Cervical Cancer Treatment Market : Global Industry Analysis 2015 - 2024 and Opportunity Assessment 2025 - 2035
Cervical Cancer Treatment Market Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Cervical Cancer Treatment Market Size and Share Forecast Outlook 2025 to 2035
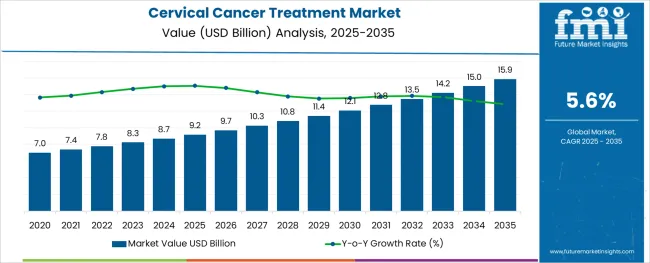
The Cervical Cancer Treatment Market is estimated to be valued at USD 9.2 billion in 2025 and is projected to reach USD 15.9 billion by 2035, registering a compound annual growth rate (CAGR) of 5.6% over the forecast period.
Quick Stats for Cervical Cancer Treatment Market
- Industry Value (2025): USD 9.2 billion
- Forecast Value (2035): USD 15.9 billion
- Forecast CAGR: 5.6%
- Leading Segment in 2025: Open system (54.2%)
- Key Growth Region: North America, Asia-Pacific, Europe
- Top Key Players: Pfizer Inc, Biocon, Alnylam Pharmaceuticals, Novartis, GlaxoSmithKline, Genentech, Hetero, Eli Lilly, ALLERGAN
Rationale for Segmental Growth in the Cervical Cancer Treatment Market
Market Overview
The cervical cancer treatment market is progressing steadily owing to the global emphasis on early diagnosis, improved treatment access, and integration of precision oncology in gynecological care. Rising awareness campaigns, enhanced screening infrastructure, and growing support from public health organizations have contributed to a more proactive approach toward cervical cancer management.
Technological advancements in radiation therapy, targeted drug delivery, and magnetic resonance guided procedures are reshaping treatment protocols. Healthcare providers are increasingly adopting patient specific treatment plans that minimize invasiveness and improve survival outcomes.
With increased funding for women’s health research and the adoption of integrated care models, the market outlook remains promising. Innovations in non invasive imaging, enhanced safety profiles of treatment systems, and demand for effective therapies with minimal side effects are driving ongoing growth and strategic investments in this space.
Segmental Analysis:
The market is segmented by Type, Treatment, and End User and region. By Type, the market is divided into Open system and Closed system. In terms of Treatment, the market is classified into Low Field Strength, Mid Field Strength, and High Field Strength.
Based on End User, the market is segmented into Hospitals, Imaging Centers, Ambulatory Surgical Centers, and Others. Regionally, the market is classified into North America, Latin America, Western Europe, Eastern Europe, Balkan & Baltic Countries, Russia & Belarus, Central Asia, East Asia, South Asia & Pacific, and the Middle East & Africa.
Insights into the Low Field Strength Treatment Segment
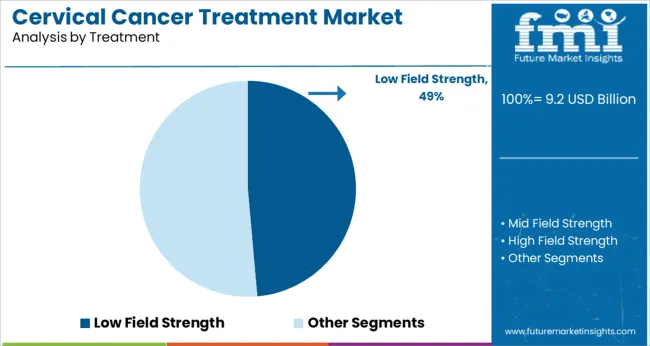
The low field strength segment is projected to represent 48.60% of total market revenue by 2025 within the treatment category, positioning it as the leading option. This dominance is attributed to its compatibility with gynecologic imaging protocols, lower operational costs, and reduced risks associated with prolonged exposure.
Low field strength systems are widely utilized in radiation therapy planning and image guided interventions, offering sufficient imaging clarity while improving patient safety. Their reliability and affordability make them particularly suitable for deployment in emerging healthcare systems where cost efficiency and accessibility are key priorities.
