemia accounts for 46% of total demand, followed by multiple myeloma, myelodysplastic syndromes, and myeloproliferative neoplasms. By treatment type, targeted therapy represents 44.0% of total usage, followed by immunotherapy, chemotherapy, and radiation therapy. Demand behavior is shaped by disease prevalence, relapse risk, treatment tolerability, and long term disease management needs within an aging population. These segments reflect how diagnostic volume, therapy effectiveness, and clinical treatment pathways guide therapeutic adoption across oncology centers, university hospitals, and specialized hematology clinics in Japan.
Why Does Leukemia Lead the Blood Cancer Application Demand in Japan?
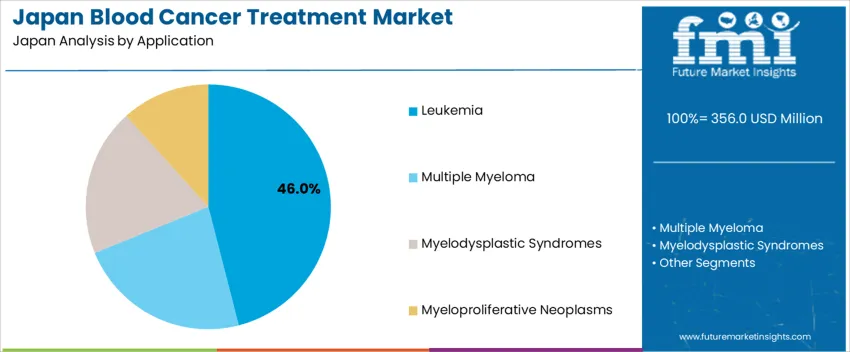
Leukemia accounts for 46% of total blood cancer treatment demand in Japan due to its high incidence across both pediatric and elderly populations. Acute and chronic leukemia cases require prolonged treatment cycles that often extend across multiple lines of therapy. Patients undergo induction, consolidation, and maintenance regimens that sustain continuous drug utilization across hospital and outpatient settings. Bone marrow transplantation pathways further expand treatment intensity for eligible patient groups. These prolonged clinical care requirements drive sustained therapy demand over long disease timelines.
Leukemia management also requires frequent monitoring, relapse intervention, and supportive care due to infection risk and treatment related complications. Japan’s national cancer screening and early diagnosis programs continue to increase detected case volume. Clinical guidelines prioritize early therapeutic initiation to improve survival outcomes. These disease burden, detection intensity, and extended treatment duration factors position leukemia as the dominant application segment within the Japan blood cancer treatment demand structure.
Why Does Targeted Therapy Dominate the Blood Cancer Treatment Type Demand in Japan?
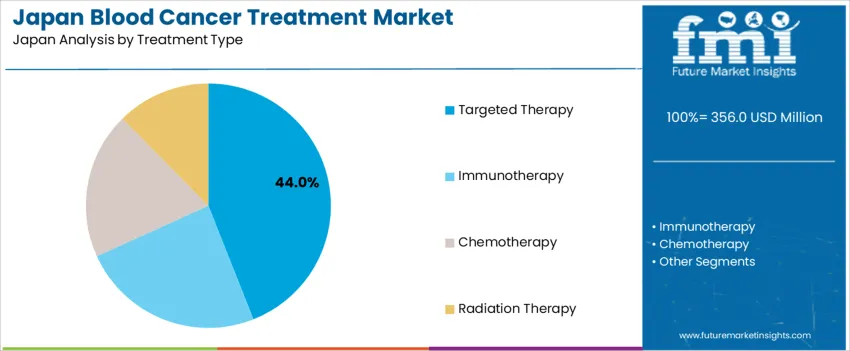
Targeted therapy accounts for 44.0% of total blood cancer treatment demand in Japan due to its disease specific mechanism of action and improved safety profile compared with traditional chemotherapy. Targeted agents inhibit defined molecular pathways involved in cancer cell proliferation and survival, which improves therapeutic precision while limiting systemic toxicity. These therapies are widely used across leukemia and multiple myeloma where genetic mutation profiling guides drug selection. Better tolerability supports long term maintenance treatment in elderly patients who may not tolerate intensive chemotherapy.
