Demand for Blood Cancer Treatment in USA
Demand for Blood Cancer Treatment in USA Size and Share Forecast Outlook 2025 to 2035
Historical Data Covered: 2015 - 2024 | Base Year: 2024 | Estimated Year: 2025 | Forecast Period: 2025 - 2035
Demand for Blood Cancer Treatment in USA 2025 to 2035
The USA blood cancer treatment demand is valued at USD 2.4 billion in 2025 and is expected to reach USD 5.6 billion by 2035, registering an 8.6% CAGR. Growth is driven by the increasing incidence of leukaemia, lymphoma, and myeloma across the ageing population, alongside wider access to specialized oncology care. Continuous approvals of advanced therapeutics and supportive reimbursement structures contribute to treatment adoption. Clinical efforts aim to enable earlier diagnosis, personalized regimens, and prolonged survival.
Chemotherapy remains the leading category due to its established role in frontline treatment protocols and its broad applicability across blood cancer types. Combination regimens, targeted drug inclusion, and engineered dosing strategies support persistent clinical use.
Demand is concentrated in the West, South, and Northeast, where major cancer centers, specialized haematology departments, and large patient pools are located. Key suppliers include Roche Holding AG, Novartis AG, Bristol-Myers Squibb, Johnson & Johnson, and Gilead Sciences, Inc. Their therapeutic portfolios span monoclonal antibodies, kinase inhibitors, CAR-T therapies, and supportive care pharmaceuticals used across acute and chronic blood malignancies.
Quick Stats for USA Blood Cancer Treatment Demand
- USA Blood Cancer Treatment Sales Value (2025): USD 2.4 billion
- USA Blood Cancer Treatment Forecast Value (2035): USD 5.6 billion
- USA Blood Cancer Treatment Forecast CAGR (2025–2035): 8.6%
- Leading Category in USA Blood Cancer Treatment Demand: Chemotherapy
- Key Growth Regions in USA Blood Cancer Treatment Demand: West USA, South USA, Northeast USA
- Top Players in USA Blood Cancer Treatment Demand: Roche Holding AG, Novartis AG, Bristol-Myers Squibb, Johnson & Johnson, Gilead Sciences, Inc.
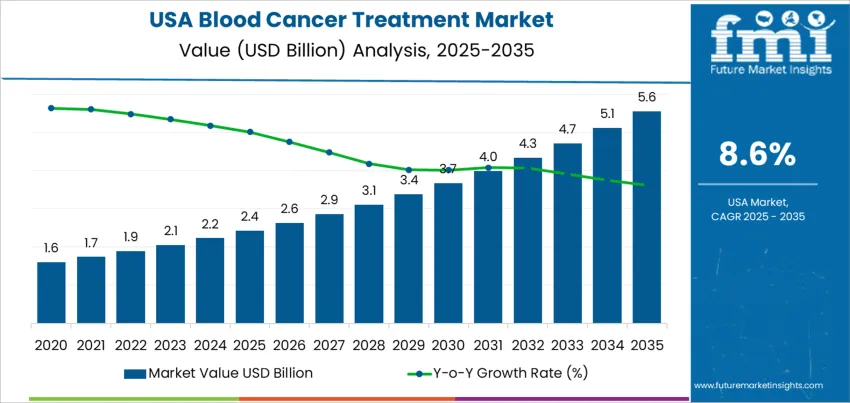
What is the Growth Forecast for Blood Cancer Treatment Industry in USA through 2035?
Ten-year growth for demand for blood cancer treatment in the United States shows stronger expansion during the first half of the forecast period. Advancements in targeted therapies and immuno-oncology drive early acceleration as new products enter treatment pathways for leukemia, lymphoma, and multiple myeloma. Wider patient access to CAR-T cell therapy and oral therapies supporting outpatient care increases utilization and spending. Clinical guidelines incorporate more precision-medicine approaches, which contributes to above-average growth in the initial years.
