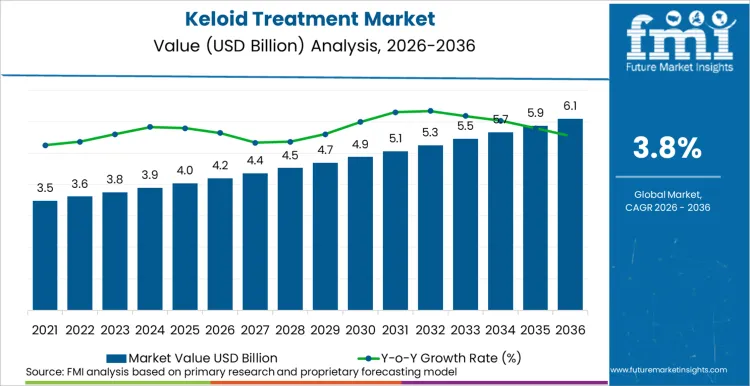
The global keloid treatment market is on track to achieve a valuation of USD 6,202.8 million by 2036, expanding from USD 4,292.2 million in 2026 at a CAGR of 3.8%. As per Future Market Insights, expansion is structurally underpinned by the growing clinical evidence base for multi-modal keloid management which combines laser therapy, pharmacological agents, and emerging molecular inhibitors to achieve success rates that single-modality treatments could not deliver.
The global prevalence of keloid scarring affects an estimated 100 million people, with significantly higher incidence in populations of African, Asian, and Hispanic descent, creating a large and underserved addressable patient population. This clinical need compels device manufacturers and pharmaceutical companies to invest in keloid-specific product development rather than repurposing general dermatology tools. Simultaneously, the regulatory environment is evolving to recognize keloid treatment as a distinct clinical category with dedicated clearance pathways.
Thomas J. Appio, CEO of Bausch Health, stated: 'This improved technology marks a significant milestone in our commitment to advancing aesthetic medicine and enhancing the patient experience.' This confirms that major medical device and pharmaceutical companies now view keloid and scar management as a strategically important growth category rather than a niche aesthetic concern. FMI is of the opinion that the elevation of keloid treatment from cosmetic to clinical medicine is the most significant structural shift in this market, as it opens pathways for insurance reimbursement and institutional procurement that were previously unavailable.
The clinical and commercial landscape in 2025 and 2026 has been reshaped by a series of regulatory milestones and product launches. Bausch Health and Solta Medical announced the launch of Fraxel FTX, a next-generation fractional laser system for skin resurfacing and keloid management, in April 2025. Sciton received FDA 510(k) clearance for its Joule 1064nm System for the reduction of red pigmentation in keloid scars in December 2025. BirchBioMed received regulatory approval from Health Canada for its FS2 (KynA) topical cream as a natural health product for mature keloid scars in February 2026.
Syntara dosed the first patient in its SATELLITE Phase 1c trial evaluating SNT-6302, a topical pan-Lysyl Oxidase inhibitor for keloid softening, in May 2025. As per FMI, this rapid succession of device clearances and drug pipeline milestones confirms that the keloid treatment market is entering a product innovation cycle that will redefine standard of care over the next decade.
The keloid treatment market is controlled by a fragmented structure where medical device companies (Bausch Health/Solta, Sciton, Candela), pharmaceutical firms (Merck, Galderma), and specialized clinics (London Keloid Scar Clinic) each hold influence over different modalities. FMI analysts observe that no single company controls the full multi-modal treatment pathway, creating both competitive tension and partnership opportunities across the device-pharma-clinic value chain.
Pricing asymmetry in the keloid treatment market is extreme. Laser-based treatments command USD 500 to USD 3,000 per session with limited insurance coverage in most markets, while corticosteroid injections cost USD 50 to USD 200 per session with broader reimbursement eligibility. As per FMI, this pricing gap restricts advanced laser and molecular therapies to self-pay patients in most geographies, creating a two-tier market where treatment modality selection is driven by ability to pay rather than clinical evidence.