Targeted therapy adoption is further supported by regulatory approvals and reimbursement coverage under the national health insurance system. Clinical outcomes show improved progression free survival in multiple blood cancer subtypes. Combination protocols that integrate targeted agents with chemotherapy also expand usage volume. These precision treatment benefits, reimbursement support, and survival improvements position targeted therapy as the leading treatment type segment in the Japan blood cancer treatment demand landscape.
Why Is Blood Cancer Treatment in Japan Closely Linked to Population Aging Rather Than Pure Incidence Growth?
Demand for blood cancer treatment in Japan is shaped primarily by demographic structure rather than sudden disease expansion. Leukemia, lymphoma, and myeloma occur disproportionately in older age groups, which now represent a large share of the population. Survival extension from earlier diagnosis also increases long-term treatment volume, as patients remain in active management for many years. Chronic blood cancers require continuous therapy rather than one-time intervention. This converts blood cancer into a sustained care burden within the national medical system, driven by longevity and repeat treatment cycles rather than short-term incidence spikes.
How Do Universal Health Coverage and Hospital Network Design Shape Treatment Access?
Japan universal health coverage ensures that blood cancer patients enter structured treatment pathways regardless of income. Regional cancer centers coordinate diagnosis, chemotherapy, stem cell transplantation, and long-term follow up under standardized national protocols. Referral systems move complex cases to high-capability hospitals while maintaining community-level monitoring. This network design produces relatively uniform access to advanced therapies across regions. At the same time, capacity concentration in large hospitals creates scheduling pressure for infusion suites, transplant units, and specialist staffing. These organizational structures directly shape real-world treatment throughput and patient flow.
What Clinical Risk, Side Effect Burden, and Cost Controls Constrain Therapeutic Intensity?
Blood cancer treatment intensity in Japan is restrained by patient frailty, toxicity management, and reimbursement discipline. Elderly patients often tolerate aggressive chemotherapy poorly, which forces dose reductions or alternative regimens. Infection risk, cytopenia, and cardiac stress influence therapy selection as strongly as tumor response. National pricing controls limit how quickly premium therapies penetrate routine first-line use. Hospitals must balance survival benefit against hospitalization length and complication management cost. These clinical and economic brakes prevent unlimited escalation of treatment complexity despite rising therapeutic options.
How Are Targeted Therapy, Cellular Medicine, and Long-Term Disease Control Redefining Demand?
Future blood cancer treatment demand in Japan is shifting toward targeted drugs, antibody therapies, and cell-based approaches that blur the line between acute treatment and chronic disease management. Oral targeted agents extend outpatient treatment duration while reducing inpatient burden. CAR-T and other cellular therapies introduce high-intensity, short-duration intervention models for refractory cases. Biomarker-driven stratification increases therapy precision but also fragments treatment pathways. Long-term relapse monitoring becomes a permanent care layer. These changes show demand evolving from episodic crisis treatment into continuous, individualized disease control systems.
What is the Demand for Blood Cancer Treatment in Japan by Region?