During the second half, growth remains positive but moderates as high-cost therapies face pricing constraints and payer scrutiny. Generic and biosimilar competition enters select drug classes, reducing revenue expansion despite stable treatment volumes. Broader adoption ensures a significant cumulative increase, although incremental gains flatten relative to the earlier momentum. Survivor populations rise due to improved outcomes, expanding long-term treatment and monitoring requirements that help sustain demand. The comparison indicates early years characterized by stronger innovation-driven gains and later years defined by steady but slower growth. This pattern reflects a maturing therapeutic environment with continued evolution in evidence-based care.
USA Blood Cancer Treatment Key Takeaways
| Metric | Value |
|---|---|
| USA Blood Cancer Treatment Sales Value (2025) | USD 2.4 billion |
| USA Blood Cancer Treatment Forecast Value (2035) | USD 5.6 billion |
| USA Blood Cancer Treatment Forecast CAGR (2025-2035) | 8.6% |
Why is the Demand for Blood Cancer Treatment in the USA Growing?
Demand for blood cancer treatment in the USA is increasing because more individuals receive timely diagnosis for leukemia, lymphoma and multiple myeloma through expanded screening and improved access to hematology services. Growing awareness of early signs such as unexplained fatigue or persistent infections leads to faster referral to specialists and earlier initiation of therapy. An ageing population also contributes to rising treatment needs since many blood cancers occur more frequently in older adults who require ongoing clinical support.
Treatment centers provide advanced options including targeted drugs, immunotherapy and cellular therapy that improve survival and reduce relapse risk. Hospitals expand transplant programs for eligible patients, while outpatient infusion centres strengthen access to maintenance therapies and follow up care. Health insurance coverage for approved therapies encourages wider adoption through community oncology networks. Constraints include high cost for advanced treatments and long care timelines that place financial and emotional pressure on families. Not all patients respond similarly to therapy, and side effect management requires continuous monitoring. Access to experienced oncologists may be limited in rural regions, leading to travel requirements for specialized care.
Which Applications and End Users Lead Demand for Blood Cancer Treatment in the USA?
Demand for blood cancer treatment in the United States is driven by strong clinical focus on improving survival rates for leukemia, lymphoma, and myeloma patients. Providers adopt advanced therapies supporting personalized treatment, immune restoration, and relapse risk reduction. Treatment selection varies by cancer type, disease stage, patient age, and prior therapy response. Hospital networks and specialized oncology centers drive innovation through integrated care models, reimbursed treatment pathways, and access to clinical trials.
By Application, Chemotherapy Holds the Largest Share
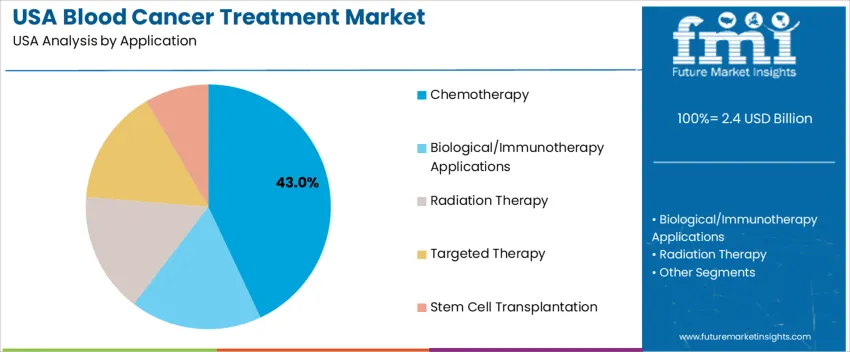
Chemotherapy accounts for 43.0%, reflecting its continued role as the primary frontline intervention for acute and chronic blood cancers, often used in combination regimens. It is widely supported by established care protocols, affordability relative to newer modalities, and availability across USA cancer care centers. Other applications, biological/immunotherapy, targeted therapy, radiation therapy, and stem cell transplantation, support precision treatment, relapse prevention, and long-term remission outcomes. Immunotherapy adoption continues to rise as CAR-T and monoclonal antibody options expand access for treatment-resistant cases. Targeted therapy demand grows in genetic mutation-specific cancers. Stem cell transplantation remains essential for eligible high-risk patients in major oncology institutions.