Sourcing fragility in the keloid treatment market is concentrated in the specialized laser systems and emerging biological agents. Fractional CO2 and Nd:YAG laser platforms require precision optical components sourced from a limited number of manufacturers. Clinical-stage molecular therapies (SNT-6302, STP705) rely on novel formulation and delivery technologies that are not yet scaled. FMI opines that the supply chain for next-generation keloid therapies will remain constrained until these pipeline products achieve commercial manufacturing scale.
Brand and compliance risks center on the distinction between cosmetic and medical claims. Manufacturers who position keloid treatments as purely cosmetic face pricing and reimbursement limitations, while those who pursue medical device or pharmaceutical classification face longer regulatory timelines and higher clinical evidence requirements. BirchBioMed's Health Canada approval of FS2 as a natural health product illustrates a middle-path regulatory strategy. FMI is of the opinion that regulatory classification will be the primary strategic decision point for keloid treatment companies over the next five years.
Geographic leverage is concentrated in North America and Western Europe, which account for the highest per-patient spending on keloid treatment driven by advanced dermatology infrastructure and higher rates of elective aesthetic procedures. Asia Pacific, particularly Japan and South Korea, is a high-growth market driven by cultural emphasis on skin aesthetics and strong dermatology technology ecosystems. Africa and the Middle East have the highest keloid prevalence but the lowest treatment penetration due to healthcare infrastructure constraints.
The executive reframing for the keloid treatment market is that this is transitioning from a cosmetic concern to a clinical fibrotic disease management category. FMI analysts emphasize that medical device and pharmaceutical executives must position keloid treatment portfolios within broader fibrotic disease platforms, leveraging shared R&D across keloid, hypertrophic scarring, and pulmonary/hepatic fibrosis to justify the clinical investment required for regulatory advancement.
Future Market Insights projects the keloid treatment market to expand at a CAGR of 3.8% from 2026 to 2036, increasing from USD 4,292.2 Million in 2026 to USD 6,202.8 Million by 2036.
FMI Research Approach: FMI proprietary forecasting model based on keloid prevalence data, dermatology procedure volume tracking, and pipeline product revenue projections.
FMI analysts perceive the market evolving toward multi-modal treatment protocols that combine fractional laser therapy, topical molecular inhibitors, and injectable biologics to achieve higher clinical success rates than any single modality.
FMI Research Approach: Clinical trial registry analysis and dermatology practice pattern surveys.
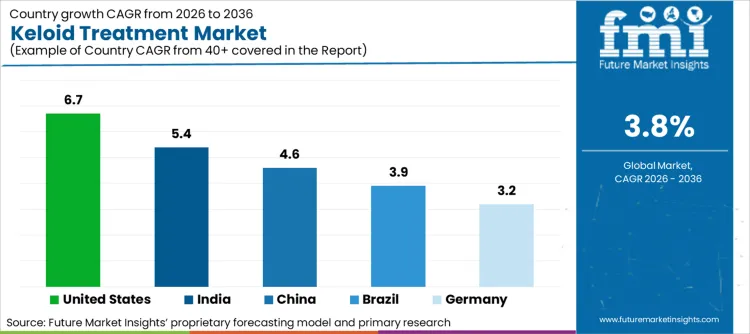
The United States holds a significant share of the global keloid treatment market by value which is supported by the highest per-patient spending on dermatological procedures and a large addressable patient population across diverse ethnic groups.
FMI Research Approach: FMI country-level revenue modeling by dermatology practice density and procedure volume per practice.
The global keloid treatment market is projected to reach USD 6,202.8 Million by 2036.
FMI Research Approach: FMI long-term revenue forecast derived from keloid prevalence growth projections and new product adoption curve modeling.
The keloid treatment market includes revenue from devices, pharmaceuticals, and clinical services used to prevent, reduce, or manage keloid scars, covering laser systems, injectable corticosteroids, silicone-based topicals, cryotherapy devices, and emerging molecular inhibitors.
FMI Research Approach: FMI market taxonomy and inclusion-exclusion framework.
Globally unique trends include the FDA clearance of keloid-specific laser wavelengths (1064nm), the clinical advancement of topical LOX inhibitors (SNT-6302), and the regulatory approval of natural health product classifications for keloid management.