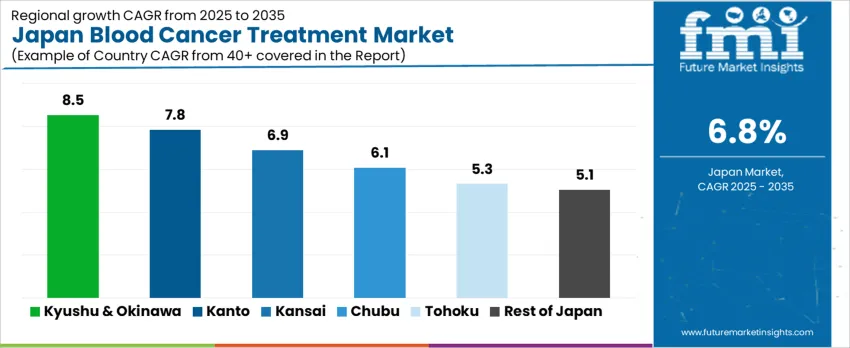
| Region | CAGR (%) |
|---|---|
| Kyushu & Okinawa | 8.5% |
| Kanto | 7.8% |
| Kansai | 6.9% |
| Chubu | 6.1% |
| Tohoku | 5.3% |
| Rest of Japan | 5.1% |
The demand for blood cancer treatment in Japan is rising steadily across all regions, with Kyushu & Okinawa leading at an 8.5% CAGR. Growth in this region is supported by expanding access to oncology services, rising diagnosis rates, and improving availability of advanced therapies in regional hospitals. Kanto follows at 7.8%, driven by a high concentration of cancer specialty centers, research hospitals, and strong adoption of targeted and immuno-oncology therapies. Kansai records 6.9% growth, supported by large urban hospital networks and ageing population needs. Chubu at 6.1% reflects moderate growth linked to regional healthcare modernization. Tohoku and Rest of Japan, at 5.3% and 5.1%, show stable expansion shaped by lower population density and limited concentration of tertiary oncology facilities.
How Is Specialized Oncology Access Driving Growth in Kyushu And Okinawa?
Momentum in Kyushu and Okinawa is advancing at a CAGR of 8.5% through 2035 for blood cancer treatment demand, supported by rising hematology referrals, expanding oncology units, and improved access to advanced therapy protocols. Regional hospitals strengthen chemotherapy infusion capacity and diagnostic screening for leukemia and lymphoma. Medical travel from nearby island communities adds to outpatient flow. Aging population trends continue to raise case incidence. Demand remains treatment driven, with steady growth linked to therapy initiation rates, hospital oncology staffing expansion, and widening access to targeted blood cancer drugs.
- Expanding hematology units support therapy access
- Leukemia and lymphoma screening drives early diagnosis
- Inter regional medical travel adds outpatient volume
- Aging population sustains long term case incidence
What Is Sustaining Strong Blood Cancer Treatment Demand Across Kanto?
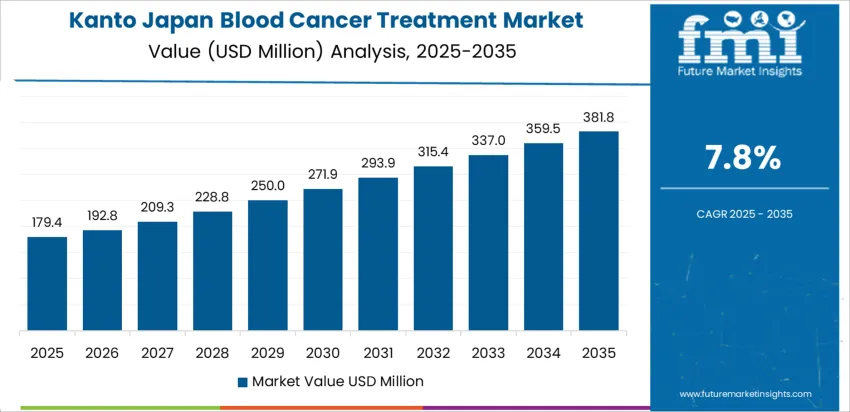
Clinical concentration in Kanto supports a CAGR of 7.8% through 2035 for blood cancer treatment demand, driven by university hospitals, national cancer centers, and large scale oncology research facilities. High patient inflow supports advanced treatment protocols including stem cell therapy and combination drug regimens. Precision diagnostics and molecular profiling improve therapy selection. Clinical trial activity also contributes to early access treatment volumes. Demand remains innovation led and referral driven, with sustained growth tied to complex case intake, continuous drug pipeline adoption, and strong reimbursement alignment across major metropolitan cancer care centers.
- National cancer centers anchor advanced therapy volumes
- Stem cell treatments support high acuity case demand
- Precision diagnostics improve therapy targeting
- Clinical trials add incremental treatment flow
Why Is Kansai Emerging as a Firm Secondary Treatment Hub?