Key Points:
- Chemotherapy remains standard frontline intervention across cancer types
- Immunotherapy and targeted therapy rapidly increase for resistant cases
- Stem cell procedures support high-risk patient survival improvement
By End User, Hospitals Represent the Largest Share
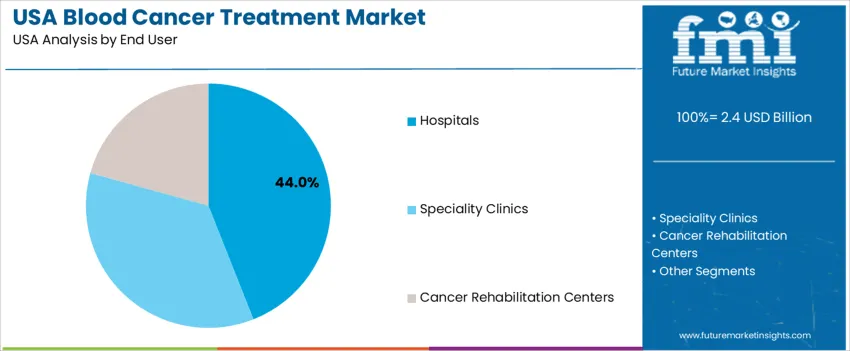
Hospitals hold 44.0%, driven by their central role in managing aggressive blood cancer cases requiring continuous monitoring, infusion services, and 24/7 inpatient care. Large oncology departments enable access to combination therapy programs, clinical trials, and advanced diagnostic tools. Specialty clinics contribute significant demand, especially for outpatient chemotherapy, immunotherapy, and post-remission care. Cancer rehabilitation centers support long-term recovery, symptom management, and therapy-related side effect reduction. Distribution across end-user settings is shaped by treatment intensity, insurance support, and the need for multidisciplinary expertise in hematological malignancies. Hospitals remain the preferred setting for critical and transplant-based care across major USA cancer centers.
Key Points:
- Hospitals lead due to critical treatment and oncology infrastructure
- Specialty clinics expand access for outpatient and maintenance therapies
- Rehabilitation centers assist recovery and quality-of-life outcomes
What are the Drivers, Restraints, and Key Trends of the Demand for Blood Cancer Treatment in the USA?
Increased survival goals, expanded immunotherapy adoption and growth of specialized oncology networks are driving demand.
In the United States, treatment demand for leukemia, lymphoma and multiple myeloma rises as hospitals and cancer centers expand access to targeted therapies that improve remission rates. CAR T cell therapy and monoclonal antibodies are used more widely in community oncology networks through partnerships with academic centers. Early diagnostic testing and widespread insurance coverage under employer plans encourage patients to pursue aggressive treatment strategies. Survivorship programs support ongoing therapy for disease maintenance and relapse prevention, generating long-term medication needs. These clinical and structural factors maintain strong purchasing of chemotherapy, biologics and supportive-care agents that manage treatment side effects.
High cost of advanced therapies, limited access in rural regions and complex toxicity management restrain demand.
CAR T cell procedures and next-generation targeted drugs involve significant expense that may produce high cost-sharing obligations even with insurance. Rural patients face travel burdens to reach specialty infusion centers, which can delay care or reduce treatment intensity. Side effects such as cytokine-release syndrome require skilled monitoring that some facilities cannot provide, limiting adoption to qualified centers. These economic and geographic barriers reduce uniform treatment access across the country.
Shift toward precision medicine, increased at-home administration for maintenance therapy and rising clinical trial participation define key trends.
