The USA keloid treatment demand is valued at USD 2.6 billion in 2025 and is expected to reach USD 5.0 billion by 2035, reflecting a CAGR of 6.7%. Rising treatment-seeking behaviour, increased diagnosis of menstrual disorders, and broader access to over-the-counter pain relief options support ongoing growth. Public health campaigns addressing menstrual wellbeing and improved availability of non-prescription therapies also contribute to wider adoption.
NSAIDs represent the leading treatment category, as they provide first-line relief from cramping and inflammation. These agents remain widely recommended by healthcare providers due to established clinical efficacy and affordability. Oral contraceptives and hormonal therapies form an additional treatment pathway for patients with recurrent or severe conditions.
Demand is concentrated in the West USA, the South USA, and the Northeast USA. These regions record higher prescription volumes through established women’s health networks and retail pharmacy penetration. Telehealth services and subscription-based medication delivery are expanding accessibility. Top suppliers include Bayer, Pfizer Inc., Johnson & Johnson, Perrigo Company plc, and GlaxoSmithKline plc. Their focus includes product availability across both OTC and prescription channels, support for gynecological guidance, and enhanced consumer awareness of treatment options.
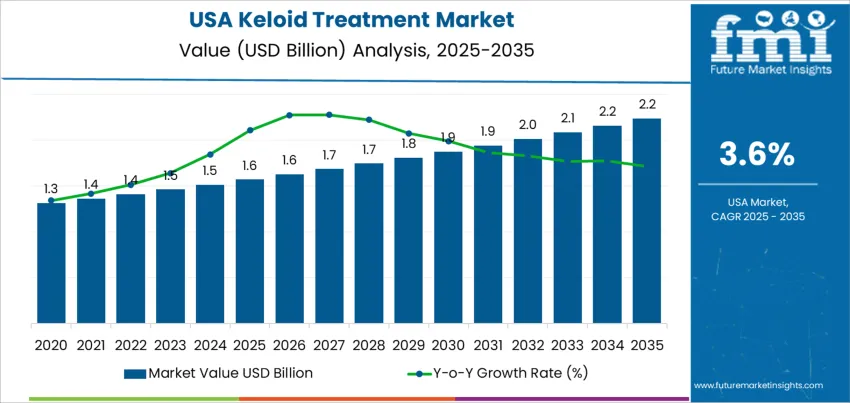
Growth rate volatility for demand for keloid treatment in the United States remains low due to steady clinical necessity and consistent patient awareness. Keloids do not resolve without intervention, which maintains recurring demand across dermatology and plastic surgery practices. Treatment methods such as corticosteroid injections, surgical excision with adjunct therapy, silicone-based products, and laser procedures provide continuous activity across both medical and aesthetic settings. These elements support a stable base with limited exposure to rapid downturns.
Variation in yearly growth can appear when new technologies gain traction. Interest in cryotherapy, radiation-supported excision, and laser platforms can temporarily raise procedure volumes. Reimbursement policy changes also create minor fluctuations because coverage varies by clinical severity. Broader consumer attention to scar aesthetics influences short-term rises in demand, particularly through direct-to-consumer channels. The volatility profile indicates predictable year-to-year expansion. Clinical demand remains independent of discretionary economic cycles while innovation adds incremental peaks rather than disruptive shifts. Stability across healthcare infrastructure and specialist availability provides a balanced growth curve without significant troughs.
| Metric | Value |
|---|---|
| USA Keloid Treatment Sales Value (2025) | USD 1.6 billion |
| USA Keloid Treatment Forecast Value (2035) | USD 2.2 billion |
| USA Keloid Treatment Forecast CAGR (2025-2035) | 3.6% |
Demand for keloid treatment in the USA is increasing because more individuals seek medical support to address visible and sometimes painful scar overgrowth following surgery, acne, burns or piercings. Dermatology and plastic surgery practices report rising consultations as patients pursue improved cosmetic outcomes, particularly for keloids located on the chest, shoulders, jawline and earlobes. Greater social awareness of scar management encourages earlier intervention, which increases use of therapies such as corticosteroid injections, cryotherapy, laser treatment and silicone-based products.
Higher prevalence of keloid formation among specific ethnic groups strengthens treatment uptake in diverse communities across the USA. Advances in laser technology and combination therapy provide more tailored approaches that can reduce recurrence frequency. Expansion of aesthetic centers and dermatology networks enhances accessibility for insured and self-paying patients. Teledermatology services also help individuals receive ongoing care without frequent in-office visits. Constraints include variable treatment response, long care timelines and high costs for multi-session therapies. Insurance coverage may be limited when treatment is considered cosmetic rather than medically necessary. Some patients delay care due to fear of recurrence, discomfort during procedures or limited knowledge of available options.