FMI Research Approach: FDA 510(k) clearance database and ClinicalTrials.gov keloid pipeline tracking.
| Metric | Details |
|---|---|
| Industry Size (2026) | USD 4,292.2 Million |
| Industry Value (2036) | USD 6,202.8 Million |
| CAGR (2026 to 2036) | 3.8% |
Source: Future Market Insights (FMI) analysis, based on proprietary forecasting model and primary research
The North American market for keloid treatments is growing as more people want cosmetic procedures. Advanced scar treatments are becoming more popular. Plus, big skincare companies are involved in launching new products.
In the USA, there’s a lot of support for certain treatments, and people have good access to laser and cryotherapy. But there are problems too. Many worry about how well these treatments work over time. Also, the costs for cosmetic procedures can be high. On the bright side, more people are using biologic therapies. AI tools for skin assessments are coming into play, and telemedicine for skin care is on the rise. These factors are likely to boost growth in the North American market.
Europe is a leading keloid treatment market owing to government-sponsored dermatology research, growing demand for non-surgical procedures, and growing application of regenerative medicine for the treatment of scars. Germany, France, and the UK are leading markets, where the advantages of structured healthcare policies and high consumer spending on aesthetic procedures kick in. Yet, strict supervision by the European Medicines Agency (EMA) regulations, cost-containment actions, and volatility in public coverage policies could potentially restrict widespread application.
The marketplace is seeing higher demand for combination therapies, arrival of sophisticated laser treatment modalities, and investment in gene-based scar treatments by research. Meanwhile, digital dermatology solutions are picking up popularity, improving initial scar detection as well as treatment planning on a personalized basis within the region.
Keloid treatment market is booming in the Asia-Pacific region on account of increasing keloid scarring incidences, more awareness about scar revision techniques, and increasing affordability for laser therapy. China, Japan, and India are the key markets where new dermatological service providers are emerging; medical tourism for aesthetic procedures is increasing, and there is a growing consumer preference for herbal-based scar management solutions.
However, challenges like inconsistent regulatory frameworks, a few trained dermatologists in remote rural areas, and affordability could impede growth. Fast penetration into the region is now being ensured by a rising presence of international skincare brands and solutions for the at-home treatment of scars, alongside the fast-paced development of non-invasive therapeutic technologies. AI-based mobile dermatology platforms are fostering early diagnosis and treatment access for keloids, especially in remote areas.
High recurrence rate after treatment is a major challenge
Effective management of keloids is also very challenging owing to a high rate of recurrence. Even with enhanced modalities that incorporate surgical removal, corticosteroid injection, laser therapy, and radiotherapy, keloids continue to recur, hence influencing long-term management. This obstacle undermines confidence on the part of the patients in the available treatments, thereby necessitating combination therapies that eventually turn out to be expensive and time-consuming.
Further complicating the dilemma is the absence of a treatment with universal effectiveness. In contrast, investigations on biologics and gene-based therapies show promise for curing keloids; the availability and affordability of such treatments could become another issue in Keloid treatment.
Additionally, new innovations in treatment and a better understanding of keloids from the molecular perspective are required to tackle this major challenge. For example, as the market will mature, any company focusing on long-term, recurrence-prevention solutions will likely gain a temporary competitive advantage.
Development of biologic and regenerative therapies present lucrative growth opportunity
There is a opportunity to enhance keloid treatments using new regenerative and biologic therapies. As of today, standard treatments such as surgery and steroid injections do not always succeed. This makes many seek stronger, longer-lasting solutions.
Biologic medications, such as monoclonal antibodies and gene therapies, are able to attack the issues at a very fundamental level. They work to prevent additional scar tissue from developing. in addition, regenerative medicine involves stem cell therapy. This might be able to make skin heal more effectively and prevent keloid development.
As technology advances, such new treatments may revolutionize how we treat keloids. Rather than merely dealing with the scarring, they may address why they occur to begin with. More funds are being put into dermatology and biotech, and patients are becoming increasingly savvy about what their choices are. This gives businesses developing these advanced treatments a tangible opportunity to head up this space and expand their companies.