Moreover, clinical acceptance has been rising due to the ability of these systems to provide adequate diagnostic and treatment guidance without compromising comfort or increasing complexity. This segment continues to lead as health systems seek dependable and budget friendly treatment solutions for managing cervical cancer.
Insights into the Open System Type Segment
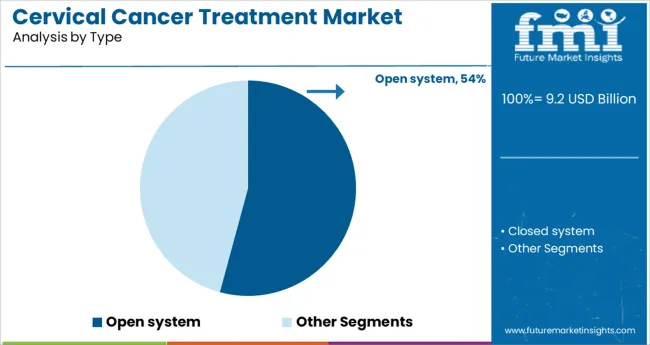
The open system segment is expected to contribute 54.20% of total market revenue by 2025 under the type category, making it the most preferred segment. This leadership is driven by enhanced patient comfort, ease of access during imaging or therapy, and reduced claustrophobia related challenges.
Open systems provide greater flexibility for positioning and accommodate a wider patient demographic, which improves diagnostic accuracy and treatment outcomes. In cervical cancer treatment, where precise targeting and continuous monitoring are critical, open systems support improved workflow integration and procedural success.
Their design also facilitates interdisciplinary collaboration between radiologists and oncologists, further enhancing treatment precision. As patient centric care models evolve and comfort becomes a priority in treatment design, open systems have gained significant adoption and continue to set the standard in cervical cancer treatment equipment.
What are the Major Drivers Boosting the Demand for Cervical Cancer Treatment Market?
The market is largely driven by developments in oncology technology, rising cervical cancer rates, new imaging devices developed by market leaders, raising awareness of the causes and risk factors for cervical cancer and its treatment, growing importance of women's healthcare, and changes in women's lifestyles.
The expansion of the cervical cancer treatment market has been driven by the rising incidence of cervical cancer and the rising awareness of cervical cancer among women. Cervical cancer is the seventh most prevalent cancer worldwide, according to the World Cancer Research Fund International. Around the world, 604,000 women were given a cervical cancer diagnosis in 2024, and about 342,000 of them died from the disease. Multiple pregnancies and unprotected sexual contact raise the risk of cervical cancer. Additionally, the number of people who develop cervical cancer is growing as a result of the rising trend of the use of alcohol and tobacco products.
Which Region Shows Strong Growth Potential in the Cervical Cancer Treatment Market?
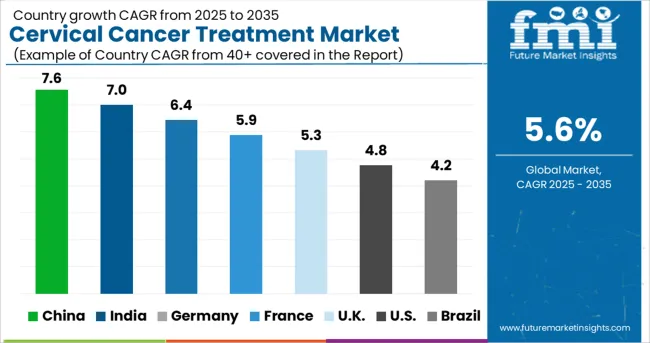
The cervical cancer treatment market is broadly divided into seven major regions, North America, Latin America, Europe, East Asia, South Asia, Oceania, and the Middle East and Africa.
North America is the leading region in the global cervical cancer treatment market owing to the better awareness among people about health care and the latest advanced healthcare technologies in the region.
Along with North America, the market for cervical cancer treatments in Europe is anticipated to develop profitably during the coming years Due to the increasing consumer awareness and the presence of regional players with a history of success.
The East Asian region is predicted to show lucrative growth owing to the rising number of health initiatives by private and government firms, and the adoption of better technologies.
Who are the Key Players in the Cervical Cancer Treatment Market?