Demand for keloid treatment in the United States is driven by rising dermatological consultations, increased cosmetic concerns, and higher incidence among individuals with darker skin types. Treatment choices depend on scar severity, recurrence risk, and access to therapy. Minimally invasive solutions and procedures supported by clinical reimbursement continue gaining adoption, while hospital-led management dominates complex and large-scar cases.
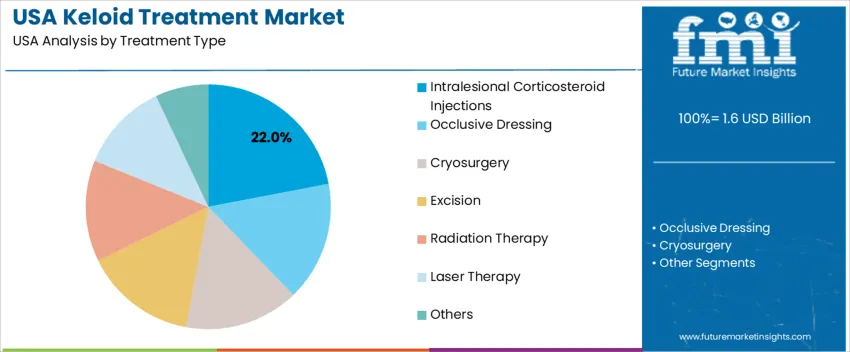
Intralesional corticosteroid injections account for 22.0% and remain the primary approach due to effectiveness in reducing inflammation and keloid size. Their familiarity among dermatologists and cost efficiency support broad USAge in outpatient care. Other modalities, including occlusive dressings, cryosurgery, excision, radiation therapy, laser therapy, and combination approaches, cover remaining demand. These methods are typically selected for persistent or complex lesions, cosmetic improvement, and long-term recurrence prevention. Clinical decisions are guided by scar location, patient sensitivity, and expected treatment downtime. Access to advanced modalities varies across facilities, with bundled therapies increasingly considered for higher treatment success.
Key Points:
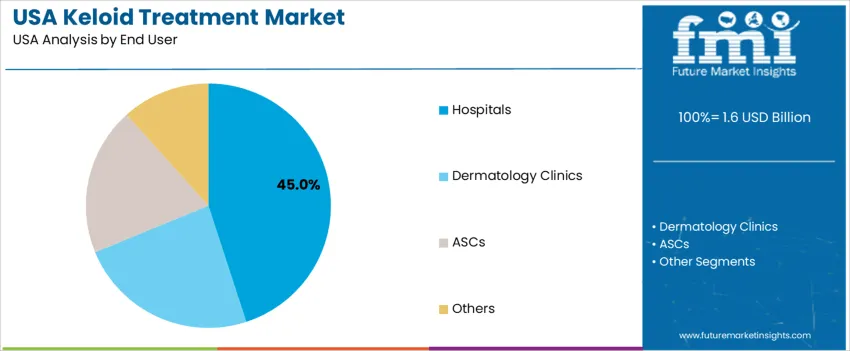
Hospitals hold 45.0%, reflecting their role in managing extensive keloids where surgical, radiological, and multi-stage interventions are necessary. Dermatology clinics and ambulatory surgical centers (ASCs) contribute the remaining segment share, driven by demand for outpatient-based skin procedures with shorter recovery times. Growth in clinic-based care aligns with cosmetic enhancements and wider access to specialist dermatology services. Treatment availability, insurance reimbursement, and physician specialization influence patient flow across facilities.
Key Points:
Growth of dermatology access, increased demand for post-surgical scar management and rising cosmetic treatment expectations are driving demand.
In the United States, keloid treatment demand increases as more patients pursue dermatology services covered by private insurance and employer health plans. Surgical procedures, ear piercings and aesthetic interventions contribute to scar formation, leading patients to seek preventive and corrective therapies. Clinics in major cities offer steroid injections, laser therapy and cryotherapy to control scar growth and improve appearance. Younger adults rely on aesthetic improvement as part of confidence and workplace readiness, supporting regular visits to dermatologists and medical spas. Teledermatology expands early evaluation and referrals, helping patients start treatment sooner and sustain adherence across multiple sessions.
Insurance coverage limitations, risk of recurrence and variable patient awareness restrain demand.