Oncologists incorporate genetic and molecular profiling to match patients with therapies that target specific mutations, improving efficacy and reducing unnecessary toxicity. Oral targeted drugs allow maintenance treatment at home with remote monitoring, reducing hospital reliance for long-term care. Patients participate in trials through decentralized study models that expand eligibility and accelerate access to investigational therapies. Healthcare networks integrate digital symptom reporting to aid early intervention and keep patients on therapy. These developments indicate sustained, innovation-focused demand for blood cancer treatment across the United States oncology ecosystem.
Analysis of the Demand for Blood Cancer Treatment in the USA by Region
Demand for blood cancer treatment in the United States continues to expand as precision therapies, early diagnostic capabilities, and access to specialized oncology centers improve patient outcomes. Treatment plans incorporate immunotherapy, targeted small-molecule drugs, and bone-marrow transplantation, requiring reliable reimbursement and strong clinical-trial participation nationwide. Rising leukemia, lymphoma, and myeloma incidence among aging populations drives higher therapeutic utilization in both inpatient and infusion-center environments. Investment decisions focus on availability of hematology specialists, genomic-testing coordination, and supportive-care frameworks to manage side effects. West USA leads growth at 9.9% CAGR, followed by South USA at 8.9%, Northeast USA at 8.0%, and Midwest USA at 6.9%, reflecting regional variation in oncology-infrastructure maturity.
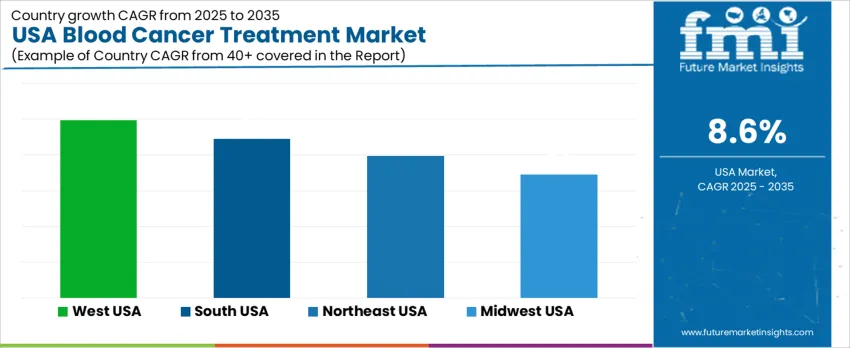
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 9.9% |
| South USA | 8.9% |
| Northeast USA | 8.0% |
| Midwest USA | 6.9% |
How is the West USA driving demand for blood cancer treatment?
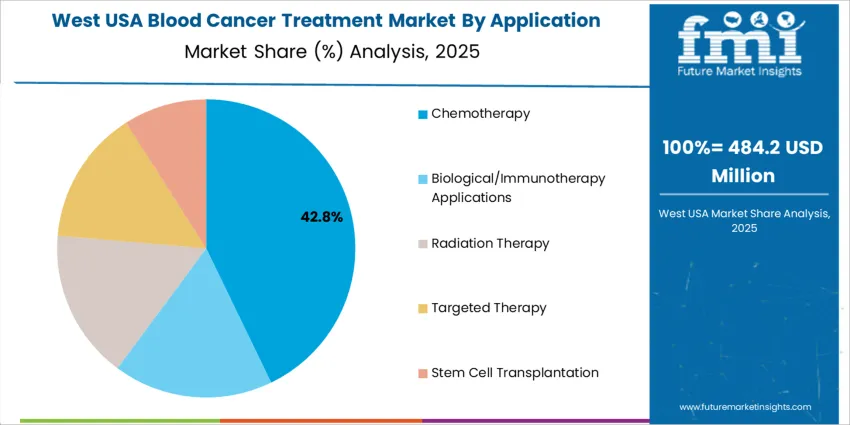
West USA demonstrates a strong 9.9% CAGR, supported by specialized hematology-oncology networks in California and Washington where clinical research accelerates uptake of CAR-T cell therapies and targeted biologics. Major cancer centers integrate genomic profiling into diagnostic workflows for accurate treatment alignment with disease subtypes. Infusion clinics maintain high throughput capacity for immunotherapies, requiring efficient supply-chain coordination for complex biologic products. Digital patient-engagement tools improve symptom reporting and therapy-adherence monitoring across large suburban populations. Payer evaluations include survival-rate improvement and hospitalization-avoidance metrics. Academic-industry partnerships increase clinical-trial enrollment, strengthening early access to investigational options. Procurement emphasizes cold-chain integrity, clinician-training requirements, and coordination with transplant organizations managing marrow-donor logistics.