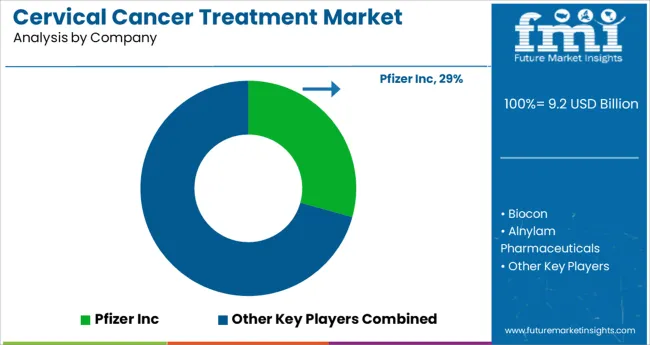
Pfizer Inc, Biocon, Alnylam Pharmaceuticals, Novartis, GlaxoSmithKline, Genentech, Hetero, Eli Lilly, ALLERGAN, Actavis Pharma Company, Bristol-Myers Squibb Company.
Scope of Report
| Report Attributes | Details |
|---|---|
| Growth Rate | CAGR of 5.6% 2025 to 2035 |
| Base Year for Estimation | 2024 |
| Historical Data | 2012 to 2024 |
| Forecast Period | 2025 to 2035 |
| Qualitative Units | Revenue in USD Billion, and CAGR from 2025 to 2035 |
| Report Coverage | Revenue Forecast, Company Ranking, Competitive Landscape, Growth Factors, and Trends |
| Segment Covered | Type, Treatment, End User, Region |
| Region Covered | North America; Latin America; Europe; East Asia; South Asia; Oceania; Middle East Africa |
| Key Countries Profiled | USA, Canada, Brazil, Mexico, Germany, UK, France, Spain, Italy, China, Japan, South Korea, Malaysia, Singapore, Australia, New Zealand, GCC, South Africa, Israel |
| Key Players | Pfizer Inc; Biocon; Alnylam Pharmaceuticals; Novartis; GlaxoSmithKline; Genentech; Hetero; Eli Lilly; ALLERGAN; Actavis Pharma Company; Bristol-Myers Squibb Company |
| Customization | Available Upon Request |
Key Segments Covered in the Cervical Cancer Treatment Market Report
By Type:
- Open system
- Closed system
By Treatment:
- Low Field Strength
- Mid Field Strength
- High Field Strength
By End User:
- Hospitals
- Imaging Centers
- Ambulatory Surgical Centers
- Others
By Region:
- North America
- Latin America
- Europe
- East Asia
- South Asia
- Oceania
- The Middle East and Africa (MEA)
Frequently Asked Questions
How big is the cervical cancer treatment market in 2025?
The global cervical cancer treatment market is estimated to be valued at USD 9.2 USD billion in 2025.
What will be the market size for the cervical cancer treatment market in 2035?
It is projected to reach USD 15.9 USD billion by 2035.
How much will be the CAGR of the cervical cancer treatment market between 2025 and 2035?
The market is expected to grow at a 5.6% CAGR between 2025 and 2035.
What are the key product types in the cervical cancer treatment market?
The key product types are open system and closed system.
Which low field strength segment is expected to dominate the cervical cancer treatment market in 2025?
low field strength segment is expected to dominate with a 48.6% industry share in 2025.