Advancements in Laser and Cryotherapy Treatments
With more fractional CO2 and pulsed dye lasers (PDLs) gaining acceptance along with cryotherapy, keloid treatment is now being approached from a more non-invasive and effective standpoint. Fractional CO2 lasers remodel collagen while eliminating keloids; the PDL attacks the blood vessels in the scar tissue to reduce redness and inflammation; cryotherapy freezes keloids with liquid nitrogen to reduce scar tissue and achieve pain minimizing effects.
Thus, the therapies appear to be more feasible alternatives to surgery or corticosteroid drug injections, with lower side effects and lower recurrence rates. With tremendous technological progress underway today, acceptance of these treatments is witnessing a quick surge in dermatologists' and patients' acceptance, which in turn becomes another significant parameter driving the upsurge of market growth.
In the last four years, from 2020 to 2024, the keloid treatment market has been growing steadily. More people are learning about scar management, and dermatology treatments are getting better. As a result, there's a higher demand for both medical and beauty solutions. This has led to more non-invasive treatments like laser therapy, cryotherapy, and silicone-based products.
Looking forward in year between 2025 to 2035, some new trends are changing the game too. There are now biologic and gene therapies that target keloids at the source. Using laser technology and combining different treatments is helping people see better results.
Plus, more folks are getting access to teledermatology and online shopping for these treatments. Healthcare spending is rising in many places, which is helping the market grow. Continued investments in research and new ideas will keep driving this growth. Personalizing treatments will also be a big part of what’s next.
| Category | 2021 to 2025 Trends |
|---|---|
| Regulatory Landscape | Prioritization of efficacy and safety of novel treatments, with regulatory authorities allowing fast-tracked approvals for treatments that target unmet medical needs. |
| Technological Advancements | Use of laser treatments and corticosteroid injections improving specificity and effectiveness of treatments, resulting in enhanced appearance of scars and patient satisfaction |
| Consumer Demand | Growing awareness leading to growing demand for less expensive yet effective therapies, wherein patients demand treatments with better cosmetic results and fewer side effects. |
| Market Growth Drivers | Increased incidence of keloid formations, high research and development expenditure, and encouraging government policies encouraging innovation in scar management therapies. |
| Sustainability | Early attempts at green manufacturing processes and lowering the environmental footprint of pharmaceutical production, with certain firms embracing green chemistry methods. |
| Supply Chain Dynamics | Dependence on mature distribution channels, with emphasis placed on the presence of therapies within urban and peri-urban medical facilities, in some cases, resulting in issues of access in remote areas. |
| Category | 2026 to 2036 Projections |
|---|---|
| Regulatory Landscape | Utilization of clear guidelines for combination treatments and advanced treatment methods, with standardized procedures and patient safety |
| Technological Advancements | Integration of cryotherapy and radiation therapy in treatment regimens, development of personalized medicine approaches, and advancements in non-invasive treatment technologies. |
| Consumer Demand | Growing demand for individually customized treatment regimens, wherein physicians and patients work together to customize therapies according to particular scar characteristics and genetic predispositions. |
| Market Growth Drivers | Growth in emerging nations with developing healthcare systems, more emphasis on early intervention and prevention measures, and collaborative alliances between pharmaceutical industry players and research institutions to promote innovation and access. |
| Sustainability | Widespread utilization of sustainable procedures, such as the utilization of biodegradable products, environmentally friendly manufacturing, and efforts at reducing the carbon footprint of treatment development and supply. |
| Supply Chain Dynamics | Supply chain optimization using digital technologies and e-commerce platforms, improving transparency, efficiency, and accessibility, providing timely delivery of therapies to a global patient base, including those in remote and underserved areas. |
Market Outlook
The United States keloid treatment market keeps growing as more people look for good ways to deal with keloid scars. More keloids in people with darker skin, mean more demand for better treatments. New ideas in laser therapy biologic drugs, and mixed treatments are making things better for patients. Also more people know about keloids now, it's easier to see a skin doctor, and personalized medicine is getting better, which all help the market grow.