Regional oncology expansion in Kansai supports a CAGR of 6.9% through 2035 for blood cancer treatment demand, shaped by strong hospital networks, rehabilitation integration, and steady lymphoma and myeloma case flow. Day care chemotherapy centers expand access for outpatient treatment cycles. Referral patterns from surrounding prefectures increase specialized case intake. Supportive care services strengthen treatment continuity. Demand remains volume driven and referral based, with growth underpinned by steady diagnosis rates, structured follow up care, and controlled expansion of hematology focused treatment infrastructure.
- Day care chemotherapy centers drive outpatient volumes
- Regional referrals increase specialized case intake
- Myeloma and lymphoma remain key therapy areas
- Supportive oncology services strengthen care continuity
How Is Industrial Workforce Demography Supporting Growth in Chubu?
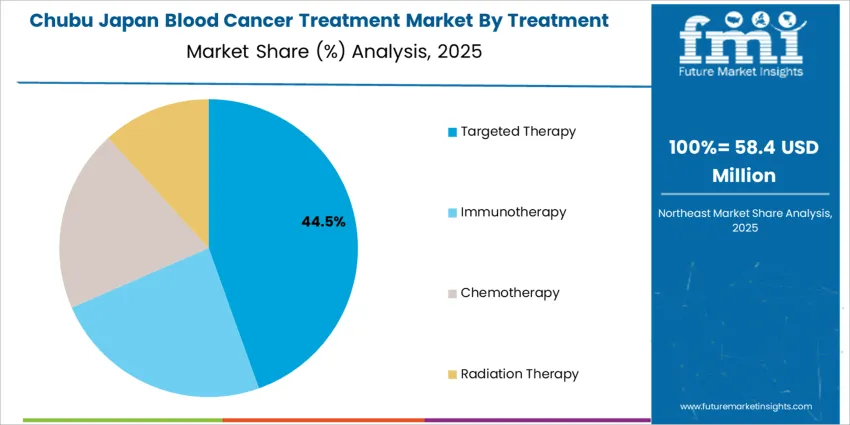
Workforce linked healthcare demand in Chubu supports a CAGR of 6.1% through 2035 for blood cancer treatment demand, influenced by employer health programs, industrial population density, and steady chronic disease diagnosis rates. Large hospitals support infusion therapy, transfusion services, and bone marrow diagnostics. Emergency oncology cases also contribute to treatment continuity. Employer sponsored insurance improves access to extended therapy regimens. Demand remains treatment focused rather than research driven, with procurement and service planning aligned to predictable patient throughput and steady diagnostic confirmation across occupational healthcare networks.
- Employer insurance sustains continuous treatment access
- Infusion and transfusion capacity supports therapy volume
- Emergency oncology adds to hospital case flow
- Industrial population density stabilizes diagnosis rates
What Is Guiding Moderate Blood Cancer Treatment Growth in Tohoku?
Healthcare consolidation in Tohoku supports a CAGR of 5.3% through 2035 for blood cancer treatment demand, shaped by aging rural populations, public hospital coverage, and gradual improvement in hematology diagnostics. Leukemia and anemia related oncology care dominate treatment volume. Limited access to advanced transplant centers moderates high complexity therapy penetration. Referral systems channel critical cases to larger metropolitan hospitals. Demand remains necessity driven and clinically focused, with steady growth tied to routine chemotherapy access, blood monitoring services, and public hospital oncology staffing stability.
- Public hospitals sustain baseline treatment access
- Leukemia care dominates regional therapy demand
- Referral systems guide complex case management
- Rural aging elevates long term treatment volume
Why Does the Rest of Japan Show Stable but Slower Treatment Expansion?
Community healthcare networks across the rest of Japan reflect a CAGR of 5.1% through 2035 for blood cancer treatment demand, supported by municipal hospitals, stable outpatient chemotherapy services, and national insurance reimbursement continuity. Treatment focuses on standard chemotherapy protocols and supportive blood transfusion care. Advanced therapies remain concentrated in major metros. Patient mobility influences referral flows for high risk cases. Demand remains steady and necessity oriented, with predictable therapy volumes linked to routine diagnosis, follow up cycles, and essential hematology care coverage across smaller urban and rural healthcare facilities.