Some treatment options such as laser therapy or radiation for severe keloids may not be fully reimbursed, resulting in significant out-of-pocket expense for cosmetic-focused visits. Recurrence after surgical removal can discourage patients from pursuing additional procedures, reducing long-term demand for intensive therapies. Awareness about keloids remains uneven, and individuals with darker skin tones who face higher risk may not seek specialty care until discomfort becomes severe. Geographic differences in dermatology availability also affect access in rural communities. These clinical and financial factors create gradual rather than rapid adoption of comprehensive keloid management.
Shift toward combination therapy protocols, increased adoption of minimally invasive approaches and rising use of digital monitoring define key trends.
Dermatologists combine steroid injections, silicone therapy and energy-based devices to improve outcomes and reduce recurrence risk. Patients prefer non-surgical methods that minimize downtime and improve cosmetic appearance without scarring from new incisions. Wearable or app-based monitoring supports treatment scheduling and adherence for silicone sheets and pressure earrings used in ear keloid care. Research into biologic and immunomodulatory approaches gains attention among academic centers seeking targeted solutions. These developments indicate sustained, patient-focused demand for keloid treatment across the United States within both medical and cosmetic care settings.
Demand for keloid treatment in the United States is influenced by population diversity, prevalence of post-surgical scarring, and better access to dermatology care. Providers are adopting multimodal therapies including corticosteroid injections, laser resurfacing, cryotherapy, silicone-based products, and emerging biologics. Digital dermatology platforms support earlier patient engagement, particularly among first-time scar-care users. Private insurance coverage affects adoption pathways for advanced interventions, while OTC topical solutions maintain significant volume. Awareness campaigns surrounding scar management contribute to broader treatment seeking. Growth differs by region: West USA leads with 4.2% CAGR, South USA follows at 3.7%, Northeast USA at 3.3%, and Midwest USA shows 2.9% gradual expansion.
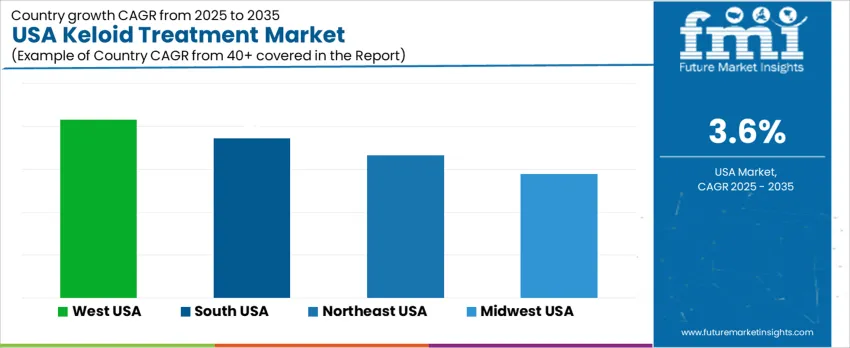
| Region | CAGR (2025-2035) |
|---|---|
| West USA | 4.2% |
| South USA | 3.7% |
| Northeast USA | 3.3% |
| Midwest USA | 2.9% |
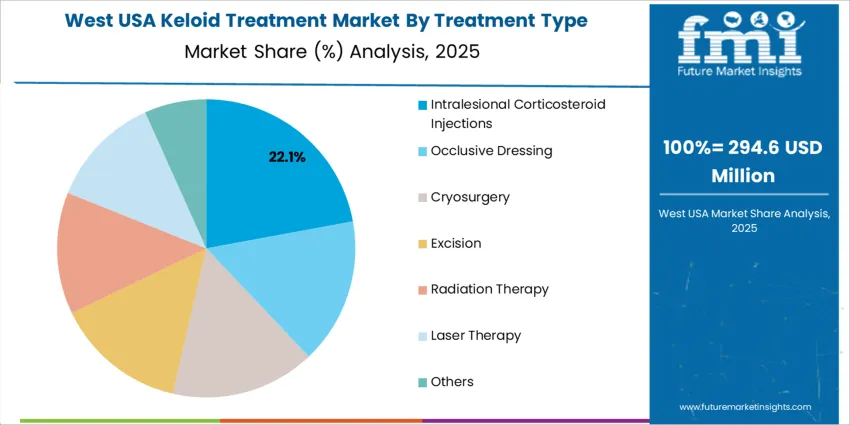
West USA records a 4.2% compound annual growth rate, the fastest among all regions. Dermatology networks across California and Washington support broad clinical access to combination therapies addressing keloid recurrence and pain. Specialists offer cryotherapy, radiation-assisted reduction, and steroid protocols aligned with updated scar-management guidelines from major medical associations. Urban populations with high cosmetic-care participation seek treatments that maintain skin appearance following piercings, tattoos, and elective surgery. Consumer-direct channels distribute silicone gels, pressure dressings, and scar-care products with clear USAge guidance. Technology adoption enables virtual evaluations that direct patients toward clinic visits only when needed, improving efficiency. Procurement teams assess effectiveness evidence, handling ease, and continuity of product supply to support daily application routines required for outcome stability.