Market Growth Factors
| Country | CAGR (2025 to 2035) |
|---|---|
| United States | 6.7% |
Market Outlook
Germany's keloid treatment market looks set to grow, thanks to its strong healthcare system and focus on medical research. The country's advanced skin care setup helps doctors use new treatments like laser therapy, freezing, and special drugs. Also ongoing skin research, plus patients knowing more and getting better care, should improve treatment choices and help the market grow.
Market Growth Factors
Market Forecast
| Country | CAGR (2025 to 2035) |
|---|---|
| Germany | 3.2% |
Market Outlook
China's keloid therapy is designed for a significant increase in the market, which increases emphasis on extended health care investments and dermatology care. With an expansion of the country's healthcare infrastructure, highly developed treatment processes such as laser therapy and biologic drugs come into play.
As awareness regarding skin diseases is on the rise, medical centers are being enhanced and dermatological research is becoming better, hence the demand for efficient keloid treatments is supposed to grow, thus leading to quick growth in the market.
Market Growth Factors
Market Forecast
| Country | CAGR (2025 to 2035) |
|---|---|
| China | 4.6% |
India's keloid remedy marketplace is noticing solid growth with the help of enhancing awareness towards skin care along with significant progress within healthcare infrastructure. With increasing people looking for efficient treatments against keloids, there's evolving need for more recent alternatives in terms of laser treatment, cryotherapy, as well as biologic capsules. Additionally, the expansion of healthcare centers is making these remedies more widely available.
Market Growth Factors
Market Forecast
| Country | CAGR (2025 to 2035) |
|---|---|
| India | 5.4% |
Brazil's Keloid treatment market is expanded, increases investments in the health care system and increase the focus on the conditions of dermatology. Health services for the country's improvement in the health care system infrastructure and increasing access to advanced treatment options, such as laser therapy and biological medicines, operate market growth. In addition, the availability of awareness of skin health and the availability of special dermal care contributes to the growing demand for effective keloid remedies and accelerates market extension.
Market Growth Factors
Market Forecast
| Country | CAGR (2025 to 2035) |
|---|---|
| Brazil | 3.9% |
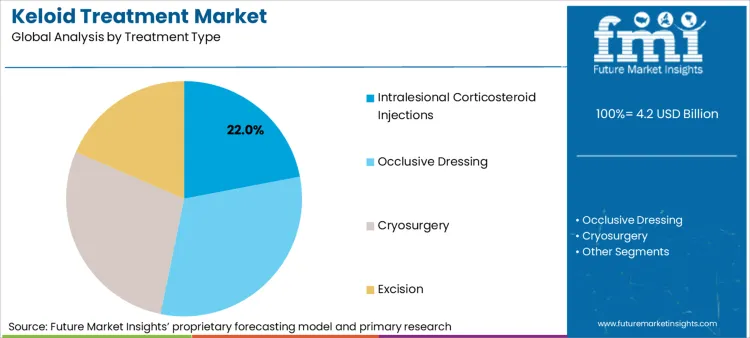
Intralesional Corticosteroid Injections are expected to dominate as they are widely used first-line treatments for keloids
Intralesional corticosteroid injection is among the most common first-line treatments of keloids that provides potent anti-inflammatory activity for decreasing the volume, redness and pain associated with the keloid marks. The injections that typically employ Triyinolone acetonide function in the inhibition of fibroblast growth and formation of collagen such that the resulting keloid markings are flattened and softened over the passage of time.
The increasing prevalence of hypertrophic and keloid scars, increasing the minimum invasive scar remedies and increasing the patient preference for non-surgical solutions is increasing. North America and Europe lead to corticosteroid injection adoption, while Asia-Pacific is looking at increasing demand due to awareness and skin care availability.
Future innovations include AI-Assisted Keloid Evaluation tools for personal treatment plans, integrate corticosteroids with laser treatment for the next generation corticosteroid formulation, and combination treatment with long-lasting effects.