Table of Content
- Executive Summary
- Global Market Outlook
- Demand-side Trends
- Supply-side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- Global Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y-o-Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- Global Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Type
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Type , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Type , 2025 to 2035
- Open system
- Closed system
- Open system
- Y-o-Y Growth Trend Analysis By Type , 2020 to 2024
- Absolute $ Opportunity Analysis By Type , 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Treatment
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Treatment, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Treatment, 2025 to 2035
- Low Field Strength
- Mid Field Strength
- High Field Strength
- Low Field Strength
- Y-o-Y Growth Trend Analysis By Treatment, 2020 to 2024
- Absolute $ Opportunity Analysis By Treatment, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2025 to 2035
- Hospitals
- Imaging Centers
- Ambulatory Surgical Centers
- Others
- Hospitals
- Y-o-Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Global Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Region
- Introduction
- Historical Market Size Value (USD Million) Analysis By Region, 2020 to 2024
- Current Market Size Value (USD Million) Analysis and Forecast By Region, 2025 to 2035
- North America
- Latin America
- Western Europe
- Eastern Europe
- East Asia
- South Asia and Pacific
- Middle East & Africa
- Market Attractiveness Analysis By Region
- North America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- USA
- Canada
- Mexico
- By Type
- By Treatment
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Treatment
- By End User
- Key Takeaways
- Latin America Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Brazil
- Chile
- Rest of Latin America
- By Type
- By Treatment
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Treatment
- By End User
- Key Takeaways
- Western Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Germany
- UK
- Italy
- Spain
- France
- Nordic
- BENELUX
- Rest of Western Europe
- By Type
- By Treatment
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Treatment
- By End User
- Key Takeaways
- Eastern Europe Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Russia
- Poland
- Hungary
- Balkan & Baltic
- Rest of Eastern Europe
- By Type
- By Treatment
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Treatment
- By End User
- Key Takeaways
- East Asia Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- China
- Japan
- South Korea
- By Type
- By Treatment
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Treatment
- By End User
- Key Takeaways
- South Asia and Pacific Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- India
- ASEAN
- Australia & New Zealand
- Rest of South Asia and Pacific
- By Type
- By Treatment
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Treatment
- By End User
- Key Takeaways
- Middle East & Africa Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Country
- Historical Market Size Value (USD Million) Trend Analysis By Market Taxonomy, 2020 to 2024
- Market Size Value (USD Million) Forecast By Market Taxonomy, 2025 to 2035
- By Country
- Kingdom of Saudi Arabia
- Other GCC Countries
- Turkiye
- South Africa
- Other African Union
- Rest of Middle East & Africa
- By Type
- By Treatment
- By End User
- By Country
- Market Attractiveness Analysis
- By Country
- By Type
- By Treatment
- By End User
- Key Takeaways
- Key Countries Market Analysis
- USA
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- Canada
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- Mexico
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- Brazil
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- Chile
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- Germany
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- UK
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- Italy
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- Spain
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- France
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- India
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- ASEAN
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- Australia & New Zealand
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- China
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- Japan
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- South Korea
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- Russia
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- Poland
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- Hungary
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- Kingdom of Saudi Arabia
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- Turkiye
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- South Africa
- Pricing Analysis
- Market Share Analysis, 2024
- By Type
- By Treatment
- By End User
- USA
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Type
- By Treatment
- By End User
- Competition Analysis
- Competition Deep Dive
- Pfizer Inc
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Biocon
- Alnylam Pharmaceuticals
- Novartis
- GlaxoSmithKline
- Genentech
- Hetero
- Eli Lilly
- ALLERGAN
- Pfizer Inc
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: Global Market Value (USD Million) Forecast by Region, 2020-2035
- Table 2: North America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 3: Latin America Market Value (USD Million) Forecast by Country, 2020-2035
- Table 4: Western Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 5: Eastern Europe Market Value (USD Million) Forecast by Country, 2020-2035
- Table 6: East Asia Market Value (USD Million) Forecast by Country, 2020-2035
- Table 7: South Asia and Pacific Market Value (USD Million) Forecast by Country, 2020-2035
- Table 8: Middle East & Africa Market Value (USD Million) Forecast by Country, 2020-2035
List of Figures
- Figure 1: Global Market Pricing Analysis
- Figure 2: Global Market Value (USD Million) Forecast 2020-2035
- Figure 3: Global Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 4: Global Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 5: Global Market Attractiveness Analysis by Region
- Figure 6: North America Market Incremental $ Opportunity, 2025-2035
- Figure 7: Latin America Market Incremental $ Opportunity, 2025-2035
- Figure 8: Western Europe Market Incremental $ Opportunity, 2025-2035
- Figure 9: Eastern Europe Market Incremental $ Opportunity, 2025-2035
- Figure 10: East Asia Market Incremental $ Opportunity, 2025-2035
- Figure 11: South Asia and Pacific Market Incremental $ Opportunity, 2025-2035
- Figure 12: Middle East & Africa Market Incremental $ Opportunity, 2025-2035
- Figure 13: North America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: Latin America Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 15: Western Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 16: Eastern Europe Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 17: East Asia Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 18: South Asia and Pacific Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 19: Middle East & Africa Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 20: Global Market - Tier Structure Analysis
- Figure 21: Global Market - Company Share Analysis