- Precision-oncology expansion driving biologic USAge
- Clinical-trial participation improving therapy access
- Cold-chain delivery supporting biologic integrity
- Large outpatient infusion capacity reinforcing volumes
How is the South USA shaping adoption?
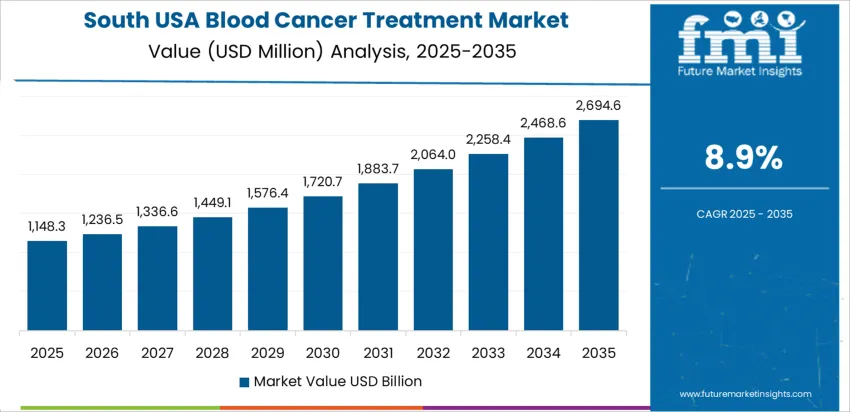
South USA posts 8.9% CAGR, driven by increasing blood cancer prevalence across Texas, Florida, and Georgia, where population aging and comorbidity rates elevate treatment demand. Regional oncology networks expand infusion-center availability to support timely initiation of immunotherapy and maintenance regimens. Hospitals adopt coordinated care pathways integrating pharmacists, oncologists, and case managers to manage complex medication schedules. Broader reimbursement access improves affordability for expensive biologic therapies. Logistics infrastructure along Gulf Coast ports enables reliable delivery of temperature-sensitive therapeutics. Education programs raise awareness of early screening among underserved communities, improving detection rates. Procurement decisions prioritize therapy outcomes, safety monitoring requirements, and compatibility with central-pharmacy oversight systems.
- Higher disease burden establishing strong therapy needs
- Expanded infusion infrastructure supporting treatment continuity
- Reimbursement alignment improving patient accessibility
- Diagnostic outreach improving early identification
How is the Northeast USA influencing therapy utilization?
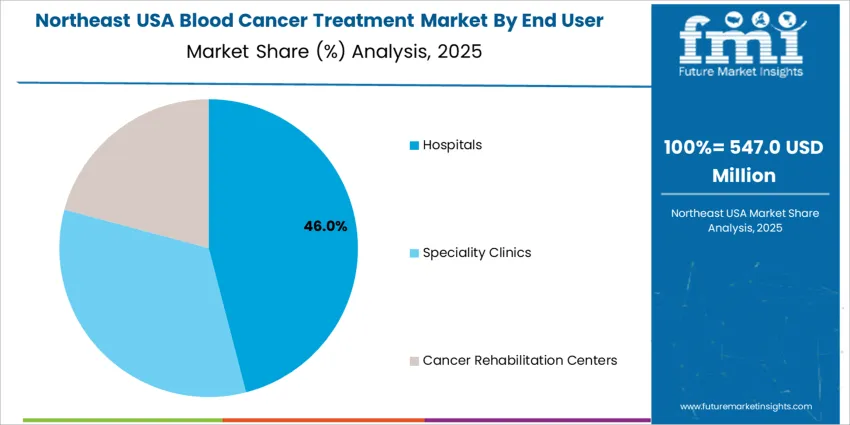
Northeast USA advances at 8.0% CAGR, anchored by leading cancer-research institutions in New York, New Jersey, and Massachusetts that validate therapeutic effectiveness through large clinical-trial cohorts. Hospitals deploy advanced cellular-therapy units enabling CAR-T administration under controlled conditions. Dense urban demographics encourage centralized oncology hubs with comprehensive genomic testing for treatment stratification. Payers enforce evidence-based therapy selection using validated clinical outcomes. Pharmaceutical-distribution centers maintain strong biologic-handling capabilities to minimize treatment schedule interruptions. Procurement leaders focus on standardized treatment protocols across multiple sites, ensuring uniform delivery of supportive-care medications and adverse-event monitoring tools.