- Municipal hospitals anchor routine oncology services
- Standard chemotherapy dominates treatment protocols
- Advanced therapies remain metro centered
- Insurance stability supports predictable care access
What Is Driving the Demand for Blood Cancer Treatments in Japan and Who Are the Key Players Shaping the Market?
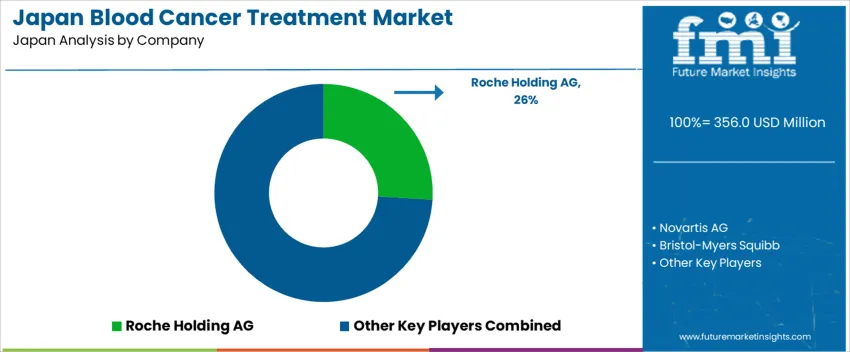
Demand for blood cancer treatments in Japan is increasing due to a rise in incidence of hematologic malignancies, including leukemia, lymphoma, and multiple myeloma, driven in part by the country’s aging population. Advances in diagnostic methods and greater access to early detection contribute to a higher number of treated cases. New therapy options-targeted drugs, immunotherapies, and cellular therapies offer improved survival rates and better tolerability, prompting physicians and patients to opt for advanced treatment regimens rather than traditional chemotherapy. Growth in clinical trials, improved regulatory approval pathways, and expanded healthcare infrastructure also support wider adoption of innovative blood cancer therapies.
Key global pharmaceutical firms influencing the Japanese blood cancer treatment landscape include Roche Holding AG, Novartis AG, Bristol Myers Squibb, Johnson & Johnson, and Gilead Sciences, Inc. These companies supply a range of therapies: monoclonal antibodies, small molecule inhibitors, and cell based treatments (including CAR T therapies) tailored for leukemia, lymphoma, and myeloma. Their breadth of oncology pipelines, regulatory experience, and global manufacturing capacity enables rapid introduction of new therapies into Japan. Through collaborations with local specialists and support for clinical trials, these firms shape patient access, treatment standards, and ongoing innovation in Japan’s hematologic oncology sector.
Key Players in Blood Cancer Treatments Industry in Japan
- Roche Holding AG
- Novartis AG
- Bristol-Myers Squibb
- Johnson & Johnson
- Gilead Sciences, Inc.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units (2025) | USD million |
| Application | Leukemia, Multiple Myeloma, Myelodysplastic Syndromes, Myeloproliferative Neoplasms |
| Treatment Type | Targeted Therapy, Immunotherapy, Chemotherapy, Radiation Therapy |
| Region | Kyushu & Okinawa, Kanto, Kansai, Chubu, Tohoku, Rest of Japan |
| Key Companies Profiled | Roche Holding AG, Novartis AG, Bristol-Myers Squibb, Johnson & Johnson, Gilead Sciences, Inc. |
| Additional Attributes | Dollar by sales by application, treatment type, and region, therapy intensity, per-patient treatment cost, hospital oncology capacity, outpatient infusion volume, clinical guideline adherence, long-term survival management, reimbursement coverage, and treatment complexity |
Blood Cancer Treatments Industry in Japan Segmentation
Application:
- Leukemia
- Multiple Myeloma
- Myelodysplastic Syndromes
- Myeloproliferative Neoplasms
Treatment Type:
- Targeted Therapy
- Immunotherapy
- Chemotherapy
- Radiation Therapy
Region:
- Kyushu & Okinawa
- Kanto
- Kansai
- Chubu
- Tohoku
- Rest of Japan
Frequently Asked Questions
How big is the demand for blood cancer treatment in Japan in 2025?