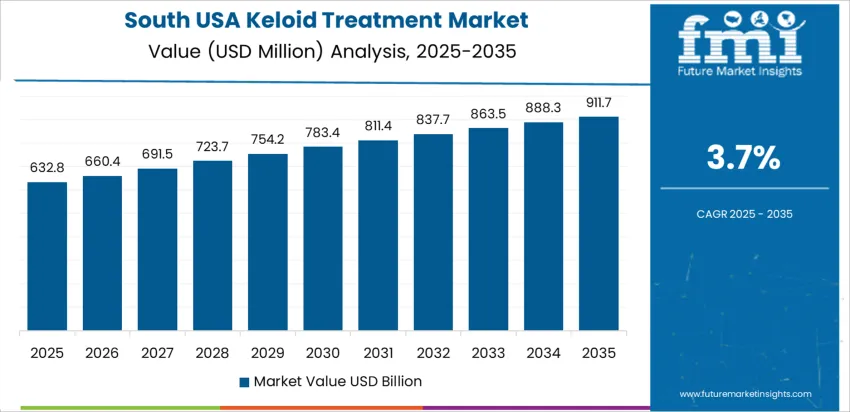
South USA exhibits approximately 3.7% CAGR, influenced by a larger share of high-melanin skin tones in Texas, Georgia, and Florida, where keloid susceptibility is elevated. Providers emphasize careful post-surgical monitoring to reduce scar overgrowth after trauma or elective procedures. Hospitals integrate preventive protocols using pressure therapy and silicone dressings during recovery. Outreach programs in suburban and rural locations improve recognition of early-stage keloid development. Retail shelves carry topical products positioned for sensitive-skin USAge, supporting self-initiated intervention. Clinic procurement focuses on storage-safe biologics and newer cryosurgical tools for controlled treatments. Economic decision-making prioritizes durable supplies and scheduling flexibility for repeat-session therapies.
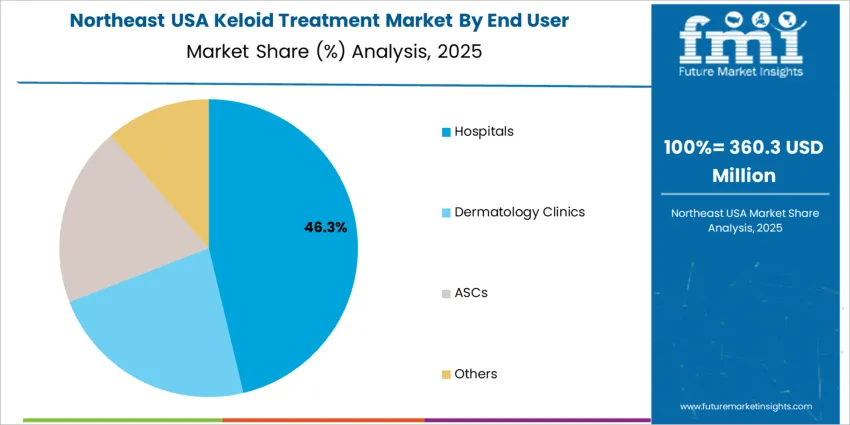
Northeast USA advances at around 3.3% CAGR, driven by dermatology specialists in New York, New Jersey, and Massachusetts who frequently manage keloids linked to surgeries and skin injuries. Hospitals use structured referral pathways enabling faster access to evidence-based therapies. Dense urban retail networks ensure extensive availability of silicone patches, anti-inflammatory topicals, and scar-softening solutions. Consumers value portable packaging for use during commutes and daily mobility. Payors increasingly review reimbursement for advanced laser therapy when medically justified. Procurement focuses on sterility standards, package clarity, and shelf-life reliability for high-turnover product ranges.