- Research-center presence advancing novel therapy access
- CAR-T infrastructure supporting high-complexity care
- Standardized clinical pathways guiding procurement decisions
- Dense service networks improving patient throughput
How is the Midwest USA contributing to demand growth?
Midwest USA grows at 6.9% CAGR, influenced by expanding oncology service networks across Illinois, Ohio, and Michigan that integrate both hospital-based and community infusion centers. Chronic-disease management programs coordinate supportive therapies to sustain tolerance for prolonged treatment courses. Employers in industrial regions encourage health-plan initiatives improving cancer-screening participation. Procurement emphasizes reliability of biologic supply, streamlined product handling, and durable equipment supporting frequent infusion cycles. Rural oncology outreach enhances accessibility for patients living far from large medical hubs, positioning tele-oncology as a supplementary follow-up tool.
- Community-infusion access strengthening treatment continuity
- Supportive-care coordination improving therapy adherence
- Supply-chain reliability influencing purchasing criteria
- Tele-oncology helping serve remote patient groups
What does the competitive landscape for blood-cancer treatment demand in the United States look like?
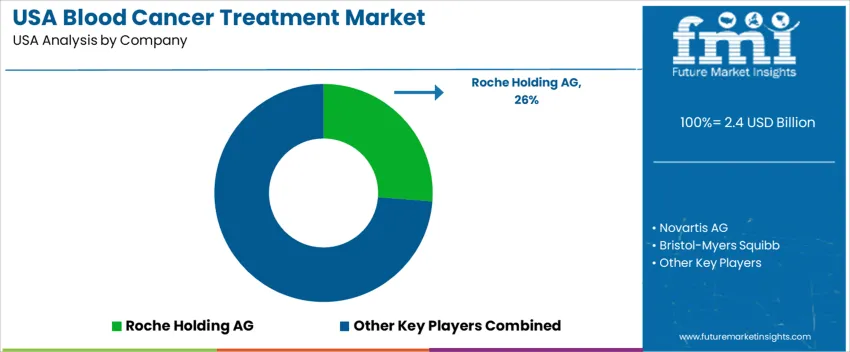
Blood-cancer treatments in the United States are driven by rising incidence rates of leukemia, lymphoma, and myeloma, together with the adoption of novel targeted therapies, immunotherapies, and cell-based therapies. Demand stems from hospitals, cancer-treatment centers, community oncology clinics, and specialized infusion facilities. Treatment choices depend on therapy effectiveness, safety profiles, regulatory approvals, and the ability to offer next-line options for resistant or relapsed disease. Roche Holding AG (through Genentech/Genomic affiliates) holds an estimated 26.2% share. Its portfolio includes monoclonal antibodies, antibody-drug conjugates, and supportive biologics used widely across hematologic malignancies, benefitting from broad clinical adoption in USA oncology centers.