Laser therapy segment holds the second largest share due to their effectiveness in managing complex keloids
Laser treatment is a novel and very effective treatment for keloids, particularly to minimize scar pigmentation, flat scars, and enhance general skin quality. Partial laser, pulsed dye laser (PDL), and COO lasers are most commonly utilized to destroy blood vessels and redundant collagen, stimulating remodeling of keloid tissue without extensive injury to surrounding skin.
Increasing demand for non-invasive scar modification increases the use of laser treatment in combination with corticosteroid injections, and fuel is fueling market expansion in laser technology for accurate targeting of fibrotic tissue.
North America and Europe dominate laser therapy, while the Asia-Pacific medical beauty is facing increasing demand due to the expansion of beauty clinics. Future progresses include AI-powered laser prohibition targeting, laser therapy equipment at home for mild keloid scars, and laser-immunotherapy treatment for prolonged scars.
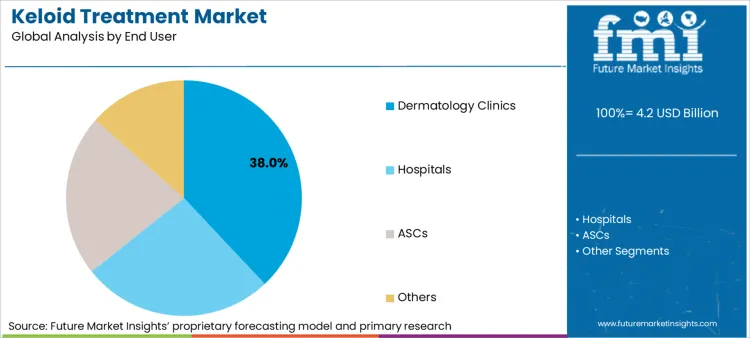
Hospitals segment will dominate due to advanced treatment options and specialized care
Hospital keloid treatment represents the largest end-user segment in the market, as they provide comprehensive scar management solutions including corticosteroid injections, cryosurgery, excitation and radiation therapy for severe cases. Dermatologists, plastic surgeons and radiation in the hospital provide access to multi -discourse teams of oncologist, which ensure individual and effective keloid treatment plans.
Increasing prevalence of surgical and post-burn keloids, government healthcare services in dermatology services, and radiation therapy and surgical sorting are increasing the increasing demand for hospital-based advanced keloid remedies.
North America and Europe lead to hospital-based keloid treatment, while Asia-Pacific hospitals are observing an increase due to increasing access to special dermal care. Future trends include integration of regenerative therapy in AI-managed keloid risk assessment tools, hospital-based 3D-affected skin graft solutions, and keloid treatment protocols.
Dermatology clinics is also a key segment as they provide non-surgical solution for keloid.
Dermatology clinics are fast emerging as the first choice for keloid treatment, particularly for non-surgical procedures such as laser therapy, cryosurgery, and corticosteroid injections. Dermatology clinics offer specialized, outpatient-based procedures with little downtime, which is why they are increasingly being sought out by patients seeking aesthetic scar revision procedures.
The increasing need for cosmetic dermatology treatments, greater access to more recent scar therapy techniques in private clinics, and rising demand for non-surgical and combination therapies are fueling market growth. North America and Europe lead in dermatology clinic-based treatment of keloid, and the Asia-Pacific region is observing aggressive growth because of the expanding availability of medical aesthetic treatments and growing patient awareness regarding scar management treatments.
Emerging innovations include AI-based digital consultations for keloid evaluation, clinic-based biophotonic treatment for scar remodeling, and hybrid laser-microneedling treatments for better collagen regulation.