Midwest USA shows a 2.9% CAGR, reflecting steady but modest expansion due to lower population density and fewer high-intensity cosmetic-care hubs compared with coastal states. Primary-care physicians manage early interventions and direct patients to dermatology specialists when keloids impact function or comfort. Local pharmacies remain key providers of OTC products with straightforward application instructions. Manufacturers emphasize cost-efficient formulations that maintain stability in variable climate conditions. Clinical teams evaluate cryotherapy and steroid-delivery devices designed for repeat treatments at predictable intervals. Employers and insurers focus on minimizing recovery-related productivity losses associated with keloid discomfort and limited mobility.
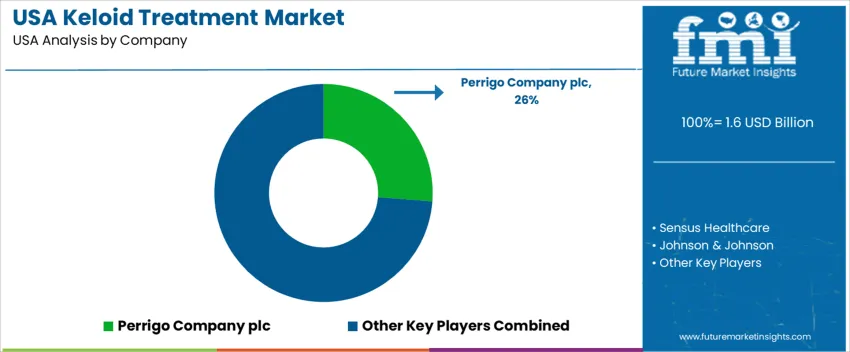
Keloid treatment demand in the United States centers on dermatology clinics, plastic surgery practices, and oncology-linked scar care pathways. Treatment strategies include corticosteroid injections, silicone-based therapy, cryotherapy, laser procedures, and emerging RNA-based approaches. Patient preferences reflect non-invasive options and sustained relief from itching, pain, and scar thickening. Providers consider recurrence risk, tissue tolerance, and compatibility with combination therapy aimed at reducing scar volume over time. Reimbursement consistency influences product adoption within dermatology and outpatient surgical settings. HRA Pharma (Perrigo Company plc) holds an estimated 26% share through silicone scar care products distributed widely in U.S. retail and clinical channels.
Distribution reach and established dermatologist familiarity support its leading role. Sensus Healthcare participates with superficial radiation therapy units used in prevention of regrowth after excision. Johnson & Johnson provides wound-care materials and topical products applied in follow-up management. Merz Aesthetics engages through injectable formulations used in dermatology, supported by established office-based treatment capabilities. RXi Pharmaceuticals (Phio Pharmaceuticals) focuses on RNA-based therapeutic programs addressing fibroblast activity, contributing to innovation pipelines aimed at lowering recurrence in challenging cases. Competitive positioning depends on demonstrated reduction in recurrence rates, clarity of clinical guidance for combination protocols, and access to treatment within insurance-approved care models across the United States.
| Items | Values |
|---|---|
| Quantitative Units | USD billion |
| Treatment Type | Intralesional Corticosteroid Injections, Occlusive Dressing, Cryosurgery, Excision, Radiation Therapy, Laser Therapy, Others |
| End User | Hospitals, Dermatology Clinics, ASCs, Others |
| Regions Covered | West USA, South USA, Northeast USA, Midwest USA |
| Key Companies Profiled | Perrigo Company plc, Sensus Healthcare, Johnson & Johnson, Merz Aesthetics, Phio Pharmaceuticals (formerly RXi) |
| Additional Attributes | Dollar sales by treatment type and end-user segments; regional variations in access to dermatologic care and cosmetic procedures; growth driven by emerging minimally invasive keloid therapies and combination treatment protocols; increased insurance reimbursement for corticosteroid injections vs. elective laser therapies; development of RNA-based therapeutics and post-surgical recurrence prevention strategies across USA. |
How big is the demand for keloid treatment in USA in 2025?
The demand for keloid treatment in USA is estimated to be valued at USD 1.6 billion in 2025.
What will be the size of keloid treatment in USA in 2035?
The market size for the keloid treatment in USA is projected to reach USD 2.2 billion by 2035.
How much will be the demand for keloid treatment in USA growth between 2025 and 2035?
The demand for keloid treatment in USA is expected to grow at a 3.6% CAGR between 2025 and 2035.
What are the key product types in the keloid treatment in USA?
The key product types in keloid treatment in USA are intralesional corticosteroid injections, occlusive dressing, cryosurgery, excision, radiation therapy, laser therapy and others.
Which end user segment is expected to contribute significant share in the keloid treatment in USA in 2025?
In terms of end user, hospitals segment is expected to command 45.0% share in the keloid treatment in USA in 2025.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.