Novartis AG contributes significantly via small-molecule inhibitors, CAR-T therapies, and targeted biologics for leukemia and lymphoma subtypes. Bristol-Myers Squibb (BMS) supports therapy demand through immune-modulating agents, cell therapies, and combination regimens for lymphoid cancers. Johnson & Johnson (Janssen) participates in both biologics and supportive-care therapies for multiple myeloma and other blood cancers, offering an established distribution and clinical support infrastructure. Gilead Sciences, Inc. adds strength through recently approved therapies and CAR-T assets, contributing to treatment options, especially for relapsed or refractory disease. Competition in the United States depends on clinical efficacy, breadth of indication, pipeline strength, insurer reimbursement alignment, and capacity to support complex treatment protocol,s including CAR-T and combination therapies. Demand continues to grow as innovative therapies and expanded screening raise treatment penetration and long-term survival outcomes.
Key Players in USA Blood Cancer Treatment Demand
- Roche Holding AG
- Novartis AG
- Bristol-Myers Squibb
- Johnson & Johnson
- Gilead Sciences, Inc.
Scope of the Report
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Application | Chemotherapy, Biological/Immunotherapy Applications, Radiation Therapy, Targeted Therapy, Stem Cell Transplantation |
| End User | Hospitals, Speciality Clinics, Cancer Rehabilitation Centers |
| Regions Covered | West USA, South USA, Northeast USA, Midwest USA |
| Key Companies Profiled | Roche Holding AG, Novartis AG, Bristol-Myers Squibb, Johnson & Johnson, Gilead Sciences, Inc. |
| Additional Attributes | Dollar sales by therapy modalities and end-user segments; clinical adoption trends in immunotherapies and targeted medicines; regional access differences across cancer care networks; focus on CAR-T therapies, stem cell transplant capacity, and survivorship support programs; biosimilar penetration impact; competitive mapping of oncology drug pipelines, reimbursement environment, and precision medicine integration in USA treatment pathways. |
USA Blood Cancer Treatment Demand by Segments
Application:
- Chemotherapy
- Biological/Immunotherapy Applications
- Radiation Therapy
- Targeted Therapy
- Stem Cell Transplantation
End User:
- Hospitals
- Speciality Clinics
- Cancer Rehabilitation Centers
Region:
- West USA
- South USA
- Northeast USA
- Midwest USA
Frequently Asked Questions
How big is the demand for blood cancer treatment in USA in 2025?
The demand for blood cancer treatment in USA is estimated to be valued at USD 2.4 billion in 2025.
What will be the size of blood cancer treatment in USA in 2035?
The market size for the blood cancer treatment in USA is projected to reach USD 5.6 billion by 2035.
How much will be the demand for blood cancer treatment in USA growth between 2025 and 2035?
The demand for blood cancer treatment in USA is expected to grow at a 8.6% CAGR between 2025 and 2035.
What are the key product types in the blood cancer treatment in USA?
The key product types in blood cancer treatment in USA are chemotherapy, biological/immunotherapy applications, radiation therapy, targeted therapy and stem cell transplantation.
Which end user segment is expected to contribute significant share in the blood cancer treatment in USA in 2025?
In terms of end user, hospitals segment is expected to command 44.0% share in the blood cancer treatment in USA in 2025.