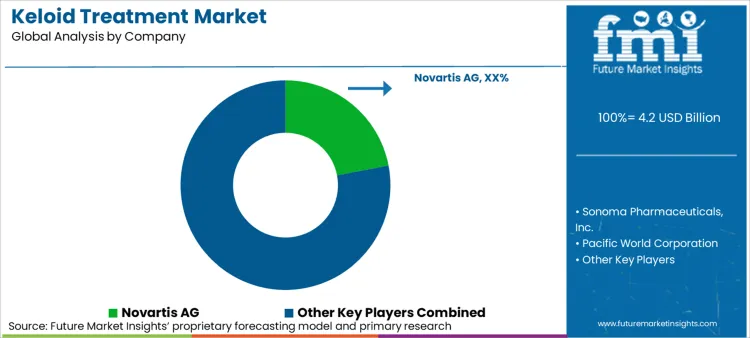
The market for keloid treatment is competitive, fueled by rising keloid scarring, innovations in dermatologic treatments, and increased demand for reconstructive and aesthetic procedures. Firms are investing in surgical procedures, corticosteroid injections, laser treatment, cryotherapy, and other treatments in order to retain a competitive market position. Well-established pharmaceutical companies, dermatology companies, and new biotech players influence the market, and each contributes to the changing scene of keloid management.
Market Share Analysis by Company
| Company Name | Estimated Market Share (%) |
|---|---|
| Novartis AG | 22-26% |
| Bristol-Myers Squibb Company | 18-22% |
| Perrigo Company plc. | 10-14% |
| Sensus Healthcare | 8-12% |
| RXi Pharmaceuticals, Inc. | 5-9% |
| Other Companies (combined) | 35-42% |
Recent Developments:
| Company Name | Key Offerings/Activities |
|---|---|
| Novartis AG | Novartis provides biologic treatments and corticosteroids for keloid treatment. Their treatment is targeted at minimizing scar formation and facilitating healing of the skin, such as topical advanced solutions with a purpose of preventing recurrence. |
| Bristol-Myers Squibb Company | Bristol-Myers Squibb creates immune modulators and biologic therapies that specifically target important biological pathways in keloid formation. Its therapies work to decrease excess scar tissue and enhance aesthetic results. |
| Perrigo Company plc. | Perrigo offers over-the-counter treatment for keloids in the form of silicone-based gels and sheets that diminish the visibility of scars and avoid keloid formation. Their solutions are non-surgical and cater to at-home scar care. |
| Sensus Healthcare | Sensus Healthcare offers non-invasive laser therapy devices for the treatment of keloids. Their focus ultrasound technology reduces scar tissue, restores skin appearance, and prevents recurrence. |
Novartis AG
Novartis AG is a top pharmaceutical firm providing innovative keloid management treatments. The firm specializes in biologic drugs, corticosteroids, and innovative topical remedies to inhibit scar formation. Novartis spends significantly on R&D to invent combination therapies that enhance efficacy while reducing side effects, responding to increasing demand for safer and long-term keloid treatment.
Bristol-Myers Squibb Company
Bristol-Myers Squibb is focusing on immune-modulating and biologic therapies specific to keloid development. Research in precision medicine is being enhanced by the firm to create keloid-prevention targeted therapies for blocking excessive deposition of collagen, which is at the center of keloid scar development. Being positioned with an extensive pipeline of dermatology-purposed biologics, Bristol-Myers Squibb is toward developing long-term keloid treatments and prevention methods.
Perrigo Company plc.
Perrigo Company plc. is a leading provider in the over-the-counter (OTC) keloid treatment segment, providing silicone-based sheets, gels, and topical solutions for managing scars. Perrigo specializes in offering cost-effective, non-surgical treatment solutions for the reduction of keloid visibility and recurrence. Perrigo continues to grow its product portfolio with new, high-level scar therapy products that are responding to the increased consumer demand for over-the-counter at-home treatment.
Sensus Healthcare
Sensus Healthcare is pioneering non-invasive keloid solution providers, dedicated to laser-based and radiation-dependent scar management technology. The highly developed devices provided by the company, including superficial radiation therapy (SRT) systems, serve as efficient solutions in place of surgical procedures to prevent recurrence while ensuring better-looking outcomes for patients with keloids. Sensus Healthcare is progressing toward further augmenting its capabilities for precision treatments.
RXi Pharmaceuticals, Inc.
RXi Pharmaceuticals, Inc. is a biopharmaceutical firm engaged in RNA-based therapeutics for the treatment of keloids. Its drug technology involves proprietary RNA interference (RNAi) that targets genes involved in keloid over-scar formation, representing a new platform for keloid recurrence prevention. RXi continues to develop its work in gene-modulating therapies with the goal of providing long-term and customized treatment options for keloid management.