The demand for blood cancer treatment in Japan is estimated to be valued at USD 356.0 million in 2025.
What will be the size of blood cancer treatment in Japan in 2035?
The market size for the blood cancer treatment in Japan is projected to reach USD 689.0 million by 2035.
How much will be the demand for blood cancer treatment in Japan growth between 2025 and 2035?
The demand for blood cancer treatment in Japan is expected to grow at a 6.8% CAGR between 2025 and 2035.
What are the key product types in the blood cancer treatment in Japan?
The key product types in blood cancer treatment in Japan are leukemia, multiple myeloma, myelodysplastic syndromes and myeloproliferative neoplasms.
Which treatment type segment is expected to contribute significant share in the blood cancer treatment in Japan in 2025?
In terms of treatment type, targeted therapy segment is expected to command 44.0% share in the blood cancer treatment in Japan in 2025.
Table of Content
- Executive Summary
- Japan Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Japan Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Japan Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Japan Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application , 2025 to 2035
- Leukemia
- Multiple Myeloma
- Myelodysplastic Syndromes
- Myeloproliferative Neoplasms
- Y-o-Y Growth Trend Analysis By Application , 2020 to 2024
- Absolute $ Opportunity Analysis By Application , 2025 to 2035
- Japan Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Treatment Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Treatment Type, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Treatment Type, 2025 to 2035
- Targeted Therapy
- Immunotherapy
- Chemotherapy
- Radiation Therapy
- Y-o-Y Growth Trend Analysis By Treatment Type, 2020 to 2024
- Absolute $ Opportunity Analysis By Treatment Type, 2025 to 2035
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Application
- By Treatment Type
- Competition Analysis
- Competition Deep Dive
- Roche Holding AG
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Novartis AG
- Bristol-Myers Squibb
- Johnson & Johnson
- Gilead Sciences, Inc.
- Roche Holding AG
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Japan Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: Japan Market Value (USD Million) Forecast by Application , 2020 to 2035
- Table 3: Japan Market Value (USD Million) Forecast by Treatment Type, 2020 to 2035
- Table 4: Japan Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 5: Japan Market Value (USD Million) Forecast by Application , 2020 to 2035
- Table 6: Japan Market Value (USD Million) Forecast by Treatment Type, 2020 to 2035
List of Figures
- Figure 1: Japan Market Pricing Analysis
- Figure 2: Japan Market Value (USD Million) Forecast 2020-2035
- Figure 3: Japan Market Value Share and BPS Analysis by Application , 2025 and 2035
- Figure 4: Japan Market Y-o-Y Growth Comparison by Application , 2025-2035
- Figure 5: Japan Market Attractiveness Analysis by Application
- Figure 6: Japan Market Value Share and BPS Analysis by Treatment Type, 2025 and 2035
- Figure 7: Japan Market Y-o-Y Growth Comparison by Treatment Type, 2025-2035
- Figure 8: Japan Market Attractiveness Analysis by Treatment Type
- Figure 9: Japan Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 10: Japan Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 11: Japan Market Attractiveness Analysis by Region
- Figure 12: Japan Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: Japan Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Japan Market Value Share and BPS Analysis by Application , 2025 and 2035
- Figure 15: Japan Market Y-o-Y Growth Comparison by Application , 2025-2035
- Figure 16: Japan Market Attractiveness Analysis by Application
- Figure 17: Japan Market Value Share and BPS Analysis by Treatment Type, 2025 and 2035
- Figure 18: Japan Market Y-o-Y Growth Comparison by Treatment Type, 2025-2035
- Figure 19: Japan Market Attractiveness Analysis by Treatment Type
- Figure 20: Japan Market - Tier Structure Analysis
- Figure 21: Japan Market - Company Share Analysis