Table of Content
- Executive Summary
- USA Market Outlook
- Demand to side Trends
- Supply to side Trends
- Technology Roadmap Analysis
- Analysis and Recommendations
- Market Overview
- Market Coverage / Taxonomy
- Market Definition / Scope / Limitations
- Market Background
- Market Dynamics
- Drivers
- Restraints
- Opportunity
- Trends
- Scenario Forecast
- Demand in Optimistic Scenario
- Demand in Likely Scenario
- Demand in Conservative Scenario
- Opportunity Map Analysis
- Product Life Cycle Analysis
- Supply Chain Analysis
- Investment Feasibility Matrix
- Value Chain Analysis
- PESTLE and Porter’s Analysis
- Regulatory Landscape
- Regional Parent Market Outlook
- Production and Consumption Statistics
- Import and Export Statistics
- Market Dynamics
- USA Market Analysis 2020 to 2024 and Forecast, 2025 to 2035
- Historical Market Size Value (USD Million) Analysis, 2020 to 2024
- Current and Future Market Size Value (USD Million) Projections, 2025 to 2035
- Y to o to Y Growth Trend Analysis
- Absolute $ Opportunity Analysis
- USA Market Pricing Analysis 2020 to 2024 and Forecast 2025 to 2035
- USA Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By Application
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By Application , 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By Application , 2025 to 2035
- Chemotherapy
- Biological/Immunotherapy Applications
- Radiation Therapy
- Targeted Therapy
- Stem Cell Transplantation
- Y to o to Y Growth Trend Analysis By Application , 2020 to 2024
- Absolute $ Opportunity Analysis By Application , 2025 to 2035
- USA Market Analysis 2020 to 2024 and Forecast 2025 to 2035, By End User
- Introduction / Key Findings
- Historical Market Size Value (USD Million) Analysis By End User, 2020 to 2024
- Current and Future Market Size Value (USD Million) Analysis and Forecast By End User, 2025 to 2035
- Hospitals
- Speciality Clinics
- Cancer Rehabilitation Centers
- Y to o to Y Growth Trend Analysis By End User, 2020 to 2024
- Absolute $ Opportunity Analysis By End User, 2025 to 2035
- Market Structure Analysis
- Competition Dashboard
- Competition Benchmarking
- Market Share Analysis of Top Players
- By Regional
- By Application
- By End User
- Competition Analysis
- Competition Deep Dive
- Roche Holding AG
- Overview
- Product Portfolio
- Profitability by Market Segments (Product/Age /Sales Channel/Region)
- Sales Footprint
- Strategy Overview
- Marketing Strategy
- Product Strategy
- Channel Strategy
- Novartis AG
- Bristol-Myers Squibb
- Johnson & Johnson
- Gilead Sciences, Inc.
- Roche Holding AG
- Competition Deep Dive
- Assumptions & Acronyms Used
- Research Methodology
List of Tables
- Table 1: USA Market Value (USD Million) Forecast by Region, 2020 to 2035
- Table 2: USA Market Value (USD Million) Forecast by Application , 2020 to 2035
- Table 3: USA Market Value (USD Million) Forecast by End User, 2020 to 2035
- Table 4: USA Market Value (USD Million) Forecast by Country, 2020 to 2035
- Table 5: USA Market Value (USD Million) Forecast by Application , 2020 to 2035
- Table 6: USA Market Value (USD Million) Forecast by End User, 2020 to 2035
List of Figures
- Figure 1: USA Market Pricing Analysis
- Figure 2: USA Market Value (USD Million) Forecast 2020-2035
- Figure 3: USA Market Value Share and BPS Analysis by Application , 2025 and 2035
- Figure 4: USA Market Y-o-Y Growth Comparison by Application , 2025-2035
- Figure 5: USA Market Attractiveness Analysis by Application
- Figure 6: USA Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 7: USA Market Y-o-Y Growth Comparison by End User, 2025-2035
- Figure 8: USA Market Attractiveness Analysis by End User
- Figure 9: USA Market Value (USD Million) Share and BPS Analysis by Region, 2025 and 2035
- Figure 10: USA Market Y-o-Y Growth Comparison by Region, 2025-2035
- Figure 11: USA Market Attractiveness Analysis by Region
- Figure 12: USA Market Incremental Dollar Opportunity, 2025-2035
- Figure 13: USA Market Value Share and BPS Analysis by Country, 2025 and 2035
- Figure 14: USA Market Value Share and BPS Analysis by Application , 2025 and 2035
- Figure 15: USA Market Y-o-Y Growth Comparison by Application , 2025-2035
- Figure 16: USA Market Attractiveness Analysis by Application
- Figure 17: USA Market Value Share and BPS Analysis by End User, 2025 and 2035
- Figure 18: USA Market Y-o-Y Growth Comparison by End User, 2025-2035
- Figure 19: USA Market Attractiveness Analysis by End User
- Figure 20: USA Market - Tier Structure Analysis
- Figure 21: USA Market - Company Share Analysis