Other Key Players
Beyond the leading companies, several other manufacturers contribute significantly to the market, enhancing product diversity and technological advancements. These include:
The keloid treatment market represents revenue generated from the sale of medical devices, pharmaceutical products, and clinical services used in the prevention, reduction, and management of keloid scars. The market measures the value of laser and light-based systems, injectable therapies (corticosteroids, 5-fluorouracil), topical products (silicone, molecular inhibitors), cryotherapy devices, and radiation-based keloid treatments.
Inclusions cover fractional CO2 and Nd:YAG laser systems with keloid indications, injectable corticosteroid and 5-FU treatment protocols, silicone gel sheeting and topical silicone products, emerging molecular inhibitor therapies (LOX inhibitors, hedgehog pathway modulators), cryotherapy devices, and superficial radiation therapy (SRT) systems for post-excision keloid prevention. AI-assisted keloid mapping and treatment planning platforms are also included.
Exclusions include general dermatology laser systems without keloid-specific clearance, plastic surgery fees for keloid excision not bundled with device or pharmaceutical product revenue, and scar camouflage cosmetic products. General wound care products not marketed for keloid management are outside the scope.
| Items | Values |
|---|---|
| Quantitative Units (2026) | USD 4,292.2 Million |
| Product Type | Laser and Light-Based Systems, Injectable Therapies, Topical Products, Cryotherapy Devices, Radiation Therapy Systems, Molecular Inhibitors |
| Material Type | Fractional CO2 Laser, Nd:YAG 1064nm, Corticosteroid Injections, Silicone Gel, LOX Inhibitors, 5-Fluorouracil |
| End-Use / Channel | Dermatology Clinics, Plastic Surgery Centers, Hospital Outpatient Departments, Specialized Keloid Clinics |
| Regions Covered | North America, Europe, Asia Pacific, Latin America, Middle East and Africa |
| Countries Covered | USA, UK, Japan, South Korea, India, Brazil, Nigeria, and 40+ countries |
| Key Companies Profiled | Bausch Health (Solta), Sciton, Candela, Merck (Dermatology), Galderma, BirchBioMed, Syntara, Sirnaomics |
| Additional Attributes | Revenue analysis by treatment modality and clinical setting, pipeline drug Phase tracking, FDA 510(k) and PMA clearance monitoring, reimbursement landscape assessment, and multi-modal protocol adoption rates |
Intralesional corticosteroid injections, occlusive dressing, cryosurgery, excision, radiation therapy, laser therapy and others
Hospitals, dermatology clinics, ASCs and others
North America, Latin America, Western Europe, Eastern Europe, East Asia, South Asia & Pacific, Middle East & Africa
What is the current global market size for Keloid Treatment?
The global market is valued at USD 4,292.2 Million in 2026, driven by next-generation laser clearances and emerging molecular inhibitor therapies entering clinical trials.
What is the projected Compound Annual Growth Rate (CAGR) for the market over the next 10 years?
The market is projected to grow at a CAGR of 3.8% from 2026 to 2036.
Which regions are experiencing the fastest expansion?
Asia Pacific leads growth driven by dermatology infrastructure expansion in Japan and South Korea, followed by North America where multi-modal treatment adoption is highest.
What are the primary market drivers?
Regulatory clearance of keloid-specific laser systems and the advancement of topical molecular inhibitors through clinical trials are the primary drivers.
Who are the leading suppliers in the industry?
Bausch Health (Solta), Sciton, Candela, and Galderma are key players, differentiating through keloid-specific device clearances and integrated treatment protocol development.
Full Research Suite comprises of:
Market outlook & trends analysis
Interviews & case studies
Strategic recommendations
Vendor profiles & capabilities analysis
5-year forecasts
8 regions and 60+ country-level data splits
Market segment data splits
12 months of continuous data updates
DELIVERED AS:
PDF EXCEL ONLINE

Thank you!
You will receive an email from our Business Development Manager. Please be sure to check your SPAM/JUNK folder too